In vitro differentiation capacity of human breastmilk stem cells: A systematic review
2019-11-28CamilaMariaRibeiroPachecoPriscilaEliasFerreiraClaudiaSayuriSaakiLuanaAlvesTannousIdibertoJosZotarelliFilhoLuizCesarGuaritaSouzaKatherineAthaydeTeixeiradeCarvalho
Camila Maria Ribeiro Pacheco,Priscila Elias Ferreira,Claudia Sayuri Saçaki,Luana Alves Tannous,Idiberto José Zotarelli-Filho,Luiz Cesar Guarita-Souza,Katherine Athayde Teixeira de Carvalho
Camila Maria Ribeiro Pacheco,Priscila Elias Ferreira,Claudia Sayuri Saçaki,Katherine Athayde Teixeira de Carvalho,Cell Therapy and Biotechnology in Regenerative Medicine Department,Pelé Pequeno Príncipe Institute,Child and Adolescent Health Research and Pequeno Príncipe Faculty,Curitiba 80.240-020,Paraná,Brazil
Luana Alves Tannous,Luiz Cesar Guarita-Souza,PUCPR-Institute of Biological and Health Sciences,CCBS,Curitiba 80.215-901,Paraná,Brazil
Idiberto José Zotarelli-Filho,Post Graduate and Continuing Education (Unipos),Department of Scientific Production,São José do Rio Preto 15.020-040,São Paulo,Brazil
Abstract
Key words: Human breastmilk; Stem cells; Cell differentiation; Pluripotency; Stemness
INTRODUCTION
Adult stem cells (ASCs) are present in small quantities in several mature tissues where they are quiescent,that is,not in the process of cell division.These cells are activated during the process of cellular replacement of tissues,being responsible for maintaining the biological homeostasis of the organism,thereby preserving the integrity of the tissues in which they are found.Similar to all other stem cells (SCs),ASCs have the property of self-renewal in that they make identical copies of themselves for long periods.Also,they may give rise to mature cell types with characteristic morphology and specialized functions[1].
Typically,SCs generate an intermediate cell type,called progenitor or precursor cells,before they become fully differentiated.These intermediate-stage cells are partially differentiated in fetal or adult tissues and give rise to different lineages after cell division.ASCs generally reach differentiation through a specific cellular development pathway; however,research has shown that their characteristics are not as definitive as previously thought[2,3].
A well-known type of ASC is mesenchymal stem cells (MSCs),which are present in several adult tissues.Among the most studied are the bone marrow (BM),adipose tissue,dental pulp,and vessel wall of the umbilical cord.MSCs are considered pluripotent cells by many researchers,because they are able to differentiate into osteoblasts,chondrocytes,adipocytes,as well as neural precursors,neurons,and glial cells.This cell population can be easily isolated and expandedin vitrobecause its potential of differentiation reflects the stimulatory and inhibitory factors to which they are subjected.Moreover,they are able to differentiate into specialized cells,with distinct phenotypes from their precursor,through inhibition and/or activation of certain molecular pathways[4].
Recently,the mammary epithelium has been the focus of a large number of studies due to its remarkable population of SCs as human breastmilk stem cells (hBSCs).It is believed that the existence of these cells in this organ is related to the ability of the mammary gland to expand significantly and regress over the adult life (Figure 1)[5].The hBSCs are found in the mammary gland in a state of latency and in low numbers;however,during gestation and lactation,they are activated and then transform the ductal structure into a secretory organ.Afterwards,when weaning occurs,milk production decays,inducing apoptosis of the mammary parenchyma cells.As a result,the ductal structure returns until a next pregnancy stimulates modifications of the tissue architecture once again[6,7].
Breastmilk has several cellular components together with hBSCs.In 2007,this population of stem cells in breastmilk was identified for the first time and when analyzed,showed Nestin (marker of neural stem cells) and different cytokeratins(CKs) such as CK5,CK14,CK18,and CK19 on their surfaces[8].CKs are intermediate filaments expressed in the mammary epithelium depending on the differentiation that the cell undergoes.For example,CK5 expression is indicative of hBSCs of mammary origin.Cells in culture were found to be positive only for Nestin or double positive for CK5/Nestin,but no co-expression of CK14,CK18,or CK19 and Nestin was observed.It is also noteworthy that the analysis of fresh milk by real-time polymerase chain reaction (RT-PCR) only detected Nestin and CK18,indicating that breastmilk contains SCs and differentiated cells[9].
Research has shown that hBSCs have the ability to generate the following three lineages:(1) Alveolar lobe,also named myoepithelial cells,a structure of the adult gland,which constitutes the basal layer of the ducts and alveoli; (2) Ductal epithelial cells,which coat the lumen of the ducts; and (3) Alveolar epithelial cells,which are responsible for protein synthesis in breastmilk[10].This evident ability of differentiation into cell types different from their tissue of origin raises the question of the real potential of differentiation of these cells.In this context,the purpose of this systematic review was to address the potential of differentiation of hBSCs into the three germ layers.
MATERIALS AND METHODS
Data sources and search strategy
In order to select papers that addressed the topic “potential of cell differentiation of hBSCs” in the literature,PubMed,EMBASE,OVID,AND COCHRANE LIBRARY databases were searched for analysis of the “potential of cell differentiation of hBSCs.” The keywords used were “HUMAN BREAST MILK” and “STEM CELLS.”Also,a combination of the keywords with AND,and the Boolean operator “NOT”were used to exclude those papers that had the word “CANCER” and its respective synonyms,which were previously consulted in medical subject heading (MeSH)terms.The title and abstracts were examined in all conditions.Papers on cell differentiation were selected manually from this broader primary search by adding the term “cell differentiation” and synonyms,and a very small number of papers were generated.The research structure used in the databases is represented in Table 1.
All duplicate papers were excluded,and the remaining ones were sorted t by title,abstract,and full text.Furthermore,papers in which the abstract was not available were analyzed in their entirety.
Study eligibility criteria
The inclusion criteria were all papers that:(1) Addressed the plasticity of stem cells derived from human breastmilk,that is,the ability of the cell to differentiate into more than one cell line; (2) Were published between 2007 and 2018; (3) Were written in the English language; and (4) Werein vitrostudies.Three authors independently assessed all papers,and the reviewers reached consensus on the eligibility of the studies after discussion.The exclusion criteria were as follows:(1) Studies in which the origin of the stem cells obtained were from other animal species; (2) Studies that used stem cells from other body tissues; (3)In vivostudies; and (4) Written in languages other than English.Subsequently,using the same criteria,the abstracts were selected.Lastly,the complete texts of the papers considered relevant in the previous steps were evaluated according to the following exclusion criteria:(1)Unpublished literature; (2) Only summary of the congress available; (3) Full text not available; and (4) Does not contemplate the process of cell differentiation.
Study selection and risk of bias in each study
Two independent reviewers (1 and 2) performed research and study selection.Data extraction was performed by reviewer 1 and fully reviewed by reviewer 2.A third investigator decided some conflicting points and made the final decision to choose the articles.Only studies reported in English were evaluated.The Cochrane instrument was adopted to assess the quality of the included studies[11].
Risk of bias
Considering the Cochrane tool for risk of bias,the overall evaluation resulted in five studies with a high risk of bias and three studies with uncertain risk.The domains that presented the highest risk of bias were related to a number of samples,volume of milk collected e income cells per milliliters (n= 3).Also,there was an absence of financial source in two studies and two studies did not disclose information on the conflict of interest statement.
RESULTS
In the review,315 papers were initially retrieved (Figure 2),21 of which were present in more than one database; therefore,they were excluded and considered only once,generating a total of 294 papers.Of these,only five papers met the eligibility criteria;their characteristics are summarized in Table 2.
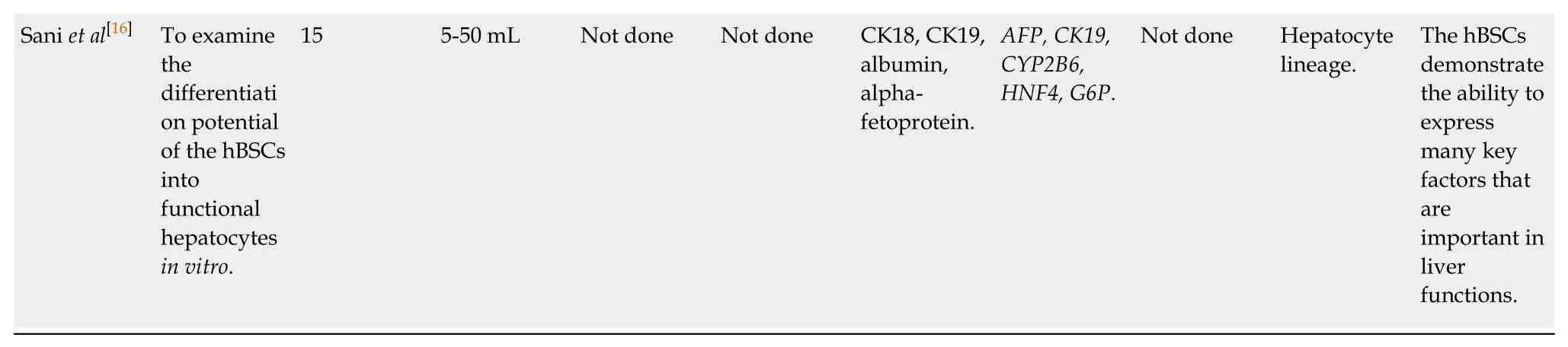
Although there are few studies in the literature on hBSCs,it is evident that this cell population has mesenchymal characteristics.Positivity for smooth muscle actin(SMA),Vimentin,and Nestin markers,along with the expression of surface markers[cluster of differentiations (CDs)]:CD44,CD29,CD73,CD90,CD106,CD133,CD146,CD271,and spinocerebellar ataxia type 1 (SCA1) support this claim.Low expression of adhesion molecules markers CD146 and CD144 was also detected.In addition,negativity was found for CD33,CD34,CD45,CD105,CD123,CD133,stage-specific embryonic antigen-4 (SSEA-4),and SSEA-1/CD15,confirming that such cells have no hematopoietic origin (Table 3).
The expression of transcription factors considered embryonic stem cell (ESC)markers such as SEEA4,TRA 60-1,Oct4,sex-determining region Y (SRY)-box 2 (Sox2),and NANOG was also evaluated by immunofluorescence staining.A subpopulation of cells expressing CK18 was found,and the frequency of positive cells for that population was 65.33% ± 6.1%.
The hBSCs differentiated into different cell types of mesodermal originin vitroincluding adipocytes,chondrocytes,and osteocytes (Figure 3).Moreover,they differentiated into three neural lines:oligodendrocytes,astrocytes,and neurons.They also differentiated into tissues of endodermal origin,as hepatocytes,β-pancreatic cells,and cardiomyocytes.Therefore,their property of differentiation in multiple lines is explicit (Figure 4).
These findings provide evidence of the existence of a subpopulation of pluripotent hBSCs,suggesting that these cells,when isolated from breastmilk,can be stimulated to create several types of tissues and be used in regenerative medicine.If hBSCs have great potential to differentiate into neural cell lines or even into neural precursor cells spontaneously,they would have great applicability in the treatment of neurodegenerative diseases.
DISCUSSION
Considering the results obtained in this review,it was possible to observe the scarcity of studies seeking to verify the potential of cellular differentiation of hBSCs.BM-derived MSCs were the first to be described in 1966[36],and even though in recent years there has been an increase in reports on the isolation of MSCs from different sources,they are the most frequently studied cell type[5].MSCs can be inducedin vitroto differentiate into various cell types found at various stages of development,as well as at specific anatomical sites[22].
Although the International Society of Cell Therapy advocates that human MSCs contain the positive surface markers CD105,CD73 and CD90; and have negativity to CD45,CD34,CD14 or CD11b,CD79a or CD19 and human leukocyte antigen-DR isotype,other surface antigens such as CD13,CD29,CD44,and CD10,which are often expressed in MSCs[37],should also be analyzed.Flow cytometry and immunocytochemistry analyses revealed that the hBSCs exhibited the positive surface markers CD29,CD44,CD90,CD146,CD271,and stem cell antigen-1.In addition,the markers CD33,CD34,CD45,CD73,CD123,and CD144 were considered negative by both techniques.Nevertheless,a discrepancy was observed concerning the CD105 marker,because it was negative (2.64% ± 0.55%) when analyzed by flow cytometry,and positive (68.3% ± 3.91%) when observed by immunocytochemistry[12,14,15].Likewise,lack of expression of CD90 (7.7% ± 0.8%) and CD73 (2.1% ± 0.41%) surface markers was reported,but a positive marker was found for CD105 (47.7% ± 2.95%)[5].
CD146-positive cells have a greater potential for cell proliferation and differentiation[21].The CD271 marker acts to maintain the clonogenicity of MSCs;however,most of these cells do not co-express CD90 and CD73,two general MSC markers.CD271 is not expressed in all MSCs,but is found at high levels in the BM,adipose tissue,and periodontal ligament.Moreover,it is expressed at low levels in MSCs derived from placenta and is not expressed in synovial membranes[22].
Regarding the transcription factors considered ESC markers that were evaluated by immunofluorescence,flow cytometry,and reverse transcription PCR (RT-PCR),the hBSCs did not express SSEA4 and SSEA1/CD15.Additionally,the markersOct4,Sox2,NANOG,andTRA-1-60/81were reactive.The genes,TRA-1-60/81andNANOGwere the most highly expressed,followed byOct4.In fact,NANOGis considered a crucial regulator of self-renewal and maintenance of pluripotentiality[38].
SSEA-4is a marker of ESCs initially used to isolate MSCs from the BM.It has been observed that when positively expressed,the cells in culture expand extensively and exhibit trilineage differentiation potency.Nevertheless,the use of SSEA-4 as a markeris questionable since its positivity was suggested to be an artificial induction ofin vitroculture using fetal bovine serum (FBS),which contains glycosphingolipids and globoserms that can be recognized by an SSEA-4 antibody.These findings raise the question of the physiological relevance and reliability of SSEA-4 as a marker for MSCs[22,39].
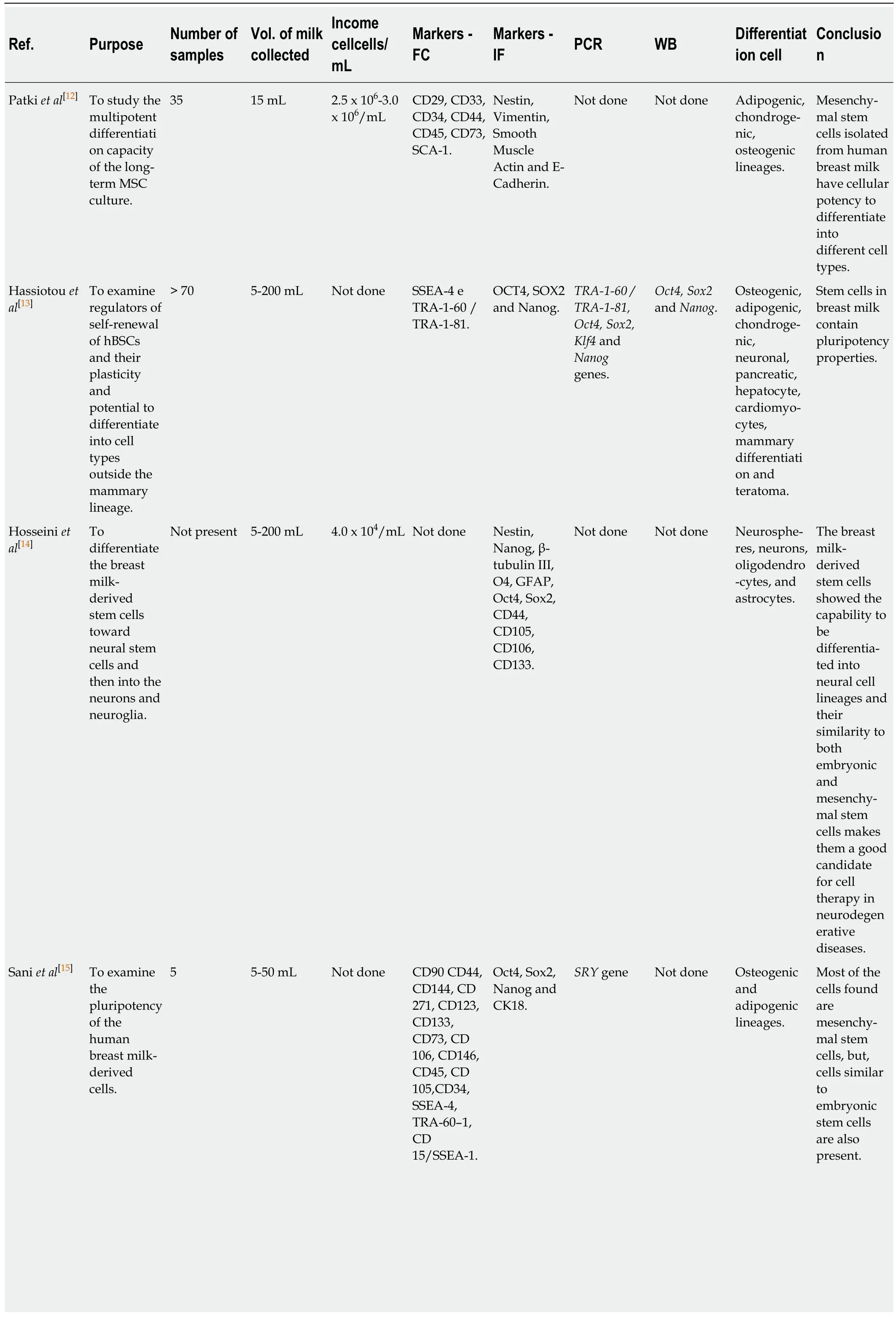
Table 2 Details of selected articles
hBSC:Human breastmilk stem cell.
The transcription factorsOct4and NANOG are indispensable for maintaining the self-renewal of ESCs.In addition,Oct4directly inhibits the signaling pathway of bone morphogenetic protein-4,which activates the mesoderm and the extra-embryonic differentiation of the ectoderm and endoderm,while NANOG acts as a repressor of the neural crest and neuroectoderma lineage and is essential for maintaining the properties of MSCs[4,28].
Pluripotency genes can be strongly correlated with and are the main transcription factors controlling the differentiation and pluripotency of multiple lines of ESCs.The presence of these genes in hBSCs allows a strong association since they play an important role in lactation and the ability to differentiate multiple breastmilk cell lines[26].
Nestin is a well-known marker of SCs of various lineages,and its expression in hBSCs indicates an increase in the characteristics of mammary progenitor cells during lactation.It is also highly expressed in lactating mammary tissues[26].
Through the observation of positivity for SMA,Vimentin,E-cadherin,and Nestin markers by flow cytochemistry,it is possible to hypothesize that the origin of the hBSCs is myoepithelial.HBSCs are positive for CK5 and SMA,and such dual positivity indicates the precursor nature of the cells.In addition,when labeling for CK18 was analyzed,it was present at a frequency of 65.33% ± 6.1% of the cell pool,revealing that there is a subpopulation of cells in breastmilk that is derived from luminal epithelial cells[8].
Currently in the MSC population,only a subpopulation contemplates the criteria recommended by the International Society of Cell Therapy since more compromised or more immature cells can be observed.There is still no consensus on the use of only one marker for the identification of MSCs from different sources,because there are phenotypic and/or functional differences that need to be better understood.It is reinforced that MSC manipulation can generate phenotypic rearrangements,losing the expression of some markers and acquiring others[2].This phenotype can be modulated in culture and does not reflect the phenotypein vivo.
By PCR in cells derived from fresh breastmilk,absence of the SRY gene in genomic DNA was shown,indicating that there are no cells originating from fetal tissues between the isolated cells; thus,there is no fetal microchemistry.Therefore,there is no exchange of stem cells between the mother and the embryo[40].
Among the criteria established for the characterization of MSCs,their ability to differentiate into osteogenic,chondrogenic and adipogenic lineage is included[13].Several studies have shown that these cells can also differentiate into unrelated germ lines in a process called transdifferentiation.Thus,MSCs can differentiate into cells of mesodermal lineage,such as bone,fat and cartilage,as well as have the potential for endodermal and neuroectodermal differentiation[41].
The use of culture medium containing FBS supplemented with ascorbic acid,βglycerophosphate,and dexamethasone is needed for osteogenic differentiation from MSCs.It induces an increase in alkaline phosphatase activity and calcium deposition.It was found that by subjecting hBSCs to the osteogenic differentiation environment,for 3 to 4 wk,the presence of osteocytes,the cell acquired cuboidal morphology,and produced mineralized material were observed.Calcium phosphate mineralization was verified by positive staining by Alizarin Red S,indicating that the calcium deposits as amorphous accumulation between the cells[12,15].Nuclear expression of Runt-related transcription factor 2(RUNX2),an essential transcription factor for osteoblastic differentiation,was also detected.SomeRUNX2-positive cells coexpressed osterix,an osteoblast-specific transcription factor required for bone formation[13].
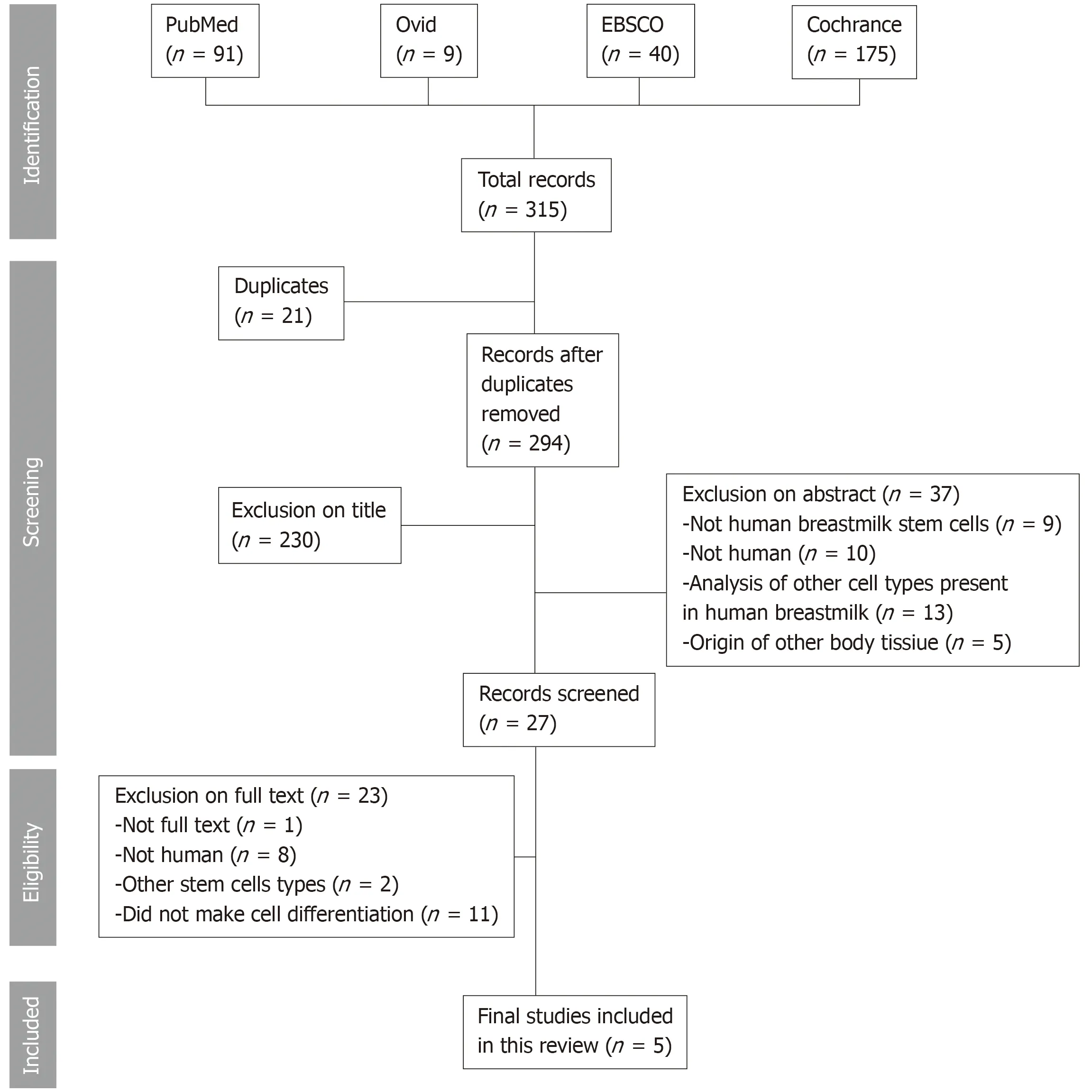
Figure 2 Flow chart demonstrating the search results with excluded and included studies.
Chondrogenic differentiation can be obtained by micromass culture utilizing an FBS-free culture medium containing transforming growth factor beta,which stimulates the production of highly sulfated proteoglycans,specific for cartilage and type II collagen.In contrast,cells synthesize extracellular matrix glycosaminoglycan,collagen type II,and aggrecan,which can be detected by immunohistological staining or by the expression of typical genes of the chondrogenic lineageviaPCR.After chondrogenic differentiation environment exposure for 21 d,the hBSCs began to show changes in their cellular morphology including acquisition of the spindle shape,the presence of larger round cell aggregates,and the accumulation of cartilage characteristic sulfate proteoglycans,being ratified by staining of Safranin-O[12,15].The transcription factors for chondrocytes,RUNX2/Sox6,were also identified in differentiated cells[13].
When exposed to culture medium containing FBS,dexamethasone,and isobutylmethylxanthine,MSCs were induced to differentiate into adipocytes.Adipogenic differentiation is characterized by the acquisition of an oval cellular morphology and the appearance of intracellular lipid droplets.Such changes can be verified in hBSCs stimulated to adipogenic differentiation for 21 d,which were confirmed by staining with Oil Red O and by positivity of the peroxisome proliferator-activated receptor gamma transcription factor,which is responsible for the regulation of the lipid metabolism[12,13,15].
Regarding the potential of hBSCs to generate different lineages,the presence of myoepithelial cells was observed in the adhered colonies after 2 wk of receiving induction medium for breast differentiation; by the 3rd wk,luminal cells were detected,some of which spontaneously synthesized milk proteins such as ß-casein,lactoferrin,and α-lactalbumin,which were detected in the culture supernatant[13].
It is interesting that many cells that constitute the liver are also the main progenitors for the formation of new hepatocytes.Moreover,when the tissue is compromised,SCs present in the bile ducts are stimulated,which leads to thedifferentiation of the hepatocytes.The differentiation of hepatocytes from non-hepatic ASCs has already been demonstrated by SCs originating from BM[42]in the umbilical cord blood[43]and adipose tissue[44].The hBSCs were able to differentiate in hepatocytes using α-fetoprotein (AFP),pyruvate-kinase M2 isoenzyme,and the albumin functional marker in this study[13].
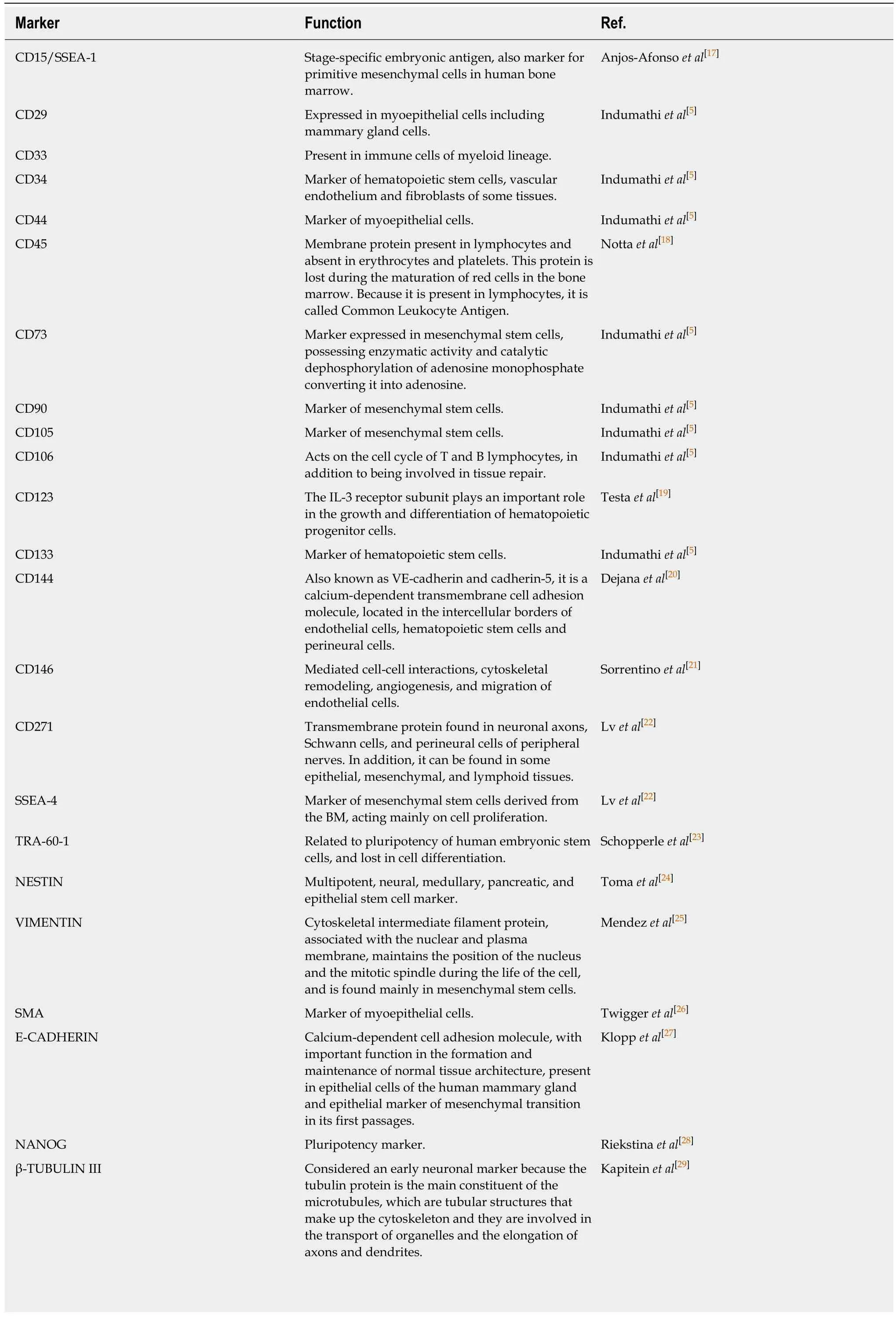
Table 3 CD and their functions

O4 Specific marker of gangliosides expressed by preoligodendrocytes and premyelinated oligodendrocytes.Girolamo et al[30]GFAP Glial fibrillary acidic protein forms subunits of the intermediate filaments of the cellular cytoskeleton,being present in the cytoplasm of the astrocytes.Hol et al[31]OCT4 Pluripotency marker.Riekstina et al[28]SOX2 Controls the undifferentiated and pluripotential state of the CTEs.Young et al[32]CK18 Expressed in the luminal cells of the breast,which synthesize proteins from breast milk.Thomas et al[33]KLF 4 Kruppel like factor 4 is a transcription factor related to pluripotency and cell proliferation potential.Li et al[34]SCA-1 The stem cell antigen 1 is found in stem cells.Holmes et al[35]
The markers CK18,CK19,and AFP were identified on differentiating hBSCs into hepatocytes on the 30th day of culture through immunocytochemistry analysis[16].They also analyzed the expression of hepatocyte-specific genes,such as hepatocyte nuclear factor 4 (HNF4),albumin,CK19,cytochrome p450 family 2 subfamily B member 6 (CYP2B6),glucose 6-phosphate (G6P),and Claudin,which were detected in differentiated cells and may indicate the formation of bile canaliculi.Semiquantitative RT-PCR showed that the amount of CK19,CYP2B6,and G6P was comparable to that of the positive control used.These cells showed the ability to store glycogen through indocyanine green clearance test,in which hepatocellular function was dynamically assessed,indicating that the differentiated hBSCs are functionalin vitro.Therefore,these cells can differentiate into endoderm lineages.
Both MSCs obtained from BM[45]and adipose tissue[46]have the ability to differentiate into multilineage,including transdifferentiation in pancreatic beta cells.RT-PCR analysis revealed that hBSCs,when exposed to an environment that induces pancreatic differentiation,positively expressed the pancreatic and duodenal homeobox 1 marker and insulin marker[13].These genes are associated with pancreatic development,as previously proven in the literature[47].From these findings,the use of these cells could be considered an alternative in the treatment of type I diabetes,with the ultimate aim of restoring the patient's glycemic levels.
The hBSCs can also stimulate the formation of cardiospheres.These cells start to express troponin T after 4 wk of culture[13].Remarkably,MSCs have been used for decades in cardiac regenerative therapy,with several sources of these cells such as the umbilical cord,placenta,and bone marrow.For example,MSC cord was seen to help in the recovery of motor function and increase strength of the cardiac muscle[48].
The ability of MSCs to differentiate into neural cells under specific culture conditions has also been reported.For instance,BM MSCs,when cultured in the presence of epidermal growth factor or brain-derived neurotrophic factor expressed glial fibrillary acidic protein (GFAP) and neuron-specific nuclear protein[49].In a previous study[14],hBSCs were cultured in DMEM/F12 with B27 1% and N2 2% for 7-10 days.After this time,some bead-like cellular aggregations were formed,which were dissociated and conditioned on a different plate containing DMEM/F12,B27,N2,beta fibroblast growth factor,and epidermal growth factor.To analyze the differentiation induction process into neural cells,and in order to compare whether there were differences between the cells before and after this protocol,the authors performed immunocytochemistry techniques for the antibody Nestin and flow cytometry for the CD133 marker.They found that a subpopulation of MSCs derived from breastmilk expressed Nestin (7.4% ± 3.30%) and CD133 (2.76% ± 1.93%),but after exposure to neurogenic induction medium,the frequency of Nestin-positive cells(58.20% ± 6.71%) and those expressing positivity for CD133 (58.74% ±3.36%)significantly increased.Since these are markers of neural stem cells,it can be inferred that hBSCs may exhibit behavior similar to that of neural stem cells.Recently,hBSCs were also observed to be prone to differentiate into the following three neural cell lines:neurons,astrocytes,and oligodendrocytes.The β-tubulin III marker was used to detect neurons,the O4 antibody for oligodendrocytes,and the GFAP antibody for astrocytes[13,14].
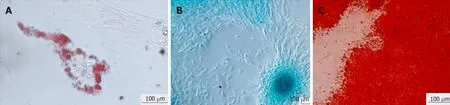
Figure 3 The hBSCs have the ability to differentiate into different cell types of mesodermal origin in vitro.
Mesenchymal breastmilk stem cells can differentiatein vitrointo cell lines of all three germ layers.Some studies have already shown thein vitrodifferentiation of MSCs to cells with neuronal and glial morphology,which express neuronal and glial markers after stimulation with retinoic acid,nerve growth factor,beta fibroblast growth factor,valproic acid,forskolin,and glial growth factor[49-51],pointing out the transdifferentiation potential of primitive mesenchymal progenitors to cells derived from other embryonic leaflets.
A theoretical model of MSC differentiation was created,in which undifferentiated cells would go through two phases before acquiring a specific phenotype.In the first one,after asymmetric divisions,the MSCs originate a population of cells less undifferentiated and a population of precursor cells with restricted self-renewal and differentiation potential.The transition from the stem cell compartment to the compromised compartment would occur with the symmetric division of tri- or bipotent progenitor cells,which would originate the unipotent progenitors.This process is well controlled and involves a change in the secretion profile of cytokines and growth factors,as well as a modification in the three-dimensional structure of the extracellular matrix,besides the activation and inactivation of transcriptional factors and consequent expression of genes related to this differentiation pathway[52].
Injecting MSCs into immunodeficient animals showed that they can respond to the different stimuliin vivo,differentiating themselves in a disorganized way and leading to the formation of cysts,teratomas,and tumors present in different types of tissues originating from each of the three embryonic leaflets[53].It is noteworthy that one of the studies performed teratoma formation testing,where freshly isolated hBSCs and cultured sphered milk cells were injected subcutaneously into SCID mice (n= 15).After 9 wk of the procedure,the mice were examined,and no tumor formation was observed[13].SCID lineage mice have great immunological compromises since they are deficient in T lymphocytes,B lymphocytes,and natural killer cells,and the use of these mice for teratoma formation tests is the most appropriate[54].In fact,the existence of pluripotent ASCs that do not form teratomas was previously described in BM,considering that these cells have unique patterns of DNA methylation in some genes and it is suggested that the non-formation of teratomas would be associated with these epigenetic characteristics[55].
Even though ESCs originate from the internal mass of the blastocysts,it is also possible to find them in adult tissues in specialized niches.Recent evidence has suggested that certain subpopulations of cells within ASC compartments can differentiate into cell types outside their dermal origin under specific microenvironmentsin vitroandin vivo[32].These results indicate that hBSCs possibly have pluripotency characteristics,with many characteristics to be still explored.
In conclusion,human breastmilk stem cells have high cellular plasticity,exhibiting the ability to generate cells from all three germ layers as demonstrated by their stemness.Thus,further studies are needed to elucidate the implication of these cells in regenerative medicine and bioengineering as well to explore these ASCs obtained from no invasive sources.
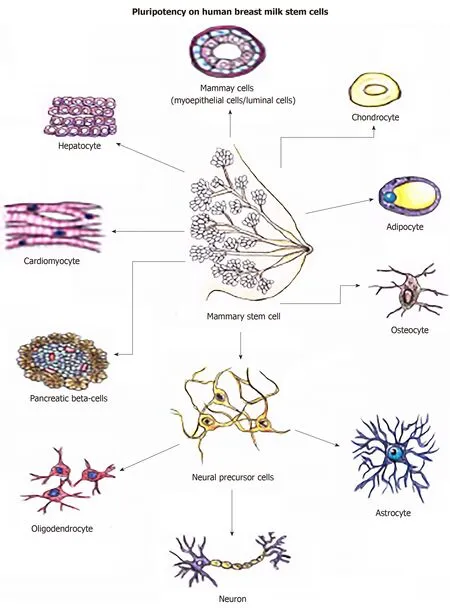
Figure 4 Multilineage potential of hBSCs.
ARTICLE HIGHLIGHTS
Research background
Mesenchymal stem cells are pluripotent cells that have the ability to generate cells from a cell line or in other cell types from different tissues but from the same origin; although those cells have a more limited differentiation capacity than embryonic stem cells,they are easily obtained from the somatic tissue and can be grown in large quantities.This characteristic of undifferentiated stem cells differentiating into different cell lines arouses strategies in regenerative medicine for the treatment of different diseases,such as neurodegenerative diseases.
Research motivation
Mammary epithelium has been the focus of studies due to its remarkable population of human breastmilk stem cells (hBSCs).It will be important to evaluate the scientific literature as to the potential for differentiation of these cells for regenerative medicine.
Research objectives
The main objective was to evaluate the cell differentiation capacity of hBSCs for the three germ layers through a systematic review.
Research methods
The searched databases were PubMed,EMBASE,OVID,and COCHRANE LIBRARY; the inclusion criteria were all papers that:(1) addressed the plasticity of stem cells derived from human breastmilk,that is,the ability of the cell to differentiate into more than one cell line; (2)published between 2007 and 2018 in the English language; and (3) werein vitrostudies for the analysis of the "cell differentiation potential" in the literature using the keywords "HUMAN BREASTMILK," "STEM CELLS," and keywords combined with the Boolean operator "NOT" used to exclude those articles that had the word "CANCER" and their respective synonyms,which were previously consulted according to the medical subject heading terms.PRISMA 2009 guidelines were followed in this study.
Research results
A total of 315 titles and abstracts of articles were examined.From these,21 were in common with more than one database; remaining 294 articles.Out of that total,5 publications met the inclusion criteria.When analyzing the publications,it was demonstrated that human breastmilk stem cells have a high cellular plasticity,exhibiting the ability to generate cells of all three germ layers:endoderm,mesoderm and ectoderm,demonstrating their stemness.Those cells expressed the genes,TRA-1-60/81,octamer-binding transcription factor 4,and NANOG; NANOG was the gene most highly expressed,which is a regulator for self-renewing and its maintenance.Those cells have the ability to differentiatein vitro:adipocytes,chondrocytes,osteocytes,oligodendrocytes,astrocytes,neurons as well hepatocytes,β-pancreatic cells,and cardiomyocytes.
Research conclusions
The hBSCs expressed the genes,TRA-1-60/81,NANOG,and Oct4.The pluripotentiality of hBSCs has been demonstrated by its NANOG expression,which is a regulator for self-renewal and its maintenance.This study opens the possibilities to use this source of adult stem cells for tissue engineering and cellular therapy.hBSCs demonstrated their stemness by high cellular plasticity and exhibiting the ability to generate cells of all three germ layers.
Research perspectives
There will be great potential of hBSCs for tissue engineering and cellular therapy,but more studies for a safe translation will be needed.The single cell analysis seems to be the best method for the next step to evaluate the genetic stability of hBSCs.
杂志排行
World Journal of Stem Cells的其它文章
- Unexpected encounter of the parasitic kind
- Colon cancer stemness as a reversible epigenetic state: Implications for anticancer therapies
- CRISPR/Cas system: An emerging technology in stem cell research
- Cytokine interplay among the diseased retina,inflammatory cells and mesenchymal stem cells - a clue to stem cell-based therapy
- Developments in cell culture systems for human pluripotent stem cells
- Monitoring maturation of neural stem cell grafts within a host microenvironment
