Delivery of genetic load during ex situ liver machine perfusion with potential for CRISPR-Cas9 gene editing: An innovative strategy for graft treatment
2021-11-08ElinoBonorsiRiniAnrewGilloolyIselBrggenwirthPuloMrtins
Elino Bonorsi-Rini , , ,Anrew Gillooly ,Isel M.A. Brüggenwirth , ,Pulo N. Mrtins , ∗
a Abdominal Transplant Unit, Cliniques Universitaires Saint Luc, UniversitéCatholique de Louvain, Brussels, Belgium
b Pôle de Chirurgie Expérimentale et Transplantation, UniversitéCatholique de Louvain, Brussels, Belgium
c Department of Surgery, Transplant Division, University of Massachusetts, Worcester-Massachusetts, USA
d Department o f Surgery, Section of hepatobiliary surgery and liver transplantation, University Medical Center Groningen, University of Groningen, Groningen, The Netherlands
TotheEditor:
Over the years, the shortage of suitable donor organs has chal-lenged the transplant community in performing life-saving liver transplantation (LT). Recent reports from European and American liver transplant registries show persistently high waitlist mortal-ity rates ranging between 10% and 18% [ 1,2 ]. To cope with this, liver transplant surgeons are increasingly forced to transplant or-gans from extended criteria donors. However, it is well known that these organs are more susceptible to the consequences of ischemia-reperfusion injury (IRI), including primary non-function (PNF) and non-anastomotic biliary strictures (NAS) after transplan-tation, which, by consequence, can increase the number of retrans-plantations, making organ shortage an endless cycle [3] . This unfa-vorable scenario has created a fertile environment for the develop-ment of organ machine perfusion (MP) strategies aiming to assess and optimize organs before transplantation [4] . A large amount of research has resulted in the development of different perfu-sion devices and protocols, which have been tested in preclini-cal and clinical studies, and, more recently, in randomized clinical trials [5] . Currently, hypothermic oxygenated machine perfusion (HOPE) is mainly used to improve mitochondrial status by decreas-ing oxidative stress and increasing cellular adenosine triphosphate (ATP) levels [ 6,7 ], whereas normothermic MP is better suited for evaluation of graft quality during the perfusion session by measur-ing different biological and physiological parameters.
Furthermore, MP technology opens a door to a new era in liver graft preservation by allowing modulation of graft function through the administration of specific therapies, such as anti-inflammatory drugs, vasodilators, defatting cocktails, and infusion of stem cells [8] . In addition, our group pioneered the use of RNA interference by demonstrating the uptake of small interfering RNA against pro-apoptotic genes during bothexsitunormothermic and hypothermic perfusion of rat liver grafts [9] . Our group started in-vestigating the potential use of CRISPR-Cas9 (clustered regularly-interspaced short palindromic repeats and CRISPR associated pro-tein 9) gene editing during liver MP as a promising strategy to fur-ther explore. The CRISPR-Cas9 is a natural immune bacterial mech-anism against viruses, which was discovered in 2012 and it is al-ready considered a revolution in the way we can manipulate DNA by making gene editing more efficient, faster, and cheaper. Indeed, Charpentier and Doudna, who first described the system, have just been awarded with the 2020 Nobel Prize in chemistry [10] .
All in all,exsitugraft modulation/optimization is a high-speed research highway, which is opening up in the field of organ preser-vation. However, as with all roads, the journey can be long and, at some point, misleading. Hence, we have many steps left to take before reaching a safe destination. In fact, in the specific case of CRISPR-Cas9 gene editing, its efficiency will largely depend on the viral delivery performance of both Cas9 nuclease and guide RNA, which may limit the results of this highly sophisticated technol-ogy. The use of CRISPR-Cas9 gene editing for graft modulation has not been reported yet.
With this in mind, we designed a proof-of-concept experiment in which adeno-associated virus (AAV) gene therapy was used to efficiently deliver genetic load during MP before liver engraftment in preparation to be used for CRISPR-Cas9 delivery. In brief, male Lewis rats, weighting 220 to 260 g, were used as donors and recip-ients. Liver grafts were procured and transplanted using an arteri-alized rat transplant model [11] . Immediately after procurement, 6 liver grafts were subjected to 2 h of HOPE at 4 °C using Uni-versity of Wisconsin MP solution, which was perfused through the portal vein as previously described [9] . At the beginning of HOPE preservation, a solution of 4 ×108Pfu/mL of AAV (serotype 8), used as a vector for the green fluorescent protein (GFP) gene, was added to the perfusion solution in 2 of the 6 grafts. After MP preservation, controls and treated livers were immediately trans-planted. All animals survived and were in good clinical conditions at 24 h after liver transplantation, when euthanasia was performed for samples collection ( Fig. 1 ). Immunohistochemistry analysis per-formed on samples of treated grafts showed 2.0 0 0 ±1.563 GFP positive cells per ×20 field ( Fig. 2 ), demonstrating transduction of liver graft cells 24 h after transplant. With this proof-of-principle study, we show for the first time adenoviral-mediated delivery of genetic cargo duringexsituHOPE liver perfusion and transplan-tation. Successful integration of viral gene therapy duringexsitumachine preservation provides an opportunity to potentially intro-duce a therapeutic cargo to ameliorate or reduce IRI. As such, viral gene therapy could be employed to deliver and induce temporary overexpression of cytoprotective, anti-apoptotic, or immunoregula-tory genes [12] .
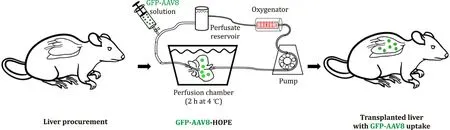
Fig. 1. Experimental design. GFP-AAV8: green fluorescent protein adeno-associated virus serotype 8. HOPE: hypothermic oxygenated machine perfusion.
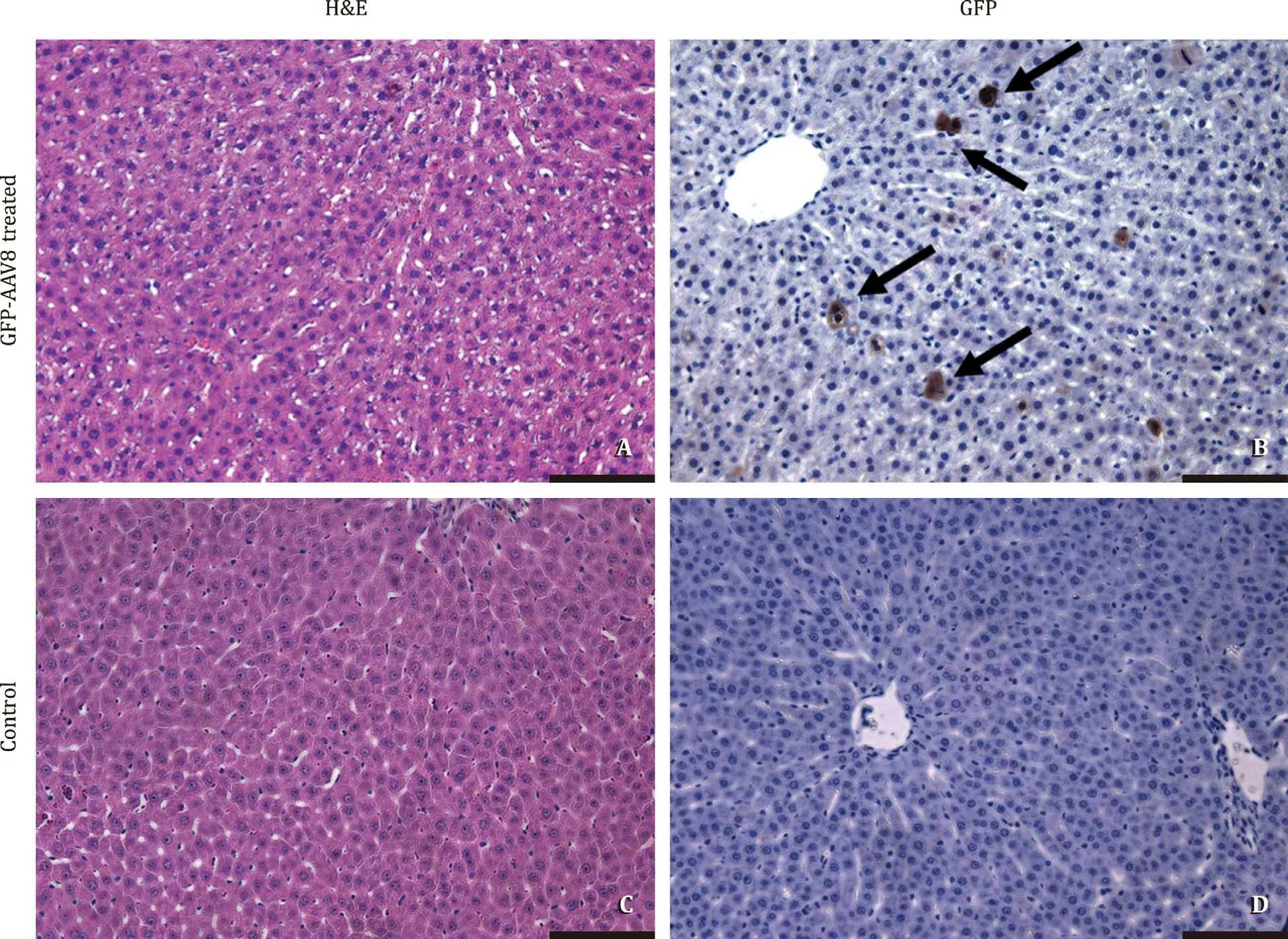
Fig. 2. Delivery of GFP-AAV8 transduces liver graft hepatocytes. Representative histology and immunohistochemistry scale bars 100 μm . Black arrows show GFP positive cells. GFP-AAV8: green fluorescent protein adeno-associated virus serotype 8.
Similarly, viral gene therapy could be employed to deliver gene-editing technologies, (i.e., CRISPR-Cas9) to knockout and reduce ex-pression of pro-ischemic, pro-apoptotic, and/or pro-inflammatory players. Indeed, CRISPR-Cas9exsitugraft editing will depend heav-ily on the efficiency of viral delivery of both Cas9 nuclease and guide RNA, guide RNA design, and frequency of potential off-target effects, including unexpected translocations, deletions, inversions, and exon skipping [13–15] . We are currently trying to apply the CRISPR-Cas9 gene-editing platform in ourexsiturat liver perfusion model.
AAV vectors have been demonstrated to have robust safety pro-files clinically. Further, AAV-delivered genetic material does not integrate into the genome. AAV vectors come in a variety of serotypes with different therapeutic profiles. Here we used AAV serotype 8, which demonstrated to be a promising candidate for hepatic gene therapy clinically [13] . However, forexsituperfusion and transplantation specifically, future studies will be needed to compare the efficiency of various AAV serotypes and to improve the robustness of transduction.
There are several important caveats to viral gene therapy that must be considered [15] . The first is packaging size of the vec-tor. For most AAV vectors, the upper limit is around 5 kilobases (kb) [13] . As such, when considering future studies to use CRISPR-Cas9 to knockout genes that promote IRI, Cas9 and the guide RNA would likely need to be packaged into and delivered by sepa-rate AAV vectors. Additionally, targeted transduction of liver cell subtypes, i.e., cholangiocytes, will likely require studies evaluat-ing multiple serotypes. Finally, while generally well tolerated, not unlike other viral vectors, AAV delivery can induce an immune response [ 14,15 ]. However, in adult mice, adenoviral delivery of CRISPR-Cas9 still generated efficient editing, despite presence of an immune response [13] . Nevertheless, the potential for AAV delivery of CRISPR-Cas9 or other genetic cargo strategy during HOPE can elicit a detrimental immune response requiring further investiga-tion. In the same way, the number of virally transduced hepato-cytes to produce a therapeutic benefit (whether overexpressing a protein of interest or editing for knock-out) and confirmation of off-target effects require more research.
In conclusion, here we show in a preliminary proof-of-concept study that AAV administration during organ machine perfusion was able to deliver genetic load to liver grafts, leading to transduc-tion in liver grafts 24 h after transplantation. Further experimental studies using AAV as vectors to deliver CRISPR-Cas9 during organ perfusion are underway to investigate the potential use of this ge-netic tool to edit genes (addition/knockout) associated with post-transplant ischemia/reperfusion injury with the goal of improving post-transplantation organ function.
Acknowledgments
We thank the Gao lab for production of the AAV-GFP. We thank JL Smith for her expertise and help with data collection. We thank the UMassMed Morphology core for assistance with histology.
CRediT authorship contribution statement
Eliano Bonaccorsi-Riani:Data acquisition, Investigation, Formal analysis, Writing –original draft.Andrew Gillooly:Data acquisi-tion, Investigation, Formal analysis.Isabel MA Brüggenwirth:For-mal analysis, Writing –original draft.Paulo N Martins:Conceptu-alization, Formal analysis, Supervision, Writing -review & editing, Funding acquisition.
Funding
Martins PN was supported by a grant from the University of Massachusetts, American Association for the Study of Liver Diseases (AASLD), and American Society of Transplant Surgeons (ASTS). Bonaccorsi-Riani E was awarded and supported by the In-ternational Travel Scholar Award (2018) by the International Liver Transplantation Society (ILTS) to develop this project. Brüggen-wirth IMA was supported by a travel grant from the European Society of Transplantation (ESOT) and a grant from Jo Kolk Studiefonds.
Ethical approval
This study was performed with approval from the University of Massachusetts Medical School Institutional Animal Care and Use Committee (IACUC) (A-2502-17). Rats were housed within the Uni-versity of Massachusetts Medical School Animal Facilities Core. Healthy male rats were used for studies.
Competing interest
No benefits in any form have been received or will be received from a commercial party related directly or indirectly to the sub-ject of this article.
杂志排行
Hepatobiliary & Pancreatic Diseases International的其它文章
- Comparison and development of advanced machine learning tools to predict nonalcoholic fatty liver disease: An extended study
- Hepatobiliary&Pancreatic Diseases International
- Influence of weight management on the prognosis of steatohepatitis in chronic hepatitis B patients during antiviral treatment
- Key factors and potential drug combinations of nonalcoholic steatohepatitis: Bioinformatic analysis and experimental validation-based study
- LC-MS-based lipidomic analysis in distinguishing patients with nonalcoholic steatohepatitis from nonalcoholic fatty liver
- Cryptococcosis in patients with liver cirrhosis: Death risk factors and predictive value of prognostic models
