Nrf2/ARE/HO-1通路是治疗帕金森病的作用新靶点
2014-11-28王玉敏崔其福赵伟丽张俊毅王洪权
王玉敏,崔其福,赵伟丽,张俊毅,王洪权*
(赤峰学院 医学院 附属医院 1.肿瘤内科;2.神经内五科;3.病理科, 内蒙古 赤峰 024000)
短篇综述
Nrf2/ARE/HO-1通路是治疗帕金森病的作用新靶点
王玉敏1,崔其福2,赵伟丽2,张俊毅3,王洪权2*
(赤峰学院 医学院 附属医院 1.肿瘤内科;2.神经内五科;3.病理科, 内蒙古 赤峰 024000)
调节Nrf2/ARE/HO-1通路在氧化应激相关性疾病中具有神经保护作用。通过药理学途径调节Nrf2/ARE/HO-1通路,可以抑制帕金森病神经毒剂诱导的神经毒性损伤。因此,筛选调节此通路的化合物有可能成为治疗帕金森病潜在的新靶点。
Nrf2;血红素加氧酶-1;帕金森病;神经保护
帕金森病(Parkinson’s disease,PD)是一种以黑质多巴胺能神经元丢失为特征的氧化损伤相关性神经退行性疾病。其主要的病理特征是种黑质多巴胺能神经元丢失以及细胞内以α-突触核蛋白(α-synuclein)为主要成分的路易小体(lewy body)形成。越来越多的证据表明氧化应激 (oxidative stress,OS) 损伤在PD的发病机制及其多巴胺能神经元死亡中发挥重要作用。PD患者脑内脂质过氧化、DNA损伤和羰基化作用蛋白的增多[1],均提示OS损伤参与PD的发病机制。鉴于此,针对抗OS的策略则成为PD有效的治疗途径。
1 Nrf2/ARE/HO-1通路
机体细胞本身具有一套复杂的抗氧化系统,此系统组成Nrf2-ARE-抗氧化酶通路。生理状态下,在胞浆内的核转录因子Nrf2 (nuclear factor-erythroid 2-related factor-2) 与细胞骨架相关性抑制蛋白Keap1(kelch-like ECH-associated protein 1)结合形成一个复合体,使Nrf2经泛素蛋白酶途径降解。当细胞或机体暴露于活性氧簇(reactive oxygen species, ROS)时,ROS可引起Keap1的修饰(如磷酸化),从而使Keap1募集Nrf2的功能发生改变,Keap1-Nrf2复合体解离,Nrf2从胞质转位到胞核,与受其调控的抗氧化酶基因的抗氧化反应元件(antioxidant response element, ARE)结合,从而启动编码解毒和抗氧化的基因表达,使其表达上调,促进细胞存活。血红素加氧酶-1(heme oxygenase, HO-1) 即是受其调控的一个非常重要的酶类。此外,Nrf2的激活除了受 Keap1调控外, 一些上游的激酶如蛋白激酶-C (protein kinase, PKC), MAPKs (mitogen-activated protein kinase) 和PI3K/Akt也可以调节Nrf2的激活[2], 因此可以形成一个Nrf2/ARE/HO-1通路 (图1)。此通路在以OS损伤为特征的PD中发挥重要的作用。
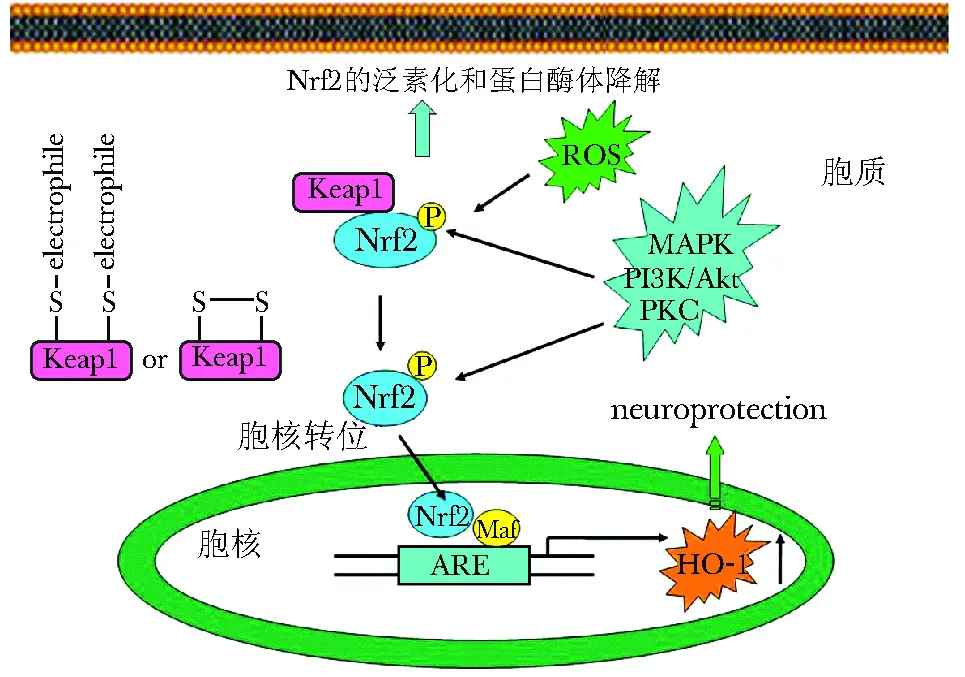
图1 Nrf2/ARE/HO-1通路及其激活机制Fig 1 The Nrf2/ARE/HO-1 pathway and a proposed mechanism responsible for its activation
2 Nrf2在PD中的神经保护作用
调控HO-1表达的上游转录因子Nrf2在PD中的发挥重要作用。第1,随着年龄(PD危险因素之一)的增加Nrf2的活性降低,而在老化动物中通过药理学途径可以恢复Nrf2的转录活性[3]。第2,Nrf2表达于黑质多巴胺能神经元胞质内,而在PD患者中Nrf2表达于存活的黑质多巴胺能神经元胞核内,表明细胞正通过Nrf2依赖的抗氧化酶来试图减弱氧化应激损伤。Nrf2在PD中的神经保护作用越来越引起研究者的注意。在动物模型中的研究也支持Nrf2在PD中发挥重要作用,在Nrf2缺失的小鼠模型中,神经元更易受MPTP诱导的神经退变和炎性反应的发生[4],Nrf2的缺失加重腹侧中脑内α-synuclein过表达引起的多巴胺能神经元的死亡,并促进α-synuclein的聚集,表明Nrf2在PD中具有神经保护作用。另外,通过siRNA抑制Keap1活性,进而激活Nrf2可以在体内抑制MPTP引起的多巴胺能神经元的损伤[5],而通过药理学途径激活Nrf2可以在体内或体外抑制6-OHDA[6]、MPP+[7]以及MPTP[8]引起的引起的神经元损伤。综上所述,Nrf2在PD中具有神经保护作用,而通过激活Nrf2可能成为治疗PD的潜在靶点。
3 HO-1在PD中的神经保护作用
HO-1为游离血红素(heme)降解的限速酶,能够催化Heme产生一氧化碳、胆绿素和游离铁,是应激早期发挥保护作用的抗氧化酶蛋白。PD患者脑内在黑质多巴胺能神经元胞质中HO-1表达升高,在路易小体内HO-1呈免疫强阳性[9]。研究者们陆续对不同PD模型中HO-1的表达进行了研究,显示引起PD的相关毒素,如百草枯(paraquat)、MPP+和MPTP等可以在体内或体外上调HO-1的表达,HO-1在PD中的作用日益凸显。HO-1的表达上调可能是对PD相关毒素引起的神经损伤具有神经保护作用, 证据如下, 1)编码HO-1蛋白的Hmox1基因启动子单核苷酸多态性与PD具有高度关联性。HO-1(-413)TT基因型较AA基因型的HO-1的表达水平低[10],暗示HO-1蛋白水平的降低可以升高氧化应激的敏感性。2)PD患者血浆内HO-1的水平升高,与对抗持续慢性氧化损伤相关。3)腺病毒介导的HO-1过表达可以保护受MPP+毒性损伤的黑质多巴胺能神经元[11]。在神经母细胞瘤M17细胞中,HO-1过表达降低α-突触核蛋白的水平[12],PINK1 G309D突变体促进H2O2诱导的SH-SY5Y凋亡的发生,而HO-1过表达可以促进细胞的存活[13]。4)上调HO-1的表达可以抑制亚铁离子(ferrous Iron, Fe2+)诱导的SK-N-SH细胞损伤[14]。
3.1上调HO-1抑制6-OHDA引起的神经毒性损伤
通过药理学手段诱导上调HO-1的表达在PD中具有神经保护作用[15- 17],可以抑制6-OHDA引起的神经毒性损伤。在SH-SY5Y细胞,人参皂甙Rb1(ginsenoside Rb1)通过上调HO-1的表达,抑制6-OHDA引起的神经毒性损伤[18]。咖啡白脂(kahweol)可通过PI3K和P38 MAPK激活来活化Nrf2,再上调HO-1的表达,从而抑制6-OHDA引起的SH-SY5Y细胞的氧化应激损伤[19]。蛋白酶体活性抑制剂MG-132可上调原代培养的中脑星形胶质细胞HO-1的表达,并抑制6-OHDA诱导的毒性损伤,而HO-1抑制剂锌原卟啉(zinc protoporphyrin IX, ZnPP)可以减弱MG-132的这种神经保护作用,这表明MG-132介导的HO-1的表达上调参与其抑制6-OHDA诱导的毒性损伤机制[20]。在Mes23.5多巴胺能细胞中,地昔帕明(desipramine)可通过ERK和JNK激活来活化Nrf2,进而上调HO-1的表达,从而抑制鱼藤酮和6-OHDA引起的神经毒性损伤[21]。莱菔硫烷(sulforaphane,SFN)能够通过PI3K/Akt通路激活Nrf2,从而上调HO-1进而抑制6-OHDA诱导的神经毒性损伤[22]。
3.2上调HO-1抑制MPP+引起的神经毒性损伤
通过药理学途径上调HO-1表达可抑MPP+引起的神经毒性损伤。在Raw 264.7巨噬细胞中,HO-1抑制剂锡原卟啉Ⅸ(tin protoporphyrin-Ⅸ,SnPP-Ⅸ)加重MPP+诱导的氧化损伤,而HO-1诱导剂钴原卟啉Ⅸ(cobalt protoporphyrin-Ⅸ,CoPP-Ⅸ)可逆转MPP+诱导的氧化损伤,最新研究表明,MPP+可以促进HO-1的表达,而抑制HO-1的表达反过来促进MPP+诱导的氧化损伤[23],为HO-1参与抑制MPP+诱导损伤提供了直接的证据。虾青素(astaxanthin) 可通过促进HO-1的表达从而保护PC12细胞受MPP+介导的NOX2来源的ROS氧化损伤[24]。1,2,3,4,6-O-五没食子酰葡萄糖(1,2,3,4,6-penta-o-galloyl-β-d-glucopyranose)通过PI3K和ERK/Nrf2通路促进HO-1的表达从而保护PC12细胞受MPP+介导的氧化损伤[25]。综上所述,上调HO-1表达在PD具有神经保护作用。
4 结语
综上所述,调控Nrf2/ARE/HO-1通路,即激活Nrf2进而上调HO-1的表达成为PD 潜在的治疗靶标。以此为切入点,筛选出具有调节Nrf2/ARE/HO-1通路的天然化合物,将成为人们探索治疗PD的新措施。
[1] Surendran S, Rajasankar S. Parkinson’s disease: oxidative stress and therapeutic approaches[J]. Neurol Sci, 2010,31:531- 540.
[2] Zenkov NK, Menshchikova EB, Tkachev VO. Keap1/Nrf2/ARE redox-sensitive signaling system as a pharmacological target[J]. Biochemistry (Mosc), 2013,78:19- 36.
[3] Sykiotis GP, Bohmann D. Stress-activated cap ‘n’ collar transcription factors in aging and human disease[J]. Science signaling, 2010,3:re3.doi: 10.1126/scisignal.3112re3
[4] Rojo AI, Innamorato NG, Martin-Moreno AM,etal. Nrf2 regulates microglial dynamics and neuroinflammation in experimental Parkinson’s disease[J]. Glia, 2010,58:588- 598.
[5] Williamson TP, Johnson DA, Johnson JA. Activation of the Nrf2-ARE pathway by siRNA knockdown of Keap1 reduces oxidative stress and provides partial protection from MPTP-mediated neurotoxicity[J]. Neurotoxicol, 2012,33:272- 279.
[6] Siebert A, Desai V, Chandrasekaran K,etal. Nrf2 activators provide neuroprotection against 6-hydroxydopamine toxicity in rat organotypic nigrostriatal cocultures[J]. J Neurosci Res, 2009,87:1659- 1669.
[7] Wruck CJ, Claussen M, Fuhrmann G,etal. Luteolin protects rat PC12 and C6 cells against MPP+induced toxicity via an ERK dependent Keap1-Nrf2-ARE pathway[J]. J Neural Transm Suppl, 2007,72:57- 67. doi:10.1007/978-3-211-73574-9_9
[8] Jazwa A, Rojo AI, Innamorato NG,etal. Pharmacological targeting of the transcription factor Nrf2 at the basal ganglia provides disease modifying therapy for experimental parkinsonism[J]. Antioxid Redox Signal, 2011,14:2347- 2360.
[9] Schipper HM. Heme oxygenase expression in human central nervous system disorders[J]. Free Radic Biol Med, 2004,37:1995- 2011.
[10] Infante J, Garcia-Gorostiaga I, Sanchez-Juan P,etal. Synergistic effect of two oxidative stress-related genes (heme oxygenase-1 and GSK3beta) on the risk of Parkinson’s disease[J]. Eur J Neurol, 2010,17:760- 762.
[11] Hung SY, Liou HC, Kang KH,etal. Overexpression of heme oxygenase-1 protects dopaminergic neurons against 1-methyl-4-phenylpyridinium-induced neurotoxicity[J]. Mol Pharmacol, 2008,74:1564- 1575.
[12] Song W, Patel A, Qureshi HY,etal. The Parkinson disease-associated A30P mutation stabilizes alpha-synuclein against proteasomal degradation triggered by heme oxygenase-1 over-expression in human neuroblastoma cells[J]. J Neurochem, 2009,110:719- 733.
[13] Chien WL, Lee TR, Hung SY,etal. Impairment of oxidative stress-induced heme oxygenase-1 expression by the defect of Parkinson-related gene of PINK1[J]. J Neurochem, 2011,117:643- 653.
[14] Du X, Xu H, Jiang H,etal. Akt/Nrf2 activated upregulation of heme oxygenase-1 involves in the role of Rg1 against ferrous iron-induced neurotoxicity in SK-N-SH cells[J]. Neurotox Res, 2013,24:71- 79.
[15] Chong CM, Zhou ZY, Razmovski-Naumovski V,etal. Danshensu protects against 6-hydroxydopamine-induced damage of PC12 cellsinvitroand dopaminergic neurons in zebrafish[J]. Neurosci Lett, 2013,543:121- 125.
[16] Bae J, Lee D, Kim YK,etal. Berberine protects 6-hydroxydopamine-induced human dopaminergic neuronal cell death through the induction of heme oxygenase-1[J]. Mol Cells, 2013,35:151- 157.
[17] Oh H, Hur J, Park G,etal. Sanguisorbae radix protects against 6-hydroxydopamine-induced neurotoxicity by regulating NADPH oxidase and NF-E2-related factor-2/heme oxygenase-1 expressions[J]. Phytother Res, 2013,27:1012- 1017.
[18] Hwang YP, Jeong HG. Ginsenoside Rb1 protects against 6-hydroxydopamine-induced oxidative stress by increasing heme oxygenase-1 expression through an estrogen receptor-related PI3K/Akt/Nrf2-dependent pathway in human dopaminergic cells[J]. Toxicol Appl Pharmacol, 2010,242:18- 28.
[19] Hwang YP, Jeong HG. The coffee diterpene kahweol induces heme oxygenase-1 via the PI3K and p38/Nrf2 pathway to protect human dopaminergic neurons from 6-hydroxydopamine-derived oxidative stress[J]. FEBS Lett, 2008,582:2655- 2662.
[20] Yamamoto N, Izumi Y, Matsuo T,etal. Elevation of heme oxygenase-1 by proteasome inhibition affords dopaminergic neuroprotection[J]. J Neurosci Res, 2010,88:1934- 1942.
[21] Lin HY, Yeh WL, Huang BR,etal. Desipramine protects neuronal cell death and induces heme oxygenase-1 expression in Mes23.5 dopaminergic neurons[J]. PLOS ONE, 2012,7(11):e50138. doi:10.1371/journal.pone.0050138.
[22] Deng C, Tao R, Yu SZ,etal. Sulforaphane protects against 6-hydroxydopamine-induced cytotoxicity by increasing expression of heme oxygenase-1 in a PI3K/Akt-dependent manner[J]. Mol Med Rep, 2012,5:847- 851.
[23] Bae JW, Kim MJ, Jang CG,etal. Protective effects of heme oxygenase-1 against MPP+-induced cytotoxicity in PC-12 cells[J]. Neurol Sci, 2010,31:307- 313.
[24] Ye Q, Huang B, Zhang X,etal. Astaxanthin protects against MPP+-induced oxidative stress in PC12 cells via the HO-1/NOX2 axis[J]. BMC Neurosci, 2012 13:156. doi:10.1186/1471-2202-13-156.
[25] Chen H, Li H, Cao F,etal. 1,2,3,4,6-penta-O-galloyl-beta-D-glucose protects PC12 Cells from MPP+-mediated cell death by inducing heme oxygenase-1 in an ERK- and Akt-dependent manner[J]. Journal of Huazhong University of Science and Technology,2012,32:737- 745.
Nrf2/ARE/HO-1 signaling pathway isa new neuroprotective target for Parkinson’s disease
WANG Yu-min1, CUI Qi-fu2, ZHAO Wei-li2, ZHANG Jun-yi3, WANG Hong-quan2*
(1.Dept. of Oncology; 2.Dept. of Neurology; 3.Dept. of Pathology, Affiliated Hospital of Chifeng Medical CollegeChifeng University, Chifeng 024000,China)
Recent studies have shown that regulation of Nrf2/ARE/HO-1 provides neuroprotection against oxidative stress-induced neurotoxicity. The specific activation of Nrf2/ARE/HO-1 gene expression by pharmacological modulation may represent a novel approach for therapeutic treatment of Parkinson’s. There is, therefore, an excellent rationale for the development of new neuroprotective agents, based on their ability to enhance activity of Nrf2 and upregulate HO-1 expression.
Nrf2;heme oxygenase-1(HO-1); Parkinson’s disease; neuroprotection
2013- 10- 23
2013- 12- 23
国家自然科学基金(81260196, 81201844);内蒙古自治区高等学校青年科技英才支持计划(NJYT-13-B20);内蒙古自治区高等学校科学研究重点项目(NJSZ12306)
*通信作者(correspondingauthor): whongquan@alu.fudan.edu.cn
1001-6325(2014)08-1125-04
R 741.05
A
