Study on related substances in ceftizoxime sodium
2022-08-18MengJunLinShunquanPuHanlinLiJinjuandYuanXiao
Meng Jun,Lin Shun-quan,Pu Han-lin,Li Jin-ju,and Yuan Xiao,4
(1 Zhuzhou Qianjin Pharmaceutical Co.,Ltd.,Zhuzhou 412000; 2 Guangzhou PI & PI Biotech,Inc.Guangzhou 510663;3 Department of Cell Biology,College of Life Science and Technology,Jinan University,Guangzhou 510632; 4 Wuhan Botanical Garden of Chinese Academy of Sciences,Wuhan 430074)
Abstract Objective To study the impurities in different batches of ceftizoxime sodium by HPLC and LC-MS.Methods The accelerated stability test of ceftizoxime sodium was carried out.The impurities in ceftizoxime sodium were detected by HPLC and LC-MS.The related impurities were prepared by preparative HPLC.The structure of each impurity was determined by pop analysis.Results A total of 12 impurities were detected,of which 2 were newly reported for the first time,and the content was high in the preparation.The impurities reported in this report are B (6R,7R)-7-((Z)-2-(2-aminothiazol-4-yl)-2-(methoxyimino)acetamido)-8-oxo-5-thia-1-azabicyclo[4,2,0]oct-3-ene-2-carboxylicacid,C 2-(R)-((Z)-2-(2-aminothiazol-4-yl)-2-(methoxyimino)acetamido)(carboxy)methyl)-3,6-dihydro-2H-1,3-thiazine-4-carboxylic acid,D 2-((R)-((Z)-2-(2-aminothiazoyl)-2-(methoxyimino)acetamido)(carboxyl)methyl)-3,6-dihydro-2H-1,3-thiazine-4-carboxylic acid,F (Z)-2-((2-(2-aminothiazol-4-yl)-2-(methoxyimino)acetamido)methyl)-3,6-dihydro-2H-1,3-thiazine-4-carboxylicacid,G 2-((R)-((E)-2-(2-aminothiazol-4-yl)-2-(methoxyimino)acetamido)(carboxy)methyl)-3,6-dihydro-2H-1,3-thiazine-4-carboxylic acid,H 2-((R)-((E)-2-(2-amino-thiazol-4-yl)-2-(methoxyimino)acetamido)(carboxy)methyl)-3,6-dihydro-2H-1,3-thiazine-4-carboxylic acid,I (6R,7R)-7-((E)-2-(2-aminothiazol-4-yl)-2-(methoxyimino)acetamido)-8-oxo-5-thia-1-azabicyclo[4,2,0]oct-3-ene-2-carboxylic acid,and J (6R)-7-((Z)-2-(2-aminothiazol-4-yl)-2-(methoxyimino)acetamido)-8-oxo-5-thia-1-azabicyclo[4,2,0]oct-2-ene-2-carboxylic acid.Conclusion The analytical method reported in this paper can detect and analyze all kinds of possible common impurities in ceftizoxime sodium.The separation and purification method developed by our laboratory can separate the impurities with purity meeting the requirements of structure confirmation.The structures of these impurities can be accurately characterized by NMR,HRMS,NOE,CD and other analytical methods.
Key words Ceftizoxime sodium; Impurity analysis and detection; Structural analysis
Ceftizoxime sodium is a third-generation semisynthetic cephalosporin developed by Fujisawa Pharmaceutical Co.,Ltd.,which can inhibit the biosynthesis of peptidoglycan of the bacterial cell wall[1].Ceftizoxime sodium is stable to a broad spectrum of β-lactamases including penicillinase and cephalosporinase,which has good bactericidal activity against many Gram-positive and Gram-negative bacteria,such asEscherichia coli,Klebsiella pneumoniae,Proteus mirabilis,etc..Clinically,it is mainly used to treat acute or moderate respiratory infections,sepsis,pneumonia,etc.,with the advantages of high curative effect and few adverse reactions[2-4].However,cephalosporins tend to produce impurities during storage,Moreover,some process impurities and intermediates also tend to remain as impurities in the drug substances and drug products.In view of these problems,a series of studies on impurities of ceftizoxime were carried out,which greatly promoted the quality control of products[5-7],Yao Lei[8]reported the test and verification of high molecular weight impurities in ceftizoxime sodium,An HPLC method for the determination of related substances in ceftizoxime sodium was developed by Yang Qian[9].The type and structure of impurities produced by the forced degradation of ceftizoxime sodium were also reported[10-11].However,some high content of ceftizoxime injection powder impurities were not included in the literature and reports,and the fine structure of some impuritiesis insufficient.To further improve the research of the related substances in the raw material and the preparations of ceftizoxime sodium,impurities of ceftizoximesodium powder injection from Bai Yun Shan Pharmaceutical Factory with different batches have been detected by LC-MS.The detected ceftizoxime impurities were prepared and compared by forced degradation and purification.These impurities obtained in this study are pure products,which can be used as the reference standard for impurities,quality inspection,and quality control of raw materials and ceftizoxime sodium preparations.
1 Materials and methods
1.1 Samples
Cefazoxime sodium was synthesized in Wuhan Zeshancheng biotechnology Co Ltd.,(China),impurities B,C,D,F,G,H,I,J were prepared in the laboratory and identification by MS and NMR.hydrogen peroxide,trifluoroacetic acid,citric acid,sodium hydrogen phosphate,sodium hydroxide and hydrochloric acid were purchased from Tianjin FuChen Chemical Reagent Factory (Analytically pure).DMSO,methanol and acetonitrile were purchased from Merck Co (Chromatographically pure and Special Mass pure).C18separation material was purchased from Jinan Bona Biotech Co.,Ltd.PIPO-02 material was provided by Guangzhou PI & PI Biotech Inc.
1.2 High performance and liquid chromatography(HPLC)
The Thermo U3000 HPLC was used to detect the ceftizoxime sodium raw material and impurities,ThermoHypersil GOLD C18was used as the columns in this experiment.The mobile phase A (buffer salt)was made up by 1.42 g citric acid and 2.31 g sodium dihydrogen phosphate diluting into the water,the mobile phase B was acetonitrile,the mobile phase for the detection was A:B=90:10,UV detection was set to 254 nm and the flow rate was kept at 1 mL/min,the temperature of column oven was set to 30℃ and the date acquisition time was 40 min.
1.3 LC-MS/MS analysis
All mass spectrometry me asurements were performed on a Thermo Scientific QE Orbitrap LCMs.Instrument operating in positive electrospray ion mode.Analysis conditions,column: Thermo Scientific ODS Hypersil(3 μm,150 mm×2.1 mm),scan type:full MS,scan range: 150.0 to 2000m/z; resolution:35000; polarity: positive,maximum inject time: 50 ms;sheath gas flow rate: 30 arb; aux gas flow rate: 10arb;spray voltage:3.8 kV; capillary temperature: 350℃;aux gas heater temperature: 320℃.mobile phase A was 1% formic acid water(V/V) and mobile phase B was acetonitrile.Detection was carried out at 254 nm and the flow rate was kept at 0.4 mL/min.0.1% formic acid water: acetonitrile (50:50) was used as diluent.Data acquisition time was 25min.The gradient program was as follows: time (min)/A(V/V):B(V/V);t0/93:7,t8.0/93:7,t14/70:30,t22/70:30,t23/93:7,andt25/93:7.
1.4 NMR analysis
Nuclear magnetic resonance (NMR) spectra were recorded on a Bruker NMR Avance Ⅲ 500MHz superconducting spectrometer with TMS as an internal standard (DMSO-d6as the solvent,actual exposure frequency: 125.76 MHz,Decoupling field frequency:500.13 MHz,δ: 0~210ppm,analysis of the results by Mnova software)
1.5 ECD(Electronic circular dichroism) calculation of D(OP1) and B(OP2)
ECD calculation for the optimized conformers was carried out utilizing time-dependent DFT methods at the[B3LYP/6-311+g(2d,p)] level in the acetonitrile-water(50:50) solution by Gaussian 09 program,the calculated ECD spectra of D and B were compared with the experimental ones and the ECD curves were generated by Origin 8.0 Software.
1.6 preparation of the impurities
Impurities B,C,D,F,G,H,I and J were prepared in the laboratory and purified by preparative chromatography.The impurities were obtained by freeze-drying after desalination.
2 Results and discussions
2.1 Impurity detection
Impurities of ceftizoxime sodium for injection were detected by LC-MS,the results were given in Tab.1 and Fig.1.The mobile phase consisted of (A) 0.1%formic acid,and (B) acetonitrile.In the detection and analysis process,twelve ceftizoxime sodium impurities were obtained,and impurities B,H,C,I,D,F were first reported by our team.
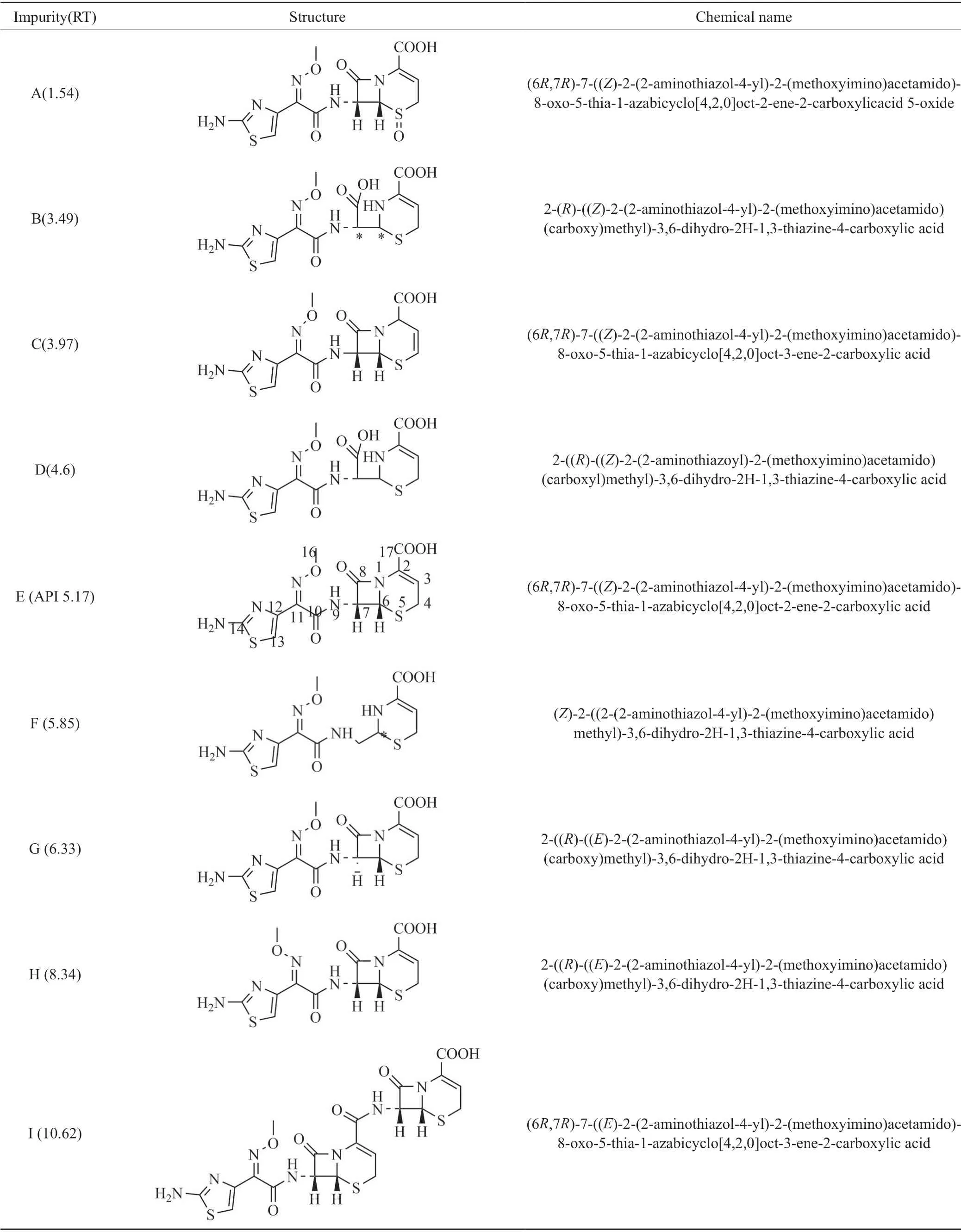
Tab.1 Chemical structureofceftizoxime sodium and their impurities表1头孢唑肟钠及其杂质列表
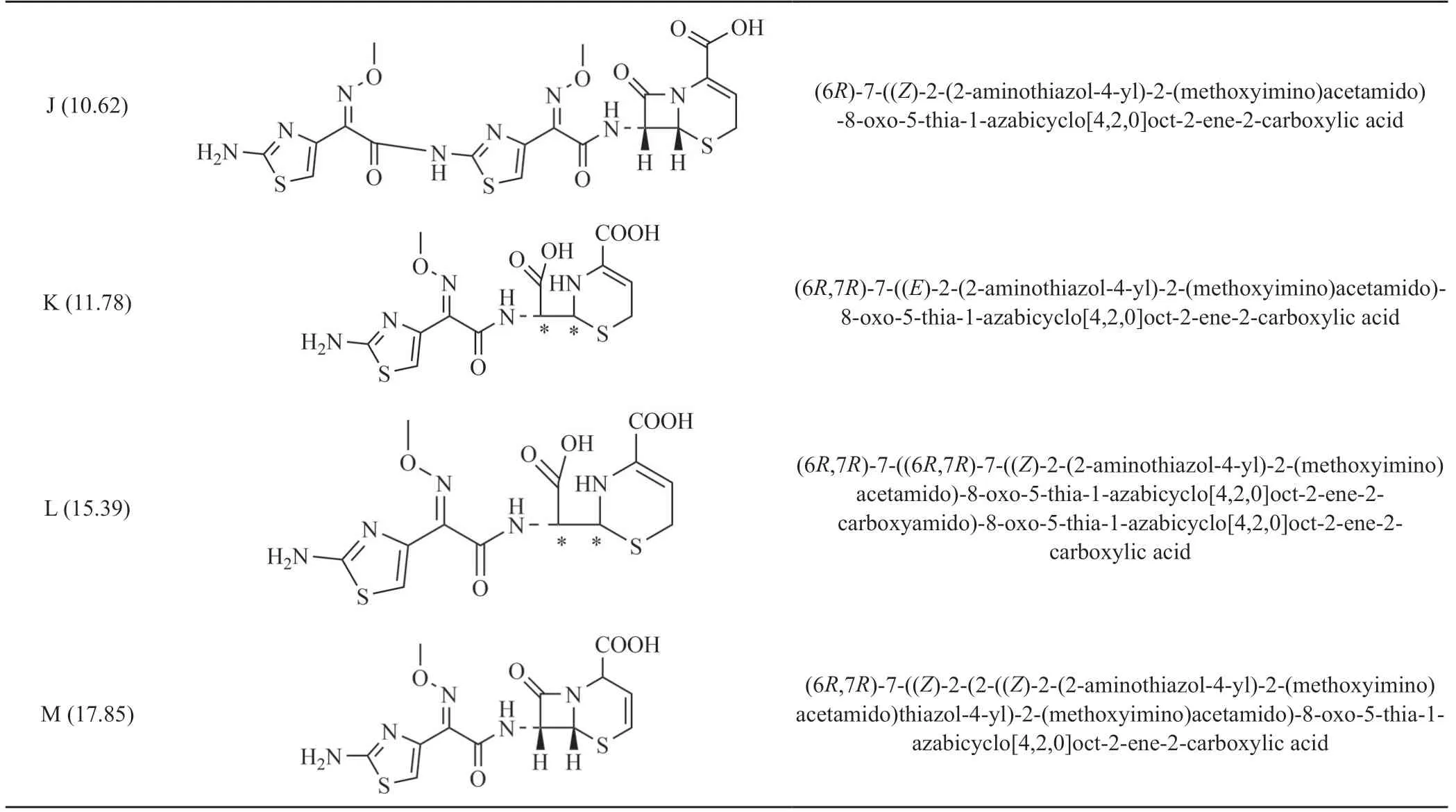
续表1
2.2 Structural elucidation
The carbon and hydrogen spectrum data of various impurities are listed in Tab.2 and Tab.3.

Tab.2 13C NMR chemical shift of the impurities表2杂质的碳谱数据
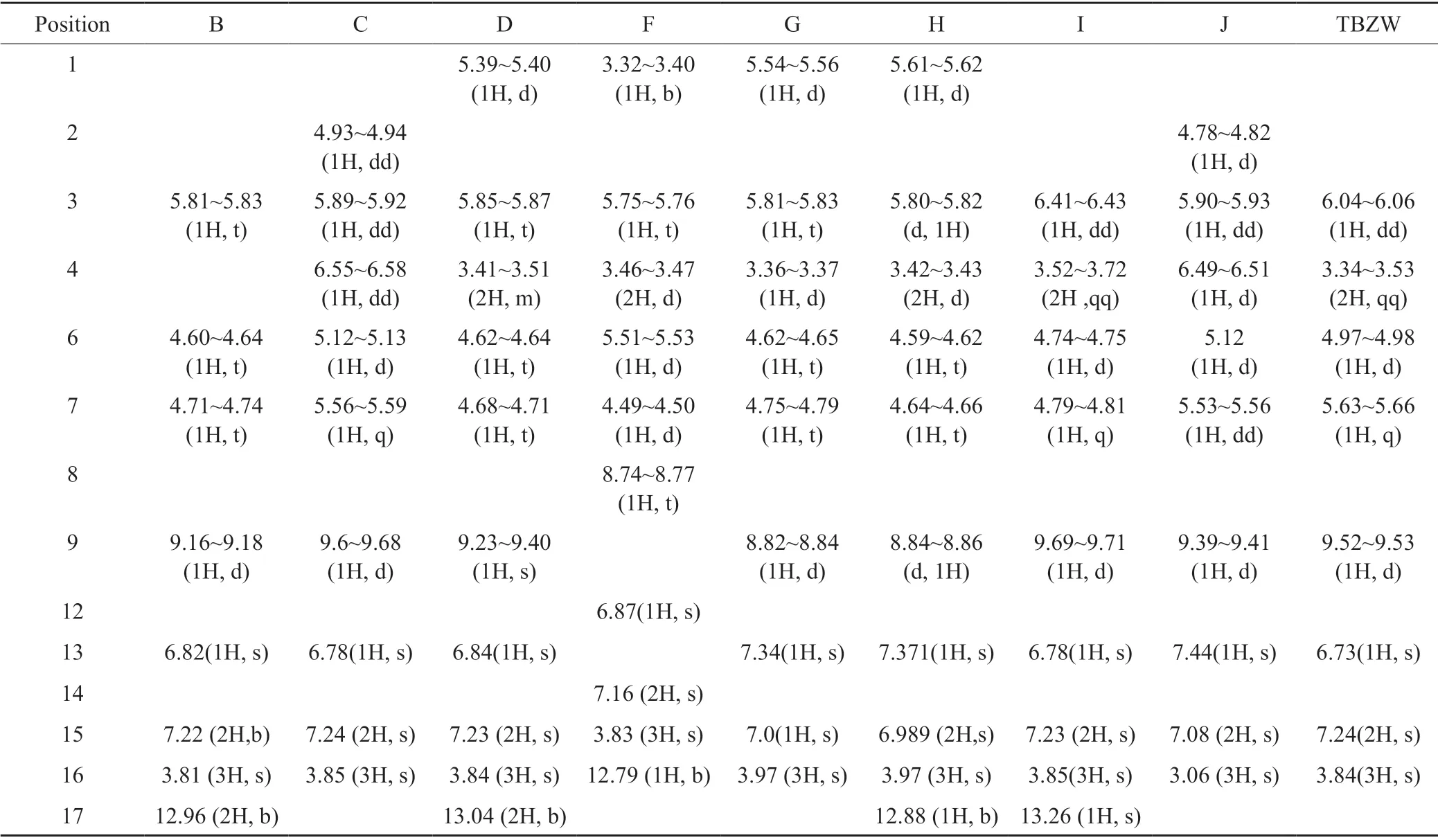
Tab.3 1H NMR chemical shift of the impurities表3杂质的氢谱数据
2.2.1 Impurity B and H
Impurity B (RT3.49) and impurity H(RT8.34)exhibited a molecular ion peak atm/z: 402.05 [M+H]+in positive ion mode in HRMS,its exact molecular weight is 401.05,an increase of 18 amu compared with ceftizoxime,comparison1H NMR and13C NMR spectra of impurity B and H with ceftizoxime,it is easy to see that B and H are ring opening isomeric impurities[12-14].B was named as 2-((R)-((Z)-2-(2-aminothiazol-4-yl)-2-(methoxyimino)acetamido)(carboxy)methyl)-3,6-dihydro-2H-1,3-thiazine-4-carboxylic acid.Impurity H(RT 8.34) can be obtained by exposing impurity B to 254nm UV lamp,impurity H was named as 2-((R)-((E)-2-(2-aminothiazol-4-yl)-2-(methoxyimino)acetamido)(carboxy)methyl)-3,6-dihydro-2-H-1,3-thiazine-4-carboxylic acid.
2.2.2 Impurity C and I
Impurity C (RT 3.97) and impurity I(RT 10.62)exhibited a molecular ion peak atm/z: 384.04 [M+H]+in positive ion mode in HRMS,which is equal to the mass of ceftizoxime,the exact molecular weight of them are 383.03,which indicating that impuity C,I and ceftizoxime are isomers.Comparison the1H NMR and13C NMR spectra data of ceftizoxime sodium with impurity C and I the chemical shifts of C-3 / C-4 in impurity C,I and its neighboring C-2 / C-6 / C-7 are significantly different from those of ceftizoxime,one CH2in the1H NMR of the impurity C,I becomes =CH,the carbon-carbon double bond of the six-membered ring in ceftizoxime molecule was transferred from C-2/C-3 to C-3/C-4,the impurity C was characterized as (6R,7R)-7-((Z)-2-(2-aminothiazol-4-yl)-2-(methoxyimino)acetamido)-8-oxo-5-thia-1-azabicyclo[4.2.0]oct-3-ene-2-carboxylic acid,impurity I was characterized as (6R,7S)-7-((Z)-2-(2-aminothiazol-4-yl)-2-(methoxyimino)acetamido)-8-oxo-5-thia-1-azabicyclo[4.2.0]oct-2-ene-2-carboxylic acid.
2.2.3 Impurity D and G
Impurity D(RT 4.6) and impurity G(RT 8.34)exhibited a molecular ion peak atm/z: 402.05 [M+H]+in positive ion mode in HRMS,the exact molecular weight of D,G was 401.05,an increase of 18 amu compared with ceftizoxime,this is equivalent to adding a molecule of water.Comparison1H NMR and13C NMR spectra of impurity D,G with ceftizoxime,the value of C-8 chemical shift of impurity D and G increased obviously,it may be related to the hydroxyl added to the carbonyl group (C-8) after the cleavage of the amide bond (C-8/N-1),in addition,1H NMR spectrum show one hydrogen atom was added on N-1,in addtion the chemical shift values of H-6 / H-7 / H-9 were significantly decreased,this also indicates that the β-lactam ring in the ceftizoxazole molecule has been opened.Therefore D was characterized as 2-(R)-((Z)-2-(2-aminothiazol-4-yl)-2-(methoxyimino)acetamido)(carboxy)methyl)-3,6-dihydro-2H-1,3-thiazine-4-carboxylic acid.Impurity G can be obtained by exposing impurity D to 254 nm UV lamp.G was characterized as 2-((R)-((E)-2-(2-aminothiazol-4-yl)-2-(methoxyimino)cetamido)(carboxy)methyl)-3,6-dihydro-2H-1,3-thiazine-4-carboxylic acid[13-14].
2.2.4 Impurity F
Impurity F(RT 5.85) exhibited a molecular ion peak atm/z: 358.05 [M+H]+in positive ion mode in HRMS,its molecular weight was 357.06,which was 26 amu less than that of ceftizoxime.Comparison1H NMR and13C NMR spectra of F with ceftizoxime,the impurity F reduced a carbonyl group and added two hydrogen atoms,indicating that the carbonyl group on the β-lactam ring was lost and N-1 / C-7 in the molecule were both added one hydrogen atom,impurity F was characterized as(Z)-2-((2-(2-aminothiazol-4-yl)-2-(methoxyimino)acetamido)methyl)-3,6-dihydro-2H-1,3-thiazine-4-carboxylic acid.
2.2.5 Impurity J
Impurity J(RT 10.62) exhibited a molecular ion peak atm/z: 384.04 [M+H]+in positive ion mode in HRMS,its exact molecular weight is 383.03,which is the same as ceftizoxime,revealing that they are isomers.Comparison the chemical shifts of1H NMR and13C NMR of the impurity J with ceftizoxime,only the chemical shift value of H-7 in the impurity J decreased more significantly from 5.63 to 4.79,the H-6 / H-7 coupling constant of impurity J became smaller (H-6: from 5.0Hz to 2.5Hz,H-7: from 5.0Hz to 2.0Hz),showing that the correlation between H-6 / H-7 in J was reduced,there was no significant correlation between H-6 / H-7 in the compound molecule(Fig.2),which demonstrated that the structure of impurity J is (6R,7S)-7-((Z)-2-(2-aminothiazol-4-yl)-2-(methoxyimino)acetamido)-8-oxo-5-thia-1-azabicyclo[4.2.0]oct-2-ene-2-carboxylic acid.
2.2.6 Determination of C-6 / C-7 configuration in ring opening impurities
Ring opening impurities D(OP1) and B(OP2) may have two enantiomers,6R7R,6S7Sand 6R7S,6S7R.the ECD calculationfor the optimized conformers of 6R7R,6S7Sand 6R7S,6S7Rwere carried out by means of time-dependent density functional theory (TDDFT)methods at the [B3LYP/6-311+g(2d,p)] level in the acetonitrile-water (50:50) solution by using Gaussian 09 software.All the ECD curves of D and B were weighted by a Boltzmann distribution of each conformer.The calculated ECD spectra of D and B were subsequently compared with the experiment ones[15],the simulated curves of 7R6Rand 7S6Sare more similar to the ECD spectral curves of D and B than that of 7S6Rand 7R6S.the ring openimpurity D corresponds to the 7R6Rconfiguration,ring open Impurity B corresponds to 7S6Sconfiguration(Fig.3~4).
Based on the NMR data of the related substances,each carbon atom and hydrogen atom in the impurity molecule were assigned.The Gauss model may simulate the stable molecular configuration of ceftizoxime and several ring open impurities,and measure the relative atomic distance and dihedral angle values.It shows that the 7R6R,7S6Sis relatively high stability configuration(adjacent atoms have the largest coupling constant when the dihedral angle is 0 or 180),Maxwell-Bolzman correlation coefficient distribution curve calculated NMR data of each impurity is obtained by Gauss program.The results also support that the ring open structure is the tendency of 7R6Rand 7S6Sgreater than 7S6Rand 7R6S[16-17].The circular dichroism of ceftizoxime,D,and B were also measured and performed circular dichroism modeling of different configurations with Gaussian calculations(Tab.4)[18].The simulated curves of 7R6Rand 7S6Sare more similar to the CD spectral curves of D and B then that of 7S6Rand 7R6S(Fig.5).NMR data of impurity D and B were assigned too[19].
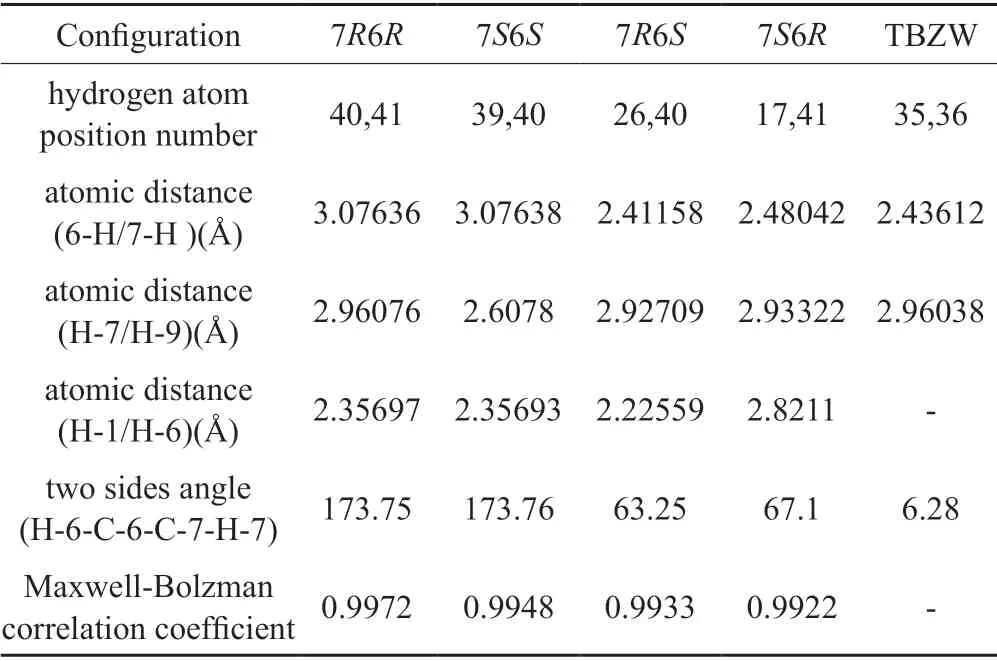
Tab.4 The calculation data of the analog configuration of the ring open impurity表4开环杂质构型模拟计算数据
3 Conclusions
The impurities in the preparation of ceftizoxime sodium injection were tested,separated,and characterized in this study.In particular,six of these impurities were first reported,which included ring open impurities with high content in the preparations and double bond transfer impurities.In this paper,the exact molecular weight and chemical structure of theseimpurities were confirmed by LC-MS,NMR,HRMS.The exact configurations of impurities D and B were deduced by ECD calculation.
Acknowledgements
The authors would like to gratefully acknowledge Yan Sujun of Chemistry Department of Sun Yat-sen University,who has contributed the ECD calculation.The authors also wish to thank Zhou Mengyi of the Department of chemistry of Jinan University for the calculation of the dominant conformation of two open loop impurities.
