Interaction between stress responses and circadian metabolism in metabolic disease☆
2017-05-07ZhoYngHyunbeKimArushnAliZeZhengKezhongZhng
Zho Yng,Hyunbe Kim,Arushn Ali,Ze Zheng,Kezhong Zhng,b,c,*
aCenter for Molecular Medicine and Genetics,Wayne State University School of Medicine,MI,USA
bDepartment of Microbiology,Immunology,and Biochemistry,Wayne State University School of Medicine,MI,USA cKarmanos Cancer Institute,Wayne State University School of Medicine,Detroit,MI,USA
1.Introduction
The circadian rhythm is the biological process that orchestrates behavior and physiology in most living organisms over a 24 h period.In mammals,circadian oscillations are generated by a network of clock-controlled genes(CCGs)that form a transcriptional auto-regulatory feedback loop.Several inter-connected transcriptional and post-translational negative feedback loops play vital roles in generating and maintaining circadian rhythms.The core circadian transcription factors CLOCK and aryl hydrocarbon receptor nuclear translocator-like protein-1(BMAL1)exist as a heterodimer and constitute the positive arm of the molecular clock.Genes that are directly regulated by the CLOCK/BMAL1 heterodimer are referred to as first-order CCGs.1,2The core clock regulators also drive circadian expression of many transcription factors,thus extending and enhancing their regulatory functions.Expression of theBmal1gene is regulated by the nuclear receptor RORα/γ and PPARγ coactivator-1α (PGC-1α),3while the major negative regulator ofBmal1mRNA expression is REV-ERBα(also known as NR1D1).4Other circadian proteins,including period 1(PER1),PER2,PER3,cryptochrome 1(CRY1),and CRY2,which are targets of BMAL1,also play roles in the negative regulation ofBmal1expression.4The components of the core clock are also regulated by post-translational modifications,including phosphorylation,acetylation,deacetylation,and ubiquitination.5
The mammalian circadian clock orchestrates diverse physiological processes by synchronizing with the nervous system,cardiovascular system,immune response,and metabolic homeostasis.The master clock oscillators residing in the suprachiasmatic nuclei(SCN)of the anterior hypothalamus orchestrate the cascade of events that control physiological rhythms and ensure temporal coordination of metabolism and behavior through the synchronization of peripheral oscillators.6The circadian oscillators in peripheral organs,such as the liver and kidney,respond differently to entraining signals and control specific physiological outputs,thus producing the coordinated response required to regulate cellular and physiological functions.In particular,circadian rhythm and liver metabolism are intrinsically linked in order to synchronize the storage and utilization of energy with the light-dark cycle.7The integration of circadian rhythm and hepatic energy metabolism is mediated through reciprocal crosstalk between these two regulatory networks.
Dysregulation of the circadian rhythm in humans is closely associated with the development of metabolic diseases,including non-alcoholic fatty liver disease(NAFLD),obesity,and type 2 diabetes(T2D).Previous work has demonstrated intimate and reciprocalinteraction between the circadian clock system and fundamental metabolic pathways.1,8,9Clockgene mutations or BMAL1 de ficiency impair lipid and glucose metabolism,evidenced by the development of hyperlipidemia,hepatic steatosis,and defective gluconeogenesis in CLOCK-or BMAL1-de ficient animals.10-13A survey of nuclear receptor mRNA pro files in metabolic tissues suggested that approximately half of the known nuclear receptors and transcriptional regulators exhibit rhythmic expression.14Bioinformatic analysis of genome-wide and phase-specific DNA-binding by the core circadian transcriptional oscillators indicated that BMAL1-binding sites are associated with carbohydrate and lipid metabolism.15In the liver,nuclear receptors and other transcription factors can be induced by metabolites or hormones,and therefore,they may serve as direct links between metabolic pathways and the circadian control of gene expression.For example,the nuclear receptor peroxisome proliferator-activated receptorα (PPARα),which binds fatty acid ligands,serves as a sensor of nutrient and energy status to temporally entrain peripheral clocks.PPARαand BMAL1 reciprocally regulate each other to provide a feedback loop that integrates lipid metabolism and circadian oscillations.16-18
The clock-controlled nuclear receptors REV-ERBs are key regulators of circadian lipid biosynthesis in the liver,and their ablation causes hepatic steatosis through de-repression of lipogenesis.19,20PGC-1αalso provides a link between the clock and energy metabolism,as PGC-1αexpression varies rhythmically and is able to stimulate expression ofBmal1and nuclear receptors of the ROR family.3Recent studies have shown that the nuclear receptor small heterodimer partner(SHP)is also critical for the control of REV-and ROR-mediated neuronal PAS domain protein 2(NPAS2)expression in NAFLD,and of the nuclear receptor FOXA1 expression in alcoholic fatty liver disease(AFLD).21,22Additionally,the clock component CRY1 functions as a circadian regulator of hepatic gluconeogenesis.23By interacting with the Gsαsubunit of G proteins,CRY1 can temporally regulate glucagon signaling,thereby activating hepatic gluconeogenesis.Furthermore,recent evidence implicates distinct signaling pathways synchronized by the circadian clock in circadian metabolism at the translational or posttranscriptional level.24,25For example,the circadian clock synchronizes the rhythmic activation of the primary endoplasmic reticulum(ER)stress sensor IRE1α(inositol requiring enzyme 1).24Lack of a functional circadian clock disrupts the rhythmic activation of the IRE1αpathway,leading to impaired lipid metabolism through aberrant activation of the sterol-regulated SREBP transcription factors.
2.ER stress response and circadian metabolism
In mammalian cells,the ER is an essential organelle that is responsible for protein folding and assembly,synthesis of sterols and lipids,and calcium storage and homeostasis.26-28As a protein folding compartment,the ER plays a crucial role in cellular protein quality control by ensuring only correctly folded proteins are transported to their final destinations,and by assembling incorrectly folded proteins into native complexes and degrading them.When cellular or environmental stressors disrupt ER function,unfolded or misfolded proteins accumulate in the ER lumen,a condition referred to as ER stress.To deal with ER stress,cells activate a sophisticated and coordinated series of signal transduction pathways known as the unfolded protein response(UPR).The UPR is a survival response which modi fies transcriptional and translational programs to restore ER homeostasis.The UPR pathways in mammals are primarily initiated by three major cell stress sensors:IRE1α,double-stranded RNA activated protein kinase-like ER kinase(PERK),and activating transcription factor 6(ATF6).Under ER stress conditions,PERK phosphorylates the serine51 residue on the alpha subunit of eIF2α,which attenuates the translation,thus reducing the workload of the ER.In addition,IRE1α,a bifunctional transmembrane protein,functions as a ribonuclease(RNase)to splice the mRNA encoding X-box binding protein(XBP1),leading to the transcriptional reprogramming of stressed cells.Also under ER stress,ATF6 translocates into the Golgi apparatus,where it is processed into its active form by the site-1 protease(S1P)and site-2 protease(S2P).Upon proteolytic activation,ATF6 travels to the nucleus,where it induces the expression of UPR target genes by binding to ER stress response elements(ERSEs)located within their promoter regions.It is important to note that the UPR pathway can also be triggered by in flammatory stimuli,chemical toxicity,infection by pathogens,and even by the differentiation of specialized cell types,such as the differentiation of B-lymphocytes into antibody-secreting plasmocytes.29,30
A number of enzymes involved in liver metabolism are located in the ER membranes.Lipid metabolism is associated with the physiological UPR,and circadian clock circuitry in fluences hepatic lipid metabolism and ER-localized enzymes.The UPR plays a key role in maintaining hepatic lipid homeostasis,and disruption of the UPR leads to hepatic steatosis and non-alcoholic steatohepatitis(NASH)under conditions of pathophysiological stress.Deletion or suppression of the UPR transducer IRE1αor ATF6 in mouse liver results in hepatic steatosis under acute ER stress or after consumption of a high fat diet.31-34Animals with a defect in the liverenriched,ER-tethered stress sensor cAMP response element binding protein-hepatic specific(CREBH)develop severe NASH when fed an atherogenic high fat diet.35-40Additionally,de ficiency of the ER stress-induced transcriptional activator XBP1 in the liver also leads to hepatic lipid accumulation.41-43Thus,a wealth of evidence indicates that UPR pathways are required to prevent hepatic lipid accumulation and the development of NAFLD under acute ER stress or chronic pathophysiological stress conditions.
ER stress and UPR signaling have signi ficant impacts on circadian activity.A recentstudy has demonstrated that activation of the IRE1α-mediated UPR pathway exhibits circadian rhythmicity in mouse liver.24Disruption of the circadian clock perturbs the circadian rhythmicity of IRE1α-mediated UPR activation and provokes deregulation of ER-localized enzymes(Fig.1).This disruption causes aberrant activation of sterol regulatory element-binding protein(SREBP)transcription factors and impaired hepatic lipid metabolism,which can potentially lead to the development of metabolic disorders.
Another example of an interaction between ER stress and circadian metabolism is the integrative role of the liver-enriched ER stress sensor CREBH in circadian regulation and hepatic energy metabolism(Fig.1).39,40The proteolytic activation of CREBH protein,but not expression ofCrebHmRNA,exhibits typical circadian rhythmicity in mouse liver.This process is controlled by the core clock oscillator BMAL1 and the AKT/glycogen synthase kinase 3β(GSK3β)signaling pathway.40Importantly,GSK3β-mediated phosphorylation of CREBH within its b-ZIP domain modulates the association between CREBH and coat protein complex II(COPII),present in the vesicles responsible for protein transport fromthe ER to the Golgi,and thus controls ER-to-Golgi transport and subsequent proteolytic cleavage of CREBH in a circadian fashion(Fig.1).
In addition to proteolytic cleavage,the post-translational modification(lysine acetylation)of CREBH protein also exhibits a circadian rhythm in the liver.39Functionally,CREBH regulates circadian expression of key genes involved in triglyceride(TG)and fatty acid(FA)metabolism,and is required to maintain circadian variation in blood TG and FA levels.37,40In mice during the light period,CREBH rhythmically regulates and interacts with the hepatic nuclear receptor PPARαto regulate expression of the key enzymes or regulators that mediate FA oxidation,TG lipolysis,and ketogenesis for energy utilization(Fig.1),while during the dark period,CREBH interacts with liver X receptorα (LXRα)to regulate expression of genes encoding functions involved inde novolipogenesis for energy storage.The coordination between CREBH and the circadian clock is also evidenced by the rhythmic interactions between CREBH and the circadian oscillation activator DBP and the repressor E4BP4 in CREBH-regulated CREBH transcriptional activity(Fig.1).Genetic defects in CREBH or impairment in CREBH activity can dysregulate lipid homeostasis,leading to NAFLD and/or hyperlipidemia.35-37,40This has been con firmed in human patients with hypertriglyceridemia,who exhibit a high rate of nonsense mutations or accumulation of rare genetic variants in theirCREBHgenes.44-46
CREBH is also required to maintain circadian homeostasis of hepatic glycogen storage and blood glucose levels.39It achieves this by regulating rhythmic expression of the genes encoding the ratelimiting enzymes for glycogenolysis and gluconeogenesis,including liver glycogen phosphorylase(PYGL),phosphoenolpyruvate carboxykinase 1(PCK1),and the glucose-6-phosphatase catalytic subunit(G6PC).CREBH de ficiency leads to lower blood glucose levels but higher hepatic glycogen levels in mice during the light period.
Circadian disruption also leads to dysregulated UPR signaling in pancreaticβcells;an intact circadian clock is required forβcells to adapt to cellular stress.Disruption of circadian control leads to oxidative stress,ER stress,and impairedβcell function.Both oxidative stress and ER stress contribute to impaired mitochondrial function andβcell failure.47Indeed,acute or chronic sleep deprivation in mice leads to ER stress in the pancreas of aged mice,and circadian disruption worsens unresolved ER stress,leading toβcell apoptosis,in diabetes-prone human islet amyloid polypeptide transgenic rats.48
3.Oxidative stress and circadian metabolism
Oxidative metabolism is a major source of reactive oxygen species(ROS).Cells have evolved many mechanisms to prevent the potential damage caused by ROS,including upregulation of antioxidant enzymes and free radical non-glutathione systems.During the day-night cycle,levels of lipid peroxidation and protein oxidation,the major components of oxidative stress responses,exhibit circadian rhythmicity in key metabolic organs or tissues,such as liver and muscle.49-52Many antioxidative enzymes,such as glutathione peroxidase(GPx),glutathione reductase(GR),superoxide dismutase(SOD),peroxiredoxins(Prxs),and melatonin,display circadian rhythmicity in their levels of expression or activity,evidencing a circadian oscillation of oxidative and antioxidative stress responses(Fig.2).ROS scavenging systems are critical for the function and survival of metabolic cells,and there is a growing body of evidence that impairment in redox regulation and circadian rhythms can lead to metabolic disorders.53Oxidative stress plays a key role in the initiation and progression of NAFLD.ROS cause lipid peroxidation,followed by in flammatory responses and activation of stellate cells during fibrogenesis in NAFLD patients.54
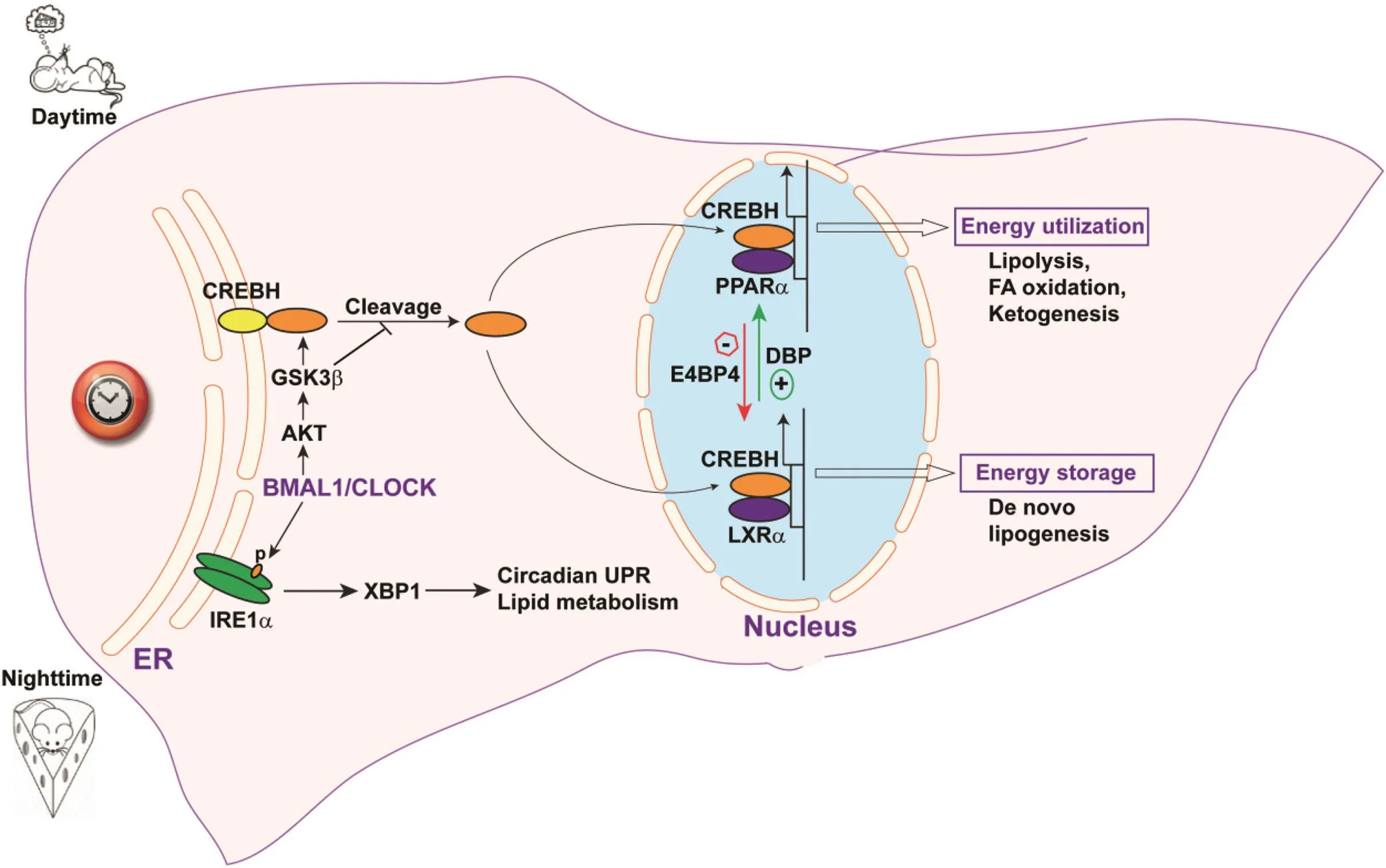
Fig.1.Integration of circadian rhythm and stress signaling in the endoplasmic reticulum regulates lipid metabolism.Activation of the ER-tethered stress sensors CREBH and IRE1α is regulated by the circadian clock.ER-to-Golgi transport and proteolytic cleavage of CREBH protein is regulated by the circadian clock through the BMAL1-AKT-GSK3β regulatory axis.Activated CREBH translocates to the nucleus,where it interacts with circadian-regulated nuclear receptors(PPARαand LXRα)or transcriptional modulators(DBP and E4BP4)to regulate expression of genes involved in lipid utilization(FA oxidation,TG lipolysis,and ketogenesis)or storage(de novo lipogenesis)pathways.The circadian clock also regulates activation of the IRE1α-XBP1 UPR pathway,which regulates the UPR and potentially metabolic pathways.Abbreviations:ER,endoplasmic reticulum;DBP,D-site-binding protein;E4BP4,E4 promoter-binding protein 4;GSK3β,glycogen synthase kinase 3 β;AKT,protein kinase B;LXRα,liver X receptorα.
Several molecular regulators of the interactions between circadian rhythm and the oxidative stress responsehave been identi fied.In particular,the stress-induced nicotinamide adenine dinucleotide(NAD+)-dependent protein deacetylase sirtuin 1(SIRT1)is linked to both the oxidative stress response and circadian rhythms.SIRT1 is involved in the core circadian mechanism because it deacetylates the circadian oscillation proteins BMAL1 and PER2(Fig.2).55,56SIRT1 mediates deacetylation of BMAL1,thereby resetting the clock to its original state.SIRT1 can also deacetylate PER2,targeting it for degradation,and subsequently preventing formation of PER2-CRY heterodimerization in the negative feedback regulation of the core circadian mechanism.
The coenzyme NAD+also plays critical roles in regulating the oxidative stress response and circadian metabolism.NAD+and its redox partner NADH are important in redox reactions,in which the phosphorylatedformsofNAD+/NADH,NADP+/NADPH,drive oxidation-reduction reactions.57Circadian oscillation of NADH/NADPH directly regulates DNA binding of the core circadian oscillation heterodimers CLOCK/BMAL1 and NPAS2/BMAL1 to their transcriptional targets,while NAD+/NADP+inhibit this activity(Fig.2).58Therefore,cellular oxidative stress and the general redox state affect the redox state of NAD+/NADH and NADP+/NADPH and circadian rhythms.Furthermore,protein deacetylation by SIRT1 involves the use of NAD+as a cofactor;57therefore,as a coenzyme ofSIRT1,NAD+functionsasakeyregulatorofcircadian metabolism.
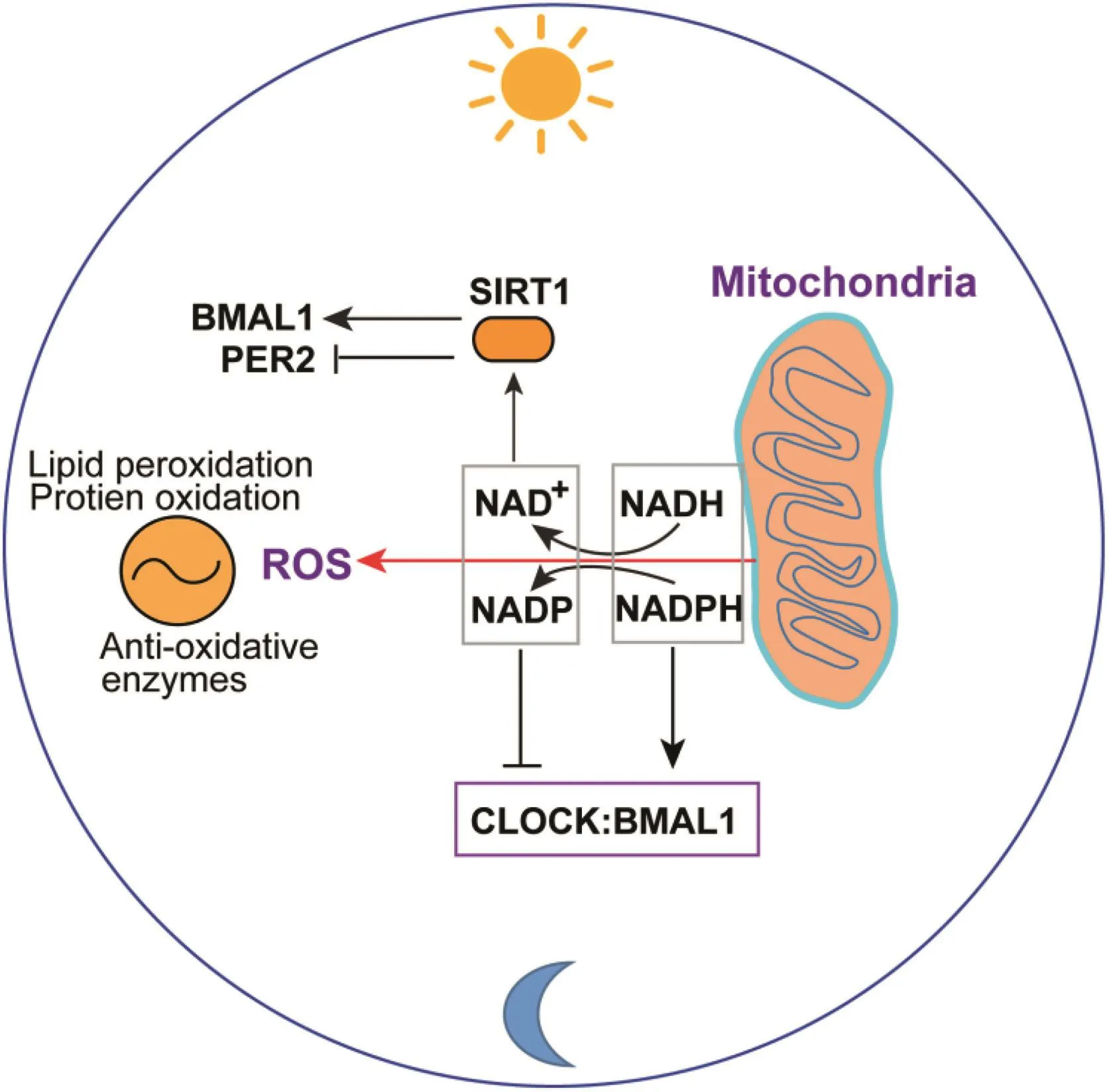
Fig.2.Interaction between oxidative stress and circadian rhythm.The circadian clock in fluences mitochondrial metabolism,leading to ROS production and changes in NADP/NADPH and NAD+/NADH ratios through oxidative phosphorylation.ROS-associated oxidative and anti-oxidative stress responses exhibit circadian rhythms because of circadian-regulated expression of anti-oxidative enzymes or oxidation of lipids and proteins.NAD+/NADH ratio modulates SIRT1 activity,which controls BMAL1 or PER2 activity through deacetylation.Furthermore,NAD+/NADP and NADH/NADPH ratios control the activity of the core circadian oscillator BMAL1/CLOCK heterodimer by modulating its binding to circadian effector genes.Abbreviations:PER2,period circadian clock 2;NAD,nicotinamide adenine dinucleotide;NADP,nicotinamide adenine dinucleotide phosphate;NADH,nicotinamide adenine dinucleotide(reduced);NADPH,nicotinamide adenine dinucleotide phosphate(reduced).
Circadian redox state has a major in fluence on the function and survival of pancreaticβcells,because they have a poor tolerance for oxidative stress due to low expression of antioxidant genes.Expression of the genes encoding many key ROS scavenging enzymesare transcriptionally regulated by the anti-oxidative response regulator Nrf2.Nrf2 expression peaksin the late light/early dark phase inβcells and is directly regulated by the circadian clock,59asBmal1can bind to the E-box element in the Nrf2 gene promoter.Indeed,the circadian expression of Nrf2 is lost inBmal1-/-βcells.47,59Disruption ofBmal1leads to defects inβcell stimulation-secretion coupling,lower ATP production,uncoupling of oxidative phosphorylation,and impaired glucose-induced insulin secretion.Genetic disruption and environmental stressors can induce oxidative stress,which is mediated by a disruption of the direct transcriptional control bythe core molecularclock andBmal1on Nrf2,the master antioxidative transcription factor inβcells.
The link between oxidative stress and circadian rhythmicity in liver metabolism is evidenced by the rhythmicity of mitochondrial dynamics and energy homeostasis.Recent studies have revealed that mitochondrial dynamics(mitochondrial fission and mitophagy)and biogenesis are transcriptional targets of the circadian regulator BMAL1 in mouse liver,and exhibit circadian rhythmicity in synchronization with diurnal energy demands.60Liver-specificBmal1knockout(LBmal1KO)mice accumulate oxidative damage in the liver and develop hepatic insulin resistance.Restoration of hepatic BMAL1 activity in high fat diet-fed mice improves metabolic outcomes.These results suggest that clock genes are evolutionarily conserved regulators of cellular energetics.
4.In flammatory responses and circadian metabolism
Chronic disruption of circadian timing during shift work or chronic jetlag leads to a higher risk of several pathologies.Many of these conditions in both shift workers and experimental models share the common risk factors of in flammation.61Impaired circadian rhythm is linked to in flammatory responses that are associated with the development of metabolic diseases.Uncoupling of peripheral and master clock gene rhythms by a reversed feeding cycle leads to exacerbated in flammatory responses in mouse models of polymicrobial sepsis.62In particular,when mice were fed during the day,rather than the night,it was found that serum levels of in flammatory cytokines,including interleukin 6(IL-6),tumor necrosis factor α (TNFα),IL-1α,IL-9,eotaxin,and granulocyte colony-stimulating factor,were higher in mice fed during the day than in those fed at night,after cecal ligation-puncture to induce sepsis.Consistent with a greater in flammatory response,expression levels of the anti-oxidative regulators SIRT1 and PPARγwere lower in the livers of day-fed mice.This study demonstrates that the uncoupling of circadian clock rhythmicity by an inverted feeding cycle exacerbates in flammatory responses in the liver.
However,in flammation can also cause dysregulation of circadian rhythms.Following a pro-in flammatory response triggered by TNF-α,the enzyme USP2a deubiquitinates and stabilizes the circadian protein CRY1.63USP2a is regulated by the circadian clock and interacts with CRY1 in the liver to enhance its stability via deubiquitination,which leads to repression of hepaticPer2gene expression.Interestingly,the pro-in flammatory cytokine TNF-α also increases CRY1 protein expression and inhibits circadian gene expression in an USP2a-dependent fashion,suggesting that USP2a maymediate circadian disruption bysuppressingCRY1 degradation during in flammation.In addition,physical and in flammatory stressors,including forced swimming,immobilization,and lipopolysaccharide injection,increased expression of the mouse circadian clock genePer1in the hypothalamic paraventricular nucleus(PVN),further demonstrating that in flammatory stressors can modulatecircadianregulationthroughupregulation ofPer1expression.64
5.Dysregulated circadian metabolism in NAFLD
NAFLD represents a spectrum of disorders ranging from relatively benign and reversible steatosis to end stage liver disease(cirrhosis).It is characterized by the accumulation of TG in hepatocytes due to an imbalance between hepatic synthesis and breakdown of lipids,and between fatty acid storage and disposal.It causes liver enlargement and manifests in the absence of alcohol ingestion.There are three distinct sources of FAs in the liver:the diet,lipolysis,andde novolipogenesis.The liver uses or shunts the excess FA by re-esteri fication to TG and storage as lipid droplets,βoxidation or export as low-density lipoprotein(VLDL).Thus,hepatic fat deposition can occur as a consequence of lower oxidation or export of lipid and/or greater synthesis.65Liver metabolic pathways are regulated by circadian biological clocks,and hepatic health is maintained by proper timing of circadian patterns of metabolic gene expression,with the alternation of anabolic processes corresponding to the feeding/active state during wakefulness,and catabolic processes characterizing the fasting/resting state during sleep.66
The pathogenesis of NAFLD is complex and multifactorial;therefore,both genetic and/or environmental disruptors may affect the circadian clock,leading to the development of metabolic diseases or exacerbation of pathological states,and indeed it has been shown that common genetic variations in theClockgene are associated with NAFLD in humans.Clockgene polymorphisms and haplotypes involving the CLOCK transcription factor are correlated with the susceptibility and severity of NAFLD.67Modern lifestyle demands force large numbers of people into asynchrony between actual time and their circadian clocks,resulting in a constant state of social jetlag.Recent findings have indicated that interactions between altered energy metabolism and disruptions in the circadian clock can lead to the development of metabolic disorders.In human NAFLD patients,disruption of the sleep-wake cycle,manifesting as daytime lethargy,is positively associated with biochemical and histologic surrogates of NAFLD severity and insulin resistance.Recent studies have demonstrated that inadequate,poor quality sleep is linked with NAFLD risk in middle-aged people.Moreover,a pilot study has shown favorable effects of using melatonin,a circadian hormone,as an antioxidant that ameliorates NAFLD.68Food intake is now more common during the night.NASH is the potential outcome of a relatively benign and reversible condition,hepatic steatosis,and is characterized by hepatocyte injury and tissue in flammation due to oxidative stress and recruitment of in flammatory mediators,such as cytokines.It has been shown that a high fat diet increases the incidence of NASH in adulthood,associated with altered cellular redox status,reduced sirtuin abundance,and desynchronized clockgene expression.69
Disruption of normal circadian rhythmicity represents an independent risk factor for hepatocellular carcinoma(HCC)in experimental animals,which has revealed opposing roles for the nuclear receptors FXR and constitutive androstane receptor(CAR)in disease progression from NAFLD to HCC.70Studies have also shown that chronic circadian misalignment,independent of germ line mutation and dietary manipulation,promotes the development of malignant hepatoma;for example,chronic jet-lag induces spontaneous HCC in wild-type mice.71This process commences with the development of simple steatosis,which progresses to steatohepatitis and fibrosis,before the onset of HCC.This pathophysiological pathway is driven by jetlag-induced genome-wide gene dysregulation and metabolic dysfunction in the liver,with nuclear receptor-controlled cholesterol/bile acid and xenobiotic metabolism among the highly dysregulated pathways.A study performed on jetlagged mice revealed higher expression of genes required for glycogenolysis,amino acid and lipid synthesis and storage,oxidative stress,glycolysis,and hepatocyte proliferation and death,together with suppression of genes responsible for stimulating tumor suppression,DNA damage repair,glycogen synthesis,β-oxidation,and FA transport.71
The deregulation of vital circadian genes(Per1,Cry1,Clock and Bmal1),together with dysregulation of molecular markers of HCC,highlights the importance of the circadian homeostasis of liver metabolism in the suppression of hepatocarcinogenesis.In addition,ablation of farnesoid X receptor dramatically increases enterohepatic bile acid levels and jetlag-induced HCC,while loss of CAR,a well-known promoter of the development of liver tumors that mediates toxic bile acid signaling,inhibits NAFLD-induced hepatocarcinogenesis.71,72Circadian disruption activates CAR by promoting cholestasis,peripheral clock disruption,and sympathetic dysfunction.The association of dysregulated circadian rhythm with HCC was con firmed by the observation that chronic circadian dysfunction disrupts liver gene expression and metabolism,promoting NAFLD and cancer.73
6.Summary and future perspectives
Dysregulation of circadian rhythms is closely associated with human cardio-metabolic diseases,such as T2D,cardiovascular disease,and NAFLD.Due to modern lifestyles,the problem of irregular circadian rhythm is not limited to shift workers,but has a wide impact on public health.The circadian rhythm aligns molecular signal transduction networks with physiological activities.While the central clock is set by theClockgene,circadian oscillators in peripheral organs,particularly in the liver,can be entrained by metabolic changes or stress signals,providing the flexibility to adapt to environmental challenges.However,under chronic stress conditions,circadian metabolism is dysregulated,which represents a signi ficant risk factor for the development of metabolic diseases.
For future research,it is still critical to understand the connections between stress responses and circadian rhythms for the maintenanceofmetabolichomeostasis.Although signi ficant progress has been made,the mechanisms by which environmental or intracellular stressors modulate circadian metabolism,and their roles in the progression of metabolic disorders,remain to be fully elucidated.In particular,recent studies have suggested that the rhythmicity of the human microbiome may affect metabolic homeostasis.74,75Feeding time has been shown to shape the rhythmicity and composition of the intestinal microbiota,and disruption of the microbiome is associated with diet,obesity,and metabolic disease.Controlling the circadian rhythmicityof human microbiota,through changes in diet and lifestyle or by the use of drugs,may represent a novel intervention for the treatment of metabolic diseases.As we gain a greater understanding of the molecular basis and the extent to which in flammatory stress responses modulate circadian metabolism,it might be possible to design novel therapies to minimize the deleterious metabolic consequences of circadian disruptions by targeting the activation of stress-associated circadian regulators and mediators.
Authors'contributions
Z.Yang,A.Ali and K.Zhang wrote the manuscript;H.Kim,Z.Zheng and K.Zhang performed related research;Z.Yang,H.Kim,Z.Zheng and K.Zhang edited the manuscript.
Con flict of interest
The authors declare that they have no con flict of interest.
Acknowledgements
This research was supported by USA National Institutes of Health(NIH)grants DK090313,ES017829,and AR066634(to K.Zhang),American HeartAssociation Grants0635423Z and 09GRNT2280479(to K.Zhang).A.Ali is an undergraduate student at the University of Houston who attended the Summer Undergraduate Research Program at Dr.K.Zhang’s lab at the Center for Molecular Medicine and Genetics of Wayne State University School of Medicine.
1.Bass J,Takahashi JS.Circadian integration of metabolism and energetics.Sci-ence.2010;330:1349-1354.
2.Hughes ME,DiTacchio L,Hayes KR,et al.Harmonics of circadian gene transcription in mammals.PLoS Genet.2009;5:e1000442.
3.Liu C,Li S,Liu T,Borjigin J,Lin JD.Transcriptional coactivator PGC-1alpha integrates the mammalian clock and energy metabolism.Nature.2007;447:477-481.
4.Ukai H,Ueda HR.Systems biology of mammalian circadian clocks.Annu Rev Physiol.2010;72:579-603.
5.Mehra A,Baker CL,Loros JJ,Dunlap JC.Post-translational modifications in circadian rhythms.Trends Biochem Sci.2009;34:483-490.
6.Reppert SM,Weaver DR.Molecular analysis of mammalian circadian rhythms.Annu Rev Physiol.2001;63:647-676.
7.Asher G,Schibler U.Crosstalk between components of circadian and metabolic cycles in mammals.Cell Metab.2011;13:125-137.
8.Green CB,Takahashi JS,Bass J.The meter of metabolism.Cell.2008;134:728-742.
9.Rutter J,Reick M,McKnight SL.Metabolism and the control of circadian rhythms.Annu Rev Biochem.2002;71:307-331.
10.Rudic RD,McNamara P,Curtis AM,et al.BMAL1 and CLOCK,two essential components of the circadian clock,are involved in glucose homeostasis.PLoS Biol.2004;2:e377.
11.Lamia KA,Storch KF,Weitz CJ.Physiological signi ficance of a peripheral tissue circadian clock.Proc Natl Acad Sci U S A.2008;105:15172-15177.
12.Turek FW,Joshu C,Kohsaka A,et al.Obesity and metabolic syndrome in circadian Clock mutant mice.Science.2005;308:1043-1045.
13.Shimba S,Ogawa T,Hitosugi S,et al.De ficient of a clock gene,brain and muscle Arnt-like protein-1(BMAL1),induces dyslipidemia and ectopic fat formation.PloS One.2011;6:e25231.
14.Yang X,Downes M,Yu RT,et al.Nuclear receptor expression links the circadian clock to metabolism.Cell.2006;126:801-810.
15.Rey G,Cesbron F,Rougemont J,Reinke H,Brunner M,Naef F.Genome-wide and phase-specific DNA-binding rhythms of BMAL1 control circadian output functions in mouse liver.PLoS Biol.2011;9:e1000595.
16.Oishi K,Shirai H,Ishida N.CLOCK is involved in the circadian transactivation of peroxisome-proliferator-activated receptor alpha(PPARalpha)in mice.Biochem J.2005;386:575-581.
17.Inoue I,Shinoda Y,Ikeda M,et al.CLOCK/BMAL1 is involved in lipid metabolism via transactivation of the peroxisome proliferator-activated receptor(PPAR)response element.J Atheroscler Thromb.2005;12:169-174.
18.Canaple L,Rambaud J,Dkhissi-Benyahya O,et al.Reciprocal regulation of brain and muscle Arnt-like protein 1 and peroxisome proliferator-activated receptor alpha de fines a novel positive feedback loop in the rodent liver circadian clock.Mol Endocrinol.2006;20:1715-1727.
19.Feng D,Liu T,Sun Z,et al.A circadian rhythm orchestrated by histone deacetylase 3 controls hepatic lipid metabolism.Science.2011;331:1315-1319.
20.Bugge A,Feng D,Everett LJ,et al.Rev-erbαand Rev-erbβcoordinately protect the circadian clock and normal metabolic function.Genes Dev.2012;26:657-667.
21.Lee SM,Zhang Y,Tsuchiya H,Smalling R,Jetten AM,Wang L.Small heterodimer partner/neuronal PAS domain protein 2 axis regulates the oscillation of liver lipid metabolism.Hepatology.2015;61:497-505.
22.Tsuchiya H,da Costa KA,Lee S,et al.Interactions between nuclear receptor SHP and FOXA1 maintain oscillatory homocysteine homeostasis in mice.Gastroenterology.2015;148:1012-1023.
23.Zhang EE,Liu Y,Dentin R,et al.Cryptochrome mediates circadian regulation of cAMP signaling and hepatic gluconeogenesis.Nat Med.2010;16:1152-1156.
24.Cretenet G,Le Clech M,Gachon F.Circadian clock-coordinated 12 Hr period rhythmic activation of the IRE1alpha pathway controls lipid metabolism in mouse liver.Cell Metab.2010;11:47-57.
25.Mauvoisin D,Wang J,Jouffe C,et al.Circadian clock-dependent and-independent rhythmic proteomes implement distinct diurnal functions in mouse liver.Proc Natl Acad Sci U S A.2014;111:167-172.
26.Zhang K,Kaufman RJ.From endoplasmic-reticulum stress to the in flammatory response.Nature.2008;454:455-462.
27.Zhang K,Kaufman RJ.Signaling the unfolded protein response from the endoplasmic reticulum.J Biol Chem.2004;279:25935-25938.
28.Ron D,Walter P.Signal integration in the endoplasmic reticulum unfolded protein response.Nat Rev Mol Cell Biol.2007;8:519-529.
29.Zhang K,Wong HN,Song B,Miller CN,Scheuner D,Kaufman RJ.The unfolded protein response sensor IRE1alpha is required at 2 distinct steps in B cell lymphopoiesis.J Clin Invest.2005;115:268-281.
30.Reimold AM,Iwakoshi NN,Manis J,et al.Plasma cell differentiation requires the transcription factor XBP-1.Nature.2001;412:300-307.
31.Zhang K,Wang S,Malhotra J,et al.The unfolded protein response transducer IRE1alpha prevents ER stress-induced hepatic steatosis.EMBO J.2011;30:1357-1375.
32.Wang S,Chen Z,Lam V,et al.IRE1α-XBP1s induces PDI expression to increase MTP activity for hepatic VLDL assembly and lipid homeostasis.Cell Metab.2012;16:473-486.
33.Rutkowski DT,Wu J,Back SH,et al.UPR pathways combine to prevent hepatic steatosis caused by ER stress-mediated suppression of transcriptional master regulators.Dev Cell.2008;15:829-840.
34.Yang L,Calay ES,Fan J,et al.METABOLISM.S-Nitrosylation links obesityassociated in flammation toendoplasmicreticulum dysfunction.Science.2015;349:500-506.
35.Kim H,Mendez R,Chen X,Fang D,Zhang K.Lysine acetylation of CREBH regulates fasting-induced hepatic lipid metabolism.Mol Cell Biol.2015;35:4121-4134.
36.Kim H,Mendez R,Zheng Z,et al.Liver-enriched transcription factor CREBH interacts with peroxisome proliferator-activated receptorαto regulate metabolic hormone FGF21.Endocrinology.2014;155:769-782.
37.Zhang C,Wang G,Zheng Z,et al.Endoplasmic reticulum-tethered transcription factor cAMP responsive element-binding protein,hepatocyte specific,regulates hepatic lipogenesis,fatty acid oxidation,and lipolysis upon metabolic stress in mice.Hepatology.2012;55:1070-1082.
38.Zhang K,Shen X,Wu J,et al.Endoplasmic reticulum stress activates cleavage of CREBH to induce a systemic in flammatory response.Cell.2006;124:587-599.39.Kim H,Zheng Z,Walker PD,Kapatos G,Zhang K.CREBH maintains circadian glucose homeostasis by regulating hepatic glycogenolysis and gluconeogenesis.Mol Cell Biol.2017:37.pii:e00048-17.
40.Zheng Z,Kim H,Qiu Y,et al.CREBH couples circadian clock with hepatic lipid metabolism.Diabetes.2016;65:3369-3383.
41.Liu X,Henkel AS,LeCuyer BE,Schipma MJ,Anderson KA,Green RM.Hepatocyte X-box binding protein 1 de ficiency increases liver injury in mice fed a high-fat/sugar diet.Am J Physiol Gastrointest Liver Physiol.2015;309:G965-G974.
42.Herrema H,Zhou Y,Zhang D,et al.XBP1s is an anti-lipogenic protein.J Biol Chem.2016;291:17394-17404.
43.Jiang S,Yan C,Fang QC,et al.Fibroblast growth factor 21 is regulated by the IRE1alpha-XBP1 branch of the unfolded protein response and counteracts endoplasmic reticulum stress-induced hepatic steatosis.J Biol Chem.2014;289:29751-29765.
44.Johansen CT,Wang J,McIntyre AD,et al.Excess of rare variants in nongenome-wide association study candidate genes in patients with hypertriglyceridemia.Circ Cardiovasc Genet.2012;5:66-72.
45.Cefalù AB,Spina R,Noto D,et al.Novel CREB3L3 nonsense mutation in a family with dominant hypertriglyceridemia.Arterioscler Thromb Vasc Biol.2015;35:2694-2699.
46.Lee JH,Giannikopoulos P,Duncan SA,et al.The transcription factor cyclic AMP-responsive element-binding protein H regulates triglyceride metabolism.Nat Med.2011;17:812-815.
47.Lee J,Liu R,de Jesus D,et al.Circadian control ofβ-cell function and stress responses.Diabetes Obes Metab.2015;17(Suppl 1):123-133.
48.Gale JE,Cox HI,Qian J,Block GD,Colwell CS,Matveyenko AV.Disruption of circadian rhythms accelerates development of diabetes through pancreatic beta-cell loss and dysfunction.J Biol Rhythms.2011;26:423-433.
49.Lapenna D,De Gioia S,Mezzetti A,et al.Circadian variations in antioxidant defences and lipid peroxidation in the rat heart.Free Radic Res Commun.1992;17:187-194.
50.Fanjul-Moles ML,Prieto-Sagredo J,L'opez DS,Bartolo-Orozco R,Cruz-Rosas H.Cray fish Procambarus clarkii retina and nervous system exhibit antioxidant circadian rhythms coupled with metabolic and luminous daily cycles.Photochem Photobiol.2009;85:78-87.
51.Kanabrocki EL,Murray D,Hermida RC,et al.Circadian variation in oxidative stress markers in healthy and type II diabetic men.Chronobiol Int.2002;19:423-439.
52.Singh R,Singh RK,Mahdi AA,et al.Circadian periodicity of plasma lipid peroxides and other anti-oxidants as putative markers in gynecological malignancies.In Vivo.2003;17:593-600.
53.Wilking M,Ndiaye M,Mukhtar H,Ahmad N.Circadian rhythm connections to oxidative stress:implications for human health.Antioxid Redox Signal.2013;19:192-208.
54.Angulo P.Nonalcoholic fatty liver disease.N Engl J Med.2002;346:1221-1231.
55.Nakahata Y,Kaluzova M,Grimaldi B,et al.The NAD+-dependent deacetylase SIRT1 modulates CLOCK-mediated chromatin remodeling and circadian control.Cell.2008;134:329-340.
56.Asher G,Gat field D,Stratmann M,et al.SIRT1 regulates circadian clock gene expression through PER2 deacetylation.Cell.2008;134:317-328.
57.Houtkooper RH,Cant'o C,Wanders RJ,Auwerx J.The secret life of NAD+:an old metabolitecontrolling new metabolicsignalingpathways.EndocrRev.2010;31:194-223.
58.Rutter J,Reick M,Wu LC,McKnight SL.Regulation of clock and NPAS2 DNA binding by the redox state of NAD cofactors.Science.2001;293:510-514.
59.Lee J,Moulik M,Fang Z,et al.Bmal1 andβ-cell clock are required for adaptation to circadian disruption,and their loss of function leads to oxidative stressinduced beta-cell failure in mice.Mol Cell Biol.2013;33:2327-2338.
60.Jacobi D,Liu S,Burkewitz K,et al.Hepatic Bmal1 regulates rhythmic mitochondrial dynamics and promotes metabolic fitness.Cell Metab.2015;22:709-720.
61.Castanon-Cervantes O,Wu M,Ehlen JC,et al.Dysregulation of in flammatory responses by chronic circadian disruption.J Immunol.2010;185:5796-5805.
62.Oyama Y,Iwasaka H,Koga H,Shingu C,Matsumoto S,Noguchi T.Uncoupling of peripheral and master clock gene rhythms by reversed feeding leads to an exacerbated in flammatory response after polymicrobial sepsis in mice.Shock.2014;41:214-221.
63.Tong X,Buelow K,Guha A,Rausch R,Yin L.USP2a protein deubiquitinates and stabilizes the circadian protein CRY1 in response to in flammatory signals.J Biol Chem.2012;287:25280-25291.
64.Takahashi S,Yokota S,Hara R,et al.Physical and in flammatory stressors elevate circadian clock gene mPer1 mRNA levels in the paraventricular nucleus of the mouse.Endocrinology.2001;142:4910-4917.
65.Dowman JK,Tomlinson JW,Newsome PN.Pathogenesis of non-alcoholic fatty liver disease.QJM.2010;103:71-83.
66.Mazzoccoli G,Vinciguerra M,Oben J,Tarquini R,De Cosmo S.Non-alcoholic fatty liver disease:the role of nuclear receptors and circadian rhythmicity.Liver Int.2014;34:1133-1152.
67.Sookoian S,Casta~no G,Gemma C,Gianotti TF,Pirola CJ.Common genetic variations in CLOCK transcription factor are associated with nonalcoholic fatty liver disease.World J Gastroenterol.2007;13:4242-4248.
68.Bernsmeier C,Weisskopf DM,P flueger MO,et al.Sleep disruption and daytime sleepiness correlating with disease severity and insulin resistance in nonalcoholic fatty liver disease:a comparison with healthy controls.PloS One.2015;10:e0143293.
69.Bruce KD,Szczepankiewicz D,Sihota KK,et al.Altered cellular redox status,sirtuin abundance and clock gene expression in a mouse model of developmentally primed NASH.Biochim Biophys Acta.2016;1861:584-593.
70.Fu T,Zhao X,Evans RM.Liver cancer checks in when bile acid clocks out.Cancer Cell.2016;30:827-828.
71.Kettner NM,Voicu H,Finegold MJ,et al.Circadian homeostasis of liver metabolism suppresses hepatocarcinogenesis.Cancer Cell.2016;30:909-924.
72.Li T,Chiang JY.Nuclear receptors in bile acid metabolism.Drug Metab Rev.2013;45:145-155.
73.Chronic jet lag promotes metabolic dysfunction and hepatocarcinogenesis.Cancer Discov.2017;7:10.
74.Thaiss CA,Zeevi D,Levy M,et al.Transkingdom control of microbiota diurnal oscillations promotes metabolic homeostasis.Cell.2014;159:514-529.
75.Mukherji A,Kobiita A,Ye T,Chambon P.Homeostasis in intestinal epithelium is orchestrated by the circadian clock and microbiota cues transduced by TLRs.Cell.2013;153:812-827.
杂志排行
Liver Research的其它文章
- Interleukin-22 in the pathogenesis and potential treatment of liver diseases☆
- Recent development and gene therapy for glycogen storage disease type Ia☆
- FOXO transcription factors in non-alcoholic fatty liver disease☆
- Long non-coding RNA in liver metabolism and disease:Current status☆
- Decoding the role of extracellular vesicles in liver diseases☆
- Guide for Authors
