Anticoagulation and antiplatelet management in gastrointestinal endoscopy: A review of current evidence
2021-01-11AndrewChanHamishPhilpottAmandaLimMinnieAuDerrickTeeDamianHardingMohamedAsifChinnarathaBijuGeorgeRajvinderSingh
Andrew Chan, Hamish Philpott, Amanda H Lim, Minnie Au, Derrick Tee, Damian Harding, Mohamed Asif Chinnaratha, Biju George, Rajvinder Singh
Abstract The role of endoscopic procedures, in both diagnostic and therapeutic purposes is continually expanding and evolving rapidly. In this context, endoscopists will encounter patients prescribed on anticoagulant and antiplatelet medications frequently. This poses an increased risk of intraprocedural and delayed gastrointestinal bleeding. Thus, there is now greater importance on optimal pre,peri and post-operative management of anticoagulant and/or antiplatelet therapy to minimise the risk of post-procedural bleeding, without increasing the risk of a thromboembolic event as a consequence of therapy interruption. Currently, there are position statements and guidelines from the major gastroenterology societies.These are available to assist endoscopists with an evidenced-based systematic approach to anticoagulant and/or antiplatelet management in endoscopic procedures, to ensure optimal patient safety. However, since the publication of these guidelines, there is emerging evidence not previously considered in the recommendations that may warrant changes to our current clinical practices. Most notably and divergent from current position statements, is a growing concern regarding the use of heparin bridging therapy during warfarin cessation and its associated risk of increased bleeding, suggestive that this practice should be avoided. In addition, there is emerging evidence that anticoagulant and/or antiplatelet therapy may be safe to be continued in cold snare polypectomy for small polyps (< 10 mm).
Key Words: Endoscopy; Anticoagulants; Antiplatelets; Antithrombotics; Bleeding;Gastrointestinal
INTRODUCTION
Contemporary management of patients with atrial fibrillation (AF), venous thromboembolism (VTE) and acute coronary syndromes (ACS) requires the use of an expanding range of anticoagulant and antiplatelet agents. Similarly, the type and range of endoscopic procedures has evolved rapidly, and screening for neoplasia has increased the frequency of procedures per se. In this context, endoscopists will encounter patients prescribed on anticoagulant and antiplatelet medications frequently, and thus an informed and systematic approach to pre, peri and postoperative management is of great importance.
The major risk of anticoagulant and antiplatelet therapy is gastrointestinal bleeding,especially within the first 30 d following an endoscopic procedure[1]. Optimal management involves minimising the risk of post-procedural bleeding (PPB) on one hand, without significantly increasing the risk of a thromboembolic event on the other.Thromboembolic events [including stroke, myocardial infarction (MI) or pulmonary embolism] often have serious, irreversible consequences compared to gastrointestinal bleeding, which if detected early and managed appropriately is of minor consequence.The old wisdom that the brain or heart cannot be replaced, whilst blood or fluid can be readily transfused holds true.
In recent years, a wealth of literature relating to anticoagulant and antiplatelet use has emerged, including a number of position statements and guidelines from the major gastroenterology societies in Europe, the United States of America and Asia.These documents, along with the research studies from which they are based, should logically form the basis of future recommendations. The purpose of this review therefore is to firstly evaluate the index bleeding risk associated with common endoscopic procedures in the absence of anticoagulant and/or antiplatelet use. We then aim to consider the major research studies relating to anticoagulant and antiplatelet use in this context, and to compare the available evidence against the relevant major guidelines mentioned, to assimilate a new working reference, and to highlight any knowledge gaps and directions for future research.
SEARCH STRATEGY
We performed a structured literature review using Ovid Medline, considering articles from January 1, 2011 to January 1, 2020, with the intention of identifying relevant research potentially not included in recent guidelines[2-4]. Medical Subject Headings (Supplementary material) were formulated relating to the anticoagulant and antiplatelet agents of interest [aspirin, thienopyridine (clopidogrel, prasugrel,ticagrelor), warfarin, direct oral anticoagulants (DOACs) (dabigatran, rivaroxaban,apixaban), heparin bridging therapy (HBT)], all relevant endoscopic procedures, and“bleeding” rates. Case reports, abstracts, commentaries, letters, and editorials were not considered. Relevant articles were retrieved and reviewed, with data tabulated (Tables 1-56)
COMMON ENDOSCOPIC PROCEDURES AND THE INDEX POSTPROCEDURE BLEEDING RISK IN THE ABSENCE OF ANTICOAGULANT AND/OR ANTIPLATELET USE
A summary of the relevant studies evaluating the index PPB risk for common endoscopic procedures, in the absence of anticoagulant and/or antiplatelet use, are outlined in Tables 1-16.
DIAGNOSTIC ENDOSCOPIC PROCEDURES
Diagnostic endoscopy and colonoscopy with biopsy (Table 1)
Endoscopic biopsy is a minimally invasive procedure that is commonly undertaken during diagnostic endoscopies and colonoscopies to diagnose a range of conditions (e.g., neoplasia, coeliac disease,Helicobacter pylori). The risk of PPB is low, ranging from 0.12%-0.98% in published studies[5-7].
Diagnostic ± therapeutic push or device assisted enteroscopy/balloon enteroscopy(Table 2)
Double balloon enteroscopy (DBE) allows for detailed and direct visualisation and assessment (diagnostic) of the small bowel and application of endoscopic intervention.The risk of PPB associated with DBE is 0.5%, but increases with therapeutic intervention[8,9]. The study by Wanget al[9]recorded seven episodes of PPB in 1531 DBEs (0.5%), with all associated with therapeutic polypectomy.There were no reported incidences of PPB in the studies for diagnostic-only DBE.
Endoscopic ultrasound ± fine needle aspiration (Table 3)
Endoscopic ultrasound-guided fine needle aspiration (EUS-FNA) with a 22G FNA needle is the gold standard diagnostic tool for pancreatic and upper gastrointestinal tract lesions. A 22G FNA needle is generally preferred, but the procedure can also be performed with either 19G or 25G needles. The reported risk of PPB varies according to needle gauge, ranging from 2.1% with 25G needles to 4.3% with 22G needles[10-17]. Of note, both the study by Vilmannet al[13]and Inoueet al[17]observed an associated immediate/intraprocedural bleeding risk of 0.7%-1%. However, in both studies, the bleeding was self-limited and did not require any further endoscopic intervention.
Published data on the use of 19G needles is more limited compared to the evidence available for both the 22G and 25G needles. A 19G needle is more rigid than its smaller gauge counterparts. This makes adequate positioning of the endoscope and manipulation technically more difficult[18]. However, successful use of 19G needles has been shown to yield superior diagnostic accuracy and better diagnostic tissue acquisition compared to the 22G and 25G needles[18,19]. There were no reported incidences of PPB in any of the studies[18-20], although two studies observed an associated immediate/intraprocedural bleeding risk of 1.0%-1.8%[19,20]with 19G needle use.
Endoscopic retrograde cholangiopancreatography (diagnostic) (Table 4)
With advancements in imaging modalities, such as magnetic resonance cholangiopancreatography (MRCP), the role for diagnostic only endoscopic retrograde cholangiopancreatography (ERCP) is rare. ERCP is now predominantly considered an interventional procedure (endoscopic sphincterotomy, papillotomy, biliary stone removal and insertion of biliary stents). Diagnostic ERCP rarely causes PPB with a rate of 0.3%-1.66% reported[21-25].
In all of the studies, PPB was most commonly observed in diagnostic ERCP whensphincterotomy was required to obtain better access. Sphincterotomy is associated with an up to five-fold increased risk of PPB[21,23-25]and will be discussed further in the“ERCP with sphincterotomy” section (Table 9).
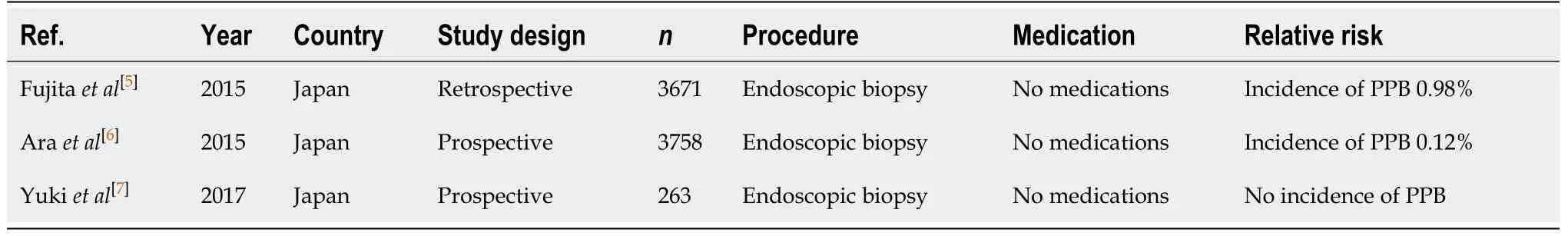
Table 1 Diagnostic endoscopy and colonoscopy with biopsy
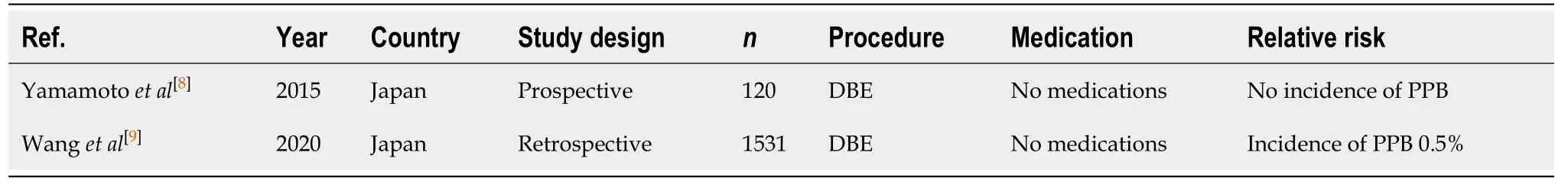
Table 2 Diagnostic ± therapeutic push or device assisted enteroscopy/balloon enteroscopy
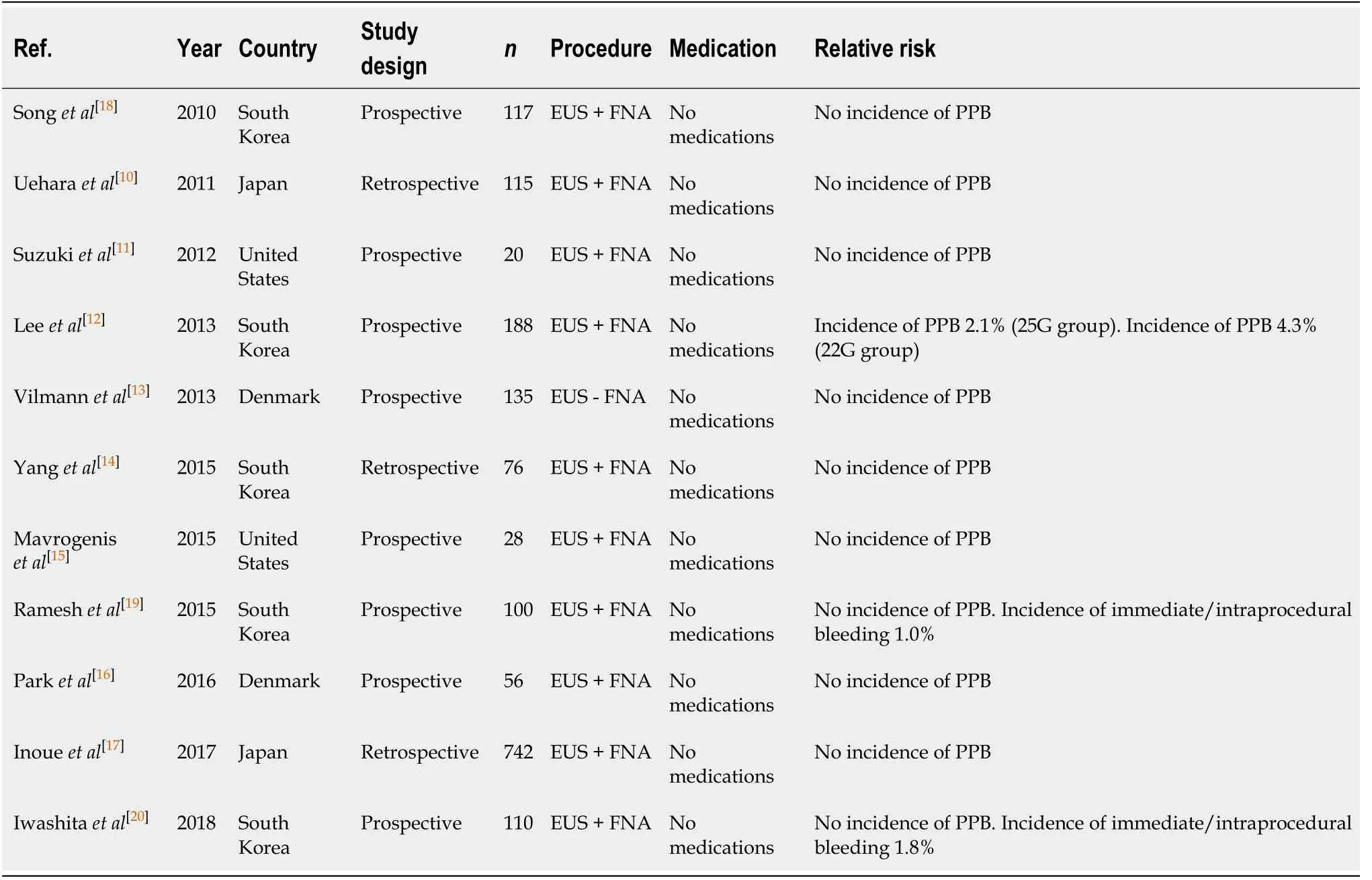
Table 3 Endoscopic ultrasound ± fine needle aspiration
THERAPEUTIC ENDOSCOPIC PROCEDURES
Conventional polypectomy/hot snare polypectomy (Table 5)
Conventional polypectomy, also referred to as hot snare polypectomy (HSP), uses electrosurgical current through a polypectomy snare and is the standard practice for polyp resection and prevention of colorectal cancer. It has been associated with a colorectal cancer mortality reduction over 30 years. Numerous published studies have identified the overall risk of PPB post conventional polypectomy to be around 0.05%-3.0%[26-42]. Larger polyp sizes (> 10 mm), polyps located in caecum and ascending colon,and pedunculated polyps are all associated with an additional increased risk of overall PPB[33,36,41,43].
Cold snare polypectomy and endoscopic mucosal resection (Tables 6 and 7)
Aside from conventional polypectomy (HSP), other polypectomy techniques are often utilised, specifically cold snare polypectomy (CSP) and endoscopic mucosal resection(EMR). The chosen method is often dependent on polyp characteristics. Hot biopsy forceps (HBF) are insulated monopolar electrocoagulating forceps, allowing for biopsy and electrocoagulating tissue simultaneously[44]. HBF were previously used for polypectomy of diminutive polyps, but have since fallen out of favour due to its pooreren-blocresection rate, and increased rate of significant injury to the pathology tissue compared to CSP[45]. HBF was not a focus for this review and will not be discussed further given it is no longer commonly practiced.
The European Society of Gastrointestinal Endoscopy (ESGE) clinical guidelines[46]recommends the use of CSP technique for removal of diminutive polyps ≤ 5mm and sessile polyps 6-9 mm in size because of its superior safety profile. Studies have shown that CSP is superior to HSP in resection of polyps ≤ 10 mm, with a shorter procedure time[27]and no statistically significant difference in complete resection rate[27,39], or delayed bleeding rates[27,37-40]. The risk of delayed PPB in CSP is shown to be very low with no incidences (0%) observed in any of the studies[27,37-39,47,48]. This is comparable to HSP with an incidence rate of 0%-0.5% for polyps ≤ 10 mm[27,37-40]. However, there is an increased risk of immediate/intraprocedural PPB in CSP for small polyps (< 10 mm),with three studies[27,39,48]showing an intraprocedural bleeding rate of 2.7%-9.1%,compared to 1%-3.5% in HSP[27,39].
Conventionally, HSP (for polyps > 10 mm in size) and EMR (for polyps > 20 mm in size, particularly if sessile) have been the standard of care in the removal of these larger polyps, as it is considered more efficacious in minimising the risk of intraprocedural bleeding. The ESGE clinical guideline on colorectal polypectomy and EMR[46]still recommends HSP as the preferred technique for polyps 10-19 mm in size and EMR for polyps ≥ 20 mm. This is due to its ability to cauterise the resected tissue,while also providing additional ablation to the residual tissue, promoting complete haemostasis[40]. The risk of intraprocedural and delayed PPB with EMR in polyps < 10 mm is 1.7%[48]and 0%-1.7%[48,49], respectively. Risk of delayed PPB is higher with increasing polyp size. Soet al[50]found an incidence of 6.3% in polyps with a mean size of 34 mm.
Recent publications suggest that HSP carries a higher risk of both PPB andperforation compared to CSP in polyps > 10 mm, likely due to the thermal injury of the intestinal wall. A study of resection specimens indicates that the increased risk of delayed bleeding was due to more extensive arterial injury in the submucosal, deep submucosa and muscularis propria layers caused by HSP[40]. In contrast, the removal of polyps > 10 mm by CSP does not cause PPB, with no evidence of bleeding in six studies[40,51-55]. The study by Hiroseet al[54]reported one case of delayed PPB, but this patient was on warfarin for AF and so was not included in the final analysis. This is compared to a delayed PPB incidence rate of 3.5%, as published in a study by Ketet al[40]in the removal of polyps > 10 mm by HSP.
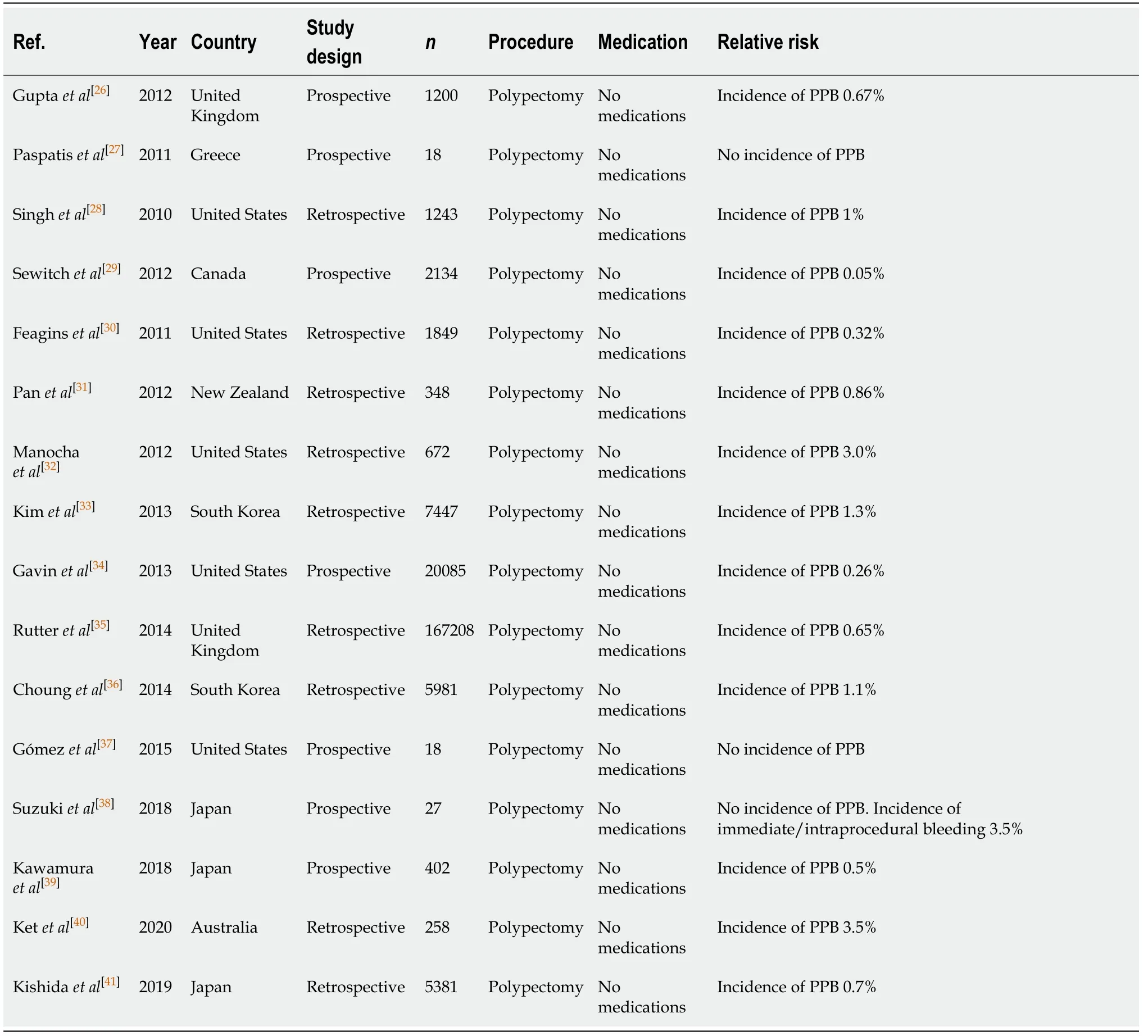
Table 5 Conventional polypectomy/hot snare polypectomy
There was limited published data on the time to PPB in patients undergoing HSP in the available studies. The study by Ketet al[40]reported the time to PPB in their patient cohort to be between 2 to 7 d post endoscopic procedure. While, the study by Sewitchet al[29]had only one complication of PPB (0.05%) which occurred 3 wk post polypectomy. However, this was thought to be more likely in the setting of follow-up treatment rather than the index colonoscopy. A potential limitation is the majority of the studies were retrospective studies, which may have missed subsequent bleeds due to an inadequate follow-up period post procedure.
Endoscopic submucosal dissection (Table 8)
The practice of endoscopic submucosal dissection (ESD) is often required for theresection of large gastrointestinal lesionsen bloc, and (compared to CSP and EMR) is associated with a significantly higher risk of PPB between 2.7% to 6.6%[56-63]irrespective of the location of the lesion. This increased risk also translates to a higher risk of immediate/intraprocedural bleeding, reportedly 6.1% in a study by Chenet al[63].
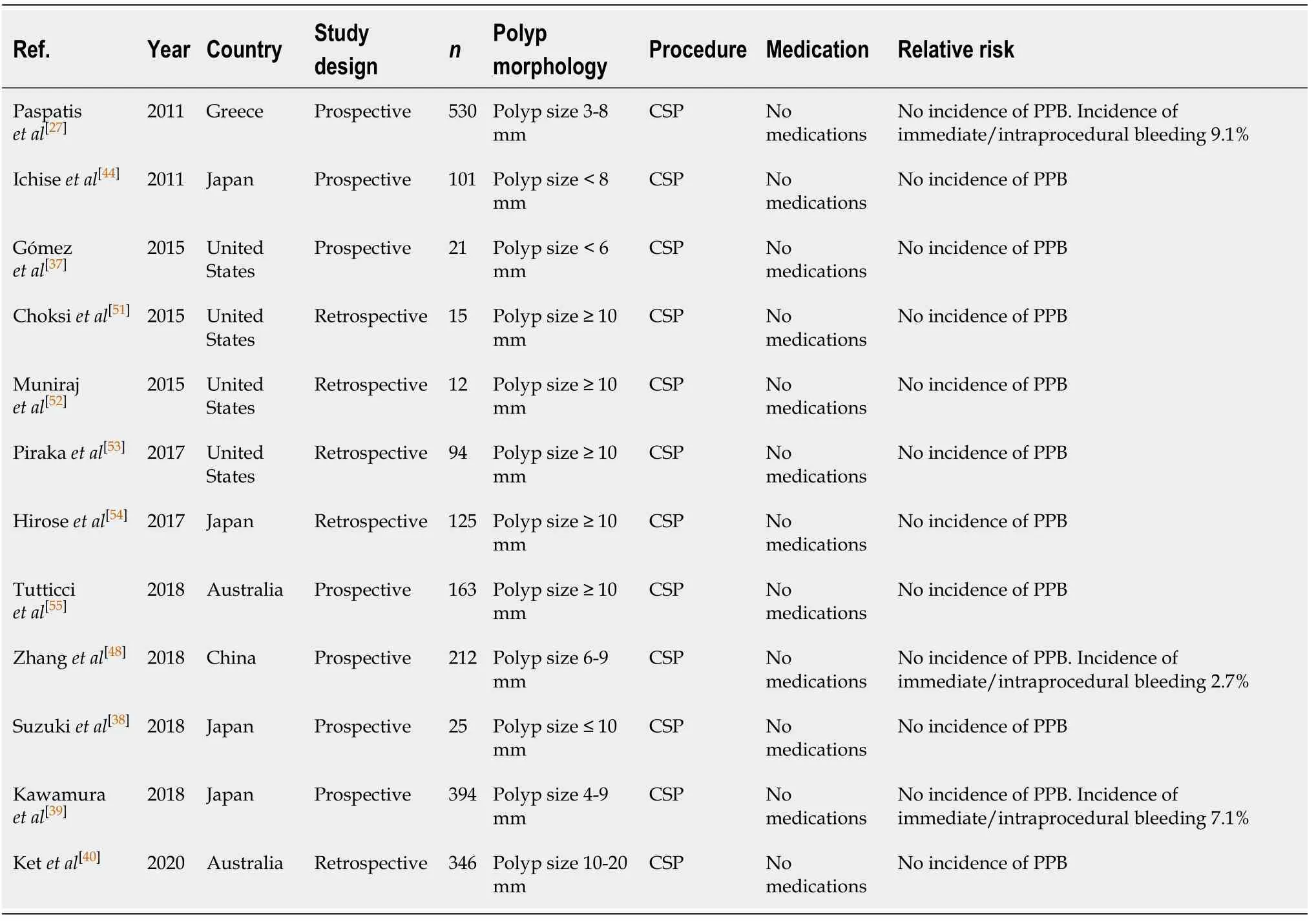
Table 6 Cold snare polypectomy
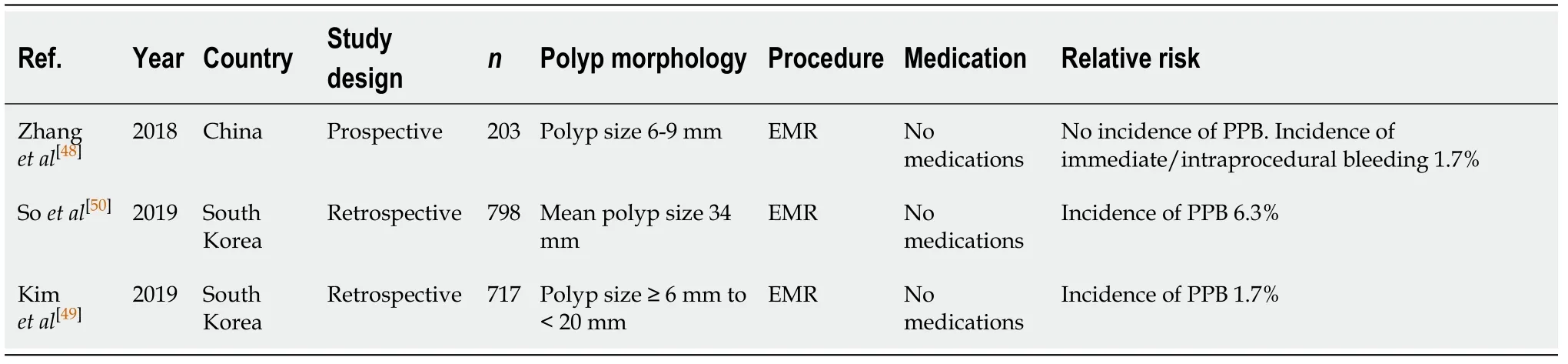
Table 7 Endoscopic mucosal resection
ERCP with sphincterotomy (Table 9)
Endoscopic sphincterotomy has now become a standard intervention during ERCP for therapy of pancreaticobiliary diseases, but is commonly associated with complications of PPB. The risk of bleeding post ERCP with sphincterotomy is between 0.45%-9.9%[21,64-71]. Timing of bleeding varied between studies, with Baeet al[69]finding the majority of their cases [95 out 108 patients (88.0%)] were from immediate/intraprocedural bleeding. Similarly, Masciet al[21]observed a higher occurrence of immediate/ intraprocedural bleeding of 1.1%, compared to only a 0.7% rate of delayed PPB. This is in contrast to the findings from Pataiet al[66], which found a higheroccurrence of delayed PPB of 6.3%, compared to only a 2.7% rate of immediate/intraprocedural bleeding.
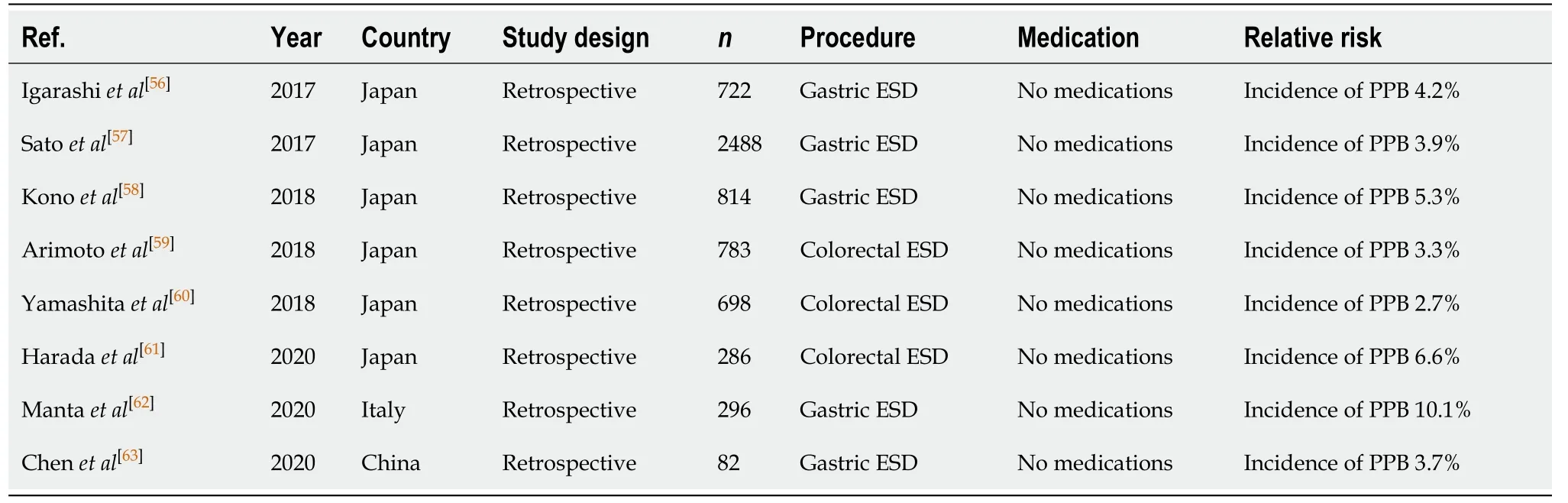
Table 8 Endoscopic submucosal dissection
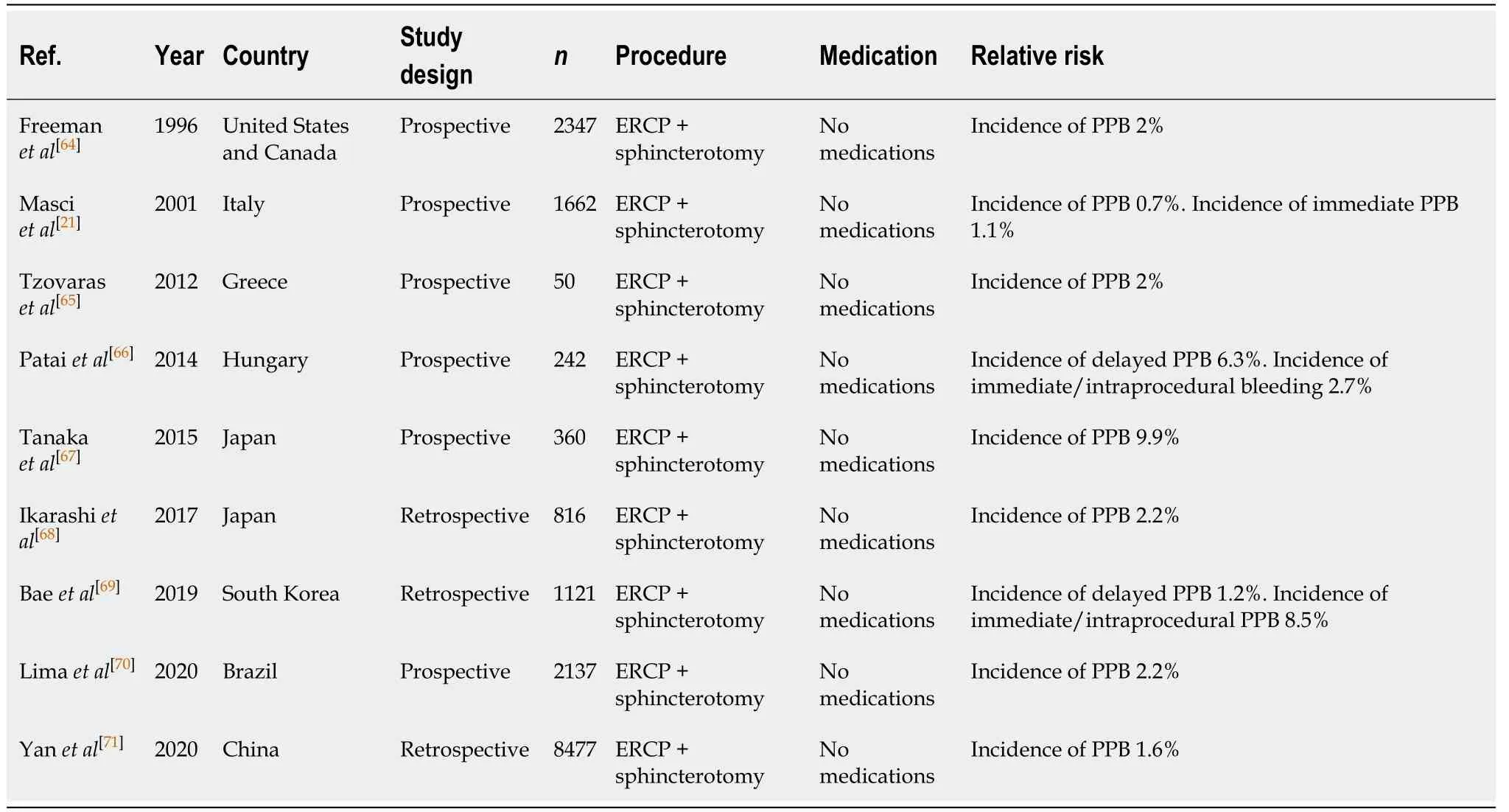
Table 9 Endoscopic retrograde cholangiopancreatography with sphincterotomy
Ampullectomy (Table 10)
Endoscopic ampullectomy allows for a minimally invasive nonsurgical intervention option for the treatment of ampullary adenomas, however is associated with significant risk of PPB between 4.9% to 30%[72-79]. The considerably high incidence of PPB of 30% reported in the study by Hopperet al[72]was observed in resections of larger sized ampullary adenomas (between 40-60 mm). A limitation of this study was a small sample size of 10. Close monitoring post endoscopic ampullectomy is important.
Endoscopic dilatation (Table 11)
Endoscopic dilatation provides an alternative to surgical intervention, reducing morbidity and prolonging the surgery-free intervals, in patients with symptomatic gastrointestinal strictures. Data from patients with eosinophilic oesophagitis who required dilatation found that PPB was rare (0%-0.3%)[80-84].
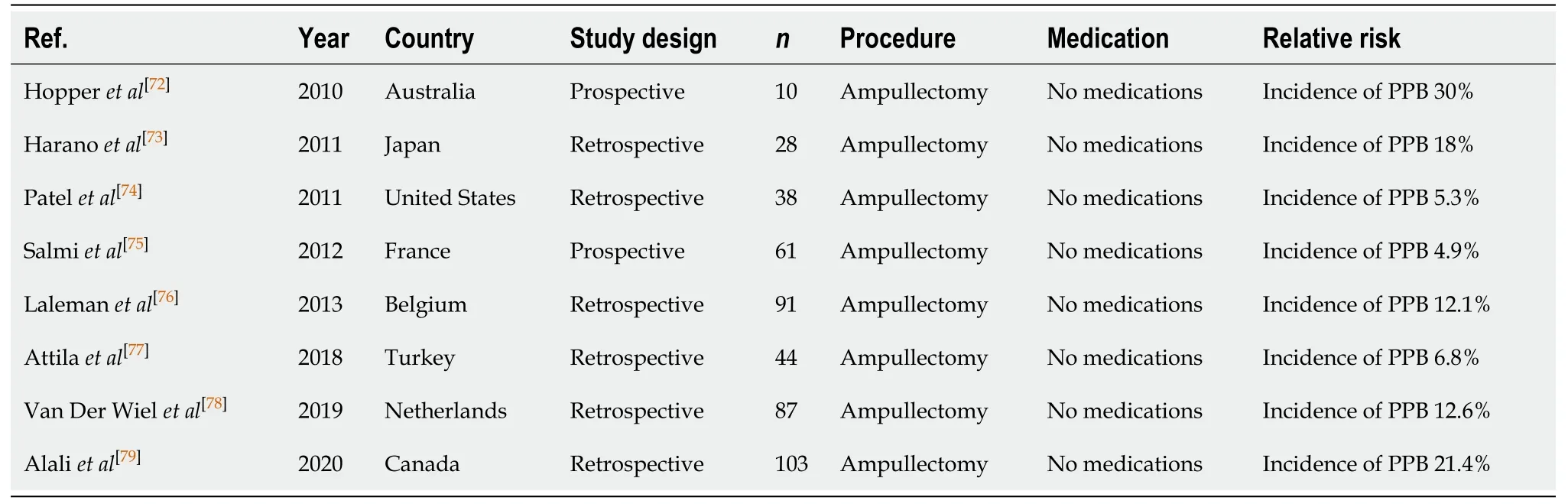
Table 10 Ampullectomy
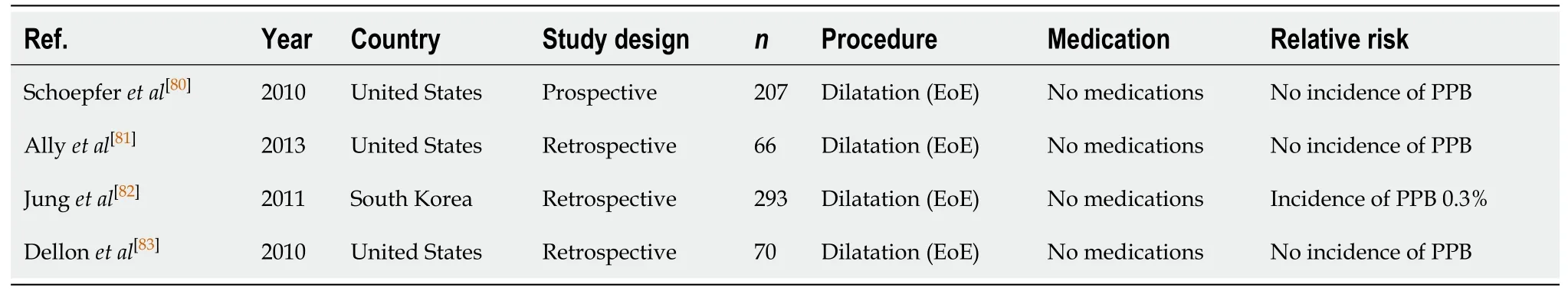
Table 11 Endoscopic dilatation
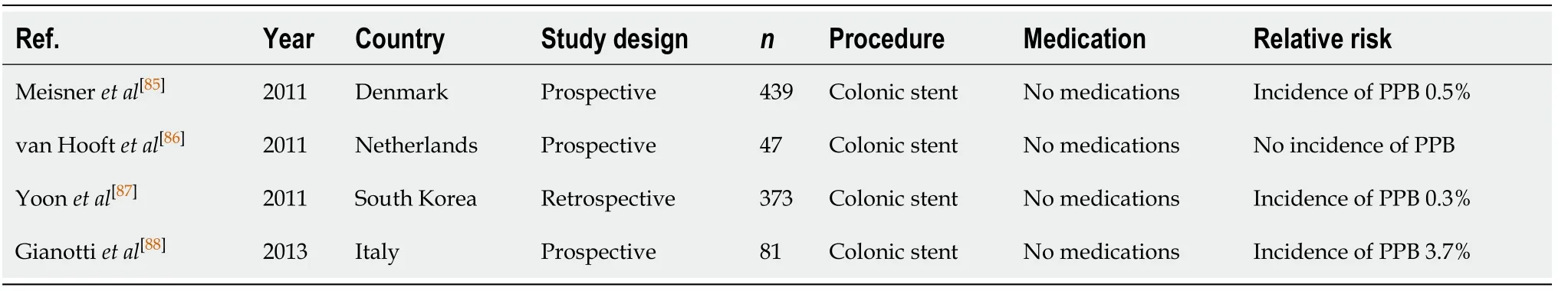
Table 12 Colonic stenting

Table 13 Enteral stenting
Colonic, enteral, and oesophageal stenting (Tables 12-14)
Endoscopic placement of self-expandable metallic stent (SEMS), or other various types of stents, is commonly indicated in patients with gastrointestinal obstructive disease secondary to malignancy. It plays an important role in either temporary bridging to surgery, or palliative management in patients with incurable disease[85]. For endoscopic colonic SEMS placement, the risk of PPB is estimated to range from 0.3%-3.7% in several publications[85-88].
A study by Costamagnaet al[89]reported a similar rate of PPB, compared to colonic stenting, of 3% post endoscopic duodenal stent insertion.

Table 14 Oesophageal stenting
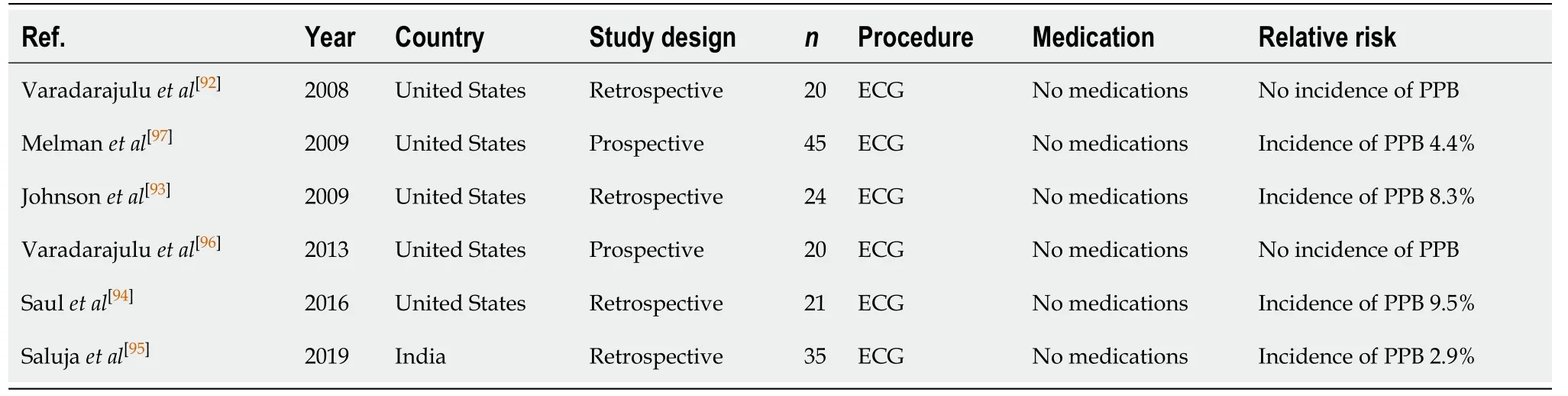
Table 15 Endoscopic cystogastrostromy
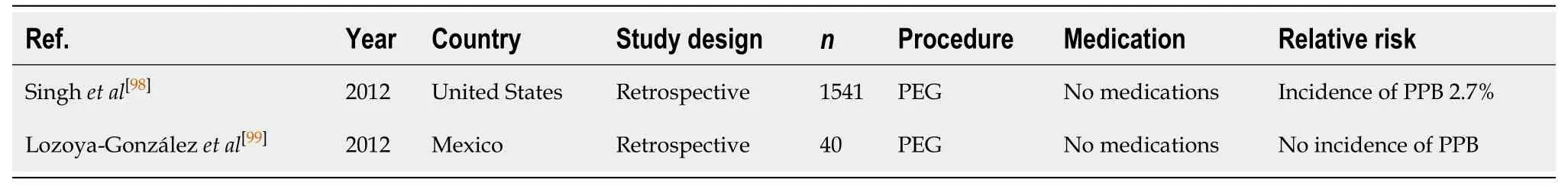
Table 16 Percutaneous endoscopic gastrostomy/percutaneous endoscopic jejunostomy insertion
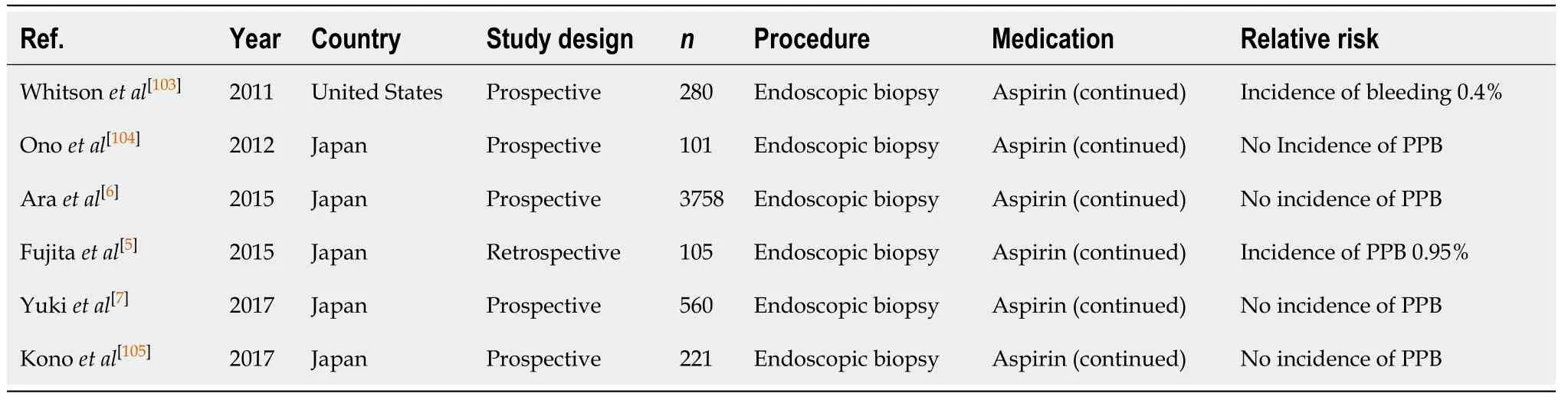
Table 17 Diagnostic endoscopy and colonoscopy with biopsy
However, oesophageal stent insertion for oesophageal obstruction has been reported to be associated with higher risk of PPB compared to both colonic and duodenal stenting, of 1.7%-10.4% in two retrospective studies[90,91]. Liuet al[91]defined massive PPB as bleeding that required > 3 units of packed red blood cells and which was complicated by haemorrhagic shock. Massive bleeding was observed in 54 out of 519 of their patients (10.4%) and was associated with fatality within 24 h. Independent risk factors contributing to an increased risk of bleeding (from highest to lowest risk)includes: The presence of accompanying tracheal stent insertion, previous history of radiotherapy and oesophageal fistulae[91].
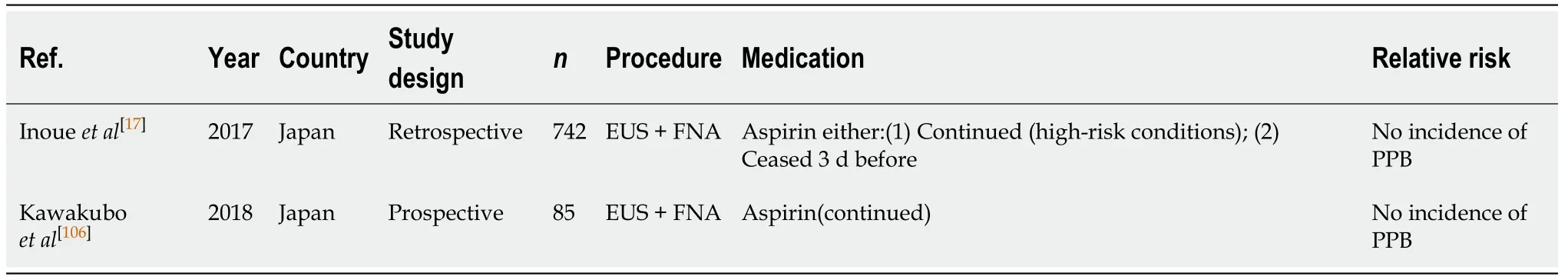
Table 18 Endoscopic ultrasound ± fine needle aspiration
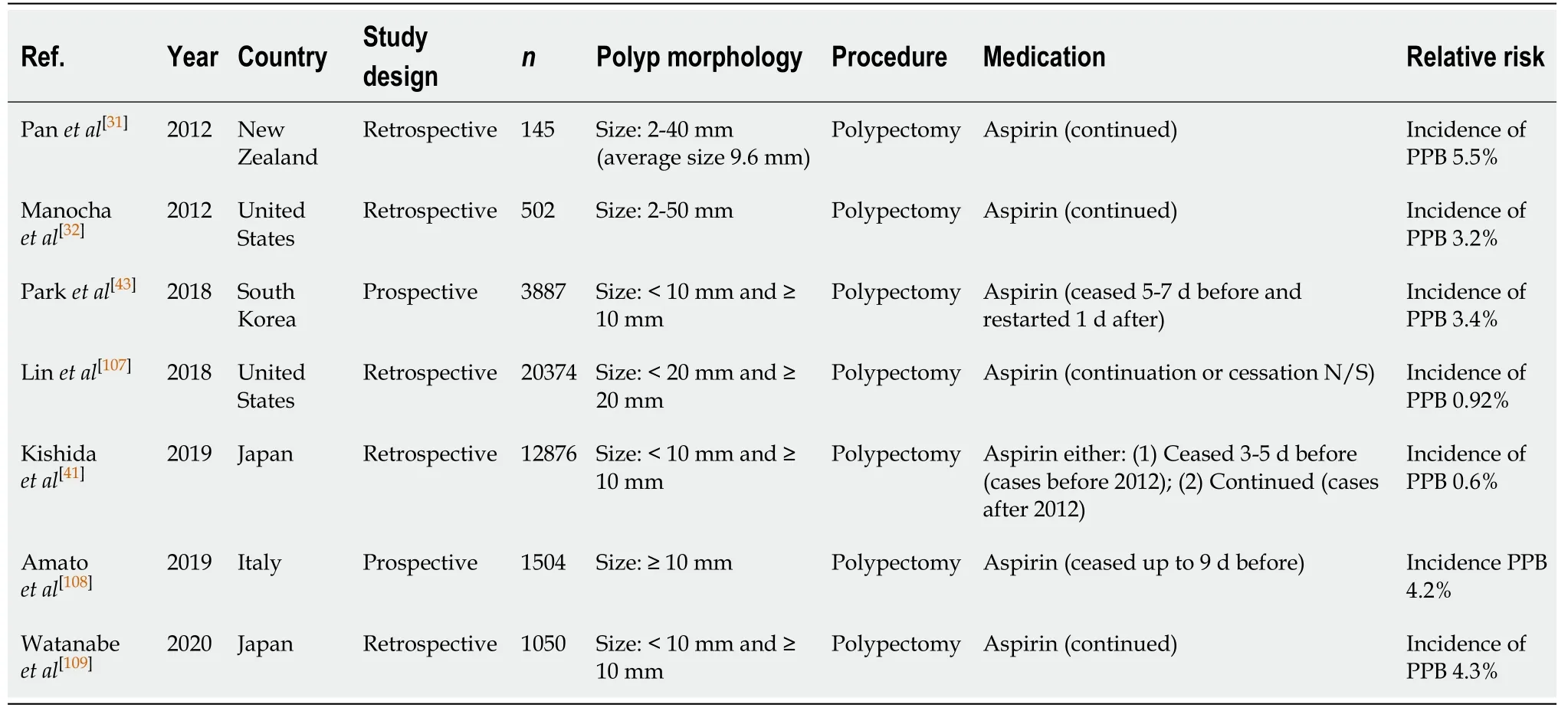
Table 19 Polypectomy
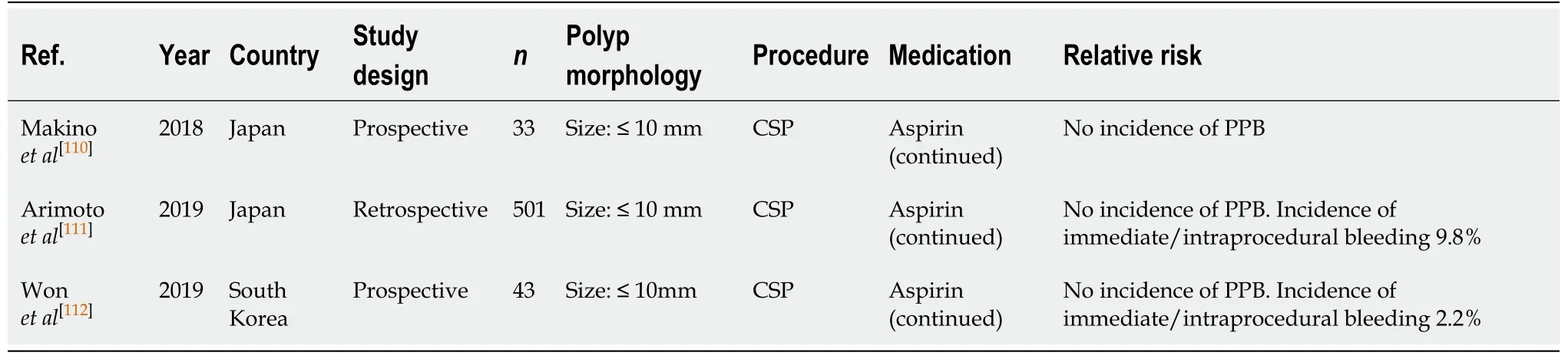
Table 20 Cold snare polypectomy
Endoscopic cystogastrostromy (Table 15)
Endoscopic drainage of contained pancreatic fluid collections (pseudocysts) as a result of acute or chronic pancreatitis, trauma or obstruction, is traditionally considered firstline management over surgical drainage[92-95]. Varadarajuluet al[96]reported no significant difference in outcomes of treatment success, complication rates, and need for re-intervention between endoscopicvssurgical drainage. Although there were significant benefits in the length of hospital stay post endoscopic cystogastrostomy[median stay of 2 d, compared to 6 d in the surgical group (P< 0.001)]. Endoscopic cystogastrostomy is however associated with a significant risk of PPB of between 2.9%-9.5%[92-97].
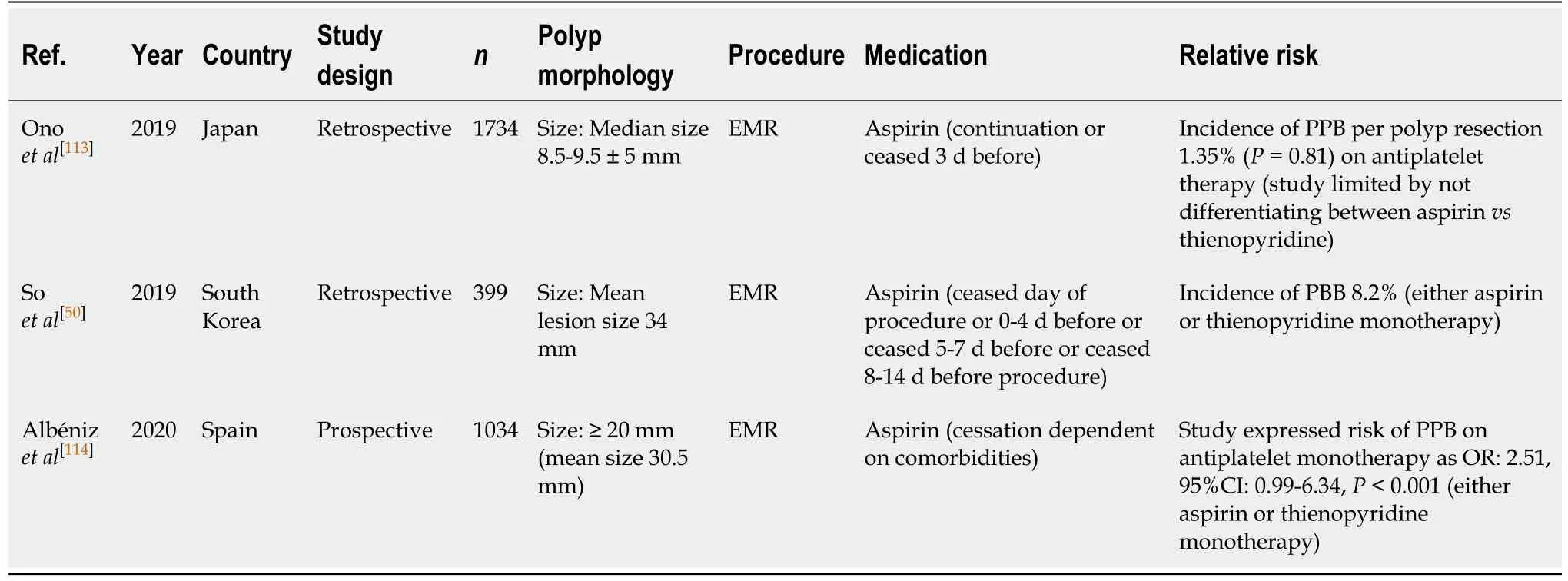
Table 21 Endoscopic mucosal resection
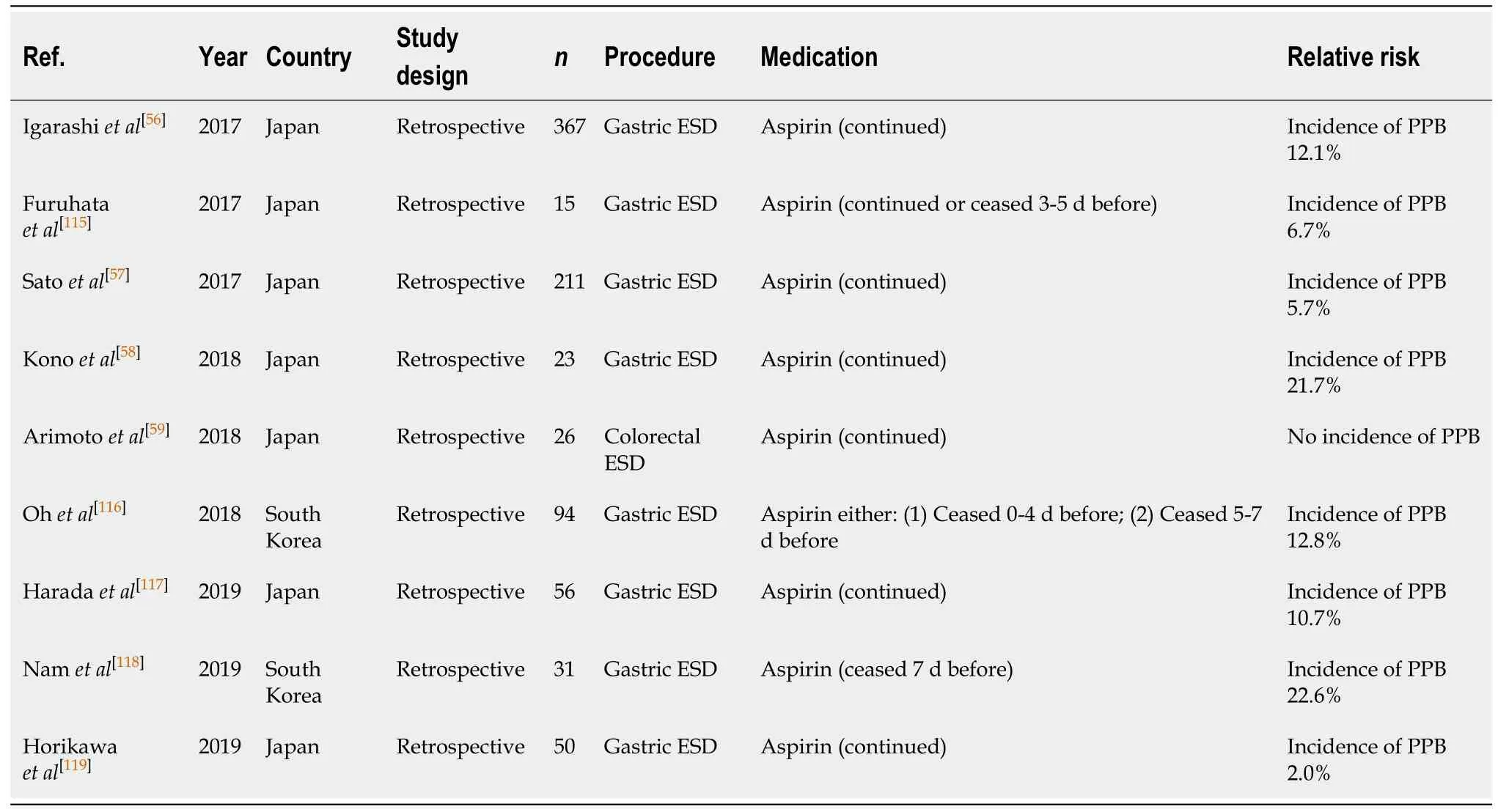
Table 22 Endoscopic submucosal dissection
Percutaneous endoscopic gastrostomy/percutaneous endoscopic jejunostomy insertion (Table 16)
The endoscopic placement of percutaneous endoscopic gastrostomy (PEG)/percutaneous endoscopic jejunostomy (PEJ) has a PPB rate of 0%-2.7%[98,99].
COMMON ENDOSCOPIC PROCEDURES AND THE RISK OF POSTPROCEDURE BLEEDING ASSOCIATED WITH EACH ANTICOAGULANT AND ANTIPLATELET AGENT
A summary of the relevant studies evaluating the bleeding risk associated with each anticoagulant and antiplatelet agent for common endoscopic procedures is outlined in Tables 17-56.
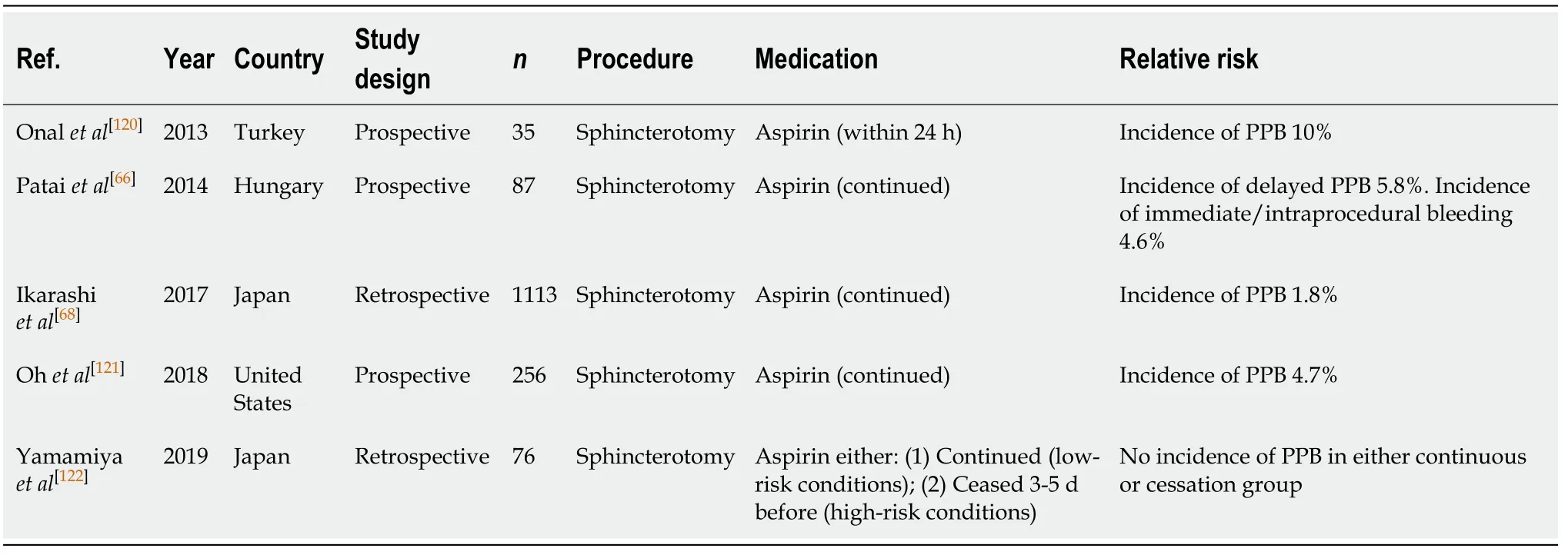
Table 23 Endoscopic retrograde cholangiopancreatography with sphincterotomy
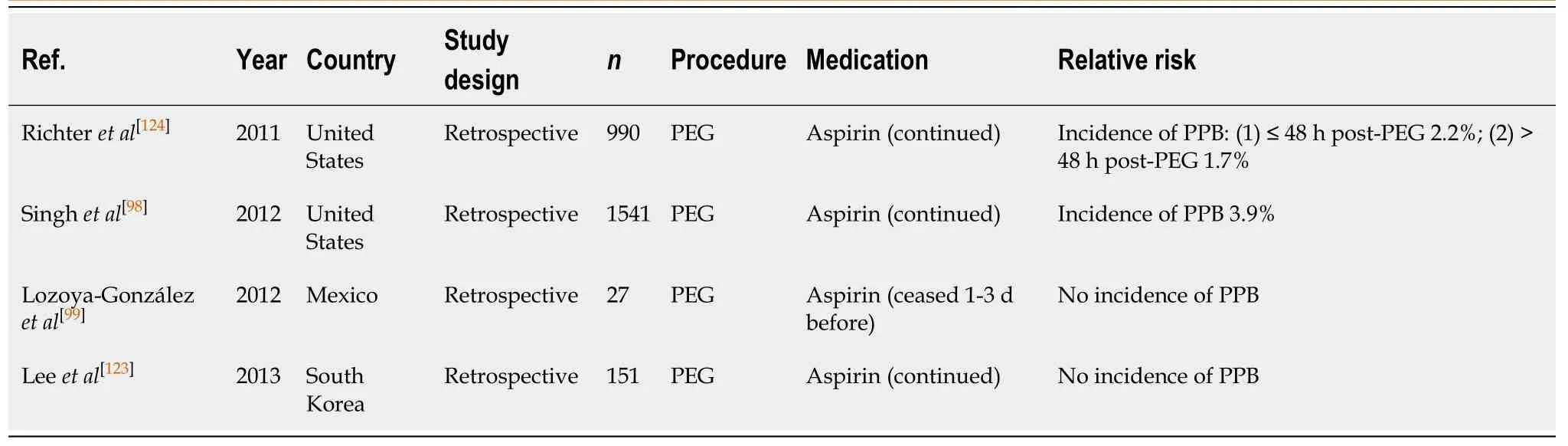
Table 24 Percutaneous endoscopic gastrostomy/percutaneous endoscopic jejunostomy insertion
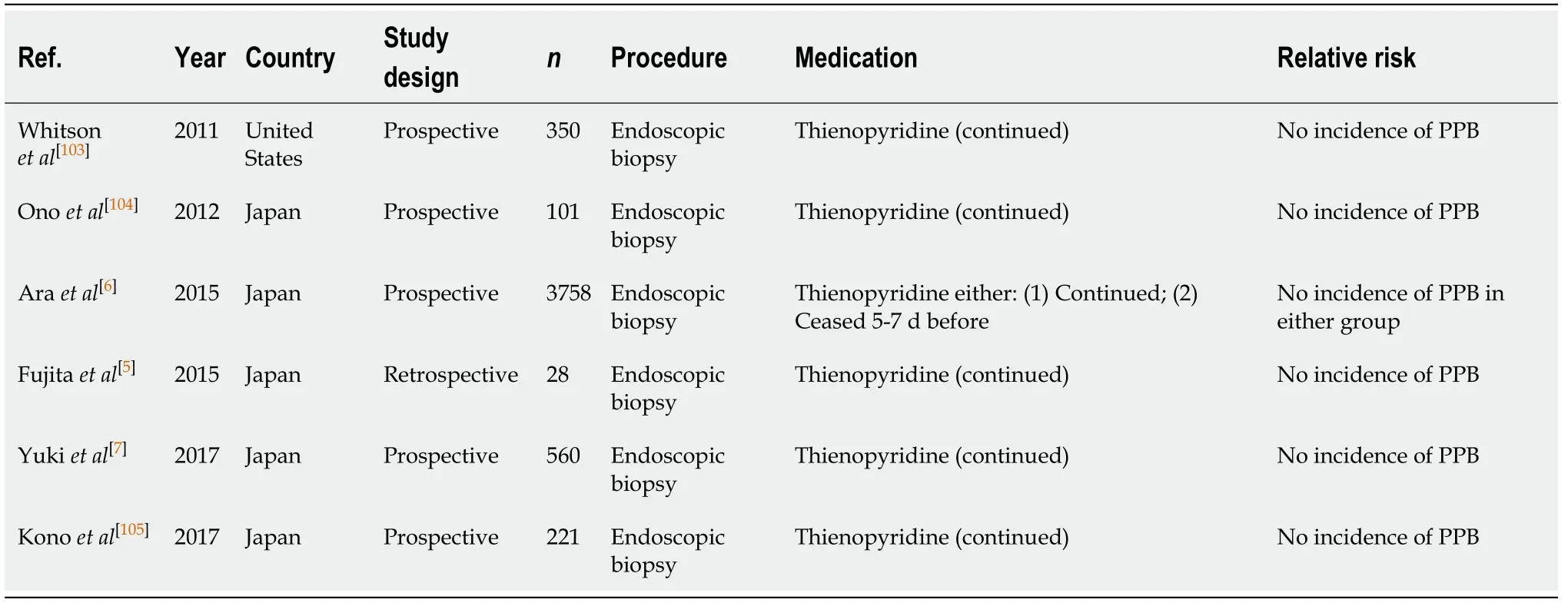
Table 25 Diagnostic endoscopy and colonoscopy with biopsy
ACETYLSALICYLIC ACID (ASPIRIN) MONOTHERAPY
Acetylsalicylic acid, also known as aspirin, acts by irreversibly inhibiting the cyclooxygenase 1 and 2 enzyme system, resulting in reduction of thromboxane A2 synthesis leading to inhibition of platelet aggregation[100].
Antiplatelet therapy, with aspirin, is first line for secondary prevention of ACS, noncardioembolic ischaemic stroke and transient ischaemic attack (TIA). In a metaanalysis of randomised controlled trials (RCTs) of aspirin therapy for secondary MI and stroke prevention, there was a 34% reduction in non-fatal MI and a 25% reduction in non-fatal strokes when on long-term aspirin therapy[101].
Interruption of aspirin, in cases of elective endoscopic procedures, is associated with a three-fold increased risk of cardiovascular or cerebrovascular event, with 70% of events occurring within the first 7 to 10 d of withholding antiplatelets[102]. Therefore,withholding aspirin therapy needs to be carefully considered.
Diagnostic endoscopy and colonoscopy with biopsy (Table 17)
Continuing aspirin monotherapy in diagnostic endoscopies and colonoscopies with biopsy is associated with an overall low risk of PPB of 0.4%-0.95% from multiple published studies[5-7,103-105]. There is minimal additive risk in continuing aspirin, as the index bleeding risk in the absence of antiplatelet use is similar, between 0.12%-0.98%(Table 1).
Continuing aspirin without interruption is considered safe in diagnostic endoscopies and colonoscopies with biopsy for patients with indication for aspirin.This recommendation concurs with previous position statements.
EUS ± FNA (Table 18)
The risk of PPB in EUS ± FNA while on continuous aspirin is low. In two recent studies there were no reported incidences of PPB[17,106]. In the study by Inoueet al[17],aspirin monotherapy was either continued, in patients considered to be at high-risk of thromboembolism secondary to drug withdrawal, or withheld 3 d before the procedure. There were no incidences of PPB in either subgroup. However, one case of immediate/intraoperative bleeding occurred in the continued aspirin group (1.6%).
Continuing aspirin in EUS ± FNA is safe and recommended to avoid the risk of a thromboembolic event. This concurs with previous position statements.
Polypectomy (Table 19)
The risk of PPB following endoscopic polypectomy in patients on aspirin monotherapy has been considered by a number of groups who performed RCTs. Aspirin use is associated with a three- to six-fold increased relative risk of PPB post endoscopic polypectomy[31], although the absolute risk of PPB is overall still low at 0.6%-5.5%[31,32,41]. Three other studies assessed the risk of PPB when aspirin was withheld at least 3-7 d before the procedure and the associated risk of PPB as a result, was reported to be 0.6%-4.2%[41,43].
The risk of PPB on aspirin monotherapy, either when continued or withheld before the procedure, is overall low at 0.6%-5.5%[31,32,41,43,107-109]and has a similar absolute risk of bleeding in the absence of anticoagulant or antiplatelet use, of 0.05%-3.0% (Table 5).Thus, continuation in all cases is recommended. This concurs with previous position statements.
CSP (Table 20)
There is emerging evidence that aspirin monotherapy in CSP is safe and not associated with an increased risk of PPB. All three studies[110-112]observed no incidences of PPB when aspirin monotherapy was continued. However, two of the studies[111,112]did observe incidences of immediate/intraprocedural bleeding, of 2.2% in the study by Wonet al[112]to 9.8% in the study by Arimotoet al[111]. However, the study by Arimotoet al[111]failed to quantify the percentage of immediate/intraprocedural PPB cases on continuous aspirin compared to thienopyridine therapy. Therefore, it is unclear the exact risk of immediate bleeding on aspirin monotherapy alone. Despite this, the reported absolute risk of immediate/intraprocedural bleeding on continued aspirin monotherapy is similar to the bleeding risk in the absence of anticoagulant or antiplatelet use (2.2%-9.8%vs2.4%-9.1%, respectively) (Table 6).

Table 26 Endoscopic ultrasound ± fine needle aspiration
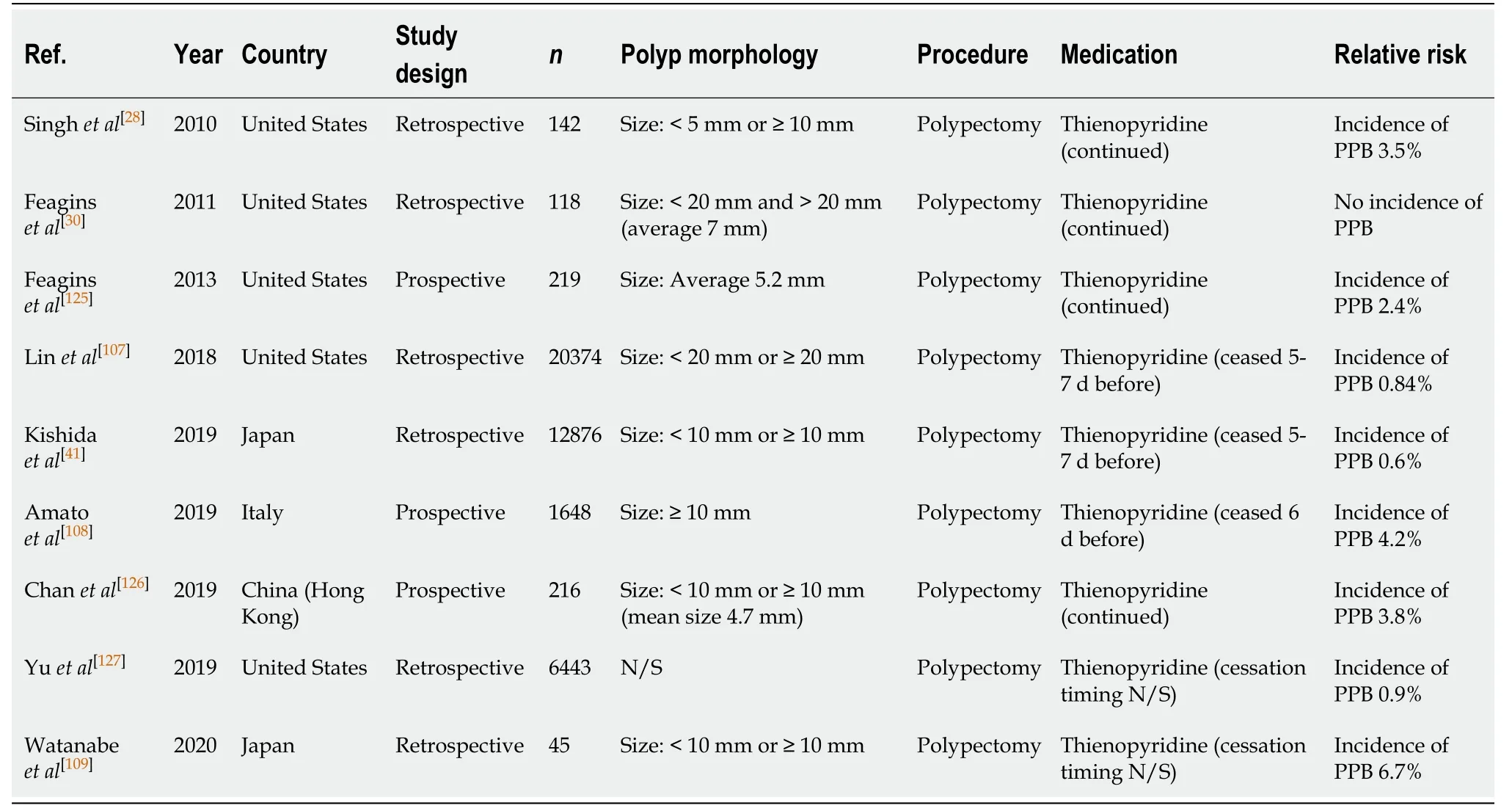
Table 27 Polypectomy

Table 28 Cold snare polypectomy
The bleeding risk with continued aspirin monotherapy is not shown to significantly increase the risk of bleeding, and continuation in all cases is recommended. This is in accordance with previous position statements.
EMR (Table 21)
Several studies have examined the effects of Aspirin monotherapy and the risk of PPB in EMR[50,113,114]. A study by Albénizet al[114]prospectively assessed the incidence of PPB post EMR in patients who either continued aspirin monotherapy, or had it withheld before EMR. They found that antiplatelet use, either aspirin or thienopyridine monotherapy before EMR, is associated with a two-fold increased relative risk of PPB(OR, 2.51; 95%CI, 2.14-9.63,P< 0.001) in lesions ≥ 20 mm. However, the study was limited by not specifying the risk of PPB associated with aspirin monotherapy only.
Another study by Soet al[50]observed a rate of PPB of 8.2% in EMR of polyps of mean size > 30 mm when on antiplatelet monotherapy. EMR in smaller polyps of < 10 mm was only associated with a 1.35% risk of PPB per polyp resection when onantiplatelet therapy (aspirin monotherapy either continued or withheld 3 d before) in the study by Onoet al[113]. Once again, both studies assessed the risk of PPB on either aspirin or thienopyridine monotherapy together and so did not specify the associated risk of aspirin monotherapy alone. Despite this, the risk of PPB is comparable to the absolute risk of bleeding in the absence of anticoagulant or antiplatelet use of respective size (1.35%vs1.7% in polyps ≤ 10 mm and 8.2%vs6.3% in polyps ≥ 20 mm,respectively) (Table 7).
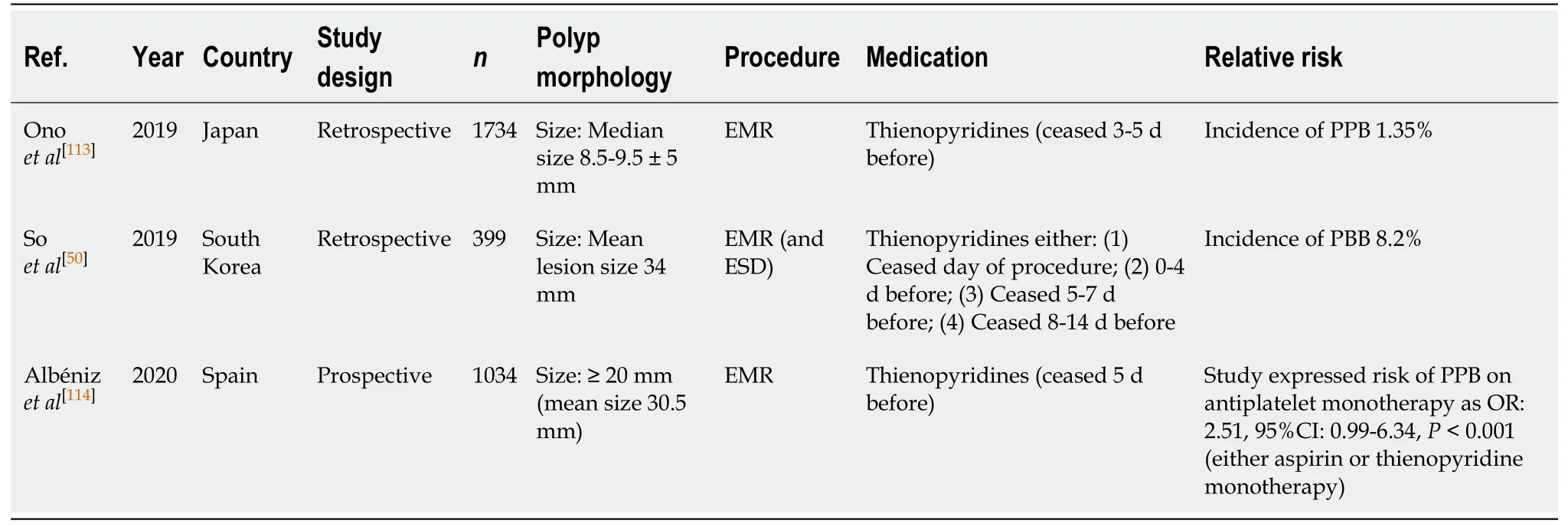
Table 29 Endoscopic mucosal resection
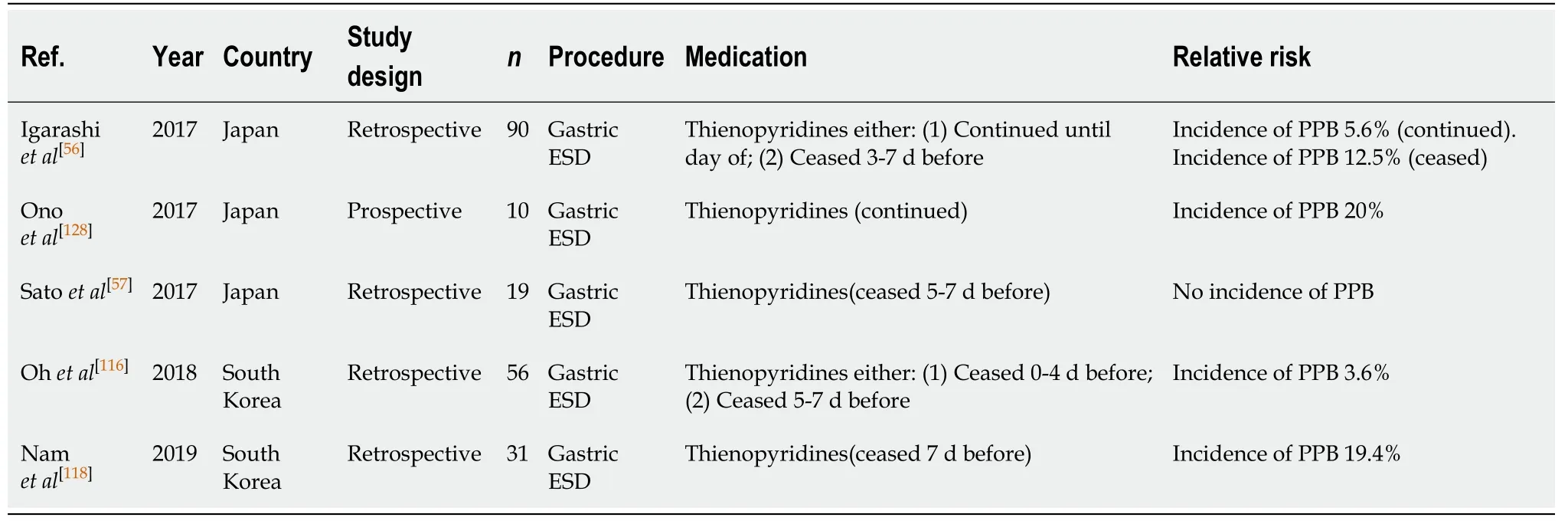
Table 30 Endoscopic submucosal dissection
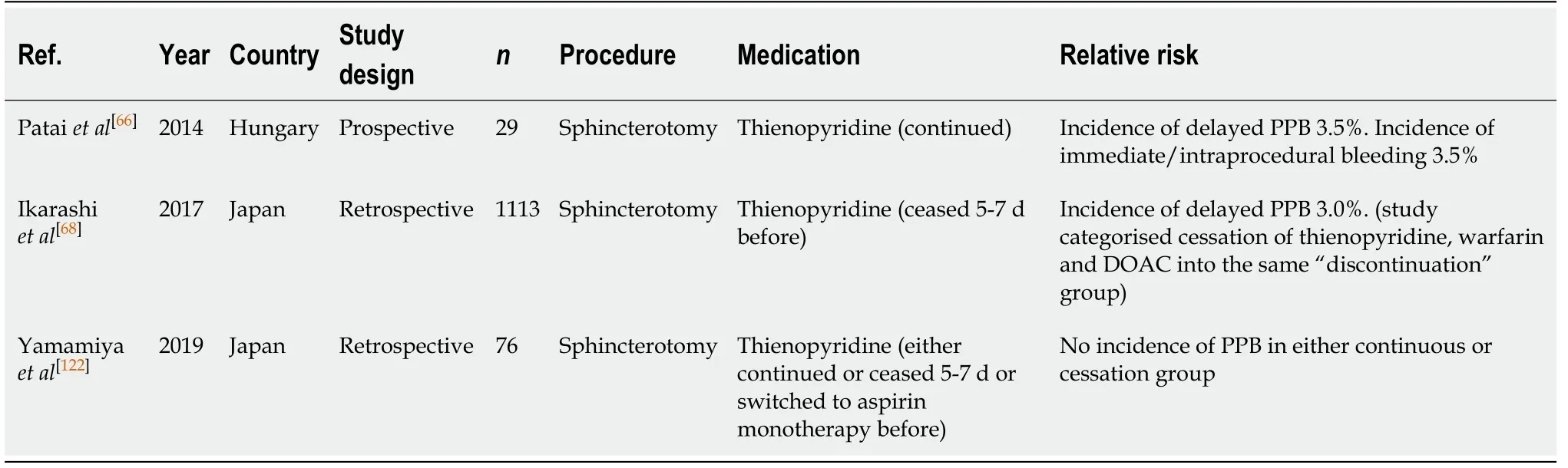
Table 31 Endoscopic retrograde cholangiopancreatography with sphincterotomy
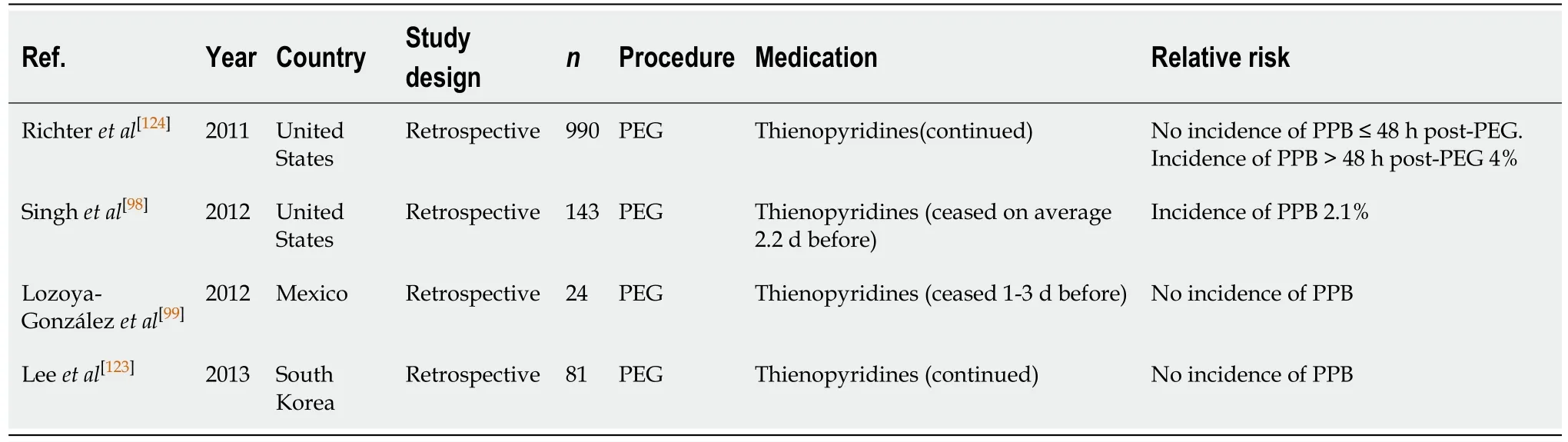
Table 32 Percutaneous endoscopic gastrostomy/percutaneous endoscopic jejunostomy insertion
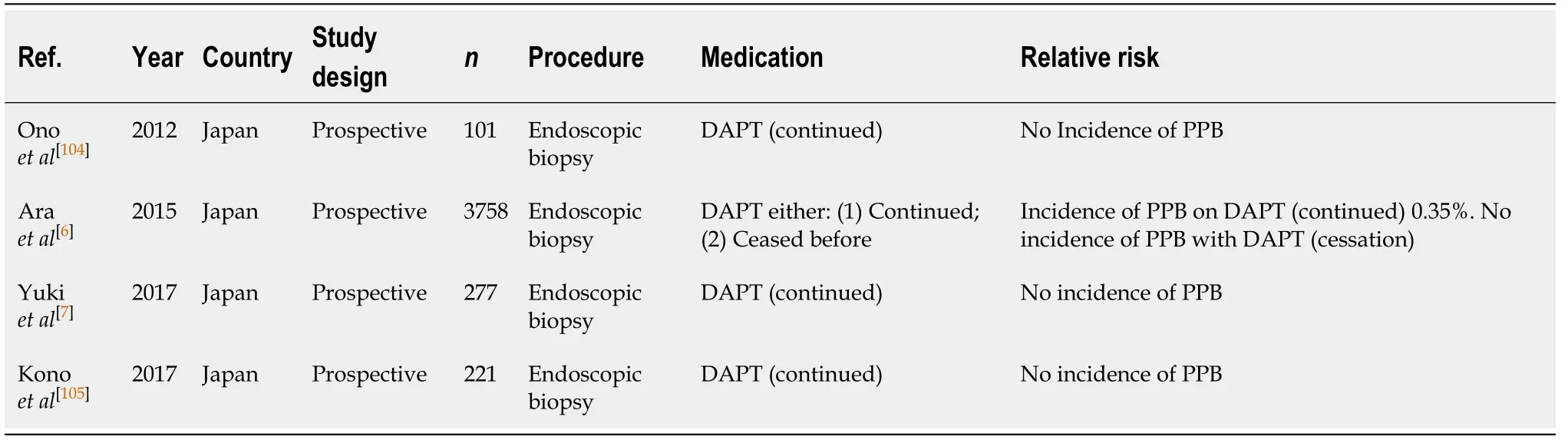
Table 33 Diagnostic endoscopy and colonoscopy with biopsy

Table 34 Endoscopic ultrasound ± fine needle aspiration
The risk of PPB with aspirin use is comparable in EMR of polyps < 10 mm[113], but the absolute risk is significantly increased in larger polyp resections ≥ 20 mm[50,114].Continuation of aspirin monotherapy is thus recommended in EMR (< 20 mm), but should be withheld 7 d before in EMRs (≥ 20 mm). This concurs with previous position statements.
ESD (Table 22)
Continued aspirin monotherapy is associated with a two-fold increased risk of PPB post ESD[58], with numerous published studies reporting the risk of bleeding to be 2.0%-22.6%[56,57,59,115-119]. This is a considerable increased absolute risk of PPB compared to the risk of bleeding in the absence of anticoagulant or antiplatelet use (2.0%-22.6%vs2.7%-6.6%, respectively) (Table 8).
Given the high risk of PPB in ESD, it is recommended aspirin monotherapy should be withheld 7 d before ESD. This concurs with previous position statements.
ERCP with sphincterotomy (Table 23)
Aspirin monotherapy in ERCP with sphincterotomy is associated with an increasedrisk of PPB of 1.8%-10%[66,68,120,121]. Three studies by Pataiet al[66], Ikarashiet al[68], and Ohet al[121]continued aspirin and reported the risk of bleeding in their studies to be 5.8%,1.8%, and 4.7%, respectively. However, the study by Onalet al[120]reported an incidence of PPB of 10.0% when aspirin monotherapy was given within the last 24 h.There were no reported incidences of PPB in the study by Yamamiyaet al[122]in either the continued or withholding aspirin 3-5 d before group.
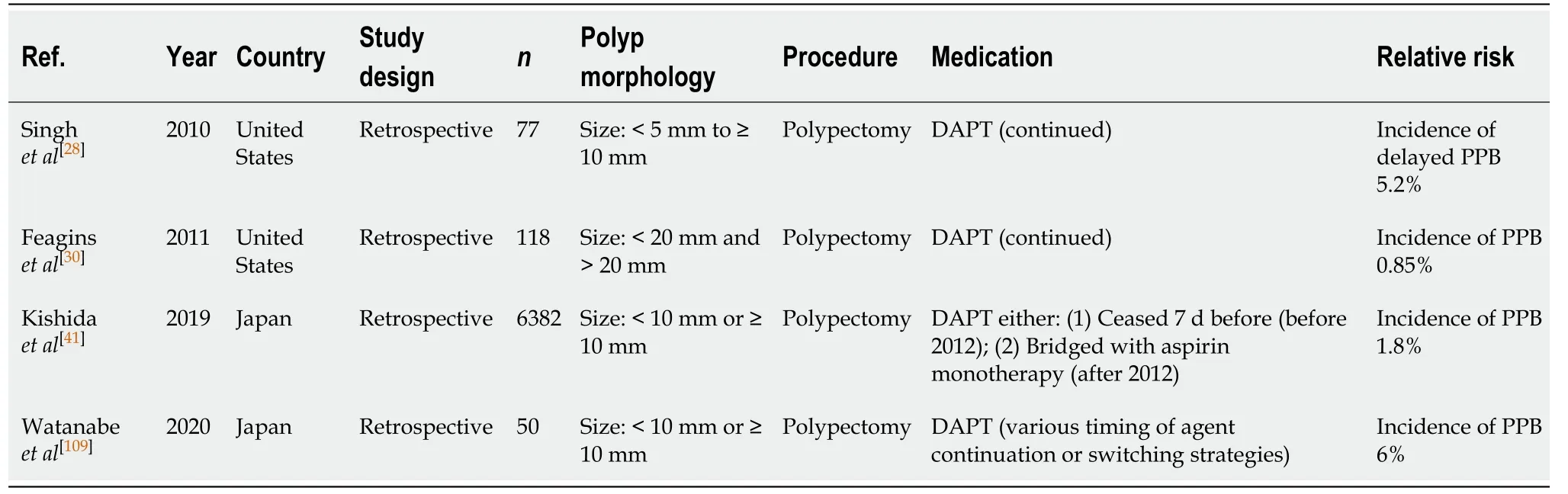
Table 35 Polypectomy

Table 36 Cold snare polypectomy
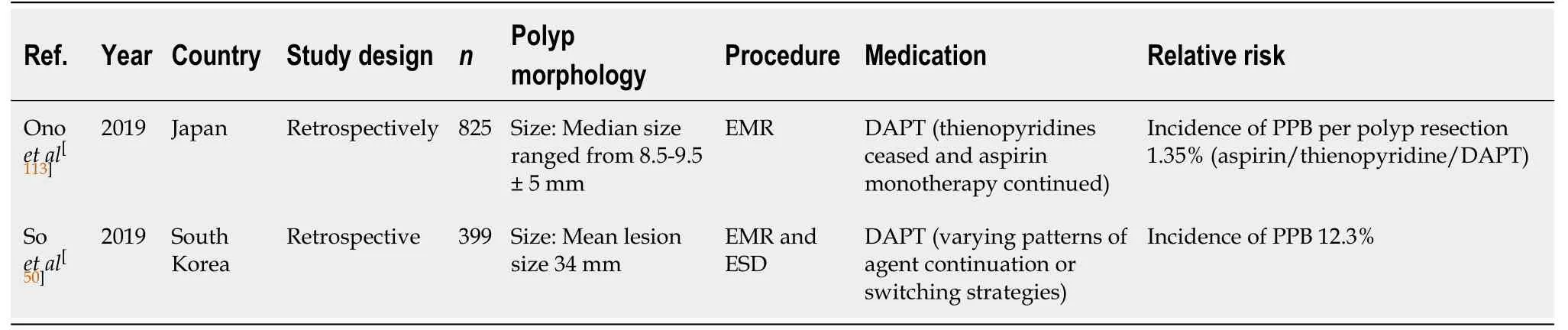
Table 37 Endoscopic mucosal resection
The absolute risk of PPB with continued aspirin use is increased compared to the absolute risk of bleeding in the absence of anticoagulant or antiplatelet use in ERCP with sphincterotomy (1.8%-10%vs0.3%-1.66%, respectively) (Table 9). However, the absolute bleeding risk on continued aspirin is still overall low. Therefore, we recommend continuing aspirin monotherapy in ERCP with sphincterotomy, but caution is advised. This concurs with previous position statements.
PEG/ PEJ insertion (Table 24)
Aspirin use, whether continued or ceased before PEG/PEJ insertions, has not been shown to be associated with an increased risk of PPB. In two retrospective studies[99,123]there were no reported incidences of PPB when aspirin monotherapy was continued.However, two other studies[98,124]observed a bleeding rate of 1.7%-3.9%. The divergent results may be explained in part by case definition, where Singhet al[98]included GI bleeding from any source post PEG insertion (as opposed to bleeding confirmed ascaused by PEG insertion).
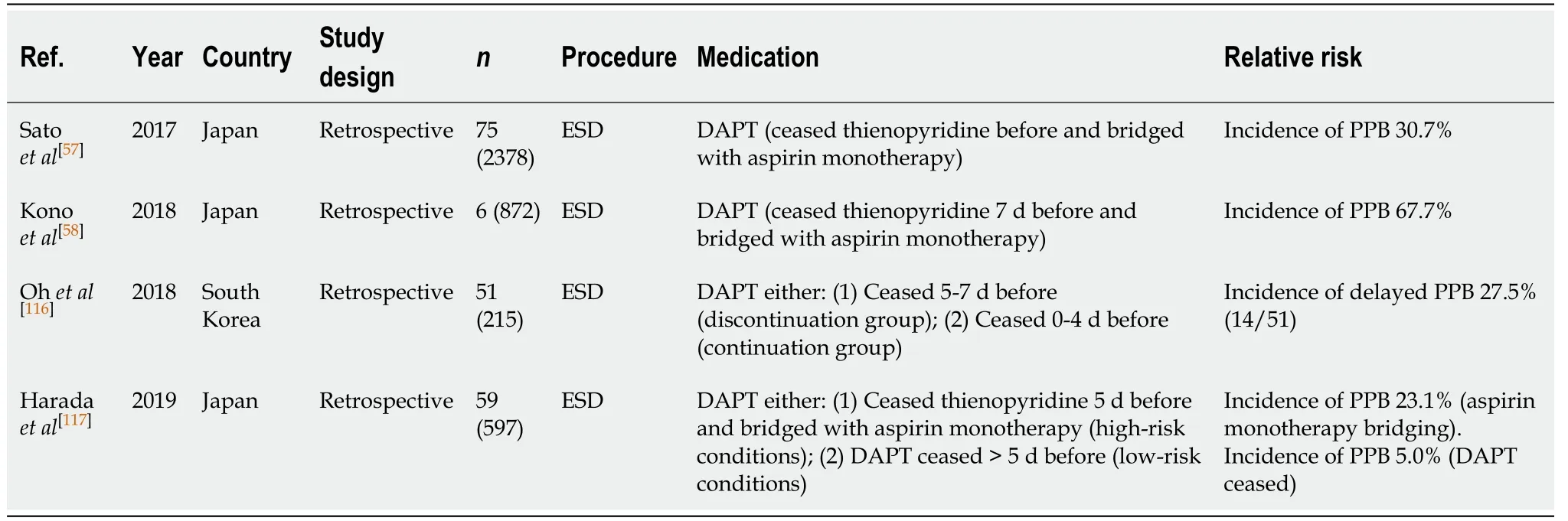
Table 38 Endoscopic submucosal dissection
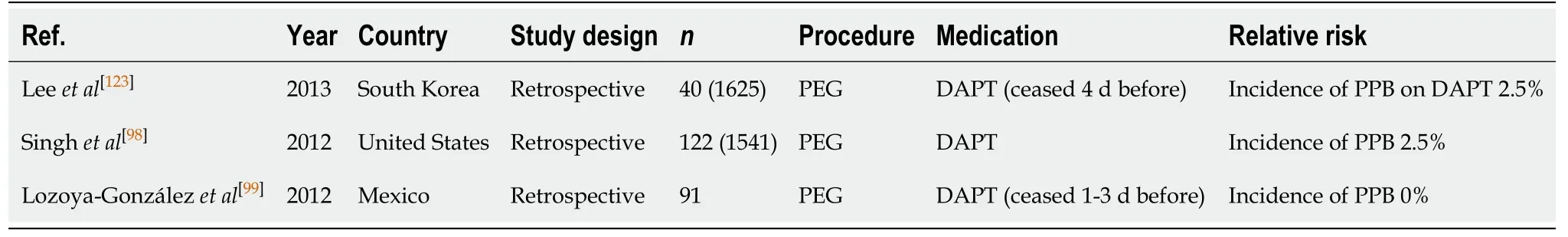
Table 40 Percutaneous endoscopic gastrostomy/percutaneous endoscopic jejunostomy insertion
The absolute risk of PPB post PEG/PEJ insertion on continued aspirin monotherapy is comparable to the overall risk of bleeding in the absence of anticoagulant or antiplatelet use (1.7%-3.9%vs2.7%, respectively) (Table 16). Thus, the overall bleeding risk is considered low and continuation of aspirin monotherapy in all cases is recommended. This concurs with previous position statements.
P2Y12 RECEPTOR ANTAGONIST/THIENOPYRIDINE (CLOPIDOGREL,PRASUGREL, TICAGRELOR) MONOTHERAPY
P2Y12 receptor antagonists includes clopidogrel, ticagrelor and prasugrel. Both clopidogrel and prasugrel are thienopyridines, an active metabolite that irreversibly binds to the P2Y12 receptor and prevents activation of the GPIIb/IIIa receptor, thereby inhibiting platelet aggregation[100]. Platelet aggregation is affected for the life of the platelet. Platelet function returns to baseline 5 to 7 d after withdrawal of clopidogrel.Ticagrelor is a different class of agent that also binds to the P2Y12 receptor but is reversible.
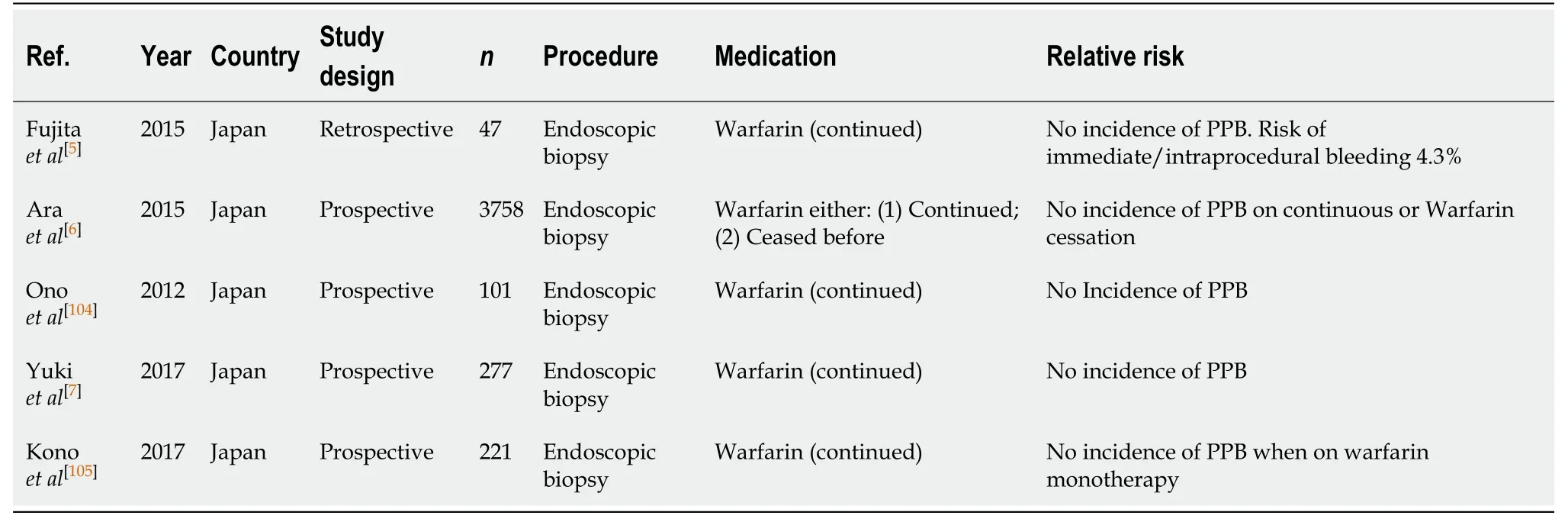
Table 41 Diagnostic endoscopy and colonoscopy with biopsy
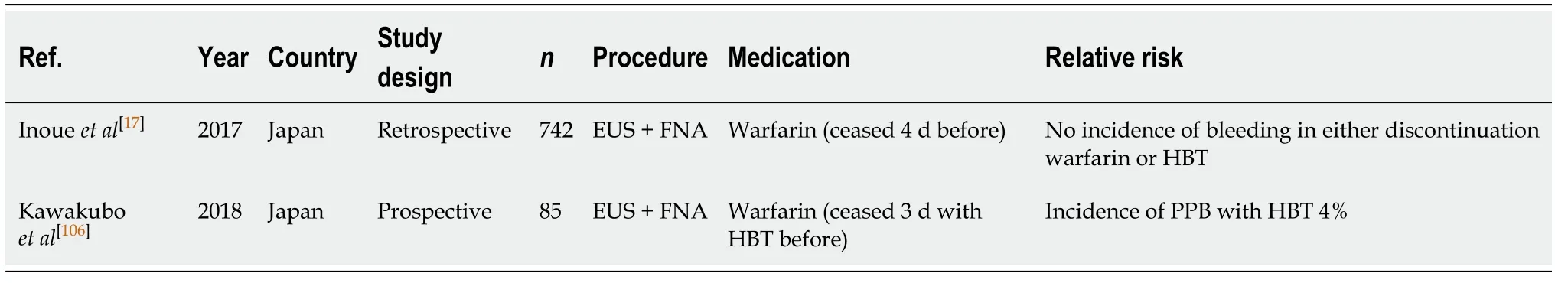
Table 42 Endoscopic ultrasound ± fine needle aspiration
Diagnostic endoscopy and colonoscopy with biopsy (Table 25)
Continued thienopyridine monotherapy is considered safe in diagnostic endoscopies and colonoscopies with biopsy. In several published studies there were no reported incidences of bleeding[5-7,103-105].
Continuing thienopyridine monotherapy is recommended in all cases. This concurs with previous position statements.
EUS ± FNA (Table 26)
Data pertaining to PPB secondary to EUS/FNA in patients where thienopyridine monotherapy is continued is limited. However, two studies from Japan[17,106]assessed the risk of bleeding on thienopyridine monotherapy when withheld 5 d before EUS ±FNA. Both studies did not observe any incidences of PPB. This is compared to a reported absolute risk of PPB between 2.1%-4.3% in the absence of anticoagulant or antiplatelet use (Table 3).
Given the current lack of high-quality evidence assessing the safety of EUS ± FNA on continued thienopyridine monotherapy, and the moderate risk of PPB associated with EUS ± FNA in the absence of anticoagulant or antiplatelet use, withholding thienopyridine 5-7 d before is recommended in all cases. This concurs with previous position statements.
Polypectomy (Table 27)
The risk of PPB attributed with conventional polypectomy while on thienopyridine monotherapy has been considered in numerous comparative studies, where the agent was ceased 5-7 d pre-procedure in the control arm. Four studies[28,107,125,126]assessing the risk of PPB on continued thienopyridine reported PPB in 2.4%-3.8%.
Continued thienopyridine is associated with a significant increased risk of immediate/intraprocedural bleeding. The study by Feaginset al[125]observed an incidence of immediate/intraprocedural bleeding of 7.3%, compared to only 4.7% in their control group. This was a similar finding in a recent RCT by Chanet al[126], which reported the risk of immediate/intraprocedural bleeding to be 8.5% when on continued thienopyridine, compared to only 5.5% in their control group.
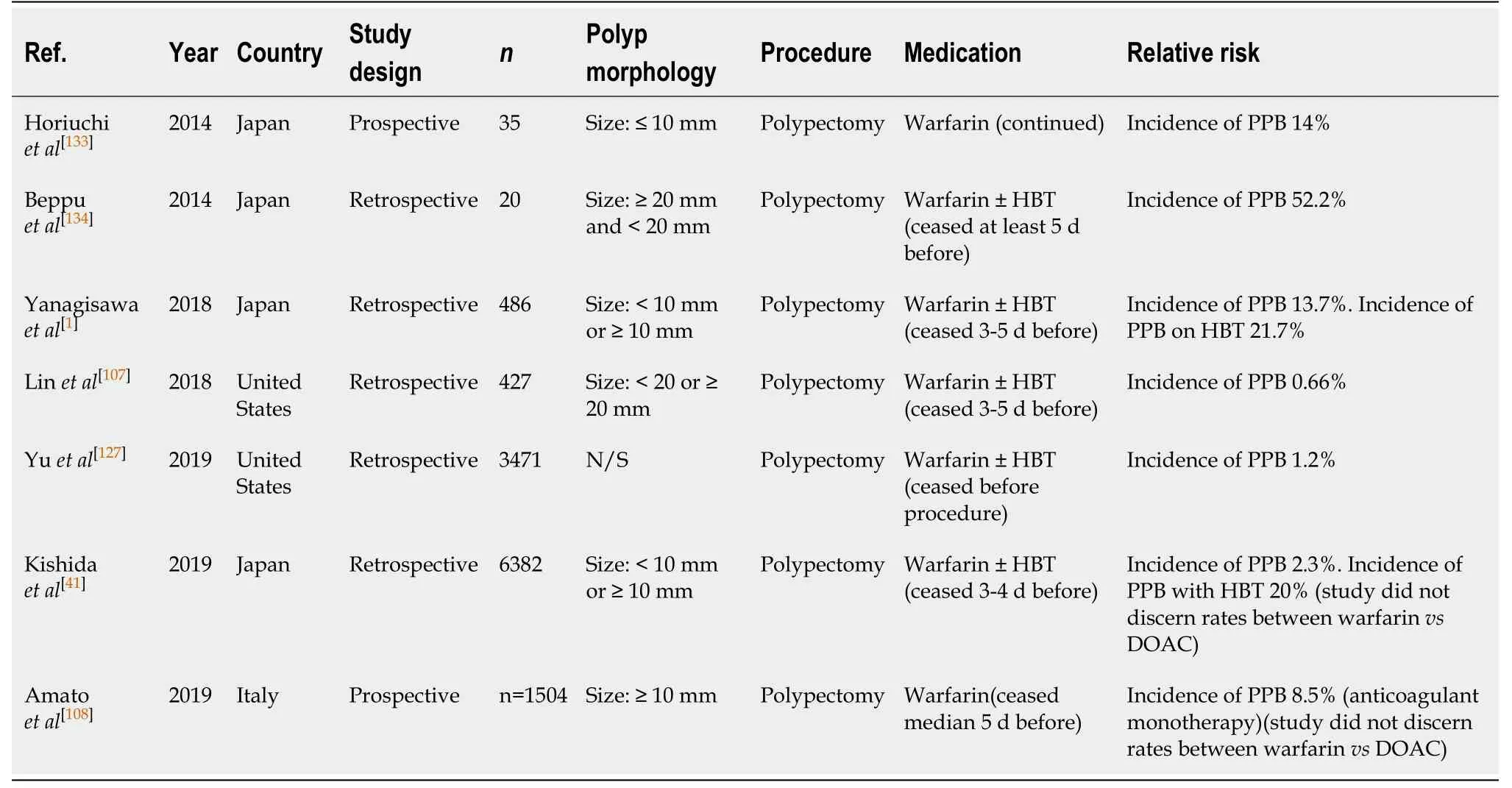
Table 43 Polypectomy
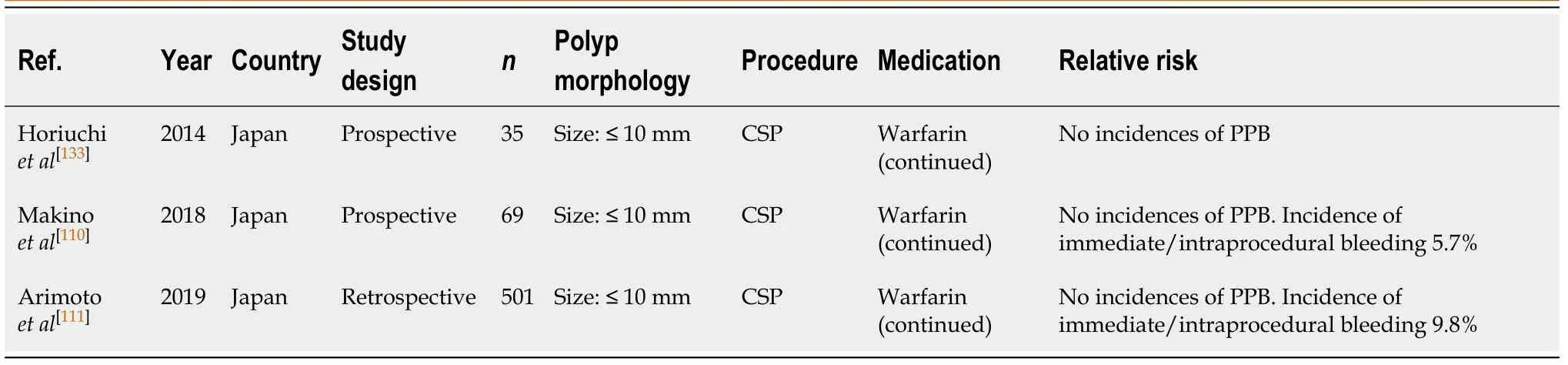
Table 44 Cold snare polypectomy
Five other studies[41,107-109,127]looked at the risk of PPB when thienopyridine was withheld 5-7 d before endoscopic polypectomy. The reported rate of PPB was between 0.6%-6.7%. Although the associated risk of PPB is still higher compared to the risk of bleeding in the absence of anticoagulant or antiplatelet use, this would be considered safer practice than continuing thienopyridine monotherapy.
The absolute risk of PPB while on thienopyridine, either when continued or when withheld 5-7 d before, is slightly increased compared to the rate of bleeding when not on any anticoagulant or antiplatelet agents (0.6%-6.7%vs0.05%-3%, respectively)(Table 6). As highlighted, there is emerging evidence to suggest the risk of delayed PPB is not greatly increased while on continuation thienopyridine monotherapy.However, given the associated high risk of immediate/intraprocedural bleeding,temporary cessation between 5-7 d before is recommended. This concurs with previous position statements.
CSP (Table 28)
There is emerging evidence to suggest that thienopyridine monotherapy may be safely continued in CSP for polyps ≤ 10 mm. Two studies[110,111]reported no incidences of PPB after CSP on continued thienopyridine monotherapy. However, both these studies were small retrospective studies. Larger, RCTs, are still required before this can be safely recommended as standard practice.
Given the current paucity of high-quality evidence, withholding thienopyridine 5-7d before CSP is recommended and concurs with previous position statements.However, with larger studies evaluating the safety of continued thienopyridine monotherapy in CSP, amendments to future position statements may be indicated.
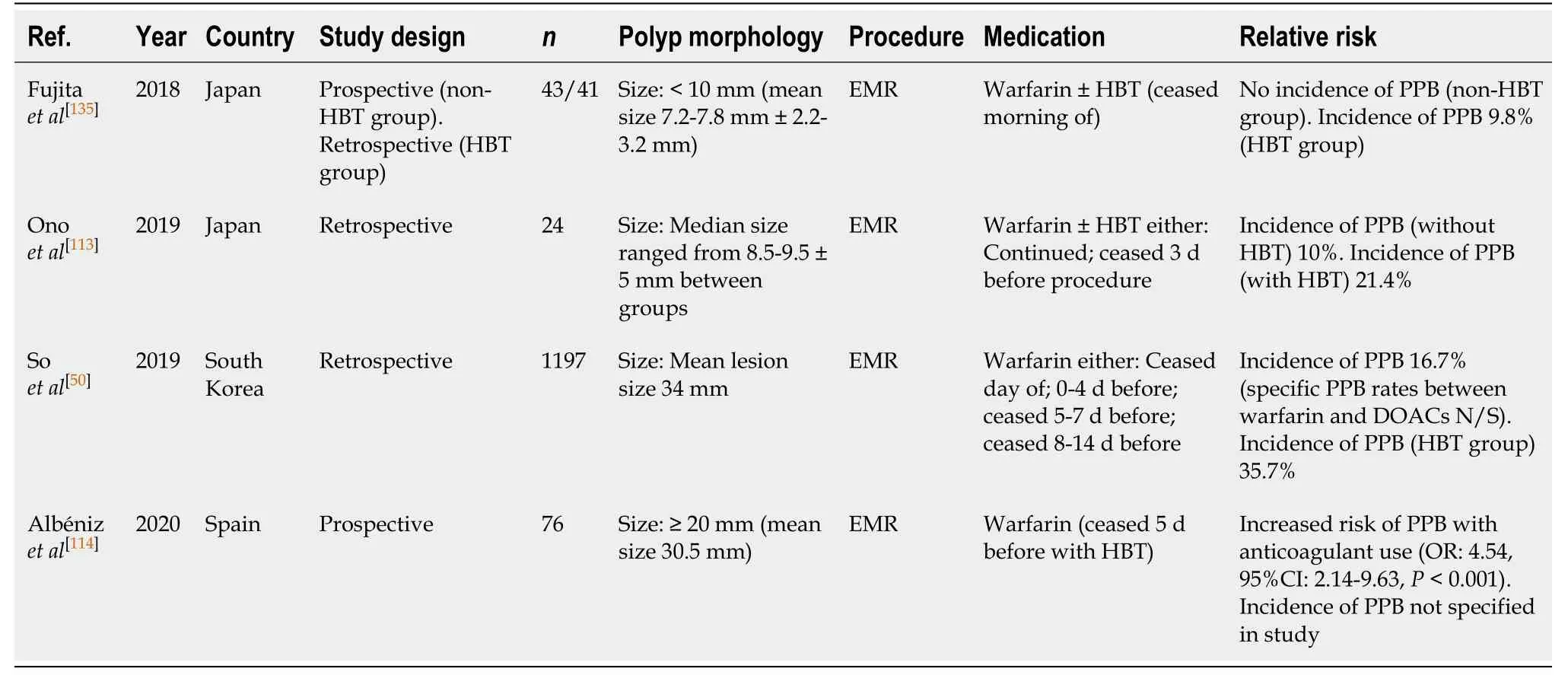
Table 45 Endoscopic mucosal resection
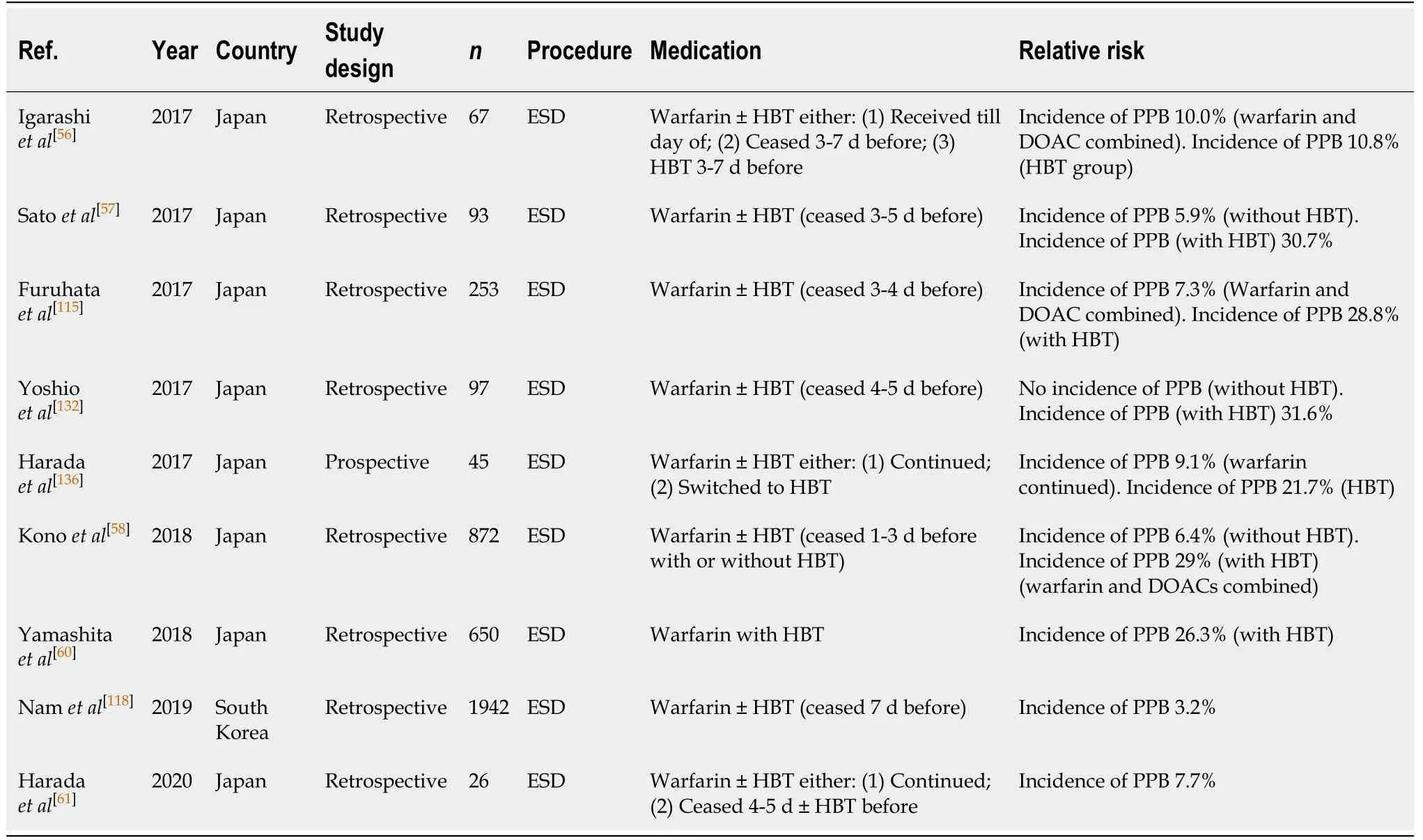
Table 46 Endoscopic submucosal dissection
EMR (Table 29)
The impact of thienopyridine monotherapy and the associated risk of PPB in EMR have not been directly evaluated in published studies. As per with aspirin monotherapy, the same three studies[50,113,114]examined the incidence of PPB associated with both aspirin and thienopyridine monotherapy, generally withheld 3-5 d before, inthe same group (antiplatelet group). Therefore, determining the direct impact of thienopyridine monotherapy can only be estimated.
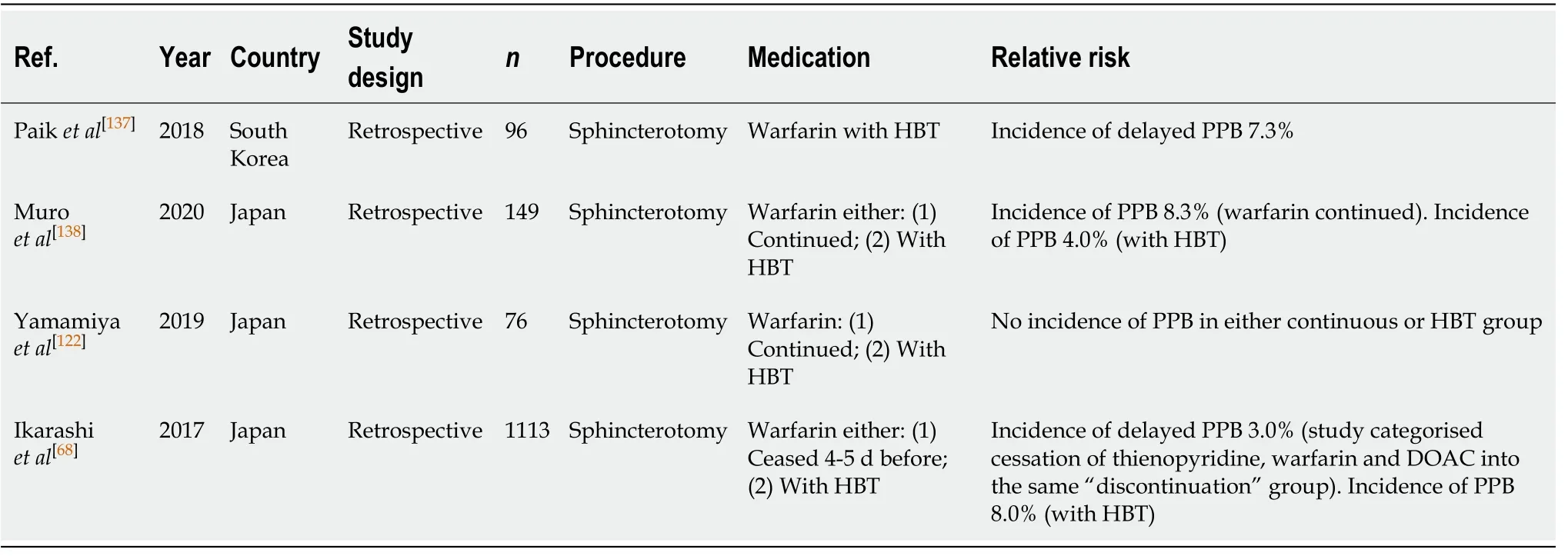
Table 47 Endoscopic retrograde cholangiopancreatography with sphincterotomy
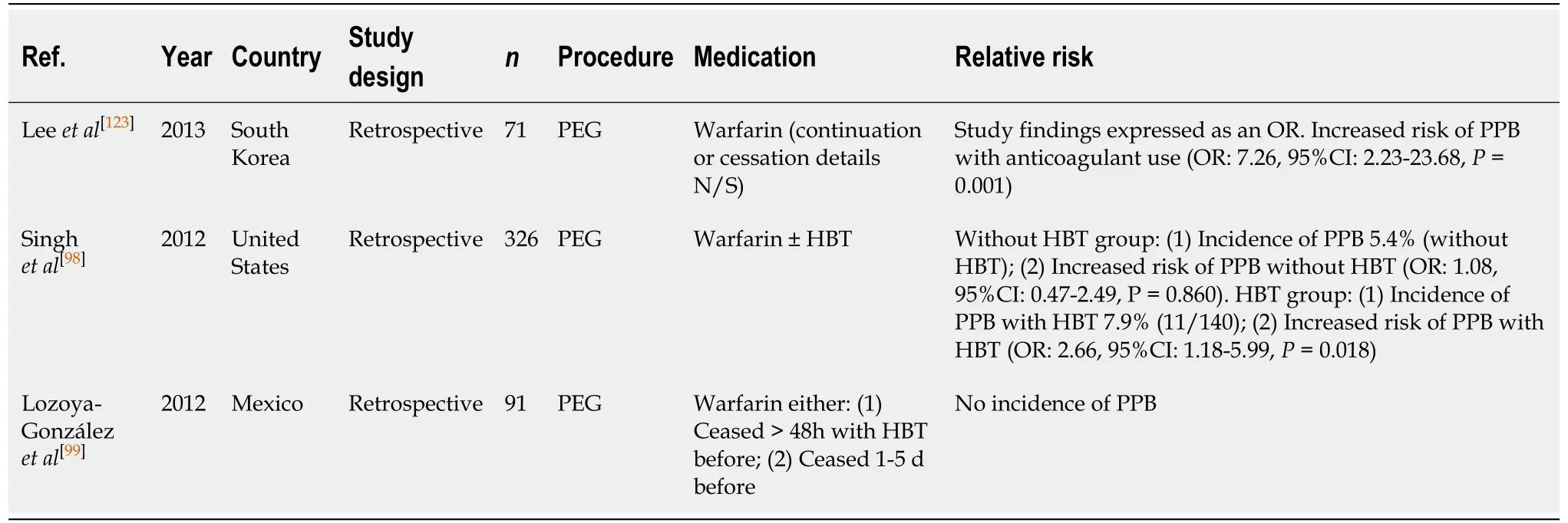
Table 48 Percutaneous endoscopic gastrostomy/percutaneous endoscopic jejunostomy insertion
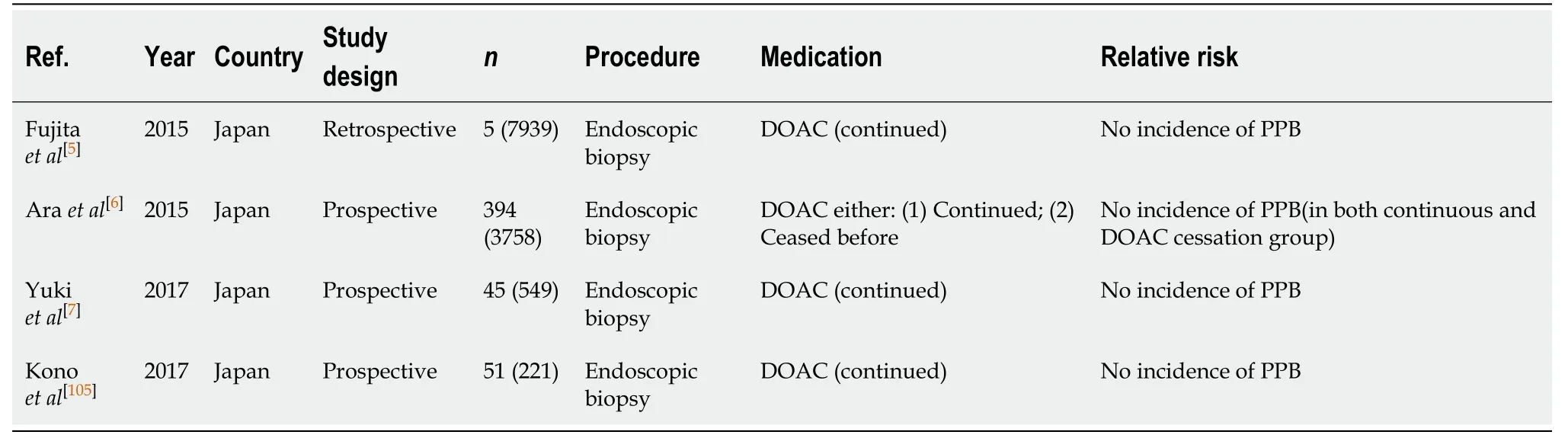
Table 49 Diagnostic endoscopy and colonoscopy with biopsy
Albénizet al[114]found that antiplatelet use with, either aspirin or thienopyridine monotherapy before EMR, is associated with a two-fold increased relative risk of PPB(OR, 2.51; 95%CI, 2.14-9.63,P< 0.001) in lesions ≥ 20 mm. Another study by Soet al[50]observed a rate of PPB of 8.2% in EMR of polyps of mean size > 30 mm when on eitheraspirin or thienopyridine monotherapy.

Table 50 Endoscopic ultrasound ± fine needle aspiration
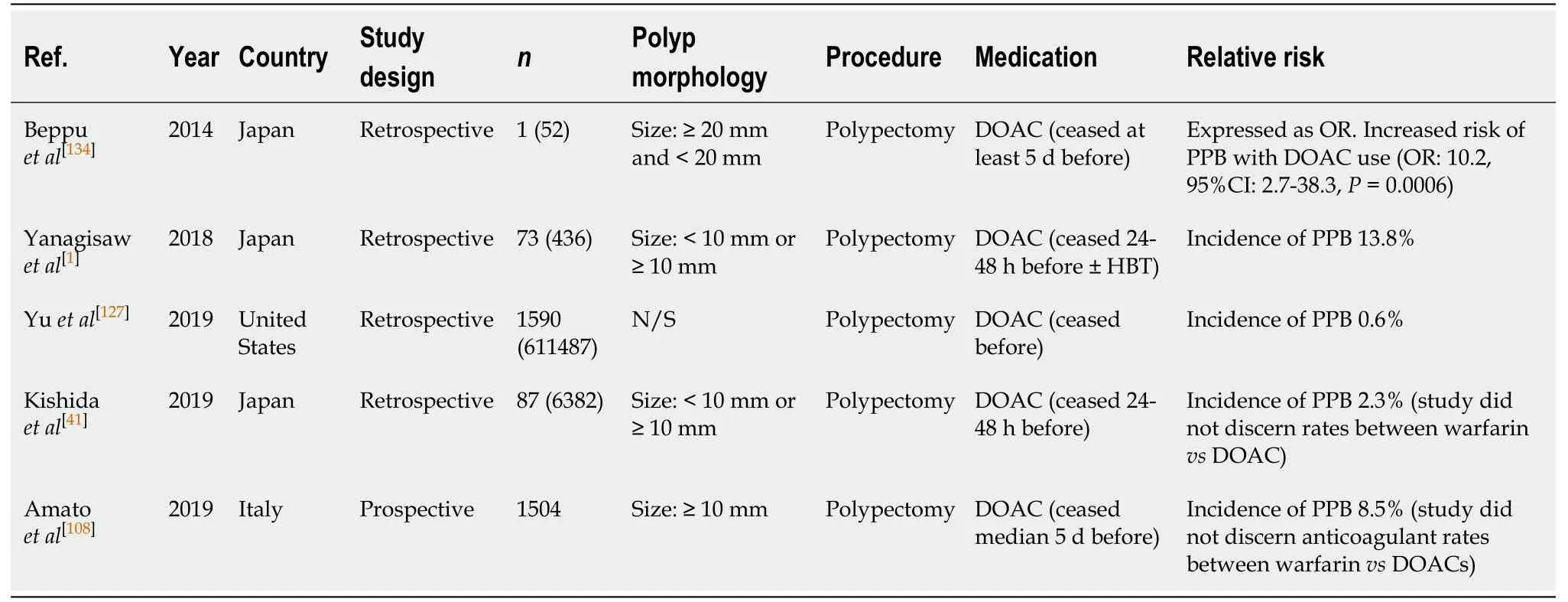
Table 51 Polypectomy

Table 52 Cold snare polypectomy
However, the risk of PPB in EMR for smaller polyps of < 10 mm, although still associated with an increased bleeding risk, is not as high when compared to larger polyp resections (≥ 20 mm). The study by Onoet al[113]reported a 1.35% risk of PPB per polyp resection when on either aspirin or thienopyridine monotherapy.
Overall, the absolute risk of PPB is increased with thienopyridine use, particularly in lesions ≥ 20 mm in size, compared to the risk of bleeding in the absence of anticoagulant or antiplatelet use of respective size (1.35%-8.2%vs1.7%-6.3%,respectively) (Table 7).
Given the increased absolute risk of PPB associated with thienopyridine use,withholding thienopyridine monotherapy 5-7 d before is recommended in all cases.This concurs with previous position statements.
ESD (Table 30)
Thienopyridine monotherapy is associated with a four-fold increased relative risk of PPB (OR, 4.26, 95%CI, 1.36-13.29,P= 0.13)[116]in ESD, with a reported incidence of 3.6%-19.4%[56,57,116,118]even when withheld 5-7 d before.
It is apparent that withholding thienopyridine monotherapy for an extended period of time is required to decrease PPB risk. A study by Ohet al[116]compared the risk of bleeding when thienopyridines were withheld at either 0-4 d or 5-7 d before EMR. The two patients in the study who developed PPB (3.6%) both had their thienopyridine ceased on the day of the EMR procedure.
Another study by Igarashiet al[56]also assessed the risk of PPB when thienopyridinewas withheld on the day of the procedure and found the risk of bleeding to be 5.6%.
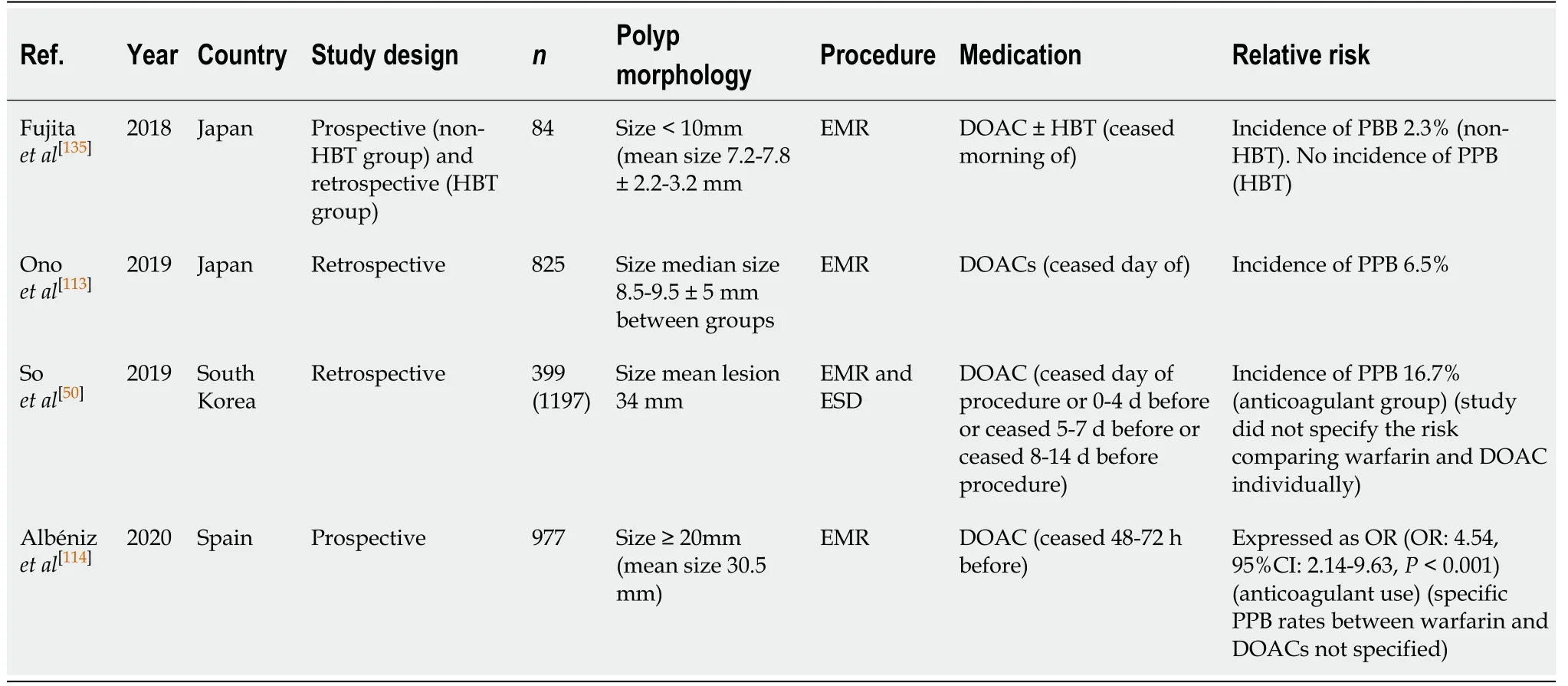
Table 53 Endoscopic mucosal resection
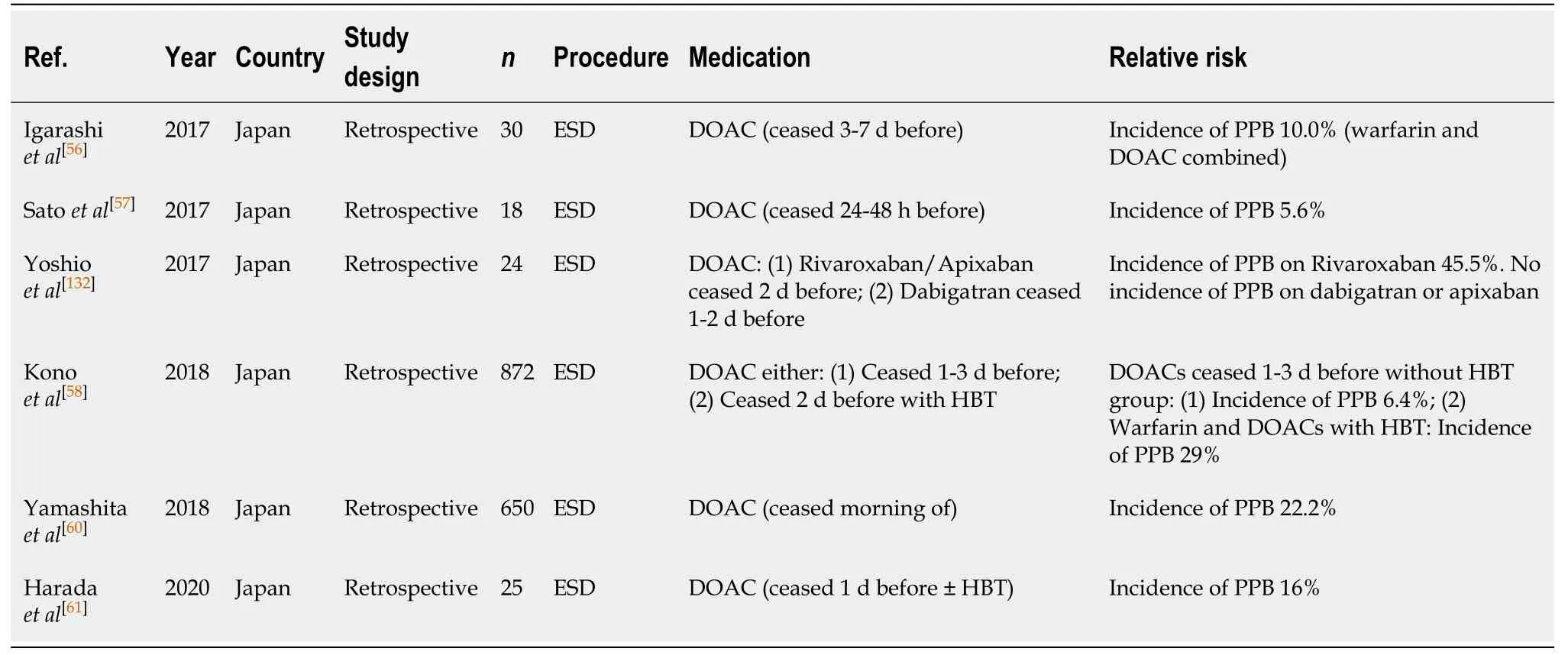
Table 54 Endoscopic submucosal dissection
Onoet al[128]observed the risk of PPB in patients on dual antiplatelet therapy (DAPT)undergoing an ESD, where aspirin was ceased but thienopyridine monotherapy continued. The observed rate of PPB reported was 20%.
The absolute risk of PPB in ESD is high irrespective of whether thienopyridine monotherapy is continued or withheld 5-7 d before the procedure and when compared to the PPB risk in the absence of anticoagulant or antiplatelet use (5.6%-20%vs2.7%-6.6%, respectively) (Table 8). In all circumstances, thienopyridine monotherapy should not be continued and withheld 5-7 d before. This concurs with previous position statements.
ERCP with sphincterotomy (Table 31)
There are currently limited studies evaluating the risk of PPB associated with thienopyridine monotherapy use in ERCP with sphincterotomy. One study by Pataiet al[66]assessed the risk of bleeding on continued thienopyridine and found the incidence of immediate/intraprocedural and delayed PPB to both be at 3.5%.
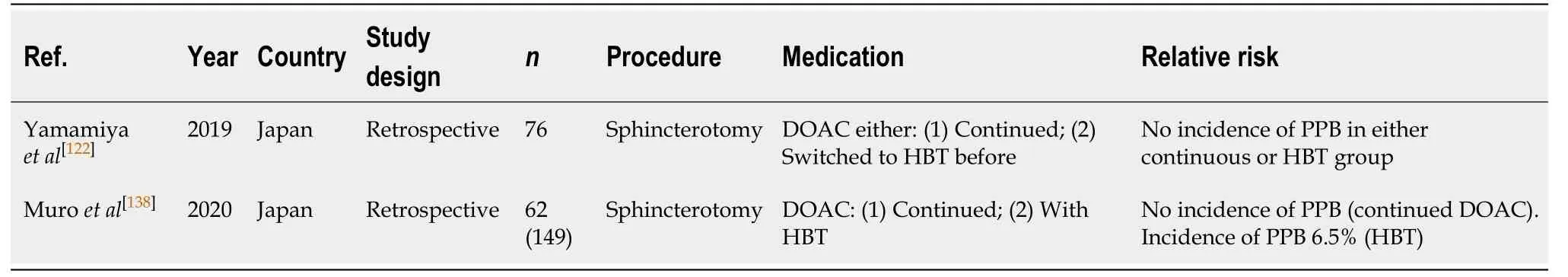
Table 55 Endoscopic retrograde cholangiopancreatography with sphincterotomy

Table 56 Percutaneous endoscopic gastrostomy/percutaneous endoscopic jejunostomy insertion
However, when thienopyridine is withheld 5-7 d before ERCP with sphincterotomy,the risk of bleeding is lower and found to be only 3.0% in one study by Ikarashiet al[68]This study was limited by analysing the risk of bleeding associated with thienopyridine, warfarin and DOAC use together. It did not directly analyse the risk thienopyridine has on PPB alone. Another study by Yamamiyaet al[122]did not observe any incidence of PPB in their study in patients on thienopyridine.
There is an increased absolute risk of PPB with thienopyridine use, when withheld 5-7 d before, compared to in the absence of anticoagulant or antiplatelet use (0%-3%vs0.3%-1.66%, respectively) (Table 9).
Given the increased absolute risk and current limited evidence of the safety on continuation thienopyridine and risk of bleeding post ERCP, it is recommended that thienopyridines should be withheld 5-7 d before the procedure. This concurs with previous position statements.
PEG/PEJ insertion (Table 32)
The estimated risk of PPB post endoscopic PEG/PEJ insertion associated with thienopyridine monotherapy, when withheld 1-3 d before, is reported to be 0%-2.1% in several published studies[99].
The study by Richteret al[124]evaluated the associated risk of PPB when thienopyridine monotherapy was continued. It reported a bleeding rate of 4%.
The absolute risk of PPB with thienopyridine use, when continued or withheld 1-3 d before, is increased when compared with the risk of bleeding in patients in the absence of anticoagulant or antiplatelet use (2.1%-4%vs2.7%, respectively) (Table 16).
Given the increased absolute risk of PPB when thienopyridine monotherapy is continued, it is recommended that thienopyridine should be withheld 5-7 d before PEG/PEJ insertion. This concurs with previous position statements.
DUAL ANTIPLATELET THERAPY (DAPT) (ASPIRIN + P2Y12 RECEPTOR ANTAGONIST/THIENOPYRIDINE)
DAPT of aspirin plus a P2Y12 receptor antagonist (thienopyridine) is most commonly indicated for the management of ACS. In percutaneous coronary intervention (PCI),such as drug eluding stent (DES) or bare metal stent (BMS) insertion, indication to remain on DAPT for a given period is paramount in order to prevent stent thrombosis.The current Cardiac Society of Australia and New Zealand (CSANZ) guidelines[129]on DAPT duration post PCI, recommends patients should remain on DAPT for 12 mo.Risk of stent thrombosis increases after 5 d without antiplatelet therapy with an approximate risk of 40% for MI and death[2]. There is emerging evidence that prolonged therapy of up to 3 years for patients with prior MI demonstrates a relative reduction in cardiovascular death (RR: 0.85, 95%CI: 0.74-0.98), and recurrent MI (RR:0.70, 95%CI: 0.55-0.88). However, there is an associated increase incidence of bleeding events (RR: 1.73, 95%CI: 1.19-2.50) with no improvement in non-cardiovascular death or overall mortality[129]. In patients with a high bleeding risk and low risk for recurrent ischaemic events, a shorter duration of treatment such as 6 mo could be considered,but not ideal. The minimum duration of uninterrupted DAPT should be at least 30 d for BMS, and 3 mo for DES.
Diagnostic endoscopy and colonoscopy with biopsy (Table 33)
Continued DAPT in diagnostic endoscopies and colonoscopies with biopsy has an overall low risk of bleeding. Three studies[7,104,105]reported no incidences of PPB post biopsy. While the study by Araet al[6]only reported one episode of bleeding post biopsy on continued DAPT (0.35%). The absolute risk on continued DAPT is comparable to the reported risk of PPB in the absence of anticoagulant or antiplatelet use (0.35%vs0.12%-0.98%) (Table 1).
Overall, DAPT is considered safe and is recommended to be continued in all cases.This concurs with previous position statements.
EUS ± FNA (Table 34)
There is currently a scarcity of evidence evaluating the risk of PPB in patients on DAPT undergoing EUS ± FNA. Although a study by Kawakuboet al[106]reported of risk of PPB of 3.6%, when thienopyridine was withheld 5 d before and bridged with aspirin monotherapy, in patients initially on DAPT. This is comparable to the absolute risk of PPB of 2.1%-4.3% in the absence of anticoagulant or antiplatelet use (Table 3).
Given the limited evidence regarding the safety of continued DAPT in EUS± FNA, it is recommended that thienopyridine should be withheld 5-7 d before with bridging aspirin monotherapy (unless contraindicated). If thienopyridine cannot be safely withheld due to contraindications, in the example of a recent PCI insertion within 12 mo, then the procedure should be postponed until it is safe to do so, if possible. This concurs with previous position statements.
Polypectomy (Table 35)
The risk of PPB is reportedly significantly increased in patients on continued DAPT undertaking endoscopic polypectomy. A study by Singhet al[28]reported a three-fold increased relative risk of PPB when DAPT is continued (OR: 3.69; 95%CI, 1.60-8.52,P=0.002), with the incidence rate of PPB on continuation DAPT between 0.85%-6%, as reported in several published studies[28,30,41,109].
The study by Kishidaet al[41]considered the risk of bleeding when either, both aspirin and thienopyridine were withheld (before 2012), or only thienopyridine withheld and bridged with aspirin monotherapy. In this study, the incidence of PPB was reported to be 1.8%.
The absolute risk of PPB post polypectomy when thienopyridine is withheld and bridged with aspirin monotherapy is comparable to the overall risk of PPB in the absence of anticoagulant or antiplatelet use (1.8%vs0.05%-3.0%, respectively)(Table 5).
Given the high risk of bleeding complications on continued DAPT, it is recommended that thienopyridine is withheld 5-7 d before and bridged with aspirin monotherapy (unless contraindicated). If thienopyridine cannot be safely withheld due to contraindications, in the example of a recent PCI insertion within 12 mo, then the procedure should be postponed until it is safe to do so, if possible. This concurs with previous position statements.
CSP (Table 36)
In CSP, there is emerging evidence to suggest the risk of bleeding on continued DAPT is overall low and estimated to be around 2.4% in a recent RCT by Wonet al[112].However, this study was limited by a small sample size of 91 patients. Thus, larger RCTs are still required before this can be safely recommended as standard practice.
In a retrospective study by Arimotoet al[111], they reported no incidences of PPB in their DAPT group. Despite this, uninterrupted DAPT appears to be associated with a significant increased risk of immediate/intraprocedural bleeding between 4.8%-17.8%[111,112]. This is significantly higher compared to the reported rates of immediate/intraprocedural bleeding in the absence of anticoagulant or antiplatelet use (2.4%-9.1%, Table 6).
Given the current paucity in high-quality evidence and significant increased risk of immediate/intraprocedural bleeding, withholding thienopyridine 5-7 d before and bridging with aspirin monotherapy is recommended in CSP (unless contraindicated).If thienopyridine cannot be safely withheld due to contraindications, in the example of a recent PCI insertion within 12 mo, then the procedure should be postponed until it is safe to do so, if possible. This concurs with previous position statements.
EMR (Table 37)
Two recent studies[50,113]retrospectively assessed the indirect effects of DAPT use, when thienopyridine was withheld and bridged with aspirin monotherapy before EMR. The study by Makinoet al[110]observed a risk of PPB per polyp resection of 1.35% when on antiplatelet therapy (monotherapy or DAPT). However, this study was limited by not quantifying the exact risk of PPB on DAPT alone.
Another study by Soet al[50]found DAPT use was associated with a two-fold increased relative risk of bleeding (OR: 2.14; 95%CI, 0.63-7.32,P= 0.226) in lesions ≥ 20 mm, with a reported incidence of PPB of 12.3% post EMR.
The relative and absolute risk of PPB with DAPT is higher compared to the risk of bleeding in the absence of anticoagulant or antiplatelet use (1.35%-12.3%vs1.7%-6.3%,respectively) (Table 7).
The risk of PPB associated with DAPT use in EMR is considerably high and precautions should be made to reduce this risk. In lesions < 20 mm, withholding thienopyridine 5-7 d before and bridging with aspirin monotherapy is recommended(unless contraindicated). In lesions ≥ 20 mm withholding both thienopyridine and aspirin is the safest recommendation with regards to bleeding risk.
If thienopyridine cannot be safely withheld due to contraindications, in the example of a recent PCI insertion within 12 mo, then the procedure should be postponed until it is safe to do so, if possible. This concurs with previous position statements.
ESD (Table 38)
The absolute risk of PPB in ESD in the absence of anticoagulant or antiplatelet use is high (2.7%-6.6%, Table 8). DAPT use before ESD is associated with a reported two- to three-fold increased relative risk of bleeding in two studies[116,117], even after withholding thienopyridine 5-7 d before and bridged with aspirin monotherapy only.The study by Satoet al[57]found that DAPT use was a significant independent risk factor for PPB than what was reported in the two other studies (OR: 10.33, 95%CI, 6.06-17.59,P< 0.001).
Several studies have reported the absolute risk of bleeding post ESD to be 23.1%-67.7%[57,58,116,117]. In the study by Haradaet al[117]they compared the risk of bleeding with bridging aspirin monotherapyvsdiscontinuation of both thienopyridine and aspirin >5 d before the procedure. The reported incidence of PPB in this study was 23.1% and 5.0%, respectively.
Continuing DAPT in ESD is not recommended given the significant increased risk of PPB. Withholding both thienopyridine and aspirin is the safest recommendation with regards to bleeding risk. However, if this cannot be undertaken due to risk of thromboembolism, then withholding thienopyridine 5-7 d before procedure and switching to bridging aspirin monotherapy is otherwise recommended (unless contraindicated). If thienopyridine cannot be safely withheld due to contraindications,in the example of a recent PCI insertion within 12 mo, then the procedure should be postponed until it is safe to do so, if possible. This concurs with previous position statements.
ERCP with sphincterotomy (Table 39)
There have been limited published studies assessing the risk of bleeding with DAPT in ERCP with sphincterotomy. Two studies by Moket al[130]and Yamamiyaet al[122]analysed the incidence of bleeding when DAPT was continued and reported an absolute risk of PPB of 0%-3.6%. This compares to an overall risk of PPB of 0.45%-9.9%in the absence of anticoagulant or antiplatelet use (Table 9).
These two studies may suggest that continued DAPT in ERCP with sphincterotomy may be safe. However, evidence is limited due to a lack of large, high-quality studies.For now, it is recommended that thienopyridine is withheld 5-7 d before and bridged with aspirin monotherapy only (unless contraindicated). If thienopyridine cannot be safely withheld due to contraindications, in the example of a recent PCI insertion within 12 mo, then the procedure should be postponed until it is safe to do so, if possible. This concurs with previous position statements.
PEG/PEJ insertion (Table 40)
Several studies have found DAPT use to be associated with a 2.5% absolute risk of PPB post PEG/PEJ insertion[98,123]. The study by Leeet al[123]ceased DAPT at least 4 d(range 4-10 d) before the PEG procedure. Whereas, the study by Singhet al[98]did not clearly specify the DAPT management regime. In the study by Lozoya-Gonzálezet al[99]there were no reported incidences of PPB in any of their patients on DAPT, which was ceased 1-3 d before the PEG procedure. The absolute risk of PPB while on DAPT is comparable to the overall risk of PPB in the absence of anticoagulant or antiplatelet use (2.5%vs2.7%, respectively) (Table 16).
Given current studies have only evaluated the risk of bleeding when DAPT is ceased before a PEG procedure, and yielded similar rates of PPB compared to in the absence of anticoagulant or antiplatelet use, it is recommended that thienopyridine is withheld 5-7 d before and bridged with aspirin monotherapy only (unless contraindicated). If thienopyridine cannot be safely withheld due to contraindications,in the example of a recent PCI insertion within 12 mo, then the procedure should be postponed until it is safe to do so, if possible. This concurs with previous position statements.
VITAMIN K ANTAGONIST (WARFARIN)
Warfarin is a vitamin K antagonist, which inhibits the synthesis of vitamin Kdependent clotting factors (II, VII, IX, X) and the antithrombotic factors protein C and S[100]. The duration of action of warfarin is 5 d. Current evidence supports the shifting trend that DOACs are more efficacious and safer than warfarin[131]. Furthermore,warfarin needs to be withheld for a longer period and generally HBT is required,further increasing the risk of PPB and the length of hospital stay[132].
Despite the rise in DOAC use, warfarin is still commonly encountered in certain conditions such as mechanical heart valve prosthesis, AF with mitral stenosis, and CKD patients where DOACs are contraindicated. Thus, its management in periendoscopic period is still very relevant.
Diagnostic endoscopy and colonoscopy with biopsy (Table 41)
Continuation of Warfarin therapy in diagnostic endoscopies and colonoscopies with biopsy is considered safe and overall is not associated with an increased risk of gastrointestinal bleeding. Four prospective and one retrospective study did not report any incidences of PPB on continued warfarin monotherapy[6,7,104,105].
The study by Konoet al[105]observed PPB in one case on continued warfarin.However, this patient was also on an antiplatelet agent and thus, had an increased overall risk of bleeding. In this case, endoscopic haemostasis was required with good clinical outcome.
Overall, continuing warfarin therapy is considered safe in diagnostic endoscopies and colonoscopies with biopsy in all cases. This concurs with previous position statements.
EUS ± FNA (Table 42)
Withholding warfarin at least 4 d before EUS ± FNA without HBT does not appear to increase the risk of PPB compared to the absolute risk of bleeding in the absence of anticoagulant or antiplatelet use (0%-4%vs2.1%-4.3%, respectively) (Table 3).
The study by Inoueet al[17]found no incidences of PPB in their cohort of patients who had warfarin ceased 4 d before EUS ± FNA. However, HBT was found to be associated with an increased risk of bleeding, without reducing the risk of thromboembolic event relating to warfarin interruption, in the study by Kawakuboet al[106]. In this study, there was one case (4%) of PPB in a patient on HBT after EUS ±FNA and none in the warfarin cessation without HBT group. No thromboembolic events occurred in either the warfarin cessation or HBT group.
We recommend withholding warfarin 5 d before EUS ± FNA based on current evidence available. HBT is associated with increased risk of bleeding and should be considered carefully in patients. Our recommendation of avoiding HBT in patients who are at high-risk of thromboembolic event differs from previous position statements.
Polypectomy (Table 43)
Warfarin use is associated with a high-risk of PPB in endoscopic polypectomy,irrespective of whether warfarin is withheld with or without HBT before the procedure. The study by Horiuchiet al[133]reported a 14% risk of PPB with continued warfarin use. However, when warfarin is withheld 3-5 d before the procedure, the absolute risk of bleeding is reported to be 0.7%-13.5%, according to several studies[1,41,107,108,127].
HBT is indicated in patients with high-thromboembolic risk patients as per current guidelines[2-4]. However, HBT has been shown to be associated with higher risk of bleeding without significantly reducing the risk of a thromboembolic event. A study by Yanagisawaet al[1]compared the risk of PPB and thromboembolic event in its analysis and found withholding warfarin with HBT, compared to withholding warfarin without HBT, yielded a higher rate of PPB (21.7%vs13.7%, respectively)without providing significant difference in the prevention of a thromboembolic event.Two cases of a thromboembolic event were reported in this study. However, this occurred in both groups, one in the HBT group and the other in the withholding warfarin without HBT.
Another study by Linet al[107]also associated HBT with a ten-fold increased relative risk of PPB in their cohort (OR: 10.3,P= 0.0001), with the incidence of bleeding on HBT reported at 14.9% compared to only 0.7% in the warfarin discontinuation without HBT. Similarly, there was no difference in the rate of thromboembolic event in both groups. No thromboembolic events occurred in the study.
Warfarin use is associated with an absolute increased risk of bleeding in endoscopic polypectomies irrespective of whether warfarin is withheld or not. The risk of bleeding while on warfarin, even when withheld 3-5 d before polypectomy, compared to the risk of bleeding in the absence of anticoagulant or antiplatelet use is significantly increased (0.7%-13.5%vs0.05%-3.0%, respectively) (Table 5). The studies also suggest that HBT is associated with a significantly increased risk of PPB, without reducing the risk of thromboembolic event in high-risk patients.
To minimise the risk of PPB, it is recommended that warfarin be withheld 5 d before the procedure. HBT is associated with an increased risk of bleeding and should be considered carefully in patients. Our recommendation of avoiding HBT in patients who are at high-risk of thromboembolic event differs from previous position statements.
CSP (Table 44)
There is emerging evidence that continuing warfarin therapy in CSP for polyps ≤ 10 mm does not increase the risk of PPB. It is theorised the reason for bleeding after polypectomy is due to submucosal vessel damage from electrocautery. CSP does not involve electrocautery and therefore, may decrease the risk of bleeding[133].
Three recent studies looking at the bleeding risk without warfarin cessation uniformly reported no incidences of PPB[110,111,133]. However, there is an associated increased risk of immediate/intraprocedural bleeding when on continued warfarin of 5.7%-9.8%[111,133].
Given the current lack of high-quality evidence evaluating the safety with continuing warfarin in CSP, withholding warfarin 5 d before should still be practiced.This concurs with previous position statements. However, with larger studies evaluating the safety of continued warfarin therapy in CSP being currently undertaken, amendments to future position statements may be needed.
EMR (Table 45)
Warfarin use in EMR is associated with over a four-fold increased relative risk of bleeding (OR: 4.54, 95%CI, 2.14-9.63,P< 0.001)[114]. The rate of PPB on warfarin therapy when ceased at least 3-5 d before EMR is between 10%-16.7%, as reported in two retrospective studies[50,113]. This represents an increased absolute risk of bleeding on warfarin therapy compared to the risk of bleeding in the absence of anticoagulant or antiplatelet use (10%-16.7%vs0%-1.7%, respectively) (Table 7).
This risk of bleeding is further increased with concurrent HBT use. HBT is considered to be a significant risk factor for PPB (OR: 5.00, 95%CI, 1.11-22.50,P=0.036)[50]. From several small studies, the overall risk of PPB is significantly increased when on HBT in EMR, reported to be 9.8%-35.7%[50,113,134,135].
To minimise the risk of PPB, it is recommended that warfarin be withheld 5 d before EMRs. HBT is associated with increased risk of bleeding and should be considered carefully in patients. Our recommendation of avoiding HBT in patients who are at high-risk of thromboembolic event differs from previous position statements.
ESD (Table 46)
The risk of PPB in warfarin usersin ESD is reported to be 3.2%-10.0% when withheld 3-5 d before the procedure[56-58,115,118]. This is similar to the absolute risk of PPB in the absence of anticoagulant or antiplatelet use (3.2%-10%vs2.7%-6.6%, respectively)(Table 8). HBT continues to be a significant independent risk factor for PPB with a four- to ten-fold increased relative risk of bleeding as estimated in some studies[57,115,132], and a reported incidence of PPB of 10.8%-31.6%[56,57,115,132,136].
Continuing warfarin, as an alternative to HBT, was assessed in two studies[61,136]and was found to have similar risk of PPB compared to when warfarin is withheld 3-5 d before the procedure (7.7%-9.1%vs3.2%-10.0%, respectively). It has been suggested that continuation of warfarin may be a safer alternative to HBT in patients of high-risk of thromboembolism. However, further larger studies are required before this can be safely recommended.
To minimise the risk of PPB, it is recommended that warfarin be withheld 5 d before ESD. HBT is associated with increased risk of bleeding and should be considered carefully in patients. Our recommendation of avoiding HBT in patients who are at high-risk of thromboembolic event differs from previous position statements.
ERCP with sphincterotomy (Table 47)
Warfarin is associated with a high risk of PPB in ERCP with sphincterotomy. Three studies analysing the incidence of PPB while withholding warfarin with HBT reported a bleeding rate of 4.0%-8.0%[68,137,138]. The study by Muroet al[138]reported the risk of bleeding on continued warfarin was slightly higher at 8.3%. This compares to an overall risk of PPB of 0.45%-9.9% in the absence of anticoagulant or antiplatelet use(Table 9).
Continuing warfarin and/or withholding warfarin with HBT are associated with an overall high-risk of PPB in ERCP with sphincterotomy. To minimise the risk of PPB, it is recommended that warfarin be discontinued 5 d before ERCP with sphincterotomy.HBT is associated with increased risk of bleeding and should be considered carefully in patients. Our recommendation of avoiding HBT in patients who are at high-risk of thromboembolic event differs from previous position statements.
PEG/PEJ insertion (Table 48)
Use of warfarin in PEG/PEJ insertion is a significant independent risk factor for PPB(OR: 7.26, 95%CI, 2.23-23.68,P= 0.001)[123]. The study by Singhet al[98]reported an incidence of PPB of 5.4% in the group who had warfarin withheld without HBT. The absolute risk increases to 7.9% with HBT. However, the study by Lozoya-Gonzálezet al[99]reported no incidences of PPB in either group.
Warfarin is a well-established risk factor for bleeding in PEG/PEJ insertion compared to the absolute risk of PPB in the absence of anticoagulant or antiplatelet use(5.4%-7.9%vs2.7%, respectively) (Table 16).
To minimise the risk of PPB, it is recommended that warfarin be withheld 5 d before the procedure. HBT is associated with increased risk of bleeding and should be considered carefully in patients. Our recommendation of avoiding HBT in patients who are at high-risk of thromboembolic event differs from previous position statements.
DIRECT ORAL ANTICOAGULANTS (DOAC) (DABIGATRAN,RIVAROXABAN AND APIXABAN)
DOAC is a collective term for direct thrombin inhibitors (dabigatran) and other direct factor Xa inhibitors (rivaroxaban and apixaban)[139-141]. DOACs offer an alternative to warfarin in the management of patients with AF and VTE. More recently, DOACs have replaced warfarin as the preferred first line therapy of choice. This is due to its noninferiority at low doses (dabigatran 110 mg BD, rivaroxaban 20 mg daily, apixaban 2.5 mg BD), but superiority at higher doses (dabigatran 150 mg BD, apixaban 5 mg BD), over warfarin in prevention of stroke and thromboembolic events, without increasing the risk of major bleeding in patients with nonvalvular AF[139-141]. DOACs also have other significant logistical benefits over warfarin. Unlike warfarin, DOACs have set doses which do not require regular monitoring with international normalisation ratio (INR) blood tests. Due to its shorter half-lives, DOACs also have a faster onset and offset of action compared to warfarin. However, both dabigatran at high dose (150 mg BD) and rivaroxaban are associated with higher rates of gastrointestinal bleeds compared to warfarin[139,140], and reversibility currently remains a significant safety concern with DOACs. Only dabigatran currently has an available antidote in idarucizumab. This is expected to change with ongoing trials and emerging evidence of antidotes for the other DOACs.
Optimal timing of DOAC cessation should take into consideration the time of maximum effect, half-life and the excretion of the agent. To minimise the risk of PPB,DOACs should be stopped for at least 2 half-lives in all high-risk endoscopic procedures[3]. Both rivaroxaban and apixaban have a relatively short time to maximum effect (2-4 h for rivaroxaban and 1-3 h for apixaban). Rivaroxaban has a half-life between 8-9 h [creatinine clearance (CrCl) > 50 mL/min] and 9-13 h (CrCl > 30-50 mL/min), with 66% of the agent excreted by the kidneys. Whereas apixaban has a halflife between 7-8 h (CrCl > 50 mL/min) and 8-15 h (CrCl 30-50 mL/min), with 25%excreted by the kidneys. Dabigatran was the first DOAC and has a time of maximum effect of 1.25-3 h and its half-life is between 12-14 h (CrCl ≥ 80 mL/min) to 22-35 h(CrCl < 30 mL/min). More cautious peri-endoscopic management is required for dabigatran as the timing of discontinuation is mostly dictated by the patient’s CrCl with 80% of the agent excreted by the kidneys[3].
Diagnostic endoscopy and colonoscopy with biopsy (Table 49)
There has been no documented increased risk of PPB in diagnostic endoscopies and colonoscopies with biopsy on continued DOAC therapy from several published studies. Four studies all observed no incidences of bleeding post biopsy in their continuation DOAC group[5-7,105]. This is compared to an already established low risk of PPB in the absence of anticoagulant or antiplatelet use (0.12%-0.98%, Table 1).
DOACs are considered safe to be continued in diagnostic endoscopies and colonoscopies with biopsy. This concurs with previous position statements.
EUS ± FNA (Table 50)
There is currently a paucity of large studies analysing the risk of bleeding while on DOAC therapy in EUS ± FNA. Only one study by Kawakuboet al[106]analysed the PPB risk when DOAC therapy was withheld 48 h before the procedure with HBT. There were no reported incidences of bleeding in this study. The absolute risk of PPB in EUS± FNA is reported to be 2.1%-4.3% in the absence of anticoagulant or antiplatelet use(Table 3).
Given the absolute risk of bleeding in the absence of anticoagulant or antiplatelet use is considerable and with currently only limited evidence of the bleeding risk with DOAC use, it is recommended that DOACs should be withheld at least 48 h before.This concurs with previous position statements.
Polypectomy (Table 51)
DOAC use in polypectomy is associated with a significant increased relative risk of PPB (OR: 17.8,P< 0.001) as reported in the study by Yanagisawet al[1]. In this study,the incidence of bleeding in their DOAC group, when DOAC therapy is withheld 24-48 h before the procedure, was 13.8%. The rates of bleeding were similar amongst the different DOAC classes, of dabigatran, rivaroxaban and apixaban, with reported rates of 11.1%, 13.2% and 13.3%, respectively. Another study by Beppuet al[134]also observed DOAC use was associated with a ten-fold increased relative risk of bleeding (OR: 10.2,95%CI, 2.7-38.3,P= 0.0006).
Several other studies that withheld DOAC therapy 24-48 h before the procedure(median 5 d in one study[108]), reported an overall incidence of bleeding of 0.6%-13.8%[1,41,108,127]. However, both the study by Kishidaet al[41]and Amatoet al[108]analysed the risk of bleeding when on either DOAC or warfarin therapy together, and not as separate agents. This limits the accuracy of the direct effect DOAC therapy has on the risk of bleeding. However regardless, it can be interpreted that DOACs are associated with a significant increased risk.
DOAC use represents a significant increased absolute risk of bleeding compared to the risk of bleeding in the absence of anticoagulant or antiplatelet use (0.6%-13.8%vs0.05%-3.0%, respectively) (Table 5). It is recommended that DOAC therapy should be withheld at least 24-48 h (72 h for dabigatran; in CrCl >50) before polypectomy to minimise the risk of bleeding. This concurs with previous position statements.
CSP (Table 52)
Similar with warfarin, there is emerging evidence from small studies that suggest continuation of DOAC therapy in CSP of polyps ≤ 10 mm is considered safe and does not significantly increase the risk of bleeding[110,111]. This is due to the hypothesis that there is minimal damage to the submucosal vessel in CSP because electrocautery is not involved[133].
The study by Makinoet al[110]only observed two cases of bleeding post CSP (1.2%).One patient was on dabigatran and the other patient was on apixaban. In the study by Arimotoet al[111]there were no reported incidences of PPB. However, this study did report complications of immediate/intraprocedural bleeding in 11.9% of cases. All cases were adequately controlled with endoscopic haemostasis and did not require further intervention with blood transfusion, admission, and/or surgery.
Although there is emerging evidence suggesting continuation DOAC therapy may be safe in CSP of polyps ≤ 10 mm, until larger studies evaluating the safety of continued DOAC therapy in CSP is undertaken, it is recommended that DOAC therapy should be withheld at least 24-48 h (72 h for dabigatran; in CrCl > 50) before CSP to minimise the risk of bleeding. This concurs with previous position statements.
EMR (Table 53)
Most published studies analysing the risk of PPB in EMR in DOAC users have done so by grouping both warfarin and DOAC monotherapy use together under the umbrella term of “anticoagulant.” The risk of bleeding in EMR while on anticoagulant therapy(either warfarin or DOAC) is reported between 5.5%-16.7%[50,113].
However, the risk of bleeding with DOAC use may be overall lower compared to warfarin therapy. In the study by Onoet al[113], the risk of bleeding when DOAC has been withheld one day before EMR was reported to be 6.5% per polyp. While another study by Fujitaet al[135]observed an incidence of 2.3% of PPB in their DOAC group when ceased the morning of EMR.
There is currently limited evidence analysing the risk of bleeding on continued DOAC therapy in EMR. Given this paucity of evidence and to minimise the risk of PPB, it is recommended that DOAC therapy should be withheld at least 24-48 h (72 h for dabigatran; in CrCl > 50) before EMR. This concurs with previous position statements.
ESD (Table 54)
ESD in patients on a DOAC, withheld at least > 24 h before, is reported to be associated with an increased relative risk of PPB compared to the bleeding risk in the absence of anticoagulant or antiplatelet use, in multiple publications[56-58,60,61,132]. The absolute risk of bleeding is, 5.6%-45.5%vs2.7%-6.6%, respectively (Table 8). There have been no studies reporting the rate of PPB on continued DOAC therapy.
The study by Yoshioet al[132]reported PPB in five cases on DOAC therapy (45.5%).All five cases were in patients on rivaroxaban. There were no observed cases of PPB in the dabigatran or apixaban group.
HBT is generally not recommended when withholding DOAC therapy, however the study by Konoet al[58]analysed the risk of bleeding with HBT during both DOAC and warfarin interruption and observed an incidence of PPB in 29% of cases.
Given the high risk of PPB in ESD procedure associated with DOAC therapy, it is recommended that DOACs should be withheld at least 24-48 h (72 h for dabigatran; in CrCl > 50) without HBT in order to minimise the risk of bleeding. This concurs with previous position statements.
ERCP with sphincterotomy (Table 55)
Two recent small retrospective studies analysing the risk of bleeding when on continued DOAC therapy in ERCP with sphincterotomy reported no incidences of PPB in their studies[122,138]. The risk of bleeding when DOAC therapy was withheld with HBT was also compared in the study by Muroet al[138]and found that HBT was a significant risk factor for bleeding. The incidence of PPB in this study was reported in 6.5% of cases. This absolute risk of bleeding when DOAC therapy is withheld compares similarly to the overall risk of bleeding in the absence of anticoagulant or antiplatelet use (6.5%vs0.45%-9.9%, respectively) (Table 9).
These two small studies may suggest that continued DOAC in ERCP with sphincterotomy may be safe. However, until larger RCTs adequately evaluate the risk of bleeding, it is still recommended that DOACs be withheld at least 24-48 h (72 h for dabigatran; in CrCl > 50) without HBT before ERCP with sphincterotomy to minimise the risk of bleeding. This concurs with previous position statements.
PEG/PEJ insertion (Table 56)
Limited data is available that considers the risk of PPB in PEG/PEJ insertion while on DOAC therapy. One study by Leeet al[123]evaluated the risk of bleeding when on either warfarin or DOAC monotherapy. It observed a seven-fold increased relative risk of PPB associated with warfarin or DOAC use (OR: 7.26, 95%CI, 2.23-23.68,P= 0.001).However, this study was limited by not specifying the bleeding risk directly related to DOAC therapy use, nor did it specify whether DOAC therapy was continued or withheld before the procedure.
Given the limited data and significant increased risk of PPB associated with anticoagulant use, it is recommended that DOACs should be withheld at least 24-48 h(72 h for dabigatran; in CrCl > 50) without HBT. This concurs with previous position statements.
DISCUSSION
The current position statements and guidelines from the major gastroenterology societies have provided endoscopists with evidenced-based systematic approaches to pre, peri and post-operative management of patients on anticoagulant and antiplatelet agents in the context of both low and high-risk endoscopic procedures. While there has been sufficient evidence on the index risk of bleeding in common endoscopic procedures in the absence of anticoagulant and/or antiplatelet use, the evidence surrounding bleeding risk while on anticoagulant and/or antiplatelet agents is still evolving.
It is well established that anticoagulant and antiplatelet therapy is associated with an increased risk of PPB in endoscopic procedures. The reported risk will vary depending on endoscopic procedure and the study in which the data was published,but overall, the rate is similar over various publications and has been emphasised in this review. This variability may be explained by the different approaches taken by each study, the patient and geographical demographics, and the technical competency of the proceduralists.
There is no doubt temporary interruption of anticoagulant and antiplatelet therapy,compared to continuation therapy, reduces the risk of PPB in endoscopic procedures.However, this needs to be carefully considered against the risk of thromboembolic event and the potential serious irreversible consequences that comes with anticoagulant and antiplatelet interruption. Careful timing of anticoagulant and antiplatelet interruption to minimise the risk of PPB, while avoiding unnecessary increased risk of thromboembolic event, is of utmost importance. The aim of this review is to provide an evidence-based framework for safe clinical application of anticoagulant and antiplatelet management in the context of both low and high-risk endoscopic procedures for all endoscopists, as outlined in Figures 1 and 2.
This article has reviewed and considered the last 10 years of originally published literature and has found the evidence largely agrees with the current position statements and guidelines from the major gastroenterology societies in anticoagulant and antiplatelet agent management in endoscopic procedures. However, as highlighted earlier, there is emerging evidence that calls attention to some discrepancies in the current recommendations.
For example, current position statements and guidelines[2-4]advise warfarin should be bridged with HBT in all patients with high risk of thromboembolic event undergoing high-risk endoscopic procedures. Peri-endoscopic management with HBT is now becoming a controversial management decision with regards to its efficacy and safety. Numerous studies highlighted in this review have demonstrated that the use of HBT is associated with a two- to three-fold increased risk of PPB[7,41,142], while being non-superior in thromboembolic event prevention, compared to warfarin cessation without HBT[1,107,143,144]. This heightened risk of PPB associated with HBT has been shown in a range of endoscopic procedures, including EMR, ESD, polypectomy, EUS ±FNA and ERCP with sphincterotomy. However, this is still emerging evidence and further larger studies directly looking at the safety of HBT compared to warfarin cessation without HBT, specifically evaluating the risk of PPB and the efficacy in thromboembolic prevention, is still very much needed. We currently recommend that HBT use should be considered carefully in all patients undergoing an endoscopic procedure despite current guidelines from major gastroenterology societies still advising for HBT in patients undergoing high-risk endoscopic procedures.
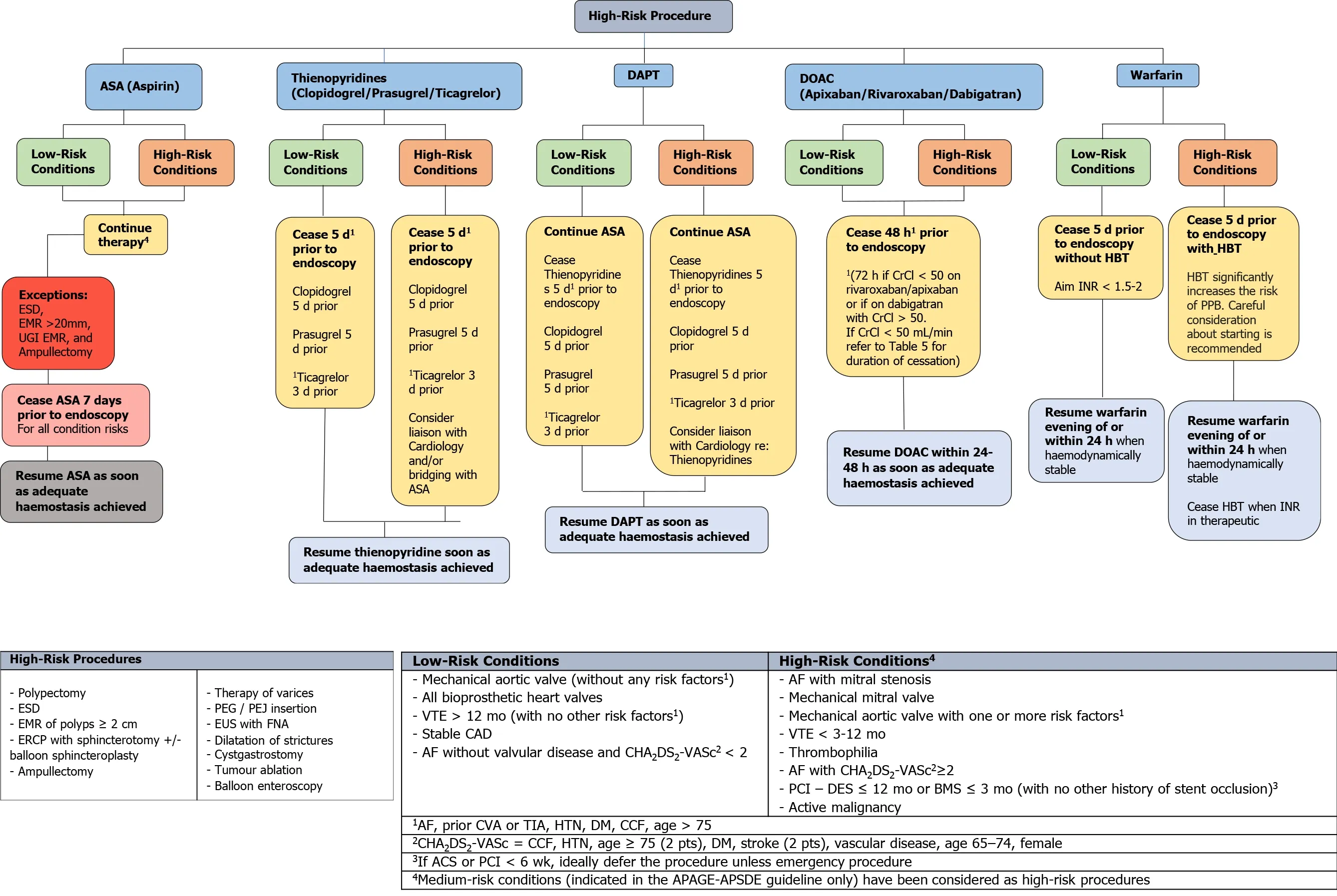
Figure 1 An evidence-based framework for safe clinical application of anticoagulant and antiplatelet management in the context of highrisk endoscopic procedures for all endoscopists. ASA: Acetylsalicylic acid; DAPT: Dual antiplatelet therapy; DOAC: Direct oral anticoagulant; ESD:Endoscopic submucosal dissection; EMR: Endoscopic mucosal resection; UGI: Upper Gastrointestinal; CrCl: Creatinine clearance; HBT: Heparin bridging therapy;INR: International normalisation ratio; PPB: Post-procedural bleeding; ERCP: Endoscopic retrograde cholangiopancreatography; PEG: Percutaneous endoscopic gastrostomy; PEJ: Percutaneous endoscopic jejunostomy; EUS: Endoscopic ultrasound; FNA: Fine needle aspiration; VTE: Venous thromboembolism; CAD:Coronary artery disease; AF: Atrial fibrillation; PCI: Percutaneous coronary intervention; DES: Drug eluding stent; BMS: Bare metal stent; CVA: Cerebrovascular accident; TIA: Transient ischaemic attack; HTN: Hypertension; DM: Diabetes mellitus; CCF: Congestive cardiac failure; ACS: Acute coronary syndrome.
In addition, current position statements and guidelines[2-4]considers CSP for polyps< 10 mm as a high-risk procedure and advises anticoagulant and antiplatelet therapy be ceased before the procedure. However, the risk of PPB on continued antiplatelet therapy of aspirin or thienopyridine (either as monotherapy or DAPT) in CSP for polyps < 10 mm has been reported to be overall low in small retrospective studies[111,113]. Even on continuation DAPT, the risk of PPB is only estimated to be around 2.4% as reported in a small RCT by Wonet al[112]. Therefore, continuing antiplatelet therapy in CSP for polyps < 10 mm may be possible in some circumstances. There is also no significantly increased risk of PPB shown when anticoagulant therapy (DOAC or warfarin) is continued in CSP for polyps < 10 mm[110,111,133]. However, this is still emerging evidence and has only been captured in a few retrospective studies and one small RCT. Further larger studies directly looking at the safety of continuation therapy is still needed. Furthermore, although the risk of PPB is not significantly increased, uninterrupted anticoagulant and antiplatelet therapy in CSP for polyps < 10 mm has shown to be associated with a significantly increased risk of immediate/intraprocedural bleeding, estimated at around 4.8%-17.8% when on DAPT[111,112], 11.9% when on a DOAC[111]and 5.7%-9.8% when on warfarin[111,133]. Given the current paucity of high-quality evidence and significant increased risk of immediate/intraprocedural bleeding, until more substantial evidence becomes available to verify the safety of continuation therapy, we recommend all anticoagulant and antiplatelet therapy be ceased before CSP for polyps < 10 mm, in accordance to the current position statements and guidelines.
CONCLUSION
This review largely agrees with the current position statements and guidelines from the major gastroenterology societies on the recommendations on anticoagulant and antiplatelet management in endoscopic procedures. Although, it has also highlighted some emerging discrepancies that requires further exploration in future guidelines,such as the two- to three-fold increased risk of PPB with HBT, and that anticoagulant and antiplatelet therapy may be safe to be continued in CSP for polyps < 10 mm.
In the meantime, we recommend strict endoscopic practice in accordance with the current major Gastroenterology guideline recommendations[2-4]be applied. Although in certain situations, anticoagulant and antiplatelet management may need to be considered on a case by case basis and tailored to the individual. Consultation with a cardiologist or haematologist is advised in these instances to ensure optimal patient safety.
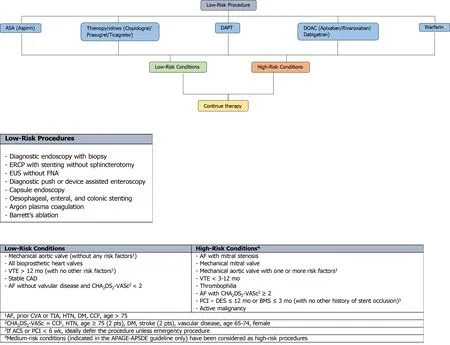
Figure 2 An evidence-based framework for safe clinical application of anticoagulant and antiplatelet management in the context of lowrisk endoscopic procedures for all endoscopists. ASA: Acetylsalicylic acid; DAPT: Dual antiplatelet therapy; DOAC: Direct oral anticoagulant; ERCP:Endoscopic retrograde cholangiopancreatography; EUS: Endoscopic ultrasound; FNA: Fine needle aspiration; VTE: Venous thromboembolism; CAD: Coronary artery disease; AF: Atrial fibrillation; PCI: Percutaneous coronary intervention; DES: Drug eluding stent; BMS: Bare metal stent; CVA: Cerebrovascular accident; TIA:Transient ischaemic attack; HTN: Hypertension; DM: Diabetes mellitus; CCF: Congestive cardiac failure; ACS: Acute coronary syndrome.
杂志排行
World Journal of Gastrointestinal Endoscopy的其它文章
- Peroral traction-assisted natural orifice trans-anal flexible endoscopic rectosigmoidectomy followed by intracorporeal colorectal anastomosis in a live porcine model
- Evaluation of the diagnostic and therapeutic utility of retrograde through-the-scope balloon enteroscopy and single-balloon enteroscopy
- Nonsteroidal anti-inflammatory drug effectivity in preventing post-endoscopic retrograde cholangiopancreatography pancreatitis: A systematic review and meta-analysis
- Endoscopic ultrasound-guided gallbladder drainage in pancreatic cancer and cholangitis: A case report
- Preemptive endoluminal vacuum therapy after pancreaticoduodenectomy: A case report
- Curling ulcer in the setting of severe sunburn: A case report
