Relationship between CD14-159C/T gene polymorphism and acute brucellosis risk
2016-07-24MehdiMoghadampourEbrahimEskandariNasabFatemehShabaniDepartmentofMicrobiologySchoolofMedicineZahedanUniversityofMedicalSciencesZahedanIranInfectiousDiseasesandTropicalMedicineResearchCenterZahedanUniversityofMedicalSciencesZah
Mehdi Moghadampour, Ebrahim Eskandari-Nasab, Fatemeh ShabaniDepartment of Microbiology, School of Medicine, Zahedan University of Medical Sciences, Zahedan, IranInfectious Diseases and Tropical Medicine Research Center, Zahedan University of Medical Sciences, Zahedan, IranDepartment of Clinical Biochemistry, School of Medicine, Zahedan University of Medical Sciences, Zahedan, IranGenetic of Non-Communicable Disease Research Center, Zahedan University of Medical Sciences, Zahedan, IranSchool of Medicine, Jiroft University of Medical Sciences, Jiroft, Kerman, Iran
Contents lists available at ScienceDirect
Relationship between CD14-159C/T gene polymorphism and acute brucellosis risk
Mehdi Moghadampour1,2, Ebrahim Eskandari-Nasab3,4*, Fatemeh Shabani5
1Department of Microbiology, School of Medicine, Zahedan University of Medical Sciences, Zahedan, Iran
2Infectious Diseases and Tropical Medicine Research Center, Zahedan University of Medical Sciences, Zahedan, Iran
3Department of Clinical Biochemistry, School of Medicine, Zahedan University of Medical Sciences, Zahedan, Iran
4Genetic of Non-Communicable Disease Research Center, Zahedan University of Medical Sciences, Zahedan, Iran
5School of Medicine, Jiroft University of Medical Sciences, Jiroft, Kerman, Iran
ABSTRACT
Objective: To investigate the association between the cluster of diff erentiation 14 (CD14)-159C/T (rs2569190) gene polymorphism and susceptibility to acute brucellosis in an Iranian population. Methods: The study included 153 Iranian patients with active brucellosis and 128 healthy individuals as the control group. Genotyping of the CD14 variant was performed using an amplifi cation refractory mutation system-polymerase chain reaction method. Results: The prevalence of CD14-159 TT and CT genotypes were associated with increased risk of brucellosis [odds ratio (OR)=1.993, 95% confi dence interval (95% CI)=1.07-3.71, P=0.03 for CT; OR=3.869, 95% CI=1.91-7.84, P=0.01 for TT genotype. Additionally, the minor allele (T) was signifi cantly more frequently present in brucellosis patients than in controls (61% vs. 45%, respectively), and was a risk factor for brucellosis (OR=3.058, 95% CI=1.507-6.315, P=0.01). Conclusions: The fi ndings provid suggestive evidence of association of the CD14-159C/T gene polymorphism with susceptibility to acute brucellosis in the Iranian population.
ARTICLE INFO
Article history:
Received in revised form 20 January 2016
Accepted 15 February 2016
Available online 20 March 2016
Acute brucellosis
Cluster of diff erentiation 14
Single nucleotide polymorphism
1. Introduction
Brucellosis (or Malta fever, Gibraltar fever, Cyprus fever, Danube fever, undulant fever, Mediterranean fever and Bang’s disease) is one of the most frequent zoonotic infections worldwide, transmitted to humans by eating contaminated, unpasteurized dairy products, direct contact with infected animal, and through the inhalation of infected aerosolized particles[1-4]. The majority of cases arise in the Mediterranean countries of Europe and Africa, Mexico, and Central and South America, India, Central Asia and Middle East including Iran[4].
Brucella spp. (pleomorphic, gram-negative, non-spore-forming coccobacillus bacteria) causes severe debilitating and disabling disease that is accompanied with manifestations such as fever, weakness, sweating, weight loss, headache, persisted joint pain, neurological complications, endocarditis and testicular or bone abscess formation[1,2,4,5]. Brucella spp. are facultative intracellular bacteria that have the ability to evade from killing mechanism of immune system and multiply within in organs with frequent macrophages such as liver, spleen, lung, bone marrow and synovium (reticuloendothelial system)[4,6]. Among all species of Brucella genus, three species including Brucella melitensis, Brucella abortus, and Brucella suis are the most commonly associated with human disease[7].
Cluster of differentiation 14 (CD14) is a glycosylphosphatidylinositol-anchored molecule that is mainly expressed on cell membrane surfaces of monocyte/macrophage lineage[8,9], and acts as a pattern recognition receptor in innate immunity fordetection of variety of ligands such as bacterial products, microbial cell wall components and even whole bacteria. Particularly, CD14 is the specific receptor for the lipopolysaccharide (LPS) of gramnegative bacteria[10,11]. LPS is the major component of the outer membrane of gram-negative bacteria and is called ‘bacterial endotoxin’[12]. LPS is the main virulence factor of Brucella and has a role in cell entry and immune evasion of the infected cell[6]. LPS also has an ability to induce the production of interleukin (IL)-12 resulting in promotion of T helper (Th) 1 responses and inhibition of Th-2 responses in the host[13]. CD14 as a co-receptor for the tolllike receptor (TLR) and together with TLR4 forms a complex that activates the innate immune system and starts a variety of eff ector functions including secretion of pro-infl ammatory cytokines such as IL-1, IL-6, tumor necrosis factor-α, nitric oxide, oxygen radicals and complement components as well as cell proliferation, subsequent binding to LPS[8,13-15]. Nevertheless, high production of these molecules may cause intense harmful consequences including severe inflammatory responses, septic shock and death[15]. Moreover, a soluble type of CD14 (sCD14) is produced by monocytes, hepatocytes and endothelial cells and is present in the circulation and other body fl uids. sCD14 is derived both from secretion of CD14 and from enzymatically cleaved glycosyl-phosphatidylinositol-anchored membrane type of CD14[10,16]. Since sCD14 plasma levels increases during infl ammation and infection, it is believed that sCD14 is an acute-phase protein, which is increased in several inflammatory processes[8,15,17].
The human CD14 gene is located on chromosome 5q31.1[14]. Several single nucleotide polymorphisms (SNPs), including -2451G/ C (rs2569192), -1855A/G (rs3138076), -1619A/G (rs2915863) and -159C/T (rs2569190), have been identifi ed in promoter region of the CD14 gene[18,19]. One of the most extensively studied SNPs, the -159 C/T (rs2569190), includes a substitution of C→T resulting in elevated transcriptional activity and expression levels of CD14[14,20].
Thus far, the CD14-159C/T gene polymorphism has been examined in several inflammatory and infectious diseases including tuberculosis[8,9,21], brucellosis[22], Chlamydia pneumoniae infection[23,24], Helicobacter pylori infection[25], hepatitis B and C[26,27] and severe acute respiratory syndrome[28], but the results have been inconsistent and confl icting. Therefore, the current study aimed to evaluate the possible association between the CD14-159C/ T (rs2569190) gene polymorphism and susceptibility to acute brucellosis in an Iranian population.
2. Materials and methods
2.1. Study population
The current case-control retrospective study included 153 patients (102 men and 51 women) suffering from active brucellosis, age range 6-76 years old and mean±SD=31.24±16.6 and 128 healthy individuals as the control group (93 men and 35 women), age range 19-64 years and mean±SD=34.04±13.69. Blood sample from each participant was collected in EDTA-containing tubes for DNA isolation after taking an informed consent from all patients and healthy individuals. All patients were either milk farmers (including diagnosed infected animals) or had a history of consuming raw milk and unpasteurized dairy products. Demographic characteristics of patients and their clinical complications are shown in Table 1. Brucellosis was diagnosed according to the clinical manifestations (including fever, night sweating, weakness, malaise, weight loss, splenomegaly, lymphadenophathy, myalgia and arthralgia) and positive blood cultures as described previously[29-32]. The control group included healthy blood donors with no evidence of brucellosis and genetic disorders and matched for age, sex, and geographic area. The control subjects had the same background as patients and were at the same risk of exposure for brucellosis.
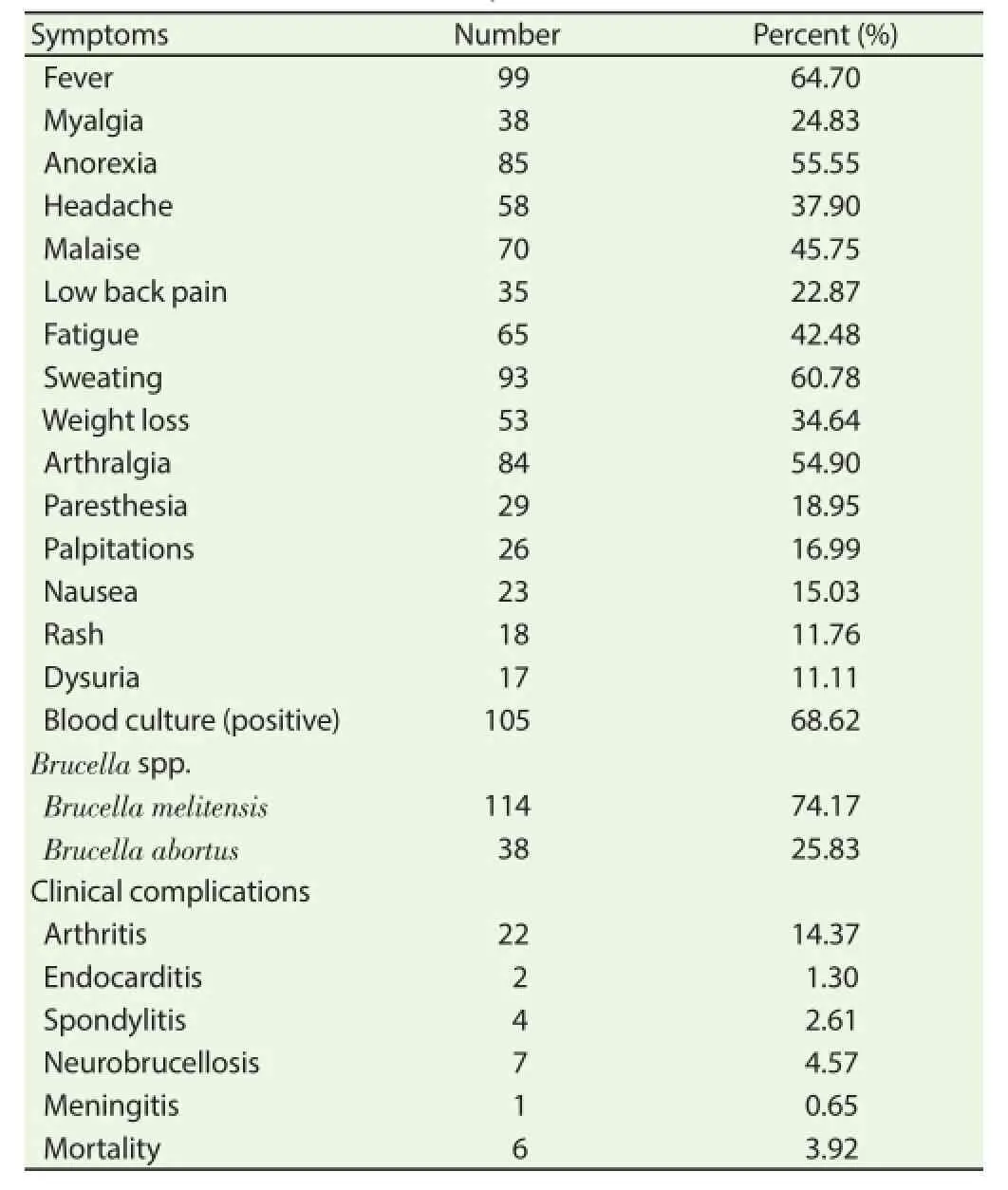
Table 1Clinical characteristics of brucellosis patients.
2.2. Culture and identification of organism
Brucella spp. were cultivated on 5% sheep blood-agar plates and incubated at 37 ℃ in the contact to 5%-10% of CO2conditions for 48 h. A typical and well-isolated Brucella like colony is tiny, transparent, raised, and convex, with an entire edge and smooth and glistening surface along the streak lines by examining macroscopically by Gram’s stain[33]. Serological tests, defined as wright titre≥1/160 plus mercaptoethanol test≥1/80 or coomb’swright≥1/320 certified the brucellosis infection. The complete details of the organism identifi cation methods have been described previously in papers by this research team[29-32].

Table 2Genotype and allele frequencies of CD14-159C/T SNP between brucellosis patients and controls.
2.3. Genotyping of CD14-159C/T (rs2569190) SNP
Genomic DNA was extracted from the peripheral blood leukocytes by the ‘salting-out’ method as explained previously[34]. The quality of the isolated DNA was checked by electrophoresis on 1% agarose gel, quantitated spectrophotometrically and stored at -20 ℃ still further use.
The CD14-159C/T SNP, rs2569190, was genotyped using an amplifi cation refractory mutation system-polymerase chain reaction method as described previously by Karhukorpi et al[25,35]. This allele-specifi c, single-tube polymerase chain reaction method uses four primers including two allele specifi c primers (cfors for the C allele and trevs for the T allele) and two outer primers (cdp1 and cdp2) for the control band. An additional mismatch was inserted at the penultimate 3’ nucleotide (underlined) of the allele-specifi c primers to increase the specifi city of the PCR reaction. The primer sequences were as follows: cfors: 5’-CTC CAG AAT CCT TCC TGT TAC GAC-3’, trevs: 5’-TGT AGG ATG TTT CAG GGA GGG GTA-3’, cdp1: 5’-TTG GTG CCA ACA GAT GAG GTT CAC-3’, cdp2: 5’-TTC TTT CCT ACA CAG CGG CAC CC-3’. The PCR conditions for the CD14-159C/T (rs2569190) polymorphism were as followed: an initial denaturation at 95 ℃ for 5 min; 30 cycles at 95 ℃ for 30 s, 60 ℃ for 30 s and 72 ℃ for 1 min; the fi nal extension step proceeded at 72 ℃ for 5 min[25,35]. The PCR products were separated by standard electrophoresis on 2% agarose gel containing ethidium bromide. The PCR products included a 381 bp band for the T allele, a 227 bp band for the C allele and a 561 bp band for the control.
2.4. Statistical analysis
All statistical analyses were performed applying the SPSS software for Windows, version 18.0 (SPSS Inc, Chicago IL, USA). The association among genotypes and brucellosis was calculated by reckoning the odds ratio (OR) and 95% confi dence interval (95% CI) from logistic regression analyses. P values below 0.05 were considered statistically signifi cant. The Hardy-Weinberg equilibrium was tested with the χ2test for the SNP under consideration.
3. Results
The CD14 polymorphism, -159C/T, was successfully genotyped in 153 brucellosis patients and 128 control subjects. No deviation from Hardy-Weinberg equilibrium was observed in the studied case and control groups (P>0.05). The genotype and allele frequency of the SNP in the studied groups were shown in Table 2.
The results showed that the CD14 polymorphism, -159C/T, was associated with the risk of brucellosis in our population. At genotypic level, the CD14 CT, and TT genotypes were more frequent in the patients group compared to cases, and were associated with increased risk of brucellosis in the codominant model (OR=1.993, 95% CI=1.07-3.71, P=0.03 for CT; OR=3.869, 95% CI=1.91-7.84, P=0.01 for TT genotype). Additionally, the CT+TT vs. CC genotype in the dominant model was a risk factor for brucellosis (OR=2.514, 95% CI=1.34-4.73, P=0.02). Likewise, in the recessive model, the TT genotype vs. CC+CT was associated with the risk of brucellosis (OR=2.379, 95% CI=1.33-4.27, P=0.02). At allelic level, the minor allele (T) was signifi cantly more frequently present in brucellosis patients than in controls (61% vs. 45%, respectively), and was a risk factor for brucellosis (OR=3.058, 95% CI=1.507-6.315, P=0.01).
4. Discussion
Our study provided data on the association between one important SNP in the 5’ fl anking region of the CD14 gene and acute brucellosis using genotype analysis. The results showed that the prevalence of -159 homozygote TT genotype as well as T allele were much higher in brucellosis patients than in controls (36.6% vs. 19.5% and 61%vs. 44%, respectively). The carriers of the TT genotype and T allele were at 3.8-fold or 3-fold higher risk of brucellosis, respectively, than those with the CC genotype or C allele.
Several studies have been conducted to show the impact of the CD14-159C/T SNP on the risk of infectious diseases[9,18,21-23]. To the best of our knowledge, there is only one study assessing the association of CD14, -159C/T polymorphism and brucellosis[22]. Haidari et al[22], in their study on brucellosis in a Western Iranian population have shown that the frequency of the TT genotype was higher in the patients than in controls (34.5% vs. 15.5%). They suggested that the homozygousity for T allele of promoter of CD14 gene may confer an increased risk of brucellosis, which supports our fi ndings. Moreover, our results regarding CD14-159C/ T polymorphism are in agreement with several infectious studies on pulmonary tuberculosis[9,18,21] and Chlamydia pneumoniae infection[23]. In contrast, other studies reported no association between CD14-159C/T SNP and Chlamydia trachomatis infection[36], Campylobacter jejuni infection[37] and severe acute respiratory syndrome[28].
Most Brucella species are highly virulent causing an acute infection in their hosts. The LPS of this bacterium is one of the essential components for the fi rst steps in the establishment of an intracellular replication niche, in which Brucella survives and multiplies[7]. LPS is a potent stimulator of monocytes and macrophages[25]. Activation of macrophages is one of the early events in the innate immunity to intracellular pathogens. CD14 is a high-affi nity receptor for bacterial LPS and is expressed mainly by cells of monocyte/macrophage lineage. The binding of LPS to CD14 results in cellular activation and initiates a range of effector functions including cytokine secretion and cell proliferation[8,19]. CD14 as a co-receptor for the TLR is transferring LPS to the TLR4/MD-2 signaling complex. Engagement of this complex results in the activation of innate immunity mechanisms such as the release of proinflammatory cytokines and subsequently severe infl ammatory responses as well as in worse circumstances of septic shock and death[15,19]. Some studies have been performed to explain the impact of the CD14-159C/T SNP on CD14 protein expression[8,38]. The CD14-159C/T polymorphism is located at a GC box close to the transcription start site, which serves as a binding site for Sp1-Sp2/Sp3 transcription factors that are involved in the regulation of CD14 gene transcription[15]. The T variant of this SNP is associated with high expression of CD14 on the surface of monocytes and macrophages, which further increases the infl ammatory response[39,40]. Thus, this SNP could be a genetic factor for inter-individual diff erences in susceptibility to infectious diseases like brucellosis. Our results highlight the role of CD14 in the infl ammatory response and the eff ect of its promoter polymorphism on the host immune defense.
In conclusion, our study provided evidence that the CD14-159, CT and TT genotypes may serve as risk factors for brucellosis infection in our population. Further studies on larger populations with different ethnicities are required to validate our findings. More inclusive understanding of the basic mechanisms underlying infections pathogenesis may lead to the development and application of effi cient products for the treatment of brucellosis.
Conflict of interest statement
The authors declare that there is no confl ict of interest.
Acknowledgements
The authors appreciate all individuals who willingly participated in the current study.
References
[1] Dean AS, Crump L, Greter H, Hattendorf J, Schelling E, Zinsstag J. Clinical manifestations of human brucellosis: a systematic review and meta-analysis. PLoS Negl Trop Dis 2012; 6(12): e1929.
[2] Dean AS, Crump L, Greter H, Schelling E, Zinsstag J. Global burden of human brucellosis: a systematic review of disease frequency. PLoS Negl Trop Dis 2012; 6(10): e1865.
[3] Galinska EM, Zagorski J. Brucellosis in humans-etiology, diagnostics, clinical forms. Ann Agric Environ Med 2013; 20(2): 233-238.
[4] Akhvlediani T, Clark DV, Chubabria G, Zenaishvili O, Hepburn MJ. The changing pattern of human brucellosis: clinical manifestations, epidemiology, and treatment outcomes over three decades in Georgia. BMC Infect Dis 2010; 10: 346.
[5] Moreno E. Retrospective and prospective perspectives on zoonotic brucellosis. Front Microbiol 2014; 5: 213.
[6] Christopher S, Umapathy BL, Ravikumar KL. Brucellosis: review on the recent trends in pathogenicity and laboratory diagnosis. J Lab Physicians 2010; 2(2): 55-60.
[7] Yumuk Z, O’ Callaghan D. Brucellosis in Turkey-an overview. Int J Infect Dis 2012; 16(4): e228-235.
[8] Ayaslioglu E, Kalpaklioglu F, Kavut AB, Erturk A, Capan N, Birben E. The role of CD14 gene promoter polymorphism in tuberculosis susceptibility. J Microbiol Immunol Infect 2013; 46(3): 158-163.
[9] Kang YA, Lee HW, Kim YW, Han SK, Shim YS, Yim JJ. Association between the -159C/T CD14 gene polymorphism and tuberculosis in a Korean population. FEMS Immunol Med Microbiol 2009; 57(3): 229-235.
[10] Martinez FD. CD14, endotoxin, and asthma risk: actions and interactions. Proc Am Thorac Soc 2007; 4(3): 221-225.
[11] Jersmann HP. Time to abandon dogma: CD14 is expressed by nonmyeloid lineage cells. Immunol Cell Biol 2005; 83(5): 462-427.
[12] Zanoni I, Granucci F. Role of CD14 in host protection against infections and in metabolism regulation. Front Cell Infect Microbiol 2013; 3: 32.
[13] Lau MY, Dharmage SC, Burgess JA, Lowe AJ, Lodge CJ, Campbell B, et al. CD14 polymorphisms, microbial exposure and allergic diseases:a systematic review of gene-environment interactions. Allergy 2014; 69(11): 1440-1453.
[14] Zhang AQ, Yue CL, Gu W, Du J, Wang HY, Jiang J. Association between CD14 promoter -159C/T polymorphism and the risk of sepsis and mortality: a systematic review and meta-analysis. PLoS One 2013; 8(8): e71237.
[15] Pacheco E, Fonseca C, Montes C, Zabaleta J, Garcia LF, Arias MA. CD14 gene promoter polymorphism in different clinical forms of tuberculosis. FEMS Immunol Med Microbiol 2004; 40(3): 207-213.
[16] Haziot A, Chen S, Ferrero E, Low MG, Silber R, Goyert SM. The monocyte differentiation antigen, CD14, is anchored to the cell membrane by a phosphatidylinositol linkage. J Immunol 1988; 141(2): 547-552.
[17] Zhao J, Lin G, Zhang WH, Ge M, Zhang Y. Contribution of CD14-159C/ T polymorphism to tuberculosis susceptibility: a meta-analysis. Int J Tuberc Lung Dis 2013; 17(11): 1472-1478.
[18] Xue Y, Zhao ZQ, Chen F, Zhang L, Li GD, Ma KW, et al. Polymorphisms in the promoter of the CD14 gene and their associations with susceptibility to pulmonary tuberculosis. Tissue Antigens 2012; 80(5): 437-443.
[19] LeVan TD, Von Essen S, Romberger DJ, Lambert GP, Martinez FD, Vasquez MM, et al. Polymorphisms in the CD14 gene associated with pulmonary function in farmers. Am J Respir Crit Care Med 2005; 171(7): 773-779.
[20] Areeshi MY, Mandal RK, Panda AK, Bisht SC, Haque S. CD14-159 C>T gene polymorphism with increased risk of tuberculosis: evidence from a meta-analysis. PLoS One 2013; 8(5): e64747.
[21] Rosas-Taraco AG, Revol A, Salinas-Carmona MC, Rendon A, Caballero-Olin G, Arce-Mendoza AY. CD14 C(-159)T polymorphism is a risk factor for development of pulmonary tuberculosis. J Infect Dis 2007; 196(11): 1698-1706.
[22] Haidari M, Hajilooi M, Rezazadeh M, Rafiei A, Alavi SA, Keramat F. Polymorphism in the promoter region of the CD14 gene and susceptibility to Brucellosis. Immunol Invest 2006; 35(2): 239-245.
[23] Eng HL, Chen CH, Kuo CC, Wu JS, Wang CH, Lin TM. Association of CD14 promoter gene polymorphism and Chlamydia pneumoniae infection. J Infect Dis 2003; 188(1): 90-97.
[24] Rupp J, Goepel W, Kramme E, Jahn J, Solbach W, Maass M. CD14 promoter polymorphism -159C>T is associated with susceptibility to chronic Chlamydia pneumoniae infection in peripheral blood monocytes. Genes Immun 2004; 5(5): 435-438.
[25] Karhukorpi J, Yan Y, Niemela S, Valtonen J, Koistinen P, Joensuu T, et al. Eff ect of CD14 promoter polymorphism and H. pylori infection and its clinical outcomes on circulating CD14. Clin Exp Immunol 2002; 128(2): 326-332.
[26] Meiler C, Muhlbauer M, Johann M, Hartmann A, Schnabl B, Wodarz N, et al. Diff erent eff ects of a CD14 gene polymorphism on disease outcome in patients with alcoholic liver disease and chronic hepatitis C infection. World J Gastroenterol 2005; 11(38): 6031-6037.
[27] Mohammad Alizadeh AH, Ranjbar M, Hajilooi M, Fallahian F. Association of promoter polymorphism of the CD14 C (-159) T endotoxin receptor gene with chronic hepatitis B. World J Gastroenterol 2006; 12(35): 5717-5720.
[28] Yuan FF, Boehm I, Chan PK, Marks K, Tang JW, Hui DS, et al. High prevalence of the CD14-159CC genotype in patients infected with severe acute respiratory syndrome-associated coronavirus. Clin Vaccine Immunol 2007; 14(12): 1644-1645.
[29] Eskandari-Nasab E, Moghadampour M, Najibi H, Hadadi-Fishani M. Investigation of CTLA-4 and CD86 gene polymorphisms in Iranian patients with brucellosis infection. Microbiol Immunol 2014; 58(2): 135-141.
[30] Eskandari-Nasab E, Moghadampour M, Hasani SS, Hadadi-fishani M, Mirghanizadeh-Bafghi SA, Asadi-Saghandi A, et al. Relationship between gamma-interferon gene polymorphisms and susceptibility to brucellosis infection. Microbiol Immunol 2013; 57(11): 785-791.
[31] Eskandari-Nasab E, Moghadampour M, Asadi-Saghandi A, Kharazi-Nejad E, Rezaeifar A, Pourmasoumi H. Levels of interleukin-(IL)-12p40 are markedly increased in Brucellosis among patients with specifi c IL-12B genotypes. Scand J Immunol 2013; 78(1): 85-91.
[32] Sepanjnia A, Eskandari-Nasab E, Moghadampour M, Tahmasebi A, Dahmardeh F. TGFbeta1 genetic variants are associated with an increased risk of acute brucellosis. Infect Dis (Lond) 2015; 47(7): 458-464.
[33] El-Koumi MA, Afify M, Al-Zahrani SH. A prospective study of brucellosis in children: relative frequency of pancytopenia. Mediterr J Hematol Infect Dis 2013; 5(1): e2013011.
[34] Eskandari-Nasab E, Hashemi M, Rezaei H, Fazaeli A, Mashhadi MA, Moghaddam SS, et al. Evaluation of UDP-glucuronosyltransferase 2B17 (UGT2B17) and dihydrofolate reductase (DHFR) genes deletion and the expression level of NGX6 mRNA in breast cancer. Mol Biol Rep 2012; 39(12): 10531-10539.
[35] Karhukorpi J, Ikaheimo I, Karvonen J, Karttunen R. Promoter region polymorphism of the CD14 gene (C-159T) is not associated with psoriasis vulgaris. Eur J Immunogenet 2002; 29(1): 57-60.
[36] Ouburg S, Spaargaren J, den Hartog JE, Land JA, Fennema JS, Pleijster J, et al. The CD14 functional gene polymorphism -260 C>T is not involved in either the susceptibility to Chlamydia trachomatis infection or the development of tubal pathology. BMC Infect Dis 2005; 5: 114.
[37] Geleijns K, Jacobs BC, Van Rijs W, Tio-Gillen AP, Laman JD, van Doorn PA. Functional polymorphisms in LPS receptors CD14 and TLR4 are not associated with disease susceptibility or Campylobacter jejuni infection in Guillain-Barre patients. J Neuroimmunol 2004; 150(1-2): 132-138.
[38] Gibot S, Cariou A, Drouet L, Rossignol M, Ripoll L. Association between a genomic polymorphism within the CD14 locus and septic shock susceptibility and mortality rate. Crit Care Med 2002; 30(5): 969-973.
[39] Chao YC, Chu HC, Chang WK, Huang HH, Hsieh TY. CD14 promoter polymorphism in Chinese alcoholic patients with cirrhosis of liver and acute pancreatitis. World J Gastroenterol 2005; 11(38): 6043-6048.
[40] Eng HL, Wang CH, Chen CH, Chou MH, Cheng CT, Lin TM. A CD14 promoter polymorphism is associated with CD14 expression and Chlamydia-stimulated TNF alpha production. Genes Immun 2004; 5(5): 426-430.
Document heading 10.1016/j.apjtm.2016.01.036
IF: 1.062
Asian Pacific Journal of Tropical Medicine
journal homepage:www.elsevier.com/locate/apjtm
15 December 2015
*Corresponding author: Ebrahim Eskandari-Nasab, Department of Clinical Biochemistry, School of Medicine, Zahedan University of Medical Sciences, Zahedan, Iran.
Tel: +98-9136326859
Fax: +98-5413229792
E-mail: eenasab@yahoo.com; eenasab@sadi.ut.ac.ir
杂志排行
Asian Pacific Journal of Tropical Medicine的其它文章
- Preinduced intestinal HSP70 improves visceral hypersensitivity and abnormal intestinal motility in PI-IBS mouse model
- Mechanism of all-transretinoic acid increasing retinoblastoma sensitivity to vincristine
- Protective effect of apoptosis signal-regulating kinase1 inhibitor against mice liver injury
- Effect of TRPV1 combined with lidocaine on cell state and apoptosis of U87-MG glioma cell lines
- Effect of miR-467b on atherosclerosis of rats
- Effect of dimethyl fumarate on rats with chronic pancreatitis
