Synergistic anti-tumor effects of oncolytic virus therapy combined with sorafenib on human hepatocellular carcinoma
2017-11-02LIYajiaoLEIWenQINYunYINGChangZHOUXiumei
LI Yajiao, LEI Wen, QIN Yun, YING Chang, ZHOU Xiumei*
(1.Xinyuan Institute of Medicine and Biotechnology, College of Life Sciences,Zhejiang Sci-Tech University, Hangzhou 310018;2.Department of Hematology, the First Affiliated Hospital, College of Medicine,Zhejiang University, Hangzhou 310003)
Synergisticanti-tumoreffectsofoncolyticvirustherapycombinedwithsorafenibonhumanhepatocellularcarcinoma
LI Yajiao1, LEI Wen2, QIN Yun1, YING Chang1, ZHOU Xiumei1*
(1.Xinyuan Institute of Medicine and Biotechnology, College of Life Sciences,Zhejiang Sci-Tech University, Hangzhou 310018;2.Department of Hematology, the First Affiliated Hospital, College of Medicine,Zhejiang University, Hangzhou 310003)
Human hepatocellular carcinoma is one of the most common and high fatal cancer in the world. In China, the liver cancer rates is the highest in the world. Many researchers put into the research of liver cancer and tried many ways and methods, but still couldn’t find effective ways to cure hepatocellular carcinoma. In the recent years, oncolytic vaccinia virus become a promising novel way in cancer treatment because of its efficient anti-tumor effect, low side effects and good safety, and has been widely used in the treatment of cancer. Sorafenib, a new tumor-targeting kinase inhibitor, has shown its effective and safe chemotherapy drugs in treatment of HCC, and has already used in clinical trials. In the present study, we combined sorafenib with VV to explore its anti-tumor effects. Through in vitro experiments like MTT, Hoechst, Flow cytometry, Western blot andinvivotransplantation of tumor model and immunohistochemical analysis, it’s demonstrated that combination of sorafenib and vaccinia virus significantly inhibited the growth of tumor cells and promoted apoptosis of tumor cells. According to the result of mice transplanted tumor model, the date showed that with the extension of time, the combined group tumor size has a tendency to decline. Thus, the combination use of sorafenib and vaccinia virus might be a novel approach for liver cancer therapy.
oncolytic vaccinia virus; sorafenib; combination therapy; liver cancer
Liver cancer is one of the most common and fatal types of cancer in the world[1]. It is especially common in Asia, where its morbidity and mortality are significantly higher than any other continent[2]. Chinese patients alone account for 55% of new HCC(human hepatocellular carcinoma) cases worldwide[3]. Traditional treatment for HCC include surgery, chemotherapy and radiotherapy alone or in combination[4]. While these treatments are effective for the patients in the early phases of the disease, they are not as effective for those who are advanced liver cancer[5]. The ineffectiveness of standard therapies, in addition to the rising mortality of liver cancer, has made development of novel strategies to treat liver cancer imperative.
Oncolytic viruses are an emerging strategy for cancer therapeutics[6]. Oncolytic viruses preferentially infect tumor cells with minimal spread into normal tissues[7-9]. Virus replication leads to lysis of the tumor cell and eventual destruction of the tumor[10]. Over the past decade, clinical trials using oncolytic adenovirus, vaccinia virus (VV), and herpes simplex virus (HSV) have shown encouraging treatment effects against various types of cancer.
One of the most promising oncolytic viruses is oVV. There is mounting evidence that oVV is safe in vivo, displays rapid replication, and exhibits the highest oncolytic virus anti-tumor activity in vitro and in vivo. oVV is a member of the well-studied Poxviridae family, and has been modified to express various antigens, cytokines, and immunostimulatory molecules[11-14]. The oVV used in our study lacks the thymidine kinase (TK) gene which is necessary for virus replication. There tends to be high expression of TK in cancer cells but little or no expression in normal cells[15]. Thus, vaccinia virus constructs that lack the TK gene replicates in tumor cells but not in normal cells. Vaccinia virus infects cells via membrane fusion rather than receptors on the surface of the host cell. Virus release begins 8 h after infection and within 72 hours there will be rapid virus replication and cell lysis allow vaccinia virus to evade the host immune response.
Another emerging cancer therapeutic is sorafenib. Sorafenib is a multi-kinase inhibitor capable of inhibiting tumor growth[16]. Sorafenib inhibits several cell surface kinases implicated in cancer, including the RAF kinase, vascular endothelial growth factor receptor 2 and receptor 3 (VEGFR-2; VEGFR-3), platelet-derived growth factor receptor beta (PDGFR-beta), KIT and PLT[17-18]. Thus, sorafenib is capable of inhibiting multiple pathways that contribute to different tumor promoting processes. In addition, sorafenib is able to inhibit several tyrosine kinase receptor that related to new blood vessels and tumor development, thus blocking tumor angiogenesis. Sorafenib is the only FDA-approved systemic chemotherapeutic agent for the treatment of HCC and is currently recognized as a standard first-line therapy for advanced HCC[19-21].
In the present study, we examined the effect of combined oVV and sorafenib therapy on anti-tumor activity in vitro and in vivo. The combinatorial treatment is demonstrated to increase liver cell death in vitro and reduce tumor growth in vivo significantly. Thus, this combinatorial treatment might provide a novel effective strategy for future liver cancer therapies.
1 Materials and Methods
1.1 Materials
The human embryonic kidney cell line HEK293, human HCC cell lines PLC, Huh7, Hep3B were purchased from Shanghai cell collection (Shanghai China). Cells were cultured in Dulbecco’s modified Eagle’s medium (DMEM, GIBCO, Carlsbad, USA) supplemented with 10% fetal bovine serum (FBS, GIBCO). All cells were incubated at 37°C humidified air atmosphere with 5% CO2. The oVV were previously constructed and stored in our laboratory. The amplification of oVV was carried out by infecting HEK293 cells integrated the viral replication related genes. The purification of vaccinia virus was through sucrose gradient centrifugation.
1.2 Methods
1.2.1 Cell viability assay Human liver cell lines Hep3B, Huh7, PLC were seeded in 96-well plates at a density of 5 000 cells / well, incubated for 12 h at 37°C before infected with oVV and sorafenib. Cells were treated with different MOI of virus and different concentration of sorafenib. After 48 h, 3-(4, 5-dimethylthiazol-2-yl)-2, 5-diphenyltetrazolium bromide (MTT) solution in PBS (5 mg/mL, 20 μL) was added into each well. Then cells were incubated at 37°C for 4 h, and the supernatant was removed with 150 μL dimethyl sulfoxide added to each well. Cell viability was assessed by measuring the absorbance at 490 nm with a micro-plate reader after mixing thoroughly.
1.2.2 Hoechst 33342 staining Human liver cancer cell line Hep3B cells were seeded in 6-well plate, and then infected with oVV (10 MOI), sorafenib (20 μmol), and oVV (10 MOI) plus sorafenib (20 μmol), respectively, with uninfected cells served as control. 48 h later, cells were stained with Hoechst 33342 (Beyotime, Shanghai) at 1 mg/mL for 30 min at 37°C and observed under the inverted fluorescence microscope.
1.2.3 Western blot analysis Cells were seeded in 6-well plates at a density of 3×105cell, and then infected with oVV (10 MOI), sorafenib (20 μmol), oVV (10 MOI) plus sorafenib (20 μmol), respectively, with uninfected cells served as control. Two days later, cells with different treatments were washed three times with ice-cold PBS, and lysed in buffer (50 mM Tris-HCl, pH8.0, 150 mM NaCl, 100 μg/mL PMSF, 1%TritonX-100) for 30 min at ice. Cells were collected and cell debris was removed by centrifugation. The protein concentration of cell lysates was measured by bicinchoninic acid (BCA) protein assay kit (Beyotime, Shanghai). Protein samples were subjected to SDS-PAGE (12%) separation and transefered to PVDF membranes. Cleaved-caspase-3, cleaved-caspase-9 and pro-PARP antibodies were purchased from Cell Signaling Techology (Danvers, MA), β-actin were purchased from Santa Cruz Biotechnology (Santa Cruz, CA). The secondary antibodies used were anti-rabbit and anti-mouse which purchased from Vazyme-Biotech (China,Nanjing).
1.2.4 Flow cytometry analysis Hep3B cells were seeded in 6-well plates at a density of 3×105cell, after cell attached, treated with oVV (10 MOI), sorafenib (20 μM), oVV (10 MOI) plus sorafenib (20 μmol), uninfected cells served as control. After 48 h, PI staining was added into each well (500 μL/tips), and incubated at 37°C for 30 min. Cell apoptosis was examined using the FACStar (BD Biosciences, Franklin Lakes).
1.2.5 Animal experiments All procedures for animal experiments were followed according to the guide for regulations and standards of experimental animal. 4-week old female nude mice were purchased from the Shanghai Experimental Animal Center of Chinese Academy of Science (Shanghai). Hep3B tumor xenografts were established by subcutaneously inoculating 5×106Hep3B cells (suspended in 100 μL PBS) into right flank of each nude mouse. When the tumors reached 80-120 mm3, mice were randomly divided into four groups (n=8). Subsequently, mice were injected with oVV, sorafenib, combination of the virus and the drug, or PBS, respectively. oVV (1×107plaque-forming units-PFUs, per mouse) was injected in an intratumor injection manner , while sorafenib (solvent in PBS) intraperitoneally injected into mice at a dose of 30 mg/kg body weight or 100 μL PBS as control for two times of continuous injection once every other day. Tumor growth was monitored by periodic measurement using calipers, and measured every 4 days. Tumor volume (mm3)=(A×B2)/2, A and B are the tumor length and width (in mm), respectively. Then tumor volume growth curve were draw.
1.2.6 HE staining and immunohistochemistry assay Tumor tissues were fixed in 4% formaldehyde, dehydrated with gradient ethanol, and embedded in paraffin wax. The sections from tumors were stained with hematoxylin and eosin (HE) for histological analysis. For immunohistochemistry (IHC), tumor sections were treated with rabbit monoclonal anti-vaccinia virus antibodies at 1∶500 dilutions. The slides were then washed with PBS and incubated with goat anti-rabbit IgG antibodies-HRP polymers with 1∶100 dilution. The slides were washed with PBS and incubated with the avidin-biotin-peroxidase complex reagent (Vector Laboratories, Burlingame, CA) detected with diaminobenzidine tetrahydrochloride solution. All sections were counter stained with hematoxylin and examined with bright-field microscopy. 1.2.7 Statistical Analysis The experimental statistical significance were expressed as mean ± standard deviation (SD) and analyzed by GraphPad 6.0 software and student’s t test. WhenP<0.05, it is considered statistically significant.
2 RESULTS
2.1 Sorafenib and oVV synergistically inhibited tumor cell proliferation
To determine whether the combination of oVV and (Fig.1A) sorafenib (Fig.1B) inhibit tumor cell growth, we performed MTT assay on human liver cell lines infected with oVV alone, sorafenib alone, and oVV plus sorafenib, respectively. For each cell line there was a significant difference in the viability of cells treated that received single or multiple treatments. However, the significant difference was only noticed at the highest levels of sorafenib and oVV application. The largest treatment effect was found in Hep3B cells with 2% viability compared with 20% and 4% viability for Huh7 and PLC cell lines, respectively (Fig.2).

Notes: (A) Schematic of oVV construction. The PCB cassette was inserted in the TK region of vaccinia by homologous recombination. (B) Schematic structure of sorafenib.Fig.1 Characterization of sorafenib and vaccinia virus
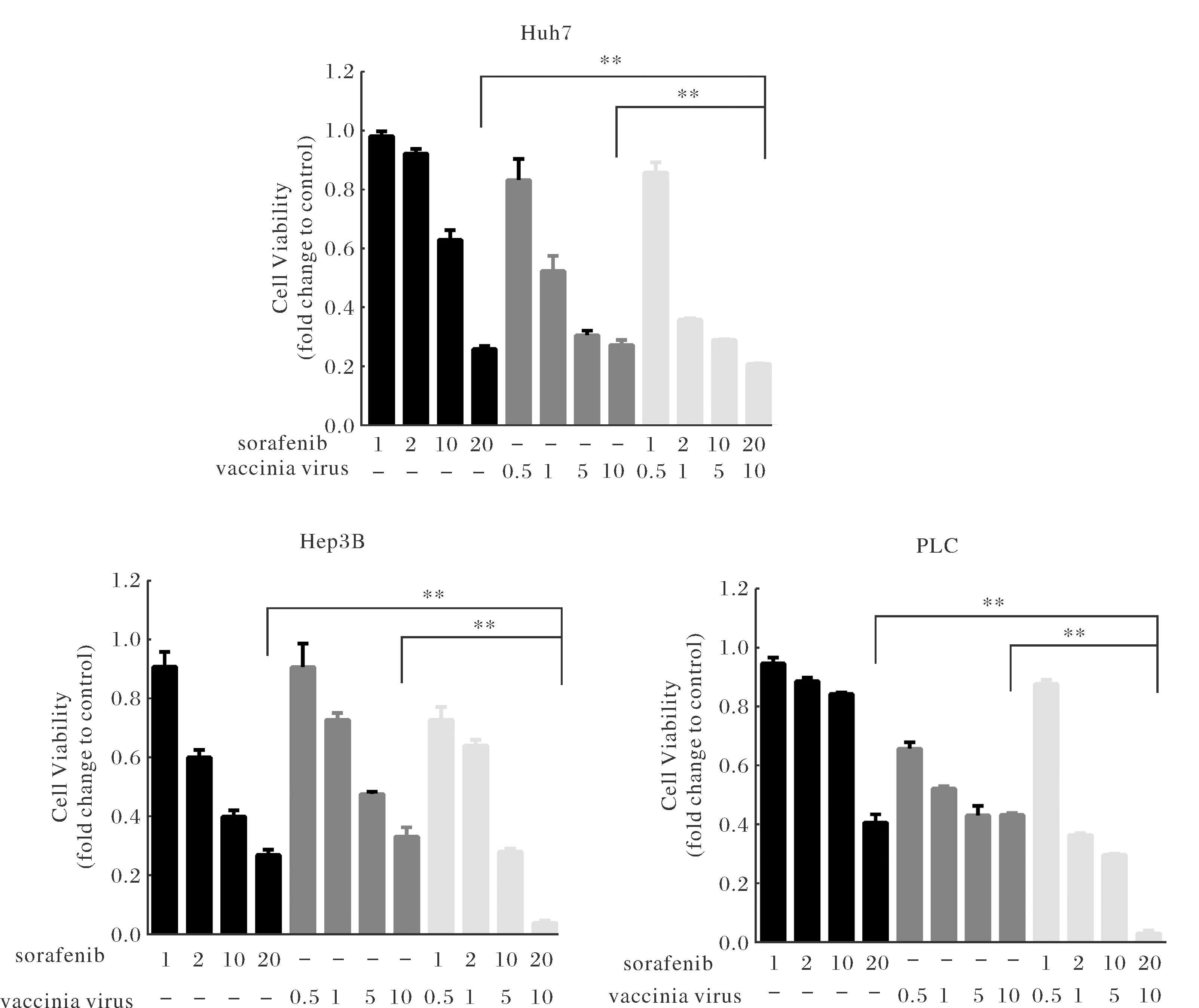
Notes: HCC cell lines Hep3B, Huh-7 and PLC were treated with oVV (0.5, 1, 5, 10 MOI), Sorafenib (1, 2, 10, 20 μmol), or combination for 48 h, respectively. The image showed three independent experiments representatively. Cell viability was evaluated by MTT assay. The image showed three independent experiments representatively.Fig.2 Sorafenib enhances oVV-mediated growth inhibition in HCC cells
2.2 Sorafenib and oVV synergistically induced apoptosis in liver cancer cells
To investigate whether combinatorial treatment with sorafenib and oVV induces tumor cell apoptosis, Hoechest 33342 staining was observed using a fluorescence microscope (Fig.3A). Compared with PBS group, other group cell morphology significantly changed. Sorafenib and oVV together group treated cells displayed more cell nuclear fragmentation and cell death.
To further examine the effect of sorafenib and oVV on cell death, PBS, sorafenib (20 μmol), oVV (10 MOI) and sorafenib+oVV treated cells underwent PI staining 48 h after treatment. Cell apoptosis rates are detected by flow cytometry, the result show that each group cell apoptosis rates is 3.08%, 4.1%, 14.7% and 38.1% (Fig.3B), The result demonstrated that the apoptosis rate of Hep3B cells combination of sorafenib and vaccinia virus was 10 times more than control group.
2.3 Sorafenib and oVV influenced cell signal pathway
To determine the mechanisms underlying sorafenib and oVV enhanced cell apoptosis, we treated Hep3B cells with PBS, sorafenib, oVV or sorafenib+oVV. Total protein was collected and expression of anti-apoptotic and pro-apoptotic proteins was examined 48 h later. Expression of pro-apoptotic proteins cleaved caspase-3 and cleaved caspase-9 were significantly upregulated in the combinatorial group. pro-PARP was also downregulated, suggesting that the pro-apoptotic protein cleaved-PARP was upregulated in sorafenib+VV exposed Hep3B cell (Fig.3C). Taken together, the data indicated that sorafenib and VV induced apoptosis by increasing expression of pro-apoptotic proteins.
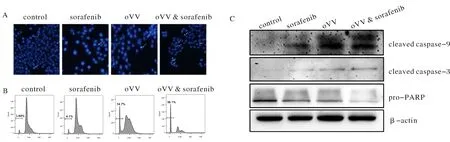
Notes: (A) Hoechst 33342 staining was used to detect apoptosis. The white arrows indicate positive apoptotic cells. Original magnification, ×200. (B) Cell apoptosis were detected by flow cytometry assay. (C) Western blot analysis of the apoptosis associated proteins. β-actin was used as a loading control.Fig.3 Sorafenib enhances oVV-induced apoptosis in HCC cells
2.4 Sorafenib and oVV significantly inhibited tumor growth in liver cancer cell-derived xenografts
To examine the effect of sorafenib and oVV on tumor growth, Hep3B cells at a density of 5×106were injected subcutaneously into the right flank of the nude mice. When the tumors grew to 80-100 mm3, PBS, sorafenib, oVV or sorafenib+oVV was injected into the mice intraperitoneally, respectively. Compared with the PBS injected mice, mice injected with sorafenib alone or oVV alone significantly inhibited tumor growth. However, the combination of sorafenib and oVV significantly inhibited tumor growth even more compared to mice treated with each therapy alone (Fig.4A).
2.5 Cytopathic effect of combination therapy on tumor growth inhibition in vivo
To determine the effect of sorafenib and oVV on tumor histology, liver cell derived-xenograft tumors of mice treated with PBS, sorafenib, oVV or sorafenib+oVV were removed 25 days after cell injection and sectioned. Hematoxylin and eosin (H&E) staining of the sections revealed that tumors from mice treated with the combinatorial therapy displayed cell death at deeper levels in the tumor and signs of necrosis compared to tumors from mice receiving one therapy or PBS. Immunohistochemical staining with anti-vaccinia virus antibodies revealed that the combination therapy and vaccinia virus group the virus relatively scattered, indicating that the virus did played an anti-cancer role in tumor tissues (Fig.4B).

Notes: (A) Tumor volumes( means±SD, n=8, **, P<0.01) were measure with caliper and estimated using the following formula: tumor volume (mm3)=(length×width2)/2. (B) Histological analysis of tumor sections. The upper rows are hematoxylin and eosin staining analysis for animal liver and tumor tissues, indicating that hepatotoxicity or cell necrotic area in tumors. The lower row showed anti-vaccinia virus expression by IHC analysis in tumor tissues. Fig.4 Synergistic effects of Sorafenib and oVV in vivo
3 Discuss
The incidence of HCC has increased worldwide in recent years. Mortality has doubled from 1990 to 2010[22]. Despite development in cancer therapies advanced liver cancer remains incurable. The discovery of sorafenib is an important breakthrough when used molecular targeted drugs treatment of liver cancer, and is the only drug that can be administered orally[23-25]. Compared with other virus, vaccinia virus replication in the cytoplasm do not integrated DNA to cellular genome, so has good security. Also vaccinia virus as medicine can through a long distance, by the blood cycle to the tumor tissue. Combination therapy is a promising anti-cancer strategy because it targets multiple factors that contribute to cancer progression[26]. This is essential given the diversity and chemoresistance of liver cancer[27]. Previous studies have shown that combining oncolytic virus with chemotherapy synergistically enhances viral burst size and apoptotic cell death[28-29].
In our study, we combined sorafenib and vaccinia virus to treat liver cancer, the results show that the combined sorafenib and oVV treatment has a stronger anti-tumor effect than either treatment alone. MTT, Hoechst 33342 and flow cytometry test found that combined sorafenib and oVV not only can inhibit the growth of tumor cells but also can promote the apoptosis of tumor cells. Western blot results show that compared with the other group, combined group cleaved-caspase-9 and cleaved-caspase-9 obviously raised, pro-PARP significantly decreased, illustrating cleaved-PARP raised obviously. When apoptosis pathway starts, the caspase-9 as a more upstream in the process of cell apoptosis signal transduction caspase, its activation can activate other caspase, for example, activated caspase-9 can activate caspase-3 which is a very key enzyme in the process of cell apoptosis, thus promote the subsequent apoptosis signaling pathways. PARP locates in the nucleus as a signal of cell apoptosis. Increase of cleaved caspase-3 is able to promote the shear of PARP, caused between Asp124 and Gly215 cut into 31 kD and 85 kD, which lost its normal function. At the established of mice transplanted tumor models, through the analysis of tumor size and pathology test, show that the therapeutic effect of combined group was obviously higher than that of other group, but the killing mechanism need further discuss. It’s reported that sorafenib induced autophagy in prostate cancer, liver cancer, gastric cancer cells[30-32]. However, deeper discussion such as autophage inducing mechanism of sorafenib remained to be done in further work.
In summary, this is the first time to report the combinational effect of vaccinia virus and sorafenib on liver cancer. This study suggests that combining sorafenib with vaccinia virus might be a promising strategy for anti-liver carcinoma therapy.
[1] JEMAL A, BRA F, CENTER M, et al. Global cancer statistics[J]. CA Cancer J Clin, 2011,61(2): 69-90.
[2] BOSCH F, RIBES J, DIAZ M, et al. Primary liver cancer: worldwide incidence and trends[J]. Gastroenterology, 2004,127(5 Suppl 1): S5-S16.
[3] MA B, WANG Y, ZHOU X, et al. Synergistic suppression effect on tumor growth of hepatocellular carinoma by combing oncolytic adenovirus carrying XAF1 with cisplatin[J]. Cancer Res Clin Oncol, 2015,141(3): 419-429.
[4] SUN S, SCHEILLER M, SPINOLA M, et al. New molecularly targeted therapies for lung cancer[J]. J Clin Invest, 2007,117(10): 2740-2750.
[5] WANG Y, HUANG F, CAI H, et al. Potent antitumor effect of TRAIL mediated by a novel adenoassociated viral vector targeting to telomerase activity for human hepatocellular carcinoma[J]. J Gene Med, 2008,10(5): 518-526.
[6] WANG S, TAN Y, LEI W, et al. Complete eradication of xenograft hepatoma by oncolytic adenovirus ZD55 harboring TRAIL-IETD-Smac gene with broad antitumor effect[J]. J Human Gene Therapy, 2012,23(9): 992-1002.
[7] HERMISTON T W, KUHN I. Armed therapeutic viruses: strategies and challenges to arming oncolytic viruses with therapeutic genes[J]. Cancer Gene Ther, 2002,9(12): 1022-1035.
[8] PAN Q, ZHONG A, LIU B, et al. Enhanced sensitivity of hepatocellular carcinoma cells to chemotherapy with a Smac-armed oncolytic adenovirus. Acta Pharmacol Sin, 2007,28(12): 1996-2004.
[9] DONG F, WANG L, DAVIS J J, et al. Eliminating established tumor in nude mice by a tumor necrosis factor-alpha-related apoptosis-inducing ligand-armed oncolytic adenovirus[J]. Clin Cancer Res, 2006,12(17): 5224-5230.
[10] SINKOVICS J, HORVATH J. New developments in the virus therapy of cancer: A historical review[J]. Intervirology. 1993,36(4): 193-214.
[11] MCANENY D, RYAN C A, BEAZLEY R M, et al. Results of a phase I trial of a recombinant vaccinia virus that expresses carcinoembryonic antigen in patients with advanced colorectal cancer[J]. Ann Surg Oncol, 1996,3(5): 3495-3500.
[12] QIN H, CHATTERJEE S. Cancer gene therapy using tumor cells infected with recombinant vaccinia virus expressing GM-CSF[J]. Hum Gene Ther, 1996,7(15): 1853-1860.
[13] MEKO J B, YIM J H, TSUNG K, et al. High cytokine production and effective antitumor activity of a recombinant vaccinia virus encoding murine interleukin-12[J]. Cancer Res, 1995,55(21): 4765-4770.
[14] ELKINS K L, ENNIST D L, WINEGAR R K, et al. In vivo delivery of interleukin-4 by a recombinant vaccinia virus prevents tumor development in mice[J]. Hum Gene Ther, 1994,5(7): 809-820.
[15] WEN L, WANG S, YANG C, et al. Combined expression of miR-34a and Smac mediated by oncolytic vaccinia virus synergistically promote anti-tumor effects in Multiple Myeloma[J/OL]. Sci Rep, 2016,6: 1-11. [2016-08-24]. https://www.nature.com/articles/srep32174. DOI: 10.1038/srep32174.
[16] BRUIX J, RAOUL J L, SHERMAN M, et al. Efficacy and safety of sorafenib in patients with advanced hepatocellular carcinoma: subanalyses of a phase III trial[J]. J Hepatol, 2012,57(4): 821-829.
[17] LIU L, CAO Y, CHEN C, et al. Sorafenib blocks the RAF/MEK/ERK pathway, inhibits tumor angiogenesis, and induces tumor cell apoptosis in hepatocellular carcinoma model PLC/PRF/5[J]. Cancer Res, 2006,66(24): 11851-11858.
[18] WILHELM S M, ADNANE L, NEWELL P, et al. Preclinical overview of sorafenib, a multikinase inhibitor that targets both Raf and VEGF and PDGF receptor tyrosine kinase signaling[J]. Mol Cancer Ther, 2008,7(10): 3129-3140.
[19] BRUIX J, SHERMAN M. Managemenr of hepatocellular carcinoma: an update[J]. Hepatology, 2011,53(3): 1020-1022.
[20] YAN J, FAN Z, WU X, et al. Circulating tumor cells are correlated with disease progression and treatment response in an orthotopic hepatocellular carcinoma model[J]. Cytometry A, 2015,87(11): 1020-1028.
[21] SHETTY K, DASH C, LAURIN. Use of adjuvant sorafenib in liver transplant recipients with high-risk hepatocellular carcinoma[J]. J Transplan, 2014,10(4): 9134-9136.
[22] LOZANO R, NAGHAVI M, FOREMAN K, et al. Global and regional mortality from 235 causes of death for 20 age groups in 1990 and 2010: a systematic analysis for the Global Burden of Disease Study 2010[J]. Lancet, 2012,380(9859): 2095-2128.
[23] KELLEY R K. Adjuvant sorafenib for liver cancer: wrong stage, wrong dose[J]. Lancet Oncol, 2015,16(13): 1279-1281.
[24] WOO H, HEO J. Sorafenib in liver cancer[J]. Expert Opin Pharmacother, 2012,13(7): 1059-1067.
[25] LANG L. FDA approves sorafenib for patients with inoperable liver cancer[J]. Gastroenterology, 2008,134(2): 379.
[26] WANG S, SHU J, CHEN L, et al. Synergistic suppression effect on tumor growth of ovarian cancer by combining cisplatin with a manganese superoxide dismutase-armed oncolytic adenovirus[J]. Onco Targets Ther, 2016,9: 6381-6388.
[27] EHRHARDT H, HACKER S, WITTMANN S, et al. Cytotoxic drug-induced, p53-mediated upregulation of caspase-8 in tumor cells[J]. Oncogene, 2008,27(6): 783-793.
[28] LIU C, ZHANG Y, LIU M M, et al. Evaluation of continuous low dose rate versus acute single high dose rate radiation combined with oncolytic viral therapy for prostate cancer[J]. Int J Radiat Biol, 2010,86(3): 220-229.
[29] EISENBERG D P, ADUSUMILLI P S, HENDERSHOTT K J, et al. 5-Fluorouracil and gemcitabine potentiate the efficacy of oncolytic herpes viral gene therapy in the treatment of pancreatic cancer[J]. J Gastrointest Sur, 2005,9(8): 1068-1079.
[30] YUAN H, LI A, MA S, et al. Inhibition of autophagy significantly enhances combination therapy with sorafenib and HDAC inhibitors for human hepatoma cells[J]. World J Gastroentero, 2014,20(17): 4953-4962.
[31] LIN J C, HUANG W, LIU C, et al. Sorafenib induces autophagy in human myeloid dendritic cells and prolongs survival of skin allografts[J]. Transplantation. 2013,95(6): 791-800.
[32] PARK M, REINEHR R, HAUSSINGER, et al. Sorafenib activates CD95 and promotes autophagy and cell death via Src family kinases in gastrointestinal tumor cells[J]. Mol Cancer Ther, 2010,9(8): 2220-2231.
索拉非尼联合溶瘤痘苗病毒对肝癌细胞抑制作用及其机制探讨
李亚姣1, 雷 文2, 秦 云1, 应 畅1, 周秀梅1
(1.浙江理工大学 生命科学学院 新元医学与生物技术研究所, 杭州 310018;2.浙江大学 医学院 第一附属医院, 杭州 310003)
人类肝细胞癌是全球最常见和具有高致命性的癌症之一.中国的肝癌患者在全球居于首位,目前仍然没有找到有效的方法来治疗肝癌.近年,溶瘤痘病毒(vaccinia virus)由于其高效的抗癌效应,副反应较低和良好的安全性已成为一种癌症治疗的新方法,且已经广泛应用于癌症的治疗中.索拉非尼,一种新型的肿瘤激酶抑制剂,作为化疗药物可安全有效地治疗肝癌,且已应用于临床试验中.本实验将索拉非尼与溶瘤痘病毒联用并探究其在肝癌细胞中的抗肿瘤的效应.通过体外MTT,Hochest,流式细胞术,Western blot等细胞实验和体内小鼠移植瘤模型的建立以及免疫组化的分析,发现索拉非尼联合溶瘤痘病毒,可显著的抑制肿瘤细胞的生长,促进肿瘤细胞的凋亡.小鼠移植瘤模型结果表明,随着时间的延长,联合处理组的肿瘤大小有下降的趋势.联合使用索拉非尼和溶瘤痘病毒可能为一项肝癌治疗的新途径.
溶瘤痘病毒; 索拉非尼; 联合治疗; 肝癌
[Q291]
A
2017-05-25.
国家自然科学基金青年科学基金项目(81602706);国家自然科学基金项目(81172137);浙江省自然科学基金项目(LY13H080005);浙江省公益性技术应用研究计划项目(2015C37035) ;浙江省中医药科学研究基金项目(2017ZB006).
1000-1190(2017)05-0638-08
*通讯联系人. E-mail: 183026077@qq.com.
10.19603/j.cnki.1000-1190.2017.05.014
