NLRC3 alleviates hypoxia/reoxygenation induced inflammation in RAW264.7 cells by inhibiting K63-linked ubiquitination of TRAF6
2020-10-23ZhongTangLiHangLiuWanQiuZhang
Zhong-Tang Li, Hang Liu, Wan-Qiu Zhang
Department of Hepatobiliary Surgery, The First Affiliated Hospital of Chongqing Medical University, Chongqing 40 0 016, China
Keywords:
ABSTRACT
Introduction
Liver transplantation is the only effective treatment for patients with end-stage liver disease worldwide [1].Ischemia-reperfusion(I/R) injury is an inevitable pathophysiological process when an ischemic liver tissue is resupplied with effective circulating blood flow during organ transplantation surgery [2 , 3], and it is the key factor that affects the prognosis of liver transplant patients [4 , 5].After liver transplantation, about 10% of early transplant failure and acute and chronic rejection are closely related to liver I/R injury [6].Moreover, due to the lack of donor liver, the application of marginal donor liver will further increase the risk of liver I/R injury [7].With the advancement of research on liver I/R injury, the key immune regulatory mechanisms affecting its occurrence and development are being continuously clarified.Accompanying the reperfusion process, complicated molecular signals are activated in immune cells, such as Kupffer cells (KCs) and lymphocytes [8], which further aggravate the ischemia related damage of liver parenchymal cells [2].KCs, as the largest macrophage population in the human, settle in liver tissues and play a key regulatory role in I/R related immune responses [9].Large amount of proinflammatory factors, such as TNF-α, IL-1β, and IL-12, produced after the activation of NF-κB and MAPK-related signaling pathways in KCs [10 , 11], as well as the release of reactive oxygen species resulting from intracellular mitochondrial damage [12 , 13], are the critical steps that initiate the inflammatory cascade effect in liver I/R injury.NF-κB signal activation is an important mechanism that triggers the inflammatory cascade effect in an innate immune response.The K63-linked ubiquitination of its upstreamTNF receptor-associated factor 6 (TRAF6) protein is particularly important for NF-κB signal activation.TRAF6 has been confirmed to be the key regulator that affects the activity of this pathway [14].
NOD-like receptor family CARD domain containing 3 (NLRC3)is highly conserved in its structure and plays an important role in suppressing inflammation in both innate and adaptive immunity [14-17].Its immune functions have received widespread attention in multiple species and disciplines [14 , 15 , 18].After stimulating mouse macrophages with lipopolysaccharide, the NLRC3 molecule can act as deubiquitinase to remove ubiquitination at the K63 site [14].Considering the important role of NF-κB signal activation in the onset and development of liver I/R injury,we selected TRAF6, a key upstream molecule of the NF-κB pathway, as a regulatory target to study the early expression changes of NLRC3 in cells and to assess its interaction with TRAF6 after hypoxia/reoxygenation (H/R) treatment of RAW264.7 macrophages.Furthermore, through constructing an H/R model of RAW264.7 cells stably overexpressing NLRC3, the effects and mechanisms of NLRC3 on the activation of TRAF6/NF-κB signaling were clarified and the possible role of NLRC3 in regulating liver I/R injury was preliminarily revealed.Our study provided a novel insight into the search of effective methods to alleviate liver I/R injury.
Materials and methods
Cell culture
RAW264.7 cells for cell experiments were kindly donated by the Department of Hepatobiliary Surgery of Chongqing Medical University.The cell experiment included the following four groups:control group of cells without H/R treatment, H/R group of cells with H/R treatment, H/R + negative control (NC) group of cells transfected with empty vector lentivirus and treated with H/R, and the H/R + Lv-NLRC3 group of cells transfected with recombinant lentivirus (HANBIO, Shanghai, China) overexpressing the NLRC3 gene and treated with H/R.Cells of each group were cultured in DMEM medium (HyClone, Utah, USA) containing 10% fetal bovine serum (HyClone) in a 37 °C incubator (Thermo Fisher Scientific,MA, USA) with 5% CO 2 , and they were routinely replaced with fresh medium, passaged, and cryopreserved.
Construction of the mouse liver I/R model
Six-eight-weeks-old male C57BL/6J mice were purchased and raised by the Experimental Animal Center of Chongqing Medical University.The experimental animals were divided into the following four groups using the random number table method: I/R groups (reperfusion time of 3, 6, and 9 h, respectively) and sham group, with 6 animals in each group.Mice in the I/R groups were anesthetized with 0.1% sodium pentobarbital (MERCK, NJ, USA), the abdomen was opened after sterilization, and the portal vein and hepatic artery of the middle and left lobes were blocked with vascular clips to build approximately 70% hepatic ischemia for 60 min.During this period, the body temperature of mice was maintained at 37 °C.In the sham group, the same anesthesia and surgical methods were employed as in the I/R group, but without blocking the hepatic blood vessels.The mice were anesthetized and euthanized after 3, 6, and 9 h of reperfusion, and fresh liver tissues and serum samples were collected.
Construction of cell H/R model
Quantitatively seeded RAW264.7 cells in a logarithmic growth phase were hypoxically cultured in a 37 °C three-gas incubator with 5% CO 2 and 1% O 2 for 12 h, and the cells were replaced with the same amount of complete culture medium and cultured in a 37 °C incubator with 5% CO 2.Further, the cells were removed from the incubator at different reoxygenation time points.The cell samples were immediately harvested for subsequent experiments.
Western blotting analysis of protein expression in cells and tissues
The proteins from mouse liver or RAW264.7 cells were collected using lysis buffer (Beyotime, Shanghai, China), and the protein concentrations were determined by the BCA method.Further, 5 ×SDS loading buffer was added at a ratio of 1:4, and the samples were mixed well and boiled for 15 min to completely denature the protein samples.After aliquoting, appropriate amount of protein samples were used in Western blotting assay to determine the protein expression levels of NLRC3 (Santa Cruz, CA, USA),IκB-α(Beyotime), ubiquitinated K63 (CST, MA, USA), NF-κB p65(Beyotime), and p-p65 (Beyotime), withβ-actin (Beyotime) used as loading control.The remaining samples were stored at -80 °C.
Co-immunoprecipitation (Co-IP) assay for detection of the interaction between NLRC3 and TRAF6
Protein extract (300 μL) was divided into two eppendorf tubes:100 μL was saved as the positive control, and 200 μL was incubated with the TRAF6 (Santa Cruz), NLRC3 (Santa Cruz) or anti-IgG(Boster, CA, USA) antibody at 4 °C overnight.Subsequently, protein G agarose beads (Beyotime) were added and the mixture was incubated at 4 °C for another 2 h.The supernatant was removed, and the pellet was washed 5 times with PBS and resuspended in a lysis buffer.Finally, the interaction between NLRC3 and TRAF6 was detected by Western blotting.
Cell transfected with empty vector lentivirus or NLRC3 over-expression lentivirus and stable cell line screening
RAW264.7 cells were seeded in a 24-well plate at 1 ×105cells/well, and they were cultured in complete medium in a 5%CO 2 incubator at 37 °C for 24 h.Before transfection, lentiviruses were removed from -80 °C freezer and slowly thawed on ice.The original cell culture medium was aspirated, half volume of the fresh medium was added, and the virus stock solutions(MOI: 30) were added to the cells and mixed well.Cells in the non-transfected group were prepared and cultured.After 4 h at 37 °C, the medium was added and cells were cultured.At 24 h after transfection, the virus-containing culture medium was aspirated, fresh complete medium was added, and the cells were cultured for another 24 h.At 48 h after transfection, the expression of the Green fluorescent protein was observed under a fluorescence microscope.Next, fresh complete culture medium containing puromycin (6 μg/mL) replaced the old medium to screen for stably transduced cell lines.The same concentration of puromycin was added to the non-transfected cells.The cells of the non-transfected group were killed by puromycin the next day.The surviving cells after the initial screening were passaged, and puromycin (3 μg/mL) was added for maintenance screening culture.Stably transduced cell lines were obtained after consecutive screening rounds and 3 passages, which were expanded in large quantities and stored in a liquid nitrogen tank for later use.
Fluorescence detection of nuclear translocation of the NF- κB p65 subunit
RAW264.7 cells were seeded on 18 mm cell slides at 2 ×105.After H/R treatment, cells were fixed with 4% paraformaldehyde.Subsequently, cells were blocked with the immunostaining blocking solution (Beyotime) at room temperature for 1 h.The immunostaining blocking solution was aspirated, and the NF-κB p65antibody was added and incubated at 4 °C overnight.Next, a sheep anti-rabbit antibody (Cy3 labeled, Beyotime) was added and incubated at room temperature for 1 h.The cells were stained with DAPI (ZSGB-Bio, Beijing, China) for 5 min, added with an appropriate amount of anti-fluorescence quenching mounting solution, and observed under a fluorescence microscope after mounting with a coverslip.The NF-κB p65 subunit showed red fluorescence, and the nuclei were stained by DAPI with blue fluorescence.
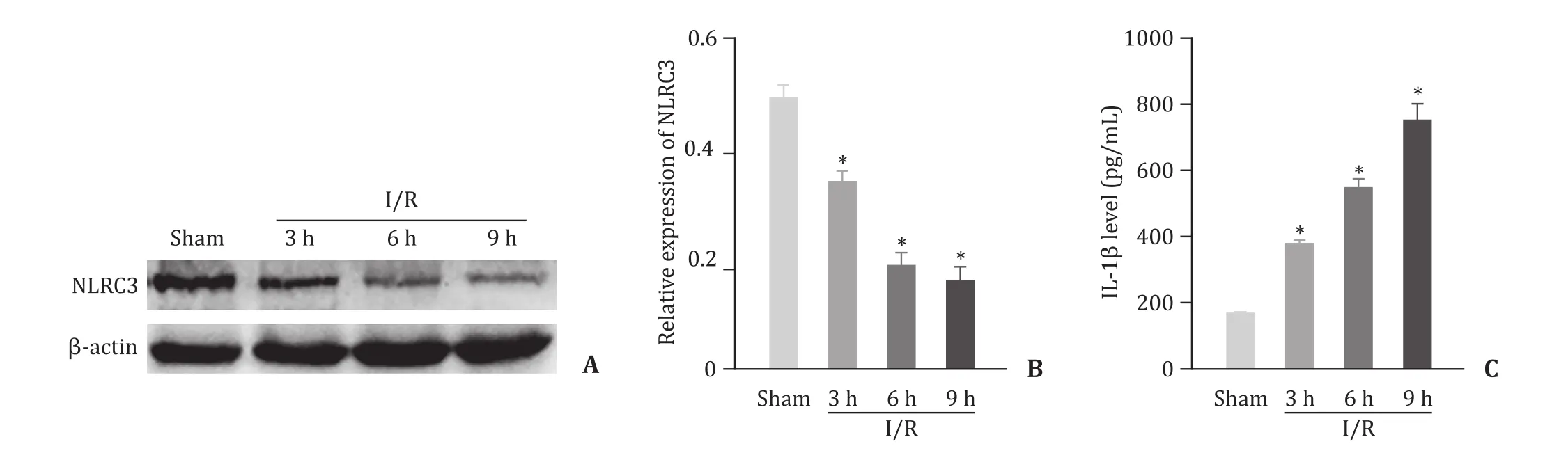
Fig.1.NLRC3 is downregulated in a mouse liver I/R model.A: Protein expression in mouse liver tissue by Western blotting; B: Semi-quantitative analysis of NLRC3 protein;C : Mouse serum IL-1β level determined by ELISA.n = 6, ∗P < 0.05 vs.sham group.
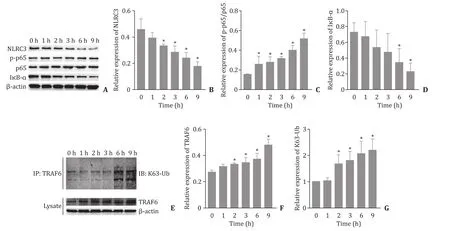
Fig.2.The correlation between NLRC3 and TRAF6 expression in H/R-induced RAW264.7 cells.A-D: The expression levels of NLRC3, I κB- α, p-p65, and p65 changed in cells after H/R treatment; E-G : The expression of TRAF6 protein and K63-linked ubiquitination level of TRAF6 changed after H/R treatment.∗P < 0.05 vs.0 h.
Enzyme-linked immunosorbent assay (ELISA) to measure IL-1 β
The cell culture supernatant and mouse serum were collected,and the ELISA kit (Boster) was used to measure the level of IL-1βin the samples according to the kit instructions.
Statistical analysis
SPSS 16.0 software (IBM Corp., Armonk, NY, USA) was used for statistical analysis.The data were expressed as mean ±standard deviation (SD).Comparison among cell groups was performed by one-way analysis of variance (ANOVA).Each experiment was repeated at least three times independently.AP<0.05 was considered statistically significant.
Results
Downregulation of NLRC3 protein in liver of I/R treated mouse
The expression of NLRC3 protein was gradually decreased with the prolongation of reperfusion ( Fig.1 A and 1 B), while IL-1βwas gradually increased following I/R injury in mice ( Fig.1 C).The differences among groups (sham, 3, 6, 9 h reperfusion) were statistically significant (P<0.05).
NLRC3 and TRAF6 expression in H/R-induced RAW264.7 cells
H/R significantly decreased the expressions of NLRC3 and IκB-αin RAW264.7 cells in time-dependent manner; the level of pp65 protein was gradually increased ( Fig.2 A-D).The expression of TRAF6 protein and K63-linked ubiquitination level of TRAF6 werealso increased gradually with the continuous extension of reoxygenation time ( Fig.2 E-G).

Fig.3.NLRC3 and TRAF6 interact directly.A: The interaction between TRAF6 and NLRC3 was detected by the Co-IP assay; B: Cell transfection efficiency was observed under an inverted fluorescence microscope; C-E : The expression levels of TRAF6 and NLRC3 protein were analyzed by Western blotting; F-H : The expression of TRAF6 protein and K63-linked ubiquitination level of TRAF6 were analyzed by Western blotting.∗P < 0.05 vs.control, # P < 0.05 vs.H/R + NC.
Interaction between NLRC3 and TRAF6
The Co-IP assay showed that NLRC3 and TRAF6 can bind to each other directly ( Fig.3 A).When transfected RAW264.7 with Lv-NLRC3 ( Fig.3 B), NLRC3 expression was increased as compared to the control group ( Fig.3 C and D,P<0.05).However,NLRC3 overexpression did not affect TRAF6 content ( Fig.3 C and E,P>0.05).Next, we assessed the TRAF6 protein expression and K63-linked ubiquitination level of TRAF6 in each group of cells after H/R treatment, and we found that K63-linked ubiquitination level and TRAF6 protein expression were higher in the H/R group and H/R + NC group than that in the control group (allP<0.05),while the K63-linked ubiquitination of TRAF6 in the H/R + Lv-NLRC3 group was significantly lower than that in the H/R + NC group (P<0.05) ( Fig.3 F-H).
NLRC3 inhibits NF- κB activation and mitigates inflammation
Compared with the control group, the p-p65 protein level was increased in the H/R group and the H/R + NC group, while the NLRC3 and IκB-αprotein levels were decreased (P<0.05).NLRC3 overexpression significantly decreased p-p65 expression and increased the protein expressions of NLRC3 and IκB-α( Fig.4 A-D,allP<0.05).Subsequently, we observed the nuclear translocation of the NF-κB p65 subunit by cellular immunofluorescence.The results showed that nuclear translocation of p65 in the H/R group and the H/R + NC group was significantly decreased compared withthat in the control group, while the nuclear translocation of p65 in the H/R + Lv-NLRC3 group was significantly decreased compared with that in the H/R group ( Fig.4 E).NLRC3 overexpression significantly decreased the level of IL-1βcompared with the H/R + NC group (P<0.05) ( Fig.4 F).

Fig.4.NLRC3 inhibits NF- κB activation and suppresses inflammation.A-D: The expression levels of NLRC3, I κB- α, p-p65, and p65 protein were analyzed by Western blotting;E: The nuclear translocation of p65 subunit was detected by immunofluorescence; F: IL -1 β level in the cell culture supernatant was determined by ELISA.∗P < 0.05 vs.control, # P < 0.05 vs.the H/R + NC group.
Discussion
Liver transplantation is the only effective treatment for endstage liver disease.Liver I/R injury is the important cause of transplant failure and acute and chronic rejection after transplantation surgery.However, the immune regulatory mechanism of liver I/R injury is unclear.This study was conducted to verify the correlation between NLRC3 expression in liver tissue and inflammatory responses in a mouse liver I/R model.We also revealed that overexpressed NLRC3 might alleviate H/R induced inflammation through inhibiting ubiquitination of the K63 site of TRAF6 protein in RAW264.7 cells, which provides a novel insight into the prevention and treatment of clinical liver I/R injury.
The molecular structure of the NLR family is highly conserved,with a typical C-terminal leucine-rich domain LRR, an N-terminal caspase activation recruitment domain CARD, and a middle oligonucleotide binding domain NBD [19].Among them, the early identified molecular subgroup is NLR molecules, which after activation combines with the intracellular apoptosis-associated speck-like protein and caspase to form a multiprotein complex the inflammasome, mainly including NLRP1, NLRP3, NLRP6, NLRC4,and NLRC5 [20-23].It is generally believed that this subgroup of molecules plays an important role in innate immunity to promote inflammation.In recent years, other non-inflammasome-forming NLRs with anti-inflammatory effects have been gradually discovered.Currently, this new subgroup mainly includes NLRP12,NLRX1, and NLRC3 [14 , 15 , 24-26 ].However, it has been reported that the inflammatory effects of NLRP12 in different experimental models are controversial, and the regulation of NLRX1 has a very complicated spatiotemporal specificity, while NLRC3, as a newly identified member of the non-inflammasome-forming NLR subfamily, has a significant anti-inflammation effect that has been confirmed by the interaction with multiple pathogen-associated or damage-associated molecular patterns (PAMPs/DAMPs) in T lymphocytes and some innate immune cells [14-16].Consistent researches above suggest that NLRC3 may have great potential application value.In this study, we also observed that NLRC3 suppressed inflammation in the RAW264.7 H/R cell model.Moreover, Allen [27]also found that although each molecule of this subgroup has different function and mechanisms, all of them inhibit the NF-κB signaling pathway to varying degrees.Other studies [28 , 29]have clearly pointed out that the K63-linked ubiq-uitination level of TRAF6 is a key upstream target that regulates the NF-κB signaling pathway.In this study, we confirmed the interaction between NLRC3 and TRAF6 in the constructed H/R inflammation model of RAW264.7 cells overexpressing NLRC3, and we found that NLRC3 decreased the release of pro-inflammatory factor IL-1βthrough reducing the K63-linked ubiquitination level of TRAF6 and inhibiting the nuclear translocation of the p65 subunit.Therefore, we preliminarily clarified the molecular mechanism of NLRC3 in the inhibition of NF-κB signal activation,which is consistent with the experimental results presented by Schneider et al.who stimulated mouse bone marrow macrophages with lipopolysaccharide [14].
Lamothe and colleagues [29]reported that TRAF6 plays an important role in cell proliferation, survival, and inflammatory responses through mediating the NF-κB, MAPK, and other signaling pathways, and that maintenance of the normal activation level of TRAF6 is critical for maintaining the physiological homeostasis of inflammatory cells.In this study, we found that in the H/R induced RAW264.7 macrophage inflammatory model, the new member of the NLR family, NLRC3, exerted a inhibiting effect on the inflammation mediated by TRAF6 through regulating the ubiquitination of TRAF6, which further confirmed the conclusion described by Schneider et al.[14]who believed that down-regulating the K63-linked ubiquitination level of TRAF6 was the key factor to regulating NF-κB signaling.However, as mentioned earlier, TRAF6 has a wide range of effects.This study did not examine more downstream signaling molecules that interact with TRAF6, and most of the data were focused on theinvitroexperiments.Related research ofinvivoanimal experiments is our future plan.
In summary, NLRC3 might inhibit H/R-mediated inflammatory responses by regulating K63-linked ubiquitination of TRAF6 in RAW264.7 cells.
Acknowledgments
None.
CRediT authorship contribution statement
Zhong-Tang Li:Data curation, Formal analysis, Writing -original draft.Hang Liu:Methodology, Writing - original draft.Wan-Qiu Zhang:Conceptualization, Funding acquisition, Supervision, Writing - review & editing.
Funding
This study was supported by grants from the National Natural Science Foundation of China ( 81873592 ) and the graduate tutor team construction project of Chongqing Municipal Education Commission Foundation ( dstd201801 ).
Ethical approval
This study was approved by the Ethics Committee of the First Affiliated Hospital of Chongqing Medical University.
Competing interest
No benefits in any form have been received or will be received from a commercial party related directly or indirectly to the subject of this article.
杂志排行
Hepatobiliary & Pancreatic Diseases International的其它文章
- Hepatobiliary&Pancreatic Diseases International
- No difference in mortality among ALPPS, two-staged hepatectomy, and portal vein embolization/ligation: A systematic review by updated traditional and network meta-analyses
- Telomerase reactivation is associated with hepatobiliary and pancreatic cancers
- Critical role of estrogen in the progression of chronic liver diseases
- Robotic isolated partial and complete hepatic caudate lobectomy: A single institution experience
- C -C motif chemokine ligand 16 inhibits the progression of liver cirrhosis via inactivating hepatic stellate cells
