Difunctional Effects of Organo-Modified T-Type Zeolite Membranes for Dewatering from Organic Solution
2021-11-18CUIXueZHANGYuPingGUITianHEMingLiangZHANGFeiHUNaLIYuQinCHENXiangShu
CUI XueZHANG Yu⁃PingGUI Tian HE Ming⁃Liang ZHANG Fei HU Na LI Yu⁃Qin CHEN Xiang⁃Shu
(Institute of Advanced Materials(IAM),State⁃Province Joint Engineering Laboratory of Zeolite Membrane Materials,College of Chemistry and Chemical Engineering,Jiangxi Normal University,Nanchang 330022,China)
Abstract:3⁃aminopropyltriethoxysilane(APTES)was introduced to modify the surface of T⁃type zeolite membranes.Characterizations,such as X⁃ray diffraction,field⁃emission scanning electron microscopy,X⁃ray photoelectron spec⁃troscopy and FT⁃IR,demonstrated that APTES was successfully modified on the membrane surface by chemical bonding.The modified APTES plays two roles:one is to improve the membrane hydrophilicity and the other is to reduce the defects of membrane layer.The modified membranes showed a high separation factor and water flux for the dewatering of a 90%isopropyl alcohol solution at 348 K.This method showed good reproducibility,and five modified membranes demonstrated an increased separation factor by about 8 times(from 359±23 to 2 934±183),and a slightly decreased total flux from(3.52±0.10)kg·m-2·h-1to(3.06±0.14)kg·m-2·h-1(a decrease of 13.07%).At 363 K,during 100 h of continuous testing,the modified membrane was very stable,and the permeation side retained a high⁃water content above 99.50%with a feed solution of 90%isopropyl alcohol.
Keywords:T⁃type zeolite membrane;3⁃aminopropyltriethoxysilane;hydrophilic;defects
0 Introduction
In the past decades,membrane separation as an energy conservation technology has been widely inves⁃tigated as a replacement for traditional high energy con⁃sumption methods such as azeotropic rectification and adsorption[1⁃3].In addition,membrane separation tech⁃nology is easily and simply fabricated compared with conventional methods[2,4⁃5].Up to now,many kinds of membranes have been reported for separation,such as polymer membranes[6]and inorganic membranes(zeo⁃lite membranes,MOF membranes,etc.)[7⁃8].As com⁃pared to flexible polymer membranes,inorganic crystal⁃line membranes supported on ceramic supports are more fragile and brittle.Researchers focus on fabricat⁃ing compact and continuous membranes for achieving excellent separation properties.However,nonzeolitic pores are usually formed during the fabrication process of crystalline membranes[9].These defects lead to the membrane showing a low separation selectivity due to that the nonzeolitic pores are larger than the zeolite pores[10⁃11].In addition,the reproducibility is a pivotal factor to cut down on expenses in the industry.Thus,reducing and/or eliminating the defects of a zeolite membrane is necessary for improving its separation properties[12⁃15]and reproducibility.
Nomura et al.[16]modified a membrane using a counter⁃diffusion CVD technique to plug the intercrys⁃talline region,and tetraethyl or thosilicate was adopted as the silica source.The CVD⁃modified silicalite membranes had a selectivity of 87.8 overn⁃butane/isobutane at 288 K.Huang et al.[12]reported that organosilica(APTES)was applied to modify zeolitic imidazolate framework ZIF⁃90 membranes to achieve both pore mouth narrowing and covering invisible defects of the polycrystalline ZIF⁃90 layer,and the selectivity of the APTES⁃modified ZIF⁃90 membrane was prominently enhanced.Lin et al.[13]eliminated the intercrystalline pores using on⁃stream counter diffusion CVD modification.At present,for polycrystalline mem⁃branes,modifications have always been applied with gas separation.For dehydrations,such as pervapora⁃tion(PV)and vapor permeation,the hydrophilicity of the membrane surface is important for preferential adsorption of water,which is beneficial for dewatering from an alcohol solution[17].To the best of our knowl⁃edge,few works have emphasized the hydrophilicity and sealing functions of a zeolite membrane except for a recent work,in which APTES was introduced on the surface to improve the hydrophilicity of a hydrophobic DDR zeolite(all⁃silica zeolite)membrane[18].So,we are proposing that if APTES was introduced to modify a hydrophilic zeolite membrane(containing aluminum),the obtained membrane may show a better separation performance,especially for the membranes with defects.
T⁃type zeolite with a Si/Al ratio(nSi/nAl)of 3⁃4 was formed by the intergrowth of offertite and erionite with an effective pore size of 0.36 nm×0.51 nm,which is between the size of water and most organic mole⁃cules[19⁃21].Thus,T⁃type zeolite membrane is a good can⁃didate for the dehydration of many organic solvents,but,as a type of polycrystalline membrane,formation of pinholes is always unavoidable during the synthesis process.In this work,T⁃type zeolite membranes were functionalized with APTES in a toluene solution.The obtained materials were used to verify that APTES was modified on the surface.The modified membranes showed a higher separation performance than that of the pristine membrane,and the modified membrane was tested for long⁃term stability.
1 Experimental
1.1 Preparation of T-type zeolite membrane
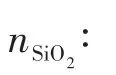
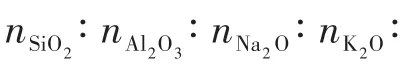
1.2 Modification of the T-type zeolite membrane
The modification method was similar to previous studies[18,23]. First, the pristine membranes were immersed in an APTES and toluene solution(5⁃20 mmol APTES in 60 mL toluene)in a Teflon⁃lined auto⁃clave.The modification was implemented in an oven at 383 K for 0.5⁃2.0 h.Then,the modified membranes were washed with toluene three times.Finally,the membranes were thermally processed at 393 K in a tube furnace by inletting argon.
1.3 Characterization and separation testing
The phases of the powders and membranes were checked by powder X⁃ray diffraction(PXRD,Rigaku UItimaⅣ)with a CuKαradiation(λ=0.154 06 nm)in a 2θrange of 5°⁃45°at a scanning speed of 4(°)·min-1.The tube voltage was 40 kV and the tube current was 40 mA.The sample morphologies were examined by field⁃emission scanning electron microscopy(FESEM,Hitachi SU8020)with the acceleration voltage of 5 kV.FT⁃IR analysis was performed using a spectrophotome⁃ter(Thermo,Nicolet 6700)in a vibration range of 600⁃4 000 cm-1.X⁃ray photoelectron spectroscopy(XPS,Thermo Fisher Scientific EscaLab Xi+)was used to in⁃vestigate the chemical state between APTES and the membrane.The water contact angle(WCA,POWE⁃REACH JC⁃2000CD)was calculated to determine the hydrophilicity of the membrane surface.
PV experiments were used to evaluate the mem⁃brane separation performance in the water/alcohol mix⁃tures.A flowchart of the PV device was illustrated as previous work,and the entire unit was a large feed⁃circulation system[22].The membrane was assembled in a module.The feed solution was provided by a meter⁃ing pump from a 3 L tank,and the permeation side was collected by a liquid nitrogen cold trap and was poured back into the feed tank after analysis to keep all compo⁃nents in the feed tank.A gas chromatograph(Shimadzu GC⁃14C)equipped with a thermal conductivity detec⁃tor(TCD)was used for analyzing the composition of the feed and permeation.The permeation flux of the mem⁃brane was defined asJ(kg·m-2·h-1).The separation factor(α)was determined asα=(Ya/Yb)/(Xa/Xb),whereXaandXbare the mass fractions of components a and b in the feed,respectively,andYaandYbare the corre⁃sponding mass fractions of components a and b in the permeation side,respectively.
The integrity of the pristine and modified mem⁃branes was checked by single gases with different dynamic diameters(H2,N2and SF6).The equipment and method were adopted from our previous work[24].
2 Results and discussion
2.1 Optimization of the modified membrane
The modified layer on the membrane surface gen⁃erally leads to increased permeate resistance and the elimination/reduction of defects.Modification usually increases the membrane selectivity but decreases the membrane flux.In addition,for dewatering,the hydro⁃philicity of the surface is important for the preferential adsorption of water,which is beneficial for the dehydra⁃tion of an alcohol solution[17].The effects of APTES con⁃tent and modification times were investigated to opti⁃mize the selectivity of modified membranes with a small decrease in the flux.
Four different APTES concentrations were used to explore the modification efficiency with a constant modification time.As shown in Table 1,the pristine membrane had a low separation factor.For the mem⁃brane modified with a relatively low APTES content,the separation factor of modified M⁃1 membrane did not show a distinct improvement.As the APTES con⁃tent increased,the separation factor of M⁃2 membrane was obviously improved from 420 to 2 910,an increase of about 7 times,while the flux only decreased 13.07%.However,by further increasing the APTES content,the separation factorofM⁃3 membrane increased slightly less than that of M⁃2 membrane,while the flux of the modified membrane was obviously reduced.For M⁃4 membrane,With the increasing of APTES concentration,the self⁃condensation reaction of APTES was strengthened besides the condensation between APTES and zeolite T membrane.The thick and uneven APTES layer which can provide large mass transfer resistance was modified on the membrane,but the uneven layer was not sufficient to reduce the de⁃fects of the membrane,leading M⁃4 membrane to show the least improvement of the separation factor and the lowest permeation flux.The probable modification mechanism of APTES layer on the membrane is illus⁃trated in Fig.1.

Fig.1 Modification mechanism of APTES layer on the membrane
Fig.2 shows the cross⁃section and surface mor⁃phology of pristine membrane and modified mem⁃branes processed with different APTES concentrations.The surface of modified M⁃1 membrane was covered by a loose APTES layer(Fig.2c and 2d),which can enhance the hydrophilicity of the membrane[18],but the very loose APTES layer did not significantly decrease the effect of defects,leading to a small increase in the selectivity.M⁃2 membrane also was modified with a loose APTES layer,but the further increasing of hydro⁃philicity might result in the desired separation factor(Fig.2e and 2f).A higher modification concentration(M⁃3,M⁃4)also existed many pores in the APTES layer(Fig.2h and 2j).Fasano et al.investigated the roles of hydrophilicity and surface barriers on the overall water transport through a zeolite membrane,and the surface barriers were proportional to the diffusion resistance[25].

Fig.2 SEM images of(a,b)the pristine membrane and modified(c,d)M⁃1,(e,f)M⁃2,(g,h)M⁃3,(i,j)M⁃4 membranes
Incorporating the PV performance,the membrane modified in high APTES solution may increase the dif⁃fusion resistance,which leads to decreased water trans⁃port through the membrane.
Modification time is another factor that influence modification efficiency[17,26⁃27].With the same APTES content(10 mmol APTES in 60 mL toluene)and modi⁃fication temperature(383 K),the membranes modified with different modification times(0.5⁃2 h)were investi⁃gated.As shown in Table 2,short treatment times(0.5 and 1.0 h)were effective but insufficient for modifying the functionalized coupling agent layer.When the pro⁃cessing time was extended to 1.5 h,the modified mem⁃brane showed a high separation factor of 2 910 with a considerable flux of 3.01 kg·m-2·h-1from a 90%iso⁃propanol solution at 348 K.The longer modified time(2.0 h)did not lead to an obvious increase of the sepa⁃ration factor but resulted in a decrease of the flux as a result of the high treatment content.

Table 1 PV results of the pristine and modified T-type zeolite membranes for a 90%isopropanol solution at 348 K

Table 2 PV results of the pristine and modified T-type zeolite membranes for a 90%isopropanol solution at 348 K
2.2 Characterization of the membranes
2.2.1 XRD analysis
The XRD patterns of pristine and modified T⁃type zeolite membrane are shown in Fig.3.The modified membrane(M⁃2)was obtained under the typical modi⁃fied conditions.The results show that the pure phase T⁃type zeolite membranes were synthesized on the sup⁃ports,and all peaks were in accordance with standard zeolite T crystals together withα⁃Al2O3signals.After modification,the location of the characteristic peaks remained well aligned with that of pristine membrane,besides some weakening of the intensity,which is attributed to the overlaid layer of APTES.The XRD pattern results demonstrate that the membranes were stable during the treatment process.

Fig.3 XRD patterns of(a)zeolite T,(b)mullite support,(c)the pristine membrane and(d)the modified membrane(M⁃2)
2.2.2 FT⁃IR analysis
To determine the interactions between APTES and the membrane,the pristine membrane and modi⁃fied membrane(M⁃2)were evaluated using FT⁃IR.As presented in Fig.4,both before and after modification,the spectral band in the vicinity of 3 450 cm-1is relat⁃ed to the stretching vibration of—OH from the adsorbed water on the membrane surface.After modifi⁃cation with APTES,the peak width of the band became wider because of overlap with the new—NH[28].Com⁃pared with pristine membrane,the peaks at 1 562 and 1 470 cm-1of M⁃2 are assigned to the N—H2vibration of the amine group(R—NH2)[23,28].The peaks around 2 928 cm-1are assigned to CH2anti⁃symmetric vibra⁃tions[29],and a new band at 1 384 cm-1corresponds to the Si—CH2bending vibration.Additionally,the C—N stretching vibration appeared in the vicinity of 1 250 cm-1[18].The bands at 1 085 and 807 cm-1are assigned to the anti⁃symmetric and the symmetric vibrations of Si—O—Si[30].The results indicate that APTES is successfully modified on the membrane surface.
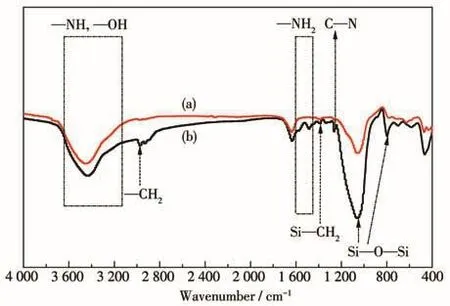
Fig.4 FT⁃IR spectra of(a)the pristine and(b)modified T⁃type zeolite membranes
2.2.3 XPS analysis
There are several different interactions between the zeolite surface and modified APTES,such as chem⁃ical bonds,hydrogen bonding,protonation of the amine and van der Waals forces[31].XPS was used to analyze the chemical state of the membrane.Comparing the XPS spectra of the membrane before and after modifi⁃cation,Fig.5a shows that the binding energies of Si2pwere 102.59 and 102.30 eV for the pristine and modi⁃fied membranes(M⁃2),respectively.Correspondingly,the binding energies of O1sin the pristine and modi⁃fied membranes were 532.05 and 531.96 eV(Fig.5b).The decreased binding energy indicates that the chemi⁃cal state on the surface changes from Si—O—H to Si—O—Si,because the electronegativity of H is 2.2,which is higher than that of Si(1.9).Fig.5c shows the XPS spectrum of N1s,with a peak at 401.11 eV attributed to hydrogen⁃bonded and protonated N,and the top peak at 399.05 eV related to the free—NH2in the terminal molecules of the chemically bonded N that may retain van der Waals forces[26,31⁃32].The results also illustrate that APTES is modified on the membrane surface by chemical bonding[26].

Fig.5 High⁃resolution XPS spectra of the pristine and modified membranes:(a)Si2p,(b)O1s and(c)N1s
2.3 Membrane stability
The stability of modified APTES layer is a critical issue during the separation process.Therefore,the modified membrane(M⁃2)was tested to evaluate the membrane stability at 363 K for 100 h.As illustrated in Fig.6,during 100 h of continual testing,the modi⁃fied membranes exhibited a good and stable perme⁃ation flux of approximately 3.8 kg·m-2·h-1together with a high separation factor about 2 000,and the wa⁃ter content of the permeation side reached above 99.50%.Notably,the separation factors of the modified membrane were retained during the entire separation process,indicating that the modified membrane shows a comparable thermal and long⁃time stability.
Furthermore,to determine the stability of the mod⁃ification layer in different water content solutions,the modified membrane(M⁃2)was used to dewater differ⁃ent isopropanol solutions.Fig.7 shows the membrane performance′s dependence on water content.With a high⁃water content of 20%,the membrane showed a higher total flux of 4.2 kg·m-2·h-1.With the water con⁃tentdecreasing,thetotalflux ofthe membrane decreased.This may be explained by the fact that un⁃der high isopropanol content,isopropanol affords a large impact resistance,which decreases the migration velocity of water and hinders the diffusion of water to the membrane surface,leading to a decrease of the total flux.It is worth noting that the water content in the permeation side stayed above 99.50%(isopropanol solution of 95%)for different isopropanol contents,showing that the modification layer has a very high sta⁃bility in high water and/or isopropanol content solu⁃tions.Therefore,we deem this processing method prom⁃ising for use as a modification membrane in industrial dewatering of isopropanol solutions.

Fig.7 PV performance of the modified membrane(M⁃2)in different isopropanol aqueous solutions at 348 K
2.4 Reproducibility of the modification method
To assess the reproducibility of the modification method,five membranes were modified under the opti⁃mal modification conditions.Table 3 shows the PV per⁃formance of the modified membranes,and the five membranes had a high separation factor of 2 934±183 and a good total flux of(3.06±0.14)kg·m-2·h-1.The average increase of the separation factor was 717.27%,while the average decrease of the total flux was only 13.07%.The results demonstrate that the simple and reproducible method described in this study is a reli⁃able method to increase the quality of hydrophilic T⁃type zeolitic membranes.

Table 3 PV results of the pristine and modified T-type zeolitic membranes for a 90%isopropanol solution at 348 K
2.5 Mechanism for improvement of the selectivity
For the dehydration of organic solutions,selectivi⁃ty is usually dominated by two factors:(1)the hydrophi⁃licity of the membrane surface,which plays a key role during the separation process due to preferential adsorption of water benefiting separation,(2)the num⁃ber of defects.Hence,eliminating and/or reducing the number of defects that are formed during the polycrys⁃talline membrane synthesis is an effective strategy for improving selectivity.
Thus,the membranes before and after modifica⁃tion were analyzed by a contact angle measurement.As shown in Fig.8,the static water contact angle of the pristine hydrophilic T⁃type zeolite membrane was 133.40°,and the modified membrane(M ⁃2)had a smaller contact angle(85.11°)related to the enhanced hydrophilic surface compared with that of pristine membrane.Namely,the hydrophilic T⁃type zeolite membranes after modification showed a more hydro⁃philic surface,which is better for the preferential adsorption of water,leading to a high separation factor.

Fig.8 Water contact angle of(a)the pristine and(b)modified T⁃type zeolite membranes
Furthermore,to investigate the decrease of defects,gases with different dynamic diameters(H2,N2and SF6)were imported as probe molecules to evaluate the contributions of the modification[33].The permeation results of the three gases are presented in Fig.9.The ideal selectivities of H2/N2and H2/SF6for pristine membrane were 2.4 and 4.3,which were lower than the corresponding Knudsen values of 3.7 and 8.5,respec⁃tively.The low ideal selectivities for H2over N2and SF6resulted from many defects in the membrane.After modification,the modified membrane showed a decrease in the permeability of H2and N2,while there was a sharp reduction of SF6,resulting in a remarkable enhancement of the H2/N2and H2/SF6idealselectivity from 2.39 to 7.33 and from 4.29 to 32.61,respectively.Compared with the pristine membrane,theideal selec⁃tivity of H2/SF6for the modified membrane was enhanced drastically,which was related to APTES layer coating which reduces the defects in the poly⁃crystalline membranes[34].
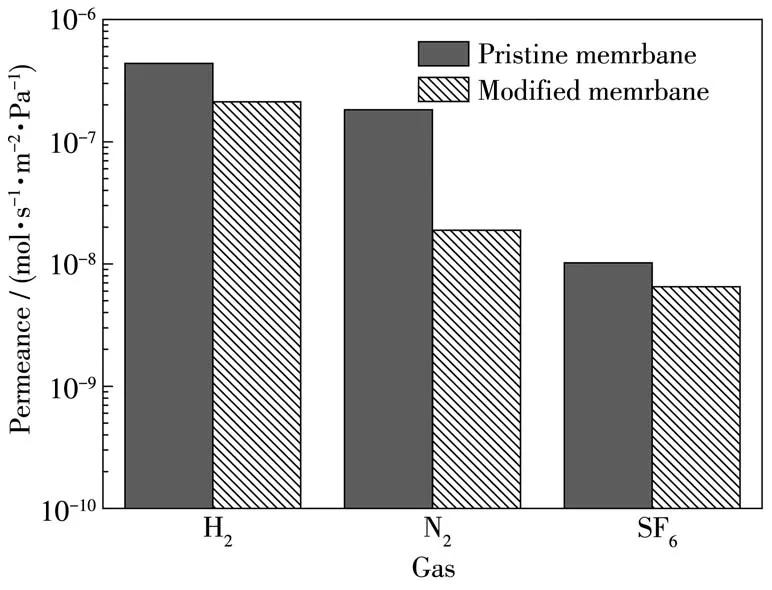
Fig.9 Single gas permeability of the pristine and modified T⁃type zeolite membranes
In conclusion,as illustrated in Fig.10,we assume the APTES layer had two functions in attaining the high separation factor in this work,and the contribu⁃tions of this modification are as follows:(1)increasing the hydrophilicity of the membrane surface for the pref⁃erential adsorption of water,and(2)reducing the de⁃fects of the polycrystalline membrane layer to achieve a more compact membrane layer.

Fig.10 Schematic diagram of the modification process and role of APTES layer
3 Conclusions
A simple and feasible modification method was introduced to modify hydrophilic T⁃type zeolite mem⁃branes.The contributions of the processing method play a double function by modifying an APTES layer.On the one hand,the APTES can reduce the defects that are formed during the synthesis process of the zeo⁃lite membrane,making the membrane more compact and continuous.On the other hand,APTES can increase the hydrophilicity of the membrane surface,which enhances dewatering by preferential adsorption.This method can effectively improve the reproducibili⁃ty of T⁃type zeolite membrane.The modified layer was resistant enough for long⁃term testing(continuous 100 h)with a stable separation factor,indicating the modifi⁃cation method has enormous potential for industrial applications.
Acknowledgements:We would like to acknowledge the financial support provided by National Scientific Foundation of China(Grants No.21766010,21868013,21968009),National High Technology Research and Development Program of China(863 program,Grants No.2009AA034801,2012AA03A609),and Jiangxi Provincial Department of Science and Technology(Grants No.20202ACBL213010,20192ACB80003).
杂志排行
无机化学学报的其它文章
- Heterometallic Ln(Na)-MOFs(Ln=Tb,Dy,Ho):Crystal Structures,Luminescent Sensing for Acetaldehyde,Fe3+,Cr2O72-,and Electrochemical Sensing for Catechol
- 自支撑多孔碳/硒复合柔性电极的制备及其电化学性能
- 同构MOFs复合氧化石墨烯电极材料构筑高性能超级电容器
- 松针状NiCo2O4@碳布复合材料在锂硫电池中的应用
- 1,4-二氮杂二环[2.2.2]辛烷-氰基合钴(Ⅲ)三维框架氢键型晶体的合成、相变及介电性质
- Ag@硅氧倍半聚合物的合成及其对对硝基苯酚的催化还原性能
