Biliary microbiota and mucin 4 impact the calcification of cholesterol gallstones
2021-03-05FengLingHuHongTanChenFangFangGuoMingYangXinJiangJingHuaYuFenMingZhangGuoQiangXu
Feng-Ling Hu , Hong-Tan Chen , Fang-Fang Guo , Ming Yang, Xin Jiang, Jing-Hua Yu,Fen-Ming Zhang, Guo-Qiang Xu
Department of Gastroenterology, the First Affiliated Hospital, Zhejiang University School of Medicine, Hangzhou 310 0 03, China
Keywords:Cholesterol gallstones Calcification Mucins Microbiota
ABSTRACT Background: Cholesterol gallstones account for over 80% of gallstones, and the pathogenesis of gallstone formation involves genetic and environmental factors. However, data on the evolution of cholesterol gallstones with various densities are limited. This study aimed to determine the roles of microbiota and mucins on the formation of calcified cholesterol gallstones in patients with cholelithiasis.Methods: Paired gallbladder tissues and bile specimens were obtained from cholelithiasis patients who were categorized into the isodense group and calcified group according to the density of gallstones.The relative abundance of microbiota in gallbladder tissues was detected. Immunohistochemistry and enzyme-linked immunosorbent assay were performed to detect the expression levels of MUC1, MUC2,MUC3a, MUC3b, MUC4, MUC5ac and MUC5b in gallbladder tissues and bile. The correlation of microbiota abundance with MUC4 expression was evaluated by linear regression.Results: A total of 23 patients with gallbladder stones were included. The density of gallstones in the isodense group was significantly lower than that of the calcified group (34.20 ±1.50 vs. 109.40 ±3.84 HU, P < 0.0 0 01). Compared to the isodense group, the calcified group showed a higher abundance of gram-positive bacteria at the fundus, in the body and neck of gallbladder tissues. The concentrations of MUC1, MUC2, MUC3a, MUC3b, MUC5ac and MUC5b in the epithelial cells of gallbladder tissues showed no difference between the two groups, while the concentrations of MUC4 were significantly higher in the calcified group than that in the isodense group at the fundus (15.49 ±0.69 vs. 10.23 ±0.54 ng/mL, P <0.05), in the body (14.54 ±0.94 vs. 11.87 ±0.85 ng/mL, P < 0.05) as well as in the neck (14.77 ±1.04 vs.10.85 ±0.72 ng/mL, P < 0.05) of gallbladder tissues. Moreover, the abundance of bacteria was positively correlated with the expression of MUC4 ( r = 0.569, P < 0.05) in the calcified group.Conclusions: This study showed the potential clinical relevance among biliary microbiota, mucins and calcified gallstones in patients with gallstones. Gram-positive microbiota and MUC4 may be positively associated with the calcification of cholesterol gallstones.
Introduction
Gallstone disease (GSD) is one of the most prevalent gastrointestinal diseases, with a substantial economic and social burden worldwide. GSD has an estimated prevalence of 10% -20% in adults in the Western world [1], ranking as the leading cause of surgical intervention of the biliary tree in the United States [2]. The incidence of GSD in China is about 13% [3], which is gradually increasing due to westernized lifestyle as well as the widespread use of ultrasonography [4]. About 75% of adult patients are asymptomatic, but GSD is the major reason of cholecystitis, pancreatitis or other complications [5]. Moreover, gallstones significantly increase the risks of cardiovascular disease [ 6 , 7 ], leading to serious public health issues.
Gallstones are comprised of cholesterol in 75% -80% cases [8],whereas the remaining 20% are pigmented stones formed by calcium bilirubinate [9]. Cholesterol gallstone formation is resulted from both genetic and environmental factors [10]. The hypersecretion of free cholesterol is the primary pathogenic factor, leading to supersaturated bile in the gallbladder [11]. Accelerated cholesterol crystallization, accumulation of mucins, and impaired gallbladder motility are the secondary pathogenic factors [12]. Intestinal factors involving slow intestinal motility, absorption of cholesterol,and altered gut microbiota are also related to the pathogenesis of gallstones [ 13 , 14 ].
Although the pathogenesis of gallstones has been widely studied recently, the molecular mechanisms still remain unclear. Treatments of symptomatic GSD are predominantly invasive and are based on surgery. There are no prophylactic drugs available for gallstones, and medical dissolution therapy used for cholesterol gallstones is usually minimally effective, with a recurrence of over 50% [15]. Moreover, the calcification of gallstones also decreases the efficiency of litholysis [16]. To improve the effectiveness of nonsurgical treatments of cholesterol gallstones, identifying the main factors that modulate the calcification of gallstones is necessary.
A recent research has linked mucin genes and microbiota in the formation of cholesterol gallstones [8]. Biliary mucins play important roles in the pathogenesis of cholesterol gallstones due to their ability to bind biliary lipids and accelerate cholesterol crystallization [17]. A study by Goral demonstrated relationships between gallstones and several mucins, such as MUC2 and MUC4 [18]. Alterations in the indigenous microbiome significantly affect the accumulation of mucin gel and normalize gallbladder weight, which may participate in the pathogenesis of cholesterol gallstones [19]. However, whether biliary microbiota and mucins are associated with the calcification of cholesterol gallstones still remains unclear.
The present study tried to figure out whether the biliary microbiota or mucins were responsible for the calcification of gallstones.16S rRNA or ribosomal DNA (rDNA) sequence analysis has become a major tool to determine the relationships between bacteria, and is widely used for identification purposes. Therefore, we detected the amount and composition of bacteria via 16S rDNA detection in gallbladder tissues of patients with isodense and calcified gallstones. Moreover, we evaluated the expression of mucins in bile and gallbladder tissues in different groups. The correlation analysis was also conducted to identify the potential correlation between biliary microbiota and mucin proteins in the formation and calcification of gallstones.
Patients and methods
Patient specimens
A total of 23 gallbladder tissues and paired bile specimens were obtained from patients who had gallbladder stones and underwent cholecystectomy in our hospital from December 2018 to June 2019.This study was approved by the Ethics Committee of the First Affiliated Hospital, Zhejiang University School of Medicine (No. 2020–0393). All the patients received abdominal dual-energy CT scan by the same machine before surgery. Gallstones with density values no more than 50 HU were identified as isodense stones, which has little calcium content. The density values of calcified gallstones are always greater than 50 HU [20]. Among the 23 patients, 10 were diagnosed as isodense gallstones, 13 were calcified gallstones.
Immunohistochemistry staining
The protein expression levels of mucins in gallbladder tissues were examined with the LSAB + Kit (Dako Cytomation, Glostrup,Denmark). Anti-MUC1 (L S-B12807), anti-MUC2 (L S-B16111), anti-MUC3a (LS-C87834), anti-MUC3b (LS-C164680), anti-MUC4 (LSB6781), anti-MUC5ac (LS-B2434) and anti-MUC5b (LS-C153937)were obtained from LSbio Corp. (Seattle, WA, USA). Slides were examined independently by two investigators. The extent and intensity grades of immunohistochemistry staining were used to define protein expressions.
Enzyme-linked immunosorbent assay (ELISA)
ELISA assays were performed according to protocols provided by the kit manufacturers. ELISA kits for detecting MUC1(Abx152393), MUC2 (Abx152395), MUC3a (Abx575985), MUC4(Abx152399), MUC5ac (Abx196059) and MUC5b (Abx250243) were obtained from Abbexa Inc. (Cambridge, UK). ELISA kit for MUC3b(LS-F29535) was obtained from LSbio Corp. (Seattle). Protein extracts were diluted appropriately until protein concentrations fell within the dynamic range. Briefly, 1 mL of liquid sample was extracted in the heated extraction buffer (60 °C). Samples were briefly mixed using a pulse-vortex technique and were extracted in a 60 °C shaking water bath for the recommended amount of time.Samples were then cooled to room temperature and 1 mL of each extract was centrifuged for 5 min at 16,0 0 0 ×g. Additional dilutions were performed as necessary. Each extract was analyzed in triplicate wells in a 96-well microtiter plate. After adding the stop solution, the absorbance of each sample was detected at 450 nm wavelength.
Gram staining of gallbladder tissues
Gram staining was performed as previously recommended [21].In brief, formalin-fixed paraffin-embedded (FFPE) gallbladder tissues were stained with Gentian violet (G2039, Sigma-Aldrich, St.Louis, MO, USA) or Safranin O reagent (S2255, Sigma-Aldrich), respectively. The results of staining were detected under oil immersion (100 ×) using a bright field microscope.
rDNA detection of gallbladder tissues
Genomic DNA (gDNA) was extracted from gallbladder specimens with the QIAamp DNA Mini Kit (QIAGEN, Hilden, Germany)as manufacturer’s instructions. gDNA was subjected to qPCR to determine the amounts of bacteria by detecting the expression of 16S rDNA. Each reaction contained 100 ng of gDNA and was assayed in triplicate in 10μL reactions in a 96-well optical PCR plate. Amplification and detection of DNA were performed with the ABI Step One Plus Real-Time PCR System (Applied Biosystems, Foster City,CA, USA) under the following reaction conditions: 10 min at 95 °C,followed by 40 cycles of denaturation at 95 °C for 15 s and at 60°C for 1 min. The primers were as follows: forward primer, 566F 5 ′ -GGG TTT AAA GGG AGC GTA GG-3 ′ and reverse primer, 692R 5 ′ -CTA CAC GAA TTC CGC CT-3 ′ . The cycle threshold (Ct) values were normalized using human endogenous 18S as a reference gene.
Statistical analysis
Statistical analyses were carried out with SPSS 22.0 (SPSS Inc.,Chicago, IL, USA). Data from at least three independent experiments were presented as mean ±SEM. Error bars in the scatter plots and the bar graphs represent SEM. Data between two groups were calculated by Student’st-test. The correlation of microbiota abundance with MUC4 expression was evaluated by linear regression. Statistical tests were two-tailed and aP<0.05 was considered statistically significant.
Results
Baseline characteristics of enrolled patients
The clinical characteristics of patients were summarized in Table 1 . There was no difference in either age or sex between thetwo groups. The density of gallstones in the isodense group was significantly lower than that in the calcified group (34.20 ±1.50 vs.109.40 ±3.84 HU,P<0.0 0 01).

Table 1 Characteristics of enrolled patients with gallstone disease.

Fig. 1. Changes in dominant flora in different groups of gallbladder tissues. A-C : Real-time PCR was performed in the fundus ( A ), body ( B ) and neck ( C ) of gallbladder tissues; D : gram staining was performed in the gallbladder tissues (arrows indicating the positive cells).
Changes in dominant flora in different groups of gallbladder tissues
The abundance of bacteria at the fundus, in the body as well as neck of gallbladder in the calcified group was significantly higher compared to that of the isodense group ( Fig. 1 A-C). In addition, Gram staining showed that Gram-positive bacteria was dominant in the calcified group, while the difference of Gram-negative bacteria between the two groups was not significantly different( Fig. 1 D).
Protein expression levels of MUC4 were associated with gallstone calcification
The primary mucins expressed in the normal gallbladder were MUC3, MUC5b, and MUC6 [22]. We also found the protein expressions of MUC1 and MUC4 in gallbladder tissues with gallstones.The protein levels of MUC2 were weak in both groups. However,the expression level of MUC4 was significantly increased in the calcified group compared to that of the isodense group ( Fig. 2 A,B), rather than other MUC proteins ( Fig. 2 C). The results indicated that MUC4 may participate in the calcification of gallstones.
The concentrations of MUC4 were significantly higher in the calcified group than that in the isodense group at the fundus(15.49 ±0.69 vs. 10.23 ±0.54 ng/mL,P<0.05), in the body(14.54 ±0.94 vs. 11.87 ±0.85 ng/mL,P<0.05) as well as in the neck (14.77 ±1.04 vs. 10.85 ±0.72 ng/mL,P<0.05) of gallbladder tissues ( Fig. 3 A-C). However, the concentrations of MUC1, MUC2,MUC3a, MUC3b, MUC5ac and MUC5b showed no significant difference between the two groups ( Fig. 3 D-I). In addition, the concentrations of MUC1, MUC2, MUC5ac and MUC5b content in bile specimens were not significantly different between the two groups( Fig. 4 ).
Correlation of MUC4 with biliary microbiota in gallbladder tissues
There was no correlation between MUC4 and the abundance of microbiota in the isodense group (r= -0.2738,P>0.05). In the calcified group, the abundance of bacteria was positively correlated with the expression of MUC4 protein (r= 0.5690,P<0.05), suggesting the synergistic effects of MUC4 and biliary microbiota in the calcification of cholelithiasis ( Fig. 5 ).
Discussion
GSD is a prevalent hepatobiliary disease worldwide and can be caused by environmental influences, genetic factors and their interactions [23]. Gallstones vary in composition and calcification patterns, while the main factors involved in the formation and calcification of gallstones have not been identified.
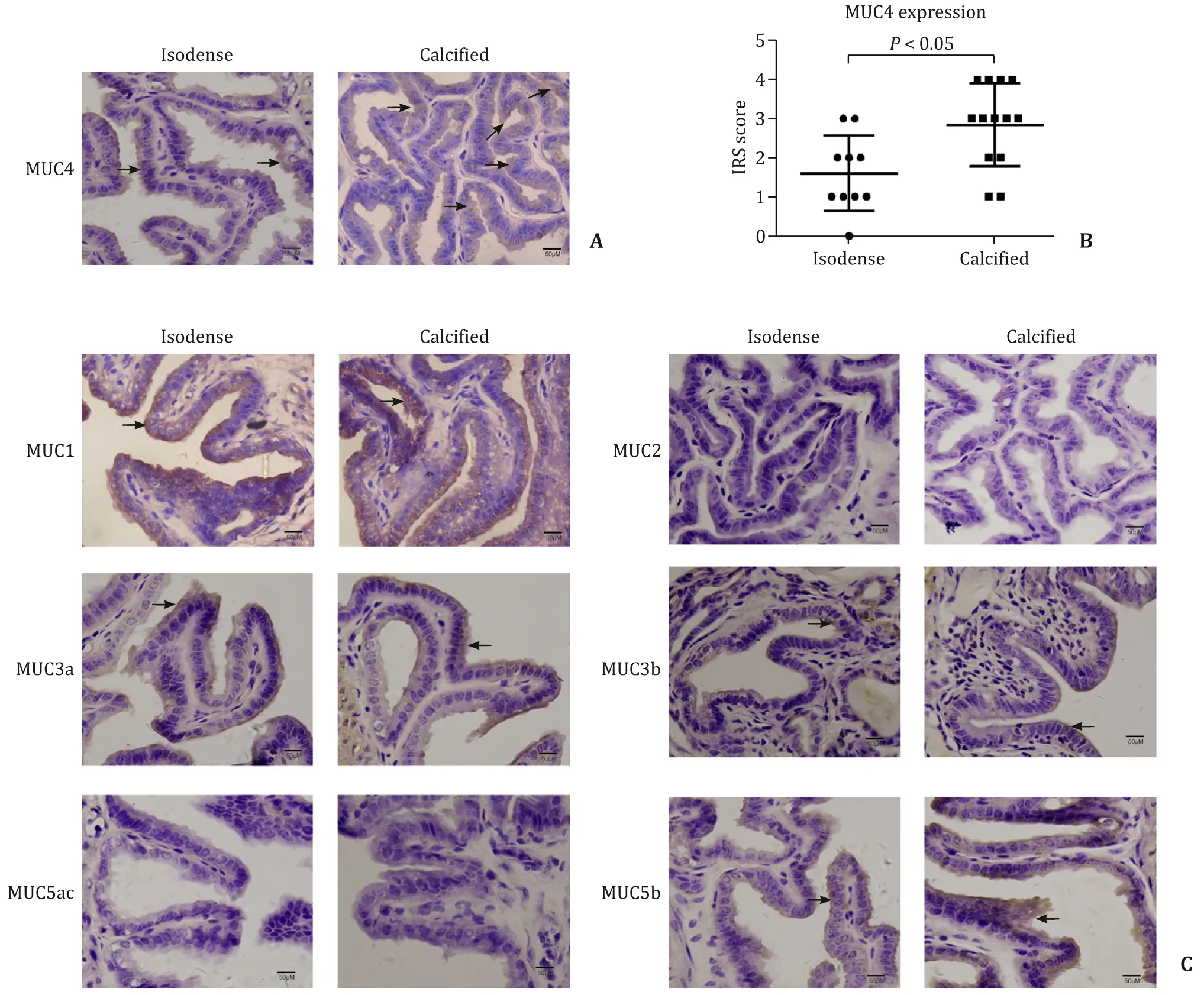
Fig. 2. The protein expression levels of MUC4 were associated with gallstone calcification (arrows indicating the positive cells). A: Representative immunohistochemistry of MUC4 in isodense and calcified groups; B: statistical analysis of immunohistochemical immunoreactive score of Remmele and Stegner (IRS) score of MUC4; C: representative immunohistochemistry of MUC1, MUC2, MUC3a, MUC3b, MUC5ac and MUC5b in isodense and calcified groups.
A previous research indicates the roles of gut microbiota in the pathogenesis of cholesterol GSD [24]. In this study, we found that gallbladder tissues with calcified gallstones contained increased abundance of biliary bacteria, especially the Gram-positive bacteria. The relevance of Gram-positive bacteria and calcified gallstones indicates the possible interaction of biliary microbiota in the calcification of gallstones.
Phospholipase and mucins are also considered bacteria-related factors in the formation of solid gallstones [23]. Alteration expressions of mucins are related to the pathogenesis of gallstone formation. Human mucin genes include membrane-bound mucins (MUC1,MUC3,MUC4) and secretory mucins (such asMUC2,MUC5ac,MUC5b,MUC6) [25]. Under physiological conditions, the content of mucins remains in a low level. Abnormal mucin secretion or mucus behavior contributes to certain diseases, such as GSD [26], cholecystitis, biliary cancer, and cystic fibrosis-associated gallbladder disease [ 27 , 28 ]. Mucin overexpression in human gallbladders favors the gallstone formation [29]. However, the potential role of mucins in the calcification of gallstones remains unclear.
In the current study, we confirmed that the expression levels of MUC4 were increased significantly in the gallbladder tissues with calcified gallstones, which was in line with the abundance of Gram-positive bacteria. The concentration of MUC4 was very low in normal gallbladder tissues as well as in bile, while the overexpression of MUC4 in bile often correlates with the malignant neoplasm [30]. We failed to detect MUC4 in bile of gallstone patients, which may be due to our small sample size and the low secretion of MUC4 in the bile of benign gallbladder disease. Nevertheless, the upregulated MUC4 mucin in gallbladder tissues of GSD patients has been identified, with even higher concentrations in the calcified group. MUC4 may favor the bacterial adhesion and growth, which may further promote the formation and calcification of gallstones.
However, due to the small sample size and the limited detection methods, our research only explored the overall abundance changes of the biliary flora in each group, but failed to isolate specific pathogenic strains. Previous researches regarding the gallstone cultures obtained from cholelithiasis patients were similar to those of bile cultures [31], andKlebsiella,E.coli,Enterococci,Enterobacter,Pseudomonas,Acinetobacter, andCitrobacterwere the dominant isolated aerobic species [32]. However, the specific bacteria species regarding calcification of gallstones and gallstone-associated infections still need further investigation. Antibiotic susceptibility may be a critical indicator for therapeutic strategies [33].
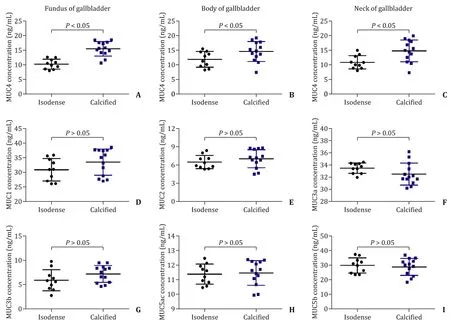
Fig. 3. The content of mucins was detected in gallbladder tissues. A-C : The concentrations of MUC4 in the fundus ( A ), body ( B ) and neck ( C ) of gallbladder tissues in two groups; D-I: the concentrations of MUC1, MUC2, MUC3a, MUC3b, MUC5ac and MUC5b in gallbladder tissues in two groups.
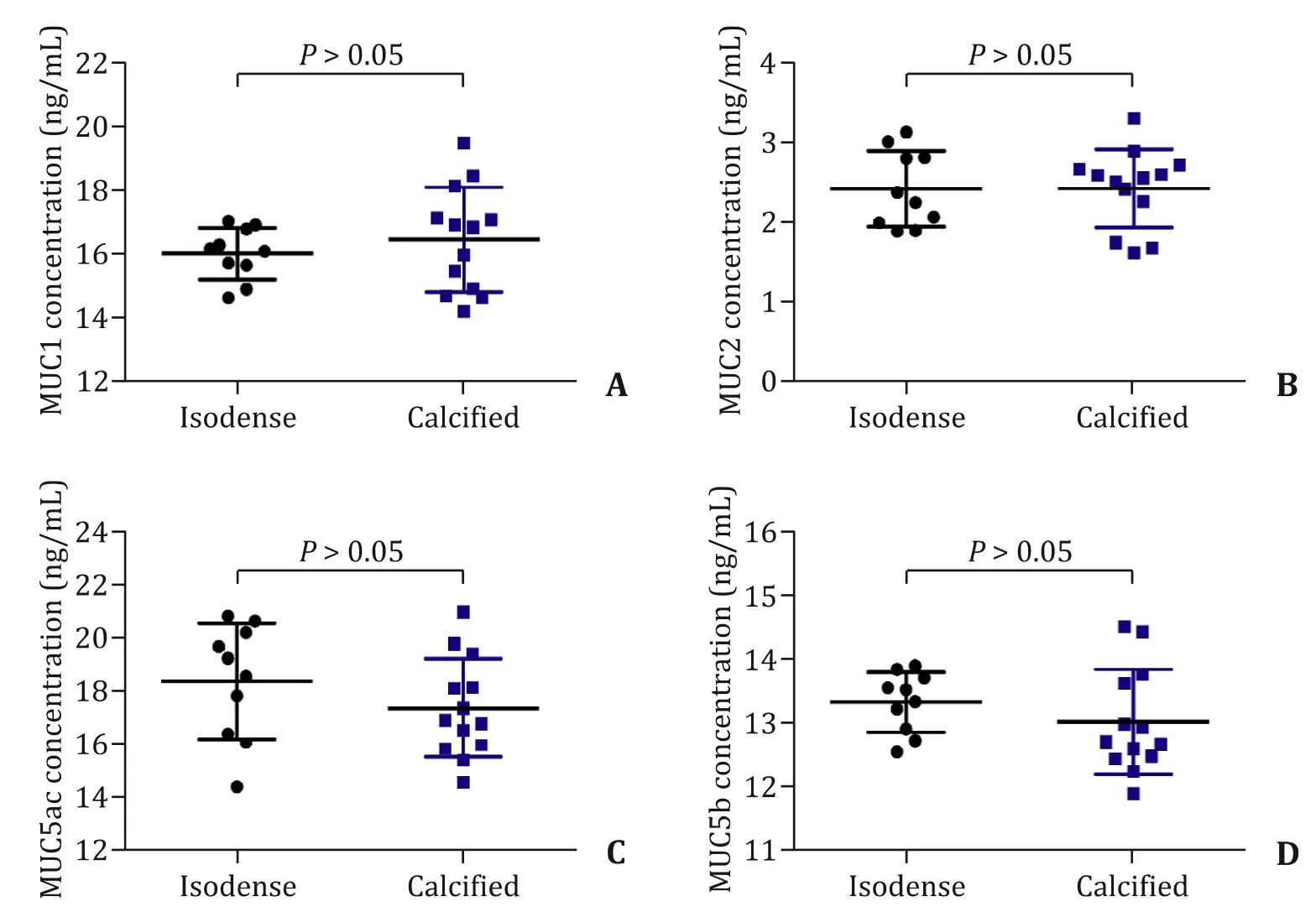
Fig. 4. The concentrations of MUC1, MUC2, MUC5ac and MUC5b were detected in the bile specimens.
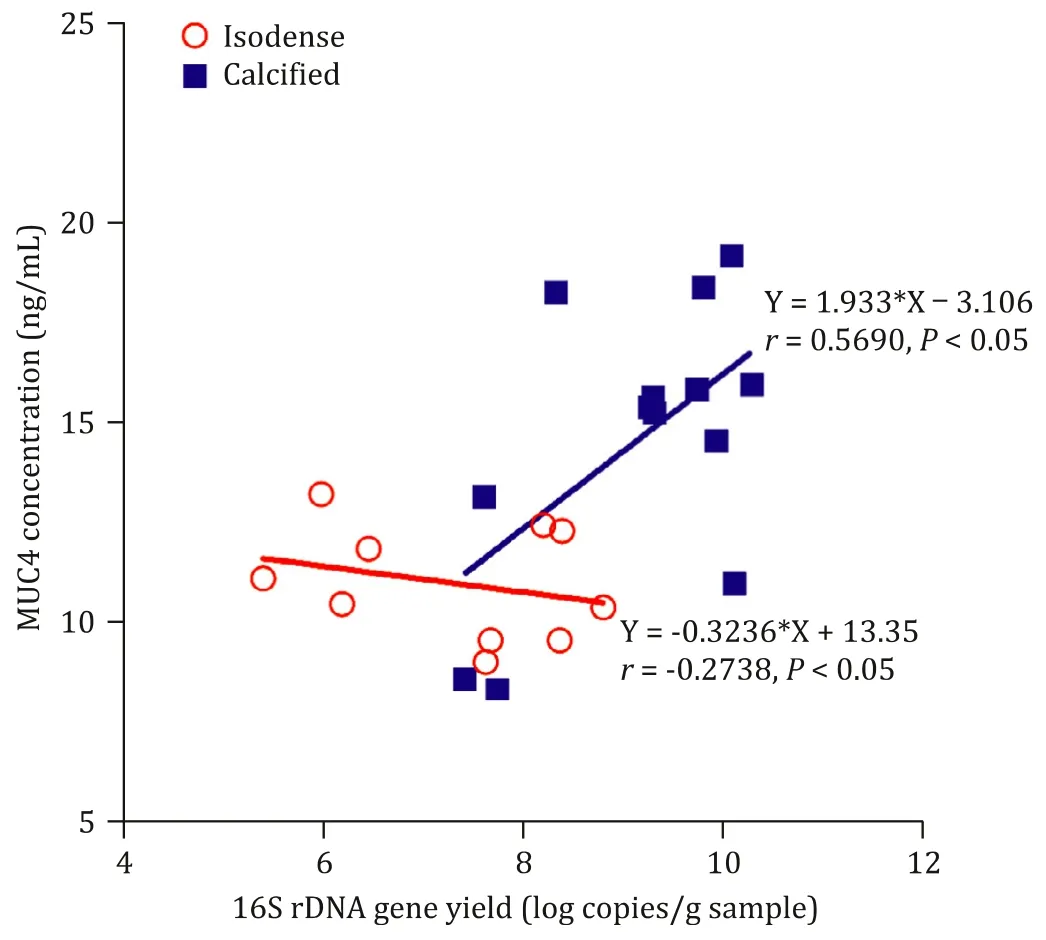
Fig. 5. Correlation of MUC4 and biliary microbiota in gallbladder tissues.
Taken together, our data indicate that alterations in the biliary microbiome may alter cholesterol gallstone pathogenesis in a manner consistent with increased mucin gene expression and mucin gel accumulation. Microbial characterization may serve as a valuable tool for predicting individuals who are vulnerable to cholesterol gallstones. Finding modifiable pathogenic factors for cholesterol gallstone calcification will also be helpful for nonsurgerical treatment of cholesterol GSD.
Acknowledgments
We thank Li-Li Wang for sample collection.
CRediT authorship contribution statement
Feng-Ling Hu:Data curation, Formal analysis, Writing - original draft.Hong-Tan Chen:Data curation, Formal analysis, Writing - original draft.Fang-Fang Guo:Data curation, Formal analysis,Writing - original draft.Ming Yang:Investigation.Xin Jiang:Data curation, Formal analysis, Methodology, Resources, Software.Jing-Hua Yu:Investigation.Fen-Ming Zhang:Investigation.Guo-Qiang Xu:Conceptualization, Funding acquisition, Supervision, Writing -review & editing.
Funding
This study was supported by grants from the National Natural Science Foundation of China ( 81870433 ), and the International Cooperation Project of Zhejiang Province Public Technology Research Program ( LGJ18H030 0 01 ).
Ethical approval
This study was approved by the Ethics Committee of the First Affiliated Hospital, Zhejiang University School of Medicine (No.2020-0393). Written informed consent was obtained from all participants.
Competing interest
No benefits in any form have been received or will be received from a commercial party related directly or indirectly to the subject of this article.
杂志排行
Hepatobiliary & Pancreatic Diseases International的其它文章
- Liver transplantation and BCLC classification: Limitations impede optimum treatment
- Do the existing staging systems for primary liver cancer apply to combined hepatocellular carcinoma-intrahepatic cholangiocarcinoma?
- Overlap of concurrent extrahepatic autoimmune diseases is associated with milder disease severity of newly diagnosed autoimmune hepatitis
- Aggressive surgical approach in patients with adrenal-only metastases from hepatocellular carcinoma enables higher survival rates than standard systemic therapy
- Integrating transcriptomes and somatic mutations to identify RNA methylation regulators as a prognostic marker in hepatocellular carcinoma
- The utility of two-dimensional shear wave elastography and texture analysis for monitoring liver fibrosis in rat model
