Testosterone therapy reduces hepatic steatosis in men with type 2 diabetes and low serum testosterone concentrations
2022-07-02RossApostolovEmilyGianattiDarrenWongNumanKutaibaPaulGowMathisGrossmannMarieSinclair
Ross Apostolov, Emily Gianatti, Darren Wong, Numan Kutaiba, Paul Gow, Mathis Grossmann, Marie Sinclair
Ross Apostolov, Darren Wong, Paul Gow, Marie Sinclair, Department of Gastroenterology and Liver Transplant Unit, Austin Health, Heidelberg 3084, VIC, Australia
Ross Apostolov, Paul Gow, Mathis Grossmann, Marie Sinclair, Department of Medicine, University of Melbourne, Parkville 3010, VIC, Australia
Emily Gianatti, Department of Endocrinology, Fiona Stanley Hospital, Murdoch 6150, WA, Australia
Numan Kutaiba, Department of Radiology, Austin Health, Heidelberg 3084, VIC, Australia
Mathis Grossmann, Department of Endocrinology, Austin Health, Heidelberg 3084, VIC, Australia
Abstract BACKGROUND Non-alcoholic fatty liver disease (NAFLD) is highly prevalent in people with diabetes with no available treatment.AIM To explore the effect of testosterone treatment on liver. Testosterone therapy improves insulin resistance and reduces total body fat, but its impact on the liver remains poorly studied.METHODS This secondary analysis of a 40 wk, randomised, double-blinded, placebocontrolled trial of intramuscular testosterone undecanoate in men with type 2 diabetes and lowered serum testosterone concentrations evaluated the change in hepatic steatosis as measured by liver fat fraction on magnetic resonance imaging (MRI).RESULTS Of 88 patients enrolled in the index study, 39 had liver MRIs of whom 20 received testosterone therapy and 19 received placebo. All patients had > 5% hepatic steatosis at baseline and 38 of 39 patients met diagnostic criteria for NAFLD. Median liver fat at baseline was 15.0% (IQR 11.5%-21.1%) in the testosterone and 18.4% (15.0%-28.9%) in the placebo group. Median ALT was 34units/L (26-38) in the testosterone and 32units/L (25-52) in the placebo group. At week 40, patients receiving testosterone had a median reduction in absolute liver fat of 3.5% (IQR 2.9%-6.4%) compared with an increase of 1.2% in the placebo arm (between-group difference 4.7% P < 0.001). After controlling for baseline liver fat, testosterone therapy was associated with a relative reduction in liver fat of 38.3% (95% confidence interval 25.4%-49.0%, P < 0.001).CONCLUSION Testosterone therapy was associated with a reduction in hepatic steatosis in men with diabetes and low serum testosterone. Future randomised studies of testosterone therapy in men with NAFLD focusing on liver-related endpoints are therefore justified.
Key Words: Hepatic steatosis; Non-alcoholic fatty liver disease; Non-alcoholic steatohepatitis; Testosterone therapy; Testosterone undecanoate; Type 2 diabetes
INTRODUCTION
Non-alcoholic fatty liver disease (NAFLD) is defined by the presence of hepatic steatosis on imaging or histology in the absence of other secondary causes of hepatic steatosis[1]. It has an estimated overall global prevalence of 25%[2]. Patients with type 2 diabetes are at significantly higher risk of NAFLD due to a bidirectional pathophysiological link between the two disease entities[3,4]. A meta-analysis of 19 observational studies found that patients with NAFLD had a higher incidence of type 2 diabetes than those without, and that the incidence of type 2 diabetes further increased in patients with radiologically higher steatosis scores[5]. Furthermore, type 2 diabetes and features of the metabolic syndrome are known to be independent risk factors for liver fibrosis progression, cirrhosis, hepatocellular carcinoma and death in patients with NAFLD[6,7].
Low serum testosterone has been associated with an increased risk of NAFLD in men after adjustment for other metabolic risk factors[8-10]. A meta-analysis of 13721 men from cross-sectional, cohort and case-control studies reported that serum total testosterone (TT) concentrations were on average 2.8nmol/L lower in men with NAFLD than those without11].[Subsequently, in a study of 159 men with NAFLD, lower testosterone concentrations were associated with a higher risk for the presence, and increasing severity, of non-alcoholic steatohepatitis, a recognised risk factor for liver disease progression in NAFLD[12].
Testosterone therapy has been shown to ameliorate hepatic steatosis and necroinflammation in animal models of male hypogonadism induced by castration [13,14] Only a small number of studies have examined the effect of exogenous testosterone in men with NAFLD and low testosterone concentrations, and these report mixed results. A placebo controlled study of obese men with severe obstructive sleep apnoea and testosterone concentrations that ranged from low normal to normal demonstrated that testosterone therapy reduced liver fat as measured by computed tomography[15]. A study of 21 men with low serum testosterone concentrations and NAFLD reported that treatment with a novel oral testosterone prodrug improved liver fat as measured by magnetic resonance imaging proton density fat fraction (MRI-PDFF) in 81% of patients[16]. Two small studies of testosterone therapy in men with type 2 diabetes and low testosterone concentrations and one in men with mobility limitation and low testosterone concentrations conversely showed no significant change in hepatic fat compared to placebo as measured by MRI[17-19].
The aim of this study was to evaluate the effect of testosterone therapy on liver fat fraction and to determine other factors associated with changes in liver fat in a population of men with low testosterone concentrations and type 2 diabetes.
MATERIALS AND METHODS
Study design
The study design, eligibility and protocol of the trial is described in full in the original publication[20]. The trial was registered on the Australian and New Zealand Clinical Trials registry (trial number NCT00613782). This was a 40 wk, randomised, double-blind placebo-controlled trial conducted at single tertiary referral centre in Australia. Ethics approval for the study was granted by the Human Research Ethics Committee, Austin Health.
Eligibility
Eligible participants were men aged 35-70 years with a history of type 2 diabetes and a fasting, early morning TT concentration of ≤ 12.0 nmol/L (346 ng/dL), as measured by electrochemiluminescence immunoassay (ECLIA) and averaged across two readings.
Exclusion criteria for the trial were testosterone therapy within five years of randomisation, screening TT concentrations < 5.0 nmol/L (144 ng/dL), established pituitary or testicular disorder, luteinising hormone level > 1.5x upper limit of normal, prostate-specific antigen level > 4 µg/L, a history of urinary obstruction, prostate cancer or breast cancer, haematocrit > 0.50, uncontrolled hypertension (> 160/90 mmHg despite treatment), untreated obstructive sleep apnoea, estimated glomerular filtration rate < 30 mL/min, cardiac insufficiency, active malignancy, unstable psychiatric disease, weight > 135 kg, use of glucagon-like peptide-1 agonist therapy or very low-calorie diet, or an HbA1c level > 8.5% (69 mmol/mol).
Randomisation and study intervention
Eligible participants were randomly assigned in a concealed 1:1 allocation to either testosterone or placebo therapy. Intramuscular testosterone undecanoate 1000 mg or a visually identical placebo was administered at 0, 6, 18 and 30 wk. Participants were followed up for a total of 40 wk.
Outcomes and measurements:
In this post-hoc analysis we assessed as the primary outcome the change in liver fat fraction, as measured using MRI liver in-phase (IP) and opposed-phase (OP) T1 sequences and expressed as percentage fat. The primary outcome measure for the index study was the change across groups and time from baseline in the homeostasis model assessment index of insulin resistance using a computerbased calculation rather than the original linear equation.
Clinical and biochemical variables that were assessed in participants at baseline and 40 wk included body weight, body mass index (BMI), waist circumference, alanine aminotransferase (ALT), gammaglutamyl transferase, alkaline phosphatase, bilirubin, albumin, international normalised ratio, TT, sex hormone-binding globulin, calculated free testosterone (cFT), fasting glucose, HbA1c and lipid profile. Free testosterone levels were calculated using Vermeulen’s formula, as previously described[21]. Additional measurements of total body mass, lean mass and fat mass were assessed by DXA scan (DXA Prodigy, Version 10.51; GE Lunar, Madison, WI) at 0 and 40 wk.
MRI assessment of liver fat
MRI scans of the abdomen were obtained at enrolment and after 40 wk of therapy using a 3 Tesla MRI scanner (Siemens, Erlangen, Germany). As part of the initial study, subcutaneous and visceral adipose tissue volume was calculated for each patient by analysis of five 10-mm slices around the L4 vertebral superior endplate using the SliceOmatic program software (version 4.2; Tomovision, Montreal, Canada) as previously described[20].
MRI images obtained before and after therapy were reanalysed for the present study by an expert liver radiologist who was blinded to treatment allocation. Liver fat fraction before and after therapy was calculated using conventional chemical shift imaging with IP and OP T1 sequences. An averaged signal intensity for IP and OP was obtained from three 5 to 10 cm2regions within hepatic parenchyma of each scan. The fat fraction was calculated using the formula (IP - OP)/2 × IP. This technique has been previously validated to accurately estimate liver fat fraction to levels of up to 50%[22-27].
Statistical analysis
Continuous data are displayed as mean (standard deviation) for normally distributed data or median [interquartile range] for skewed data. Categorical data are presented as number (percentage). The Studentt-test and Mann-Whitney test were used for normal and non-normal data, respectively. Exploratory data analysis included pairwise examination for correlation that may introduce multicollinearity into the final regression model. Linear regression modelling was used to identify variables associated with week 40 Liver fat proportion, which was the primary outcome variable, whilst controlling for baseline liver fat proportion (analysis of covariance). The outcome variable was natural log-transformed for these analyses as use of the untransformed values violated model assumptions. Univariate regression with all clinically relevant variables was performed and those withPvalues < 0.20 were selected for inclusion in the multivariable model. A manual elimination process was undertaken to arrive at the final model. The coefficients in the final model were back transformed to report relative change in liver fat proportion between groups (geometric mean). Standard regression diagnostics were performed to ensure non-violation of the underlying model assumptions. Analyses were performed in R v4.02[28].
RESULTS
Baseline characteristics
A total of 39 men with type 2 diabetes and low serum testosterone were included in our analysis, of whom 20 received testosterone therapy and 19 received placebo. They represent a subset of 88 patients from the original trial who underwent MRI liver scanning both at the start and end of treatment. Remaining study participants did not undertake MRI scans either due to incompatible or unverifiable metal implants, inability to fit in the MRI machine or claustrophobia.
The baseline characteristics of our study participants are shown in Table 1 and are similar between the testosterone and placebo therapy group with the exception of lean mass which was lower in the testosterone group and HDL which was higher in the testosterone group compared to placebo. Compared to the participants in the index study who did not have MRI scans, our cohort had lower baseline BMI, visceral adiposity, waist circumference and higher cholesterol levels, but otherwise had comparable baseline characteristics as shown in Table 2. All patients had hereditary haemochromatosis and other causes of low testosterone excluded and no patients were taking medications were taking steatogenic medications during the study period. Five participants in the testosterone group and three in the placebo group were taking glucagon-like peptide-1 analogues or thiazolidinediones at baseline which continued throughout the study at stable doses. No other patients were taking medications known to directly influence liver fat (Supplementary Table 1). Median alcohol consumption was 3.5 (IQR 0-8.5) standard drinks per week in the testosterone group and 4 (IQR 0.5-7) standard drinks per week in the placebo group. One patient in the testosterone group had heavy alcohol consumption (defined as > 14 units per week), reporting 28 standard drinks per week during the study period. No patients in the placebo group had heavy alcohol consumption.

Table 1 Baseline characteristics of study participants

Table 2 Baseline characteristics of our participants who had magnetic resonance imaging scans compared to index study participants who did not have magnetic resonance imaging scans
All patients in the study had significant hepatic steatosis, defined as a liver fat fraction ≥ 5%. All patients but the one patient with heavy alcohol consumption had secondary causes of hepatic steatosis excluded and hence met diagnostic criteria for NAFLD as defined by the American Association for the Study of Liver Diseases[1]. Median liver fat fraction at baseline was 15.0% (IQR 11.5%-21.1%) in the testosterone group and 18.4% (IQR 15.0%-28.9%) in the placebo group (P= 0.14). The median values of all liver function tests were within normal limits for both groups at baseline (Table 1).
Descriptive analysis of change in hepatic steatosis
In the testosterone group, the median absolute reduction in liver fat fraction was 3.5% (IQR 2.9%-6.4%) and median relative reduction was 27.3% (IQR 18.0%-37.6%). Liver fat fraction increased in the placebo group, with a median absolute increase in liver fat fraction of 1.2% (IQR -2.6-3.0%) and median relative increase of 6.8% (IQR -7.3-15.3%). At week 40, the median liver fat fraction was 9.8% (IQR 8.6%-13.5%) in the testosterone group and 19.6% (IQR 17.8%-29.1%) in the placebo group.
The change in absolute liver fat ranged from -15.3% to +3.6% in the testosterone group, with 18 of 20 individuals achieving a reduction in liver fat (Figure 1). The change in absolute liver fat ranged from -5.9% to +27.5% in the placebo group, with eight of 19 individuals achieving a reduction in liver fat (Figure 1). One patient from the placebo group was an outlier who had a 27.5% absolute increase in liver fat associated with a 32% increase in visceral adipose tissue (VAT) and a 7% increase in body weight. At week 40, there were no significant changes in liver function tests in either treatment group.
Factors associated with change in liver fat
Univariate regression analysis found that testosterone therapy, BMI, baseline liver fat and week 40 TT and cFT concetrations were all significantly associated with changes in liver fat. Week 40 cFT and TT concentrations were, however, highly correlated ( hence a decision to use only cFT in the development of the multivariate model.
The multivariate model was significant (F(2,35)(54.13),R2= 0.807,P <0.001) and included treatment arm controlled for baseline liver fat proportion. In detail, those in the testosterone treatment group had a significantly lower follow-up liver fat proportion than those in the placebo group (P <0.001). The Beta co-efficient of -0.48 (-0.67, -0.29) is on the log-transformed scale. Back-transformation of the co-efficient and associated 95% confidence interval returned values of -0.383 (-0.490, -0.254). Thus, in this model, testosterone therapy was associated with a 38.3% relative reduction in liver fat proportion (95% confidence interval 25.4% to 49.0% reduction) after controlling for baseline liver fat proportion compared to those receiving placebo. No other covariates reduced the unexplained residual variance in the model significantly to this estimate of between-group difference.
Changes in testosterone concentrations
Baseline median TT concentrations as measured by ECLIA were 9.8 [7.2-12.00] nmol/L and 7.8 [5.9-10.8] nmol/L in the testosterone and placebo groups, respectively. After 40 wk of therapy, trough TT concentrations increased to 12.9 [11.9-15.0] nmol/L in the testosterone group and 8.8 [7.3-10.8] nmol/L in the placebo group (P <0.001). Baseline median cFT concentrations were 216 [151-272] pmol/L and 176 [146-224] pmol/L in the testosterone and placebo groups respectively. After 40 wk of therapy cFT increased to 322 [231-392] pmol/L in the testosterone group and 193 [167-250] pmol/L in the placebo group (P <0.001).
Changes body weight, glycaemic control, liver function tests and body composition:
At 40 wk patients receiving testosterone therapy had no statistically significant changes in overall body weight, BMI, waist circumference, fasting glucose or HbA1c compared to placebo (Table 3).

Table 3 Changes in body composition and metabolic parameters

aP values are an exploratory univariate analysis of between-group week 40 values after adjusting for baseline. No adjustment for multiple comparisons were made given the exploratory nature. Data are median (IQR). BMI: Body mass index; ALT: Alanine aminotransferase; ALP: Alkaline phosphatase; GGT: Gamma-glutamyl transferase; TT: Total testosterone; cFT: Calculated free testosterone; SHBG: Sex hormone-binding globulin; LH: Luteinising hormone; LDL: Low-density lipoprotein; HDL: High density lipoprotein; SAT: Subcutaneous adipose tissue; VAT: Visceral adipose tissue.
At week 40, compared to placebo, patients receiving testosterone therapy had significantly increased lean mass (mean change 2111 g (1148-3073) relative to placebo,P <0.001) and significantly decreased fat mass (mean change -2969 g (-3998 to -1941) relative to placebo,P <0.001). All patients receiving testosterone therapy had reductions in fat mass and 19/20 patients receiving testosterone therapy had increases in lean mass (Supplementary Figure 1). Subcutaneous adipose tissue (SAT) was significantly decreased at week 40 in patients receiving testosterone therapy compared to placebo (mean change -359 cm3(-570 to -147) relative to placebo,P= 0.002) but VAT was unchanged (Supplementary Figure 2).
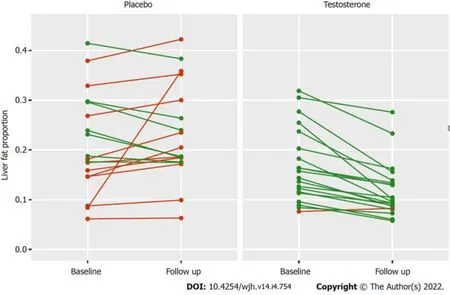
Figure 1 Absolute change in liver fat in all patients receiving placebo and testosterone therapy from baseline to 40 wk follow up, reported as proportion of fat. Lines connect observations from the same participant. Red indicates an increase, green a decrease in liver fat proportion.
Adverse events
Testosterone therapy was well-tolerated with rare serious adverse events that were not significantly different compared to placebo as outlined in Table 4 of the index study[20]. Patients receiving testosterone therapy had significant increases in haemoglobin and haematocrit at week 40 compared to placebo (P <0.001 for both).
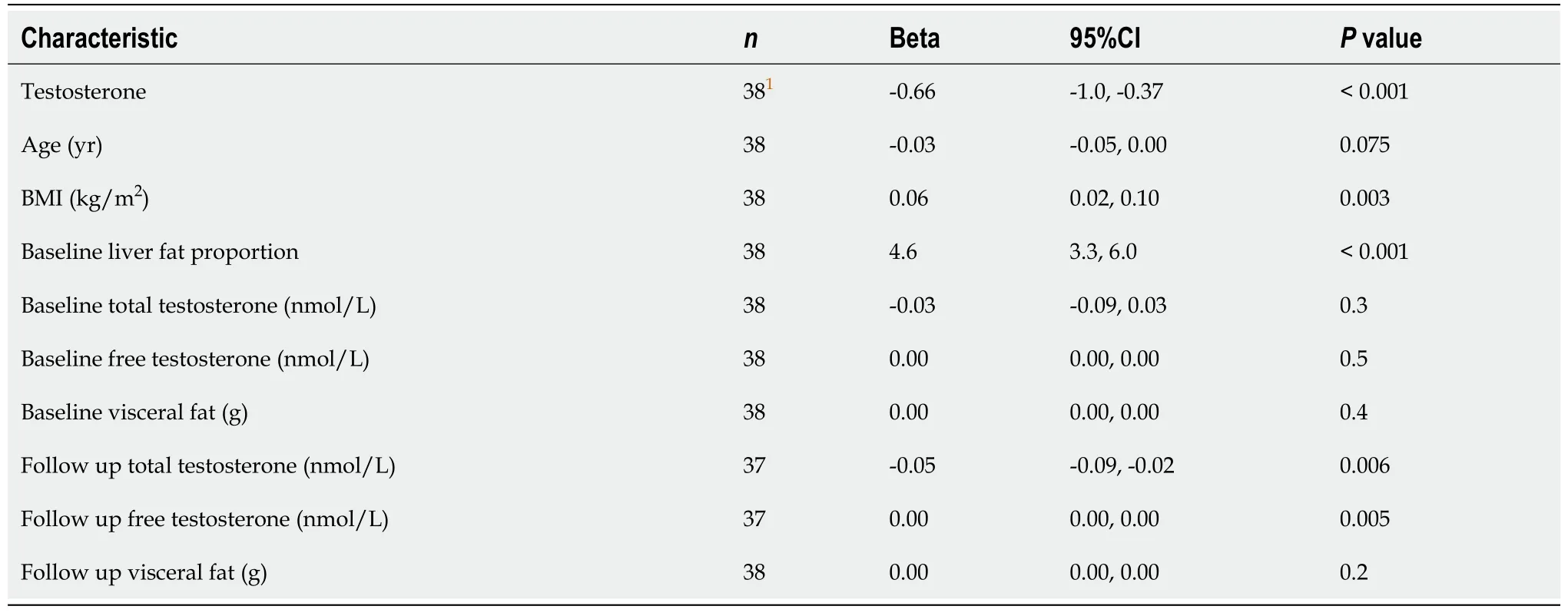
Table 4 Univariate analysis table of factors affecting change in hepatic steatosis
DISCUSSION
In this study of men with type 2 diabetes and low testosterone concentrations, intramuscular testosterone undecanoate over 40 wk significantly reduced liver fat as measured by MRI in an adjusted model compared to placebo.
Our results are consistent with a recent study in which 32 men with low serum testosterone, of whom 8 (25%) had type 2 diabetes, were given a novel oral testosterone preparation LPCN 1144 for 16 wk. In this study, 21/32 patients met diagnostic criteria for NAFLD, and a mean relative reduction in liver fat of 33% as measured by MRI-PDFF was demonstrated in 17 of these 21 patients[16]. However, in contrast to our study, the LPCN 1144 study did not include a control group. Whilst three previous studies reported no change in liver fat in men with low testosterone concentrations after testosterone treatment[17-19], these all had some limitations including short follow-up time of six months, small sample size [17,18], and one study estimated liver fat changes by a less accurate method of measuring change in liver volume and relaxometry[19]. Additionally, patients in our study received intramuscular testosterone administered by study investigators, hence eliminating adherence issues whereas testosterone gel was used in two of the three studies, making adherence difficult to assess[17,19].
Secondary benefits of testosterone therapy in this study included increases in lean muscle mass and reduction in total fat mass, a result consistent with numerous previous testosterone trials across multiple population groups[29,30]. Interestingly, despite a reduction in subcutaneous adiposity, visceral adiposity was unchanged after testosterone therapy suggesting that testosterone may have a direct effect on hepatic steatosis. This is consistent with animal studies showing that the androgen receptor is expressed in the liver and testosterone has biological effects in the liver affecting lipid metabolism and glucose homeostasis[31]. Furthermore, animal models show that testosterone differentially regulates the expression of key targets of lipid and glucose metabolism in a tissue specific manner, with regional differences in action of testosterone on SAT compared to VAT[32]. Total cholesterol levels were also significantly lower in patients treated with testosterone therapy. NAFLD most frequently occurs in the context of the metabolic syndrome, with associated visceral adiposity, dyslipidaemia, and other cardiovascular risk factors[33]. The primary cause of death in such patients is cardiovascular disease, and thus the changes in body composition and lipid profile observed in our study may favourably change cardiovascular risk in patients with NAFLD, but this study was not powered or designed to assess this.
All patients in our study had significant steatosis, and 38/39 patients met diagnostic criteria for a diagnosis of NAFLD. The global prevalence of NAFLD in men with type 2 diabetes is 55%, markedly lower than our cohort’s prevalence[34]. This difference is unsurprising as our cohort consisted of older, obese men with a longstanding history of type 2 diabetes and low serum testosterone concentrations[8]. Given concurrent type 2 diabetes is the strongest risk factor for progression of NAFLD to significant fibrosis[35], demonstrating efficacy in this population represents an important finding with potentially important clinical implications.
Our study has several limitations. This study was a sub-analysis of a randomised controlled trial that was not designed to evaluate liver-related endpoints and not all index cases had liver MRI for fat assessment. However, those included in this study did not appear to significantly differ from those who were not, apart from our cohort having lower weight and waist circumference. Baseline and end of treatment median liver function tests were within the normal ranges for both treatment groups, suggesting that patients in our study did not have significant hepatic inflammation. Finally, fat fraction was assessed using conventional MRI-IP and OP T1 sequences rather than MRI-PDFF, which was not widely available at the time of the index study. Although changes in liver fat MRI-IP and OP T1 sequences do not have the same evidence for correlation with steatohepatitis improvement, it has been shown to accurately quantify liver fat up to 50%, a threshold which was not exceeded in our study[22-24].
Despite these limitations, we identified patients who met diagnostic criteria for NAFLD who had a significant reduction in hepatic steatosis with testosterone treatment while liver fat in most placebo patients increased. The change in hepatic steatosis was measured by MRI scan, which is increasingly recognised as a reasonable surrogate endpoint for histologic response[36]. The two cohorts were well matched with respect to medication use, in particular pertaining to diabetic agents that are known to impact hepatic steatosis and all 39 patients in our study maintained a stable diabetic regimen throughout the trial.
CONCLUSION
This study shows an association between testosterone treatment and a significant reduction in liver fat in men with type 2 diabetes and low testosterone concentrations. These data provide a strong rationale to perform large-scale randomised studies testing the effect of testosterone therapy in high-risk NAFLD patients with low serum testosterone, focusing on liver-specific endpoints including liver inflammation and fibrosis progression.
ARTICLE HIGHLIGHTS
Research background
Testosterone levels are commonly low in men with type 2 diabetes and most men with type 2 diabetes have hepatic steatosis from non-alcoholic fatty liver disease (NAFLD). Animal models show that low testosterone states from castration results in hepatic steatosis and that testosterone replacement improves hepatic steatosis.
Research motivation
Hepatic steatosis occurs in NAFLD which currently has no readily available effective treatment. This study was conducted to provide rationale for future prospective studies of testosterone therapy for NAFLD.
Research objectives
To evaluate the effect of testosterone therapy on liver fat fraction as measured by magnetic resonance imaging (MRI) in a cohort of diabetic men with lowered testosterone levels. We further aimed to determine other factors associated with changes in liver fat in this population.
Research methods
We performed a secondary analysis of a previous 40 wk, randomised, double-blinded, placebocontrolled trial of intramuscular testosterone undecanoate in men with type 2 diabetes and lowered serum testosterone levels. Liver fat as determined by MRI scan before and after therapy was analyzed in addition to blood tests and body composition scans.
Research results
Patients who received testosterone therapy had an absolute reduction of liver fat fraction by 3.5% and patients who received placebo had an absolute increase in liver fat fraction by 1.2%, with a between group difference of 4.7%,P <0.001. After controlling for baseline liver fat, testosterone therapy was associated with a relative reduction in liver fat of 38.3% (P< 0.001).
Research conclusions
Testosterone therapy was associated with a reduction in hepatic steatosis in a cohort of men with type 2 diabetes and lowered serum testosterone levels.
Research perspectives
This study provides rationale for future prospective clinical trials of testosterone therapy for the treatment of NAFLD focusing on liver related endpoints.
FOOTNOTES
Author contributions:Apostolov R, Gow P, Grossmann M and Sinclair M designed the research; Apostolov R, Gianatti E and Kutaiba N were involved in data acquisition; Kutaiba N interpreted and analysed radiological data; Apostolov R, Wong D and Sinclair M drafted the manuscript; Gianatti E, Wong D, Kutaiba N, Gow P, Grossmann M and Sinclair M revised the manuscript for important intellectual content; all authors read and approved the final manuscript.
Institutional review board statement:Human Research Ethics Committee, Research Ethics Unit, Level 8 HSB - Room 8322, Austin Hospital.
Informed consent statement:There are no conflicts of interest to report.
Conflict-of-interest statement:Apostolov R, Darren Wong and Numan Kutaiba have no conflicts of interest to declare. Emily Gianatti, Paul Gow and Marie Sinclair have received financial support for research from Bayer Pharma AG more than five years ago. Mathis Grossmann has received research funding from Bayer Pharma AG, Otzuka and speaker’s honoraria from Besins Health Care and Novartis.
Data sharing statement:No additional data are available.
Open-Access:This article is an open-access article that was selected by an in-house editor and fully peer-reviewed by external reviewers. It is distributed in accordance with the Creative Commons Attribution NonCommercial (CC BYNC 4.0) license, which permits others to distribute, remix, adapt, build upon this work non-commercially, and license their derivative works on different terms, provided the original work is properly cited and the use is noncommercial. See: https://creativecommons.org/Licenses/by-nc/4.0/
Country/Territory of origin:Australia
ORCID number:Ross Apostolov 0000-0002-4827-8795; Emily Gianatti 0000-0002-5219-3241; Darren Wong 0000-0003-1490-0547; Numan Kutaiba 0000-0003-4627-9847; Paul Gow 0000-0001-6505-7233; Mathis Grossmann 0000-0001-8261-3457; Marie Sinclair 0000-0003-0657-3048.
S-Editor:Wang LL
L-Editor:A
P-Editor:Wang LL
杂志排行
World Journal of Hepatology的其它文章
- Revolution in the diagnosis and management of hepatitis C virus infection in current era
- Evidence-based approach to management of hepatic encephalopathy in adults
- Direct oral anticoagulant administration in cirrhotic patients with portal vein thrombosis: What is the evidence?
- Noninvasive diagnosis of periportal fibrosis in schistosomiasis mansoni: A comprehensive review
- Review on hepatitis B virus precore/core promoter mutations and their correlation with genotypes and liver disease severity
- Assessment of periportal fibrosis in Schistosomiasis mansoni patients by proton nuclear magnetic resonance-based metabonomics models
