HBV感染与复制模型的建立及应用
2017-08-30王宝菊郭伟娜杨东亮
王宝菊, 朱 彬, 郭伟娜, 杨东亮
(华中科技大学同济医学院附属协和医院 感染病科, 武汉 430022)
HBV感染与复制模型的建立及应用
王宝菊, 朱 彬, 郭伟娜, 杨东亮
(华中科技大学同济医学院附属协和医院 感染病科, 武汉 430022)
乙型肝炎是危害人类健康的重要传染病,目前的抗病毒治疗,如干扰素、核苷和核苷酸类药物仍无法治愈慢性乙型肝炎。因此,亟待阐明HBV复制和致病机制,探索新的治疗靶点,进而研发新的治疗药物或方案。合适的HBV感染与复制模型是上述研究的基础。由于HBV具有严格的种属限制性及组织亲嗜性,使得HBV感染与复制模型的研发受到一定限制。在国家传染病科技重大专项资助下,国内研究者建立了一系列的细胞和动物模型,就此并结合近年来国内外研究进展进行简要综述。
肝炎病毒, 乙型; 细胞模型; 疾病模型, 动物
全球有1/3的人口正在或曾经感染HBV,其中3.5亿为慢性感染者。我国是乙型肝炎大国,虽然乙型肝炎疫苗的计划接种大大减少了青少年人群HBV感染率,但仍有约9300万慢性HBV感染者[1]。现有的抗HBV药物如干扰素、核苷和核苷酸类药物存在应答率不高或不能有效清除HBV等问题。因此,为了使更多患者实现临床治愈,探索新的药物或治疗方案是乙型肝炎研究领域亟待解决的问题。由于HBV感染具有严格的种属限制性和组织亲嗜性,HBV感染与复制模型研究受到很大限制,尤其我国在该领域的研究相对滞后。在国家传染病科技重大专项课题的资助下,国内学者近年来在HBV感染与复制模型的建立及应用方面取得了较大进展,本文就此并结合国内外近年来进展简要总结如下。
1 细胞模型
HBV感染与复制体外模型包括人/树鼩原代肝细胞、来源于肝癌组织的HepaRG细胞和HLCZ01细胞、表达钠离子-牛磺胆酸共转运蛋白(sodium taurocholate cotransporting polypeptide, NTCP)的肝癌细胞系、HepG2.2.15和HepAD38细胞系,以及HepG2.4D14、HepG2.A64、HepG2.C5细胞系等。这些HBV感染与复制细胞模型在阐明HBV与肝细胞膜表面受体及相关分子结构相互作用,HBV被细胞膜内吞并进入细胞的过程、HBV cccDNA合成、病毒基因复制和转录,病毒包装和病毒释放机制,以及HBV致病机制等方面发挥了重要作用。目前已经被广泛使用。当然,上述细胞模型也存在不同的问题与不足(表1)。
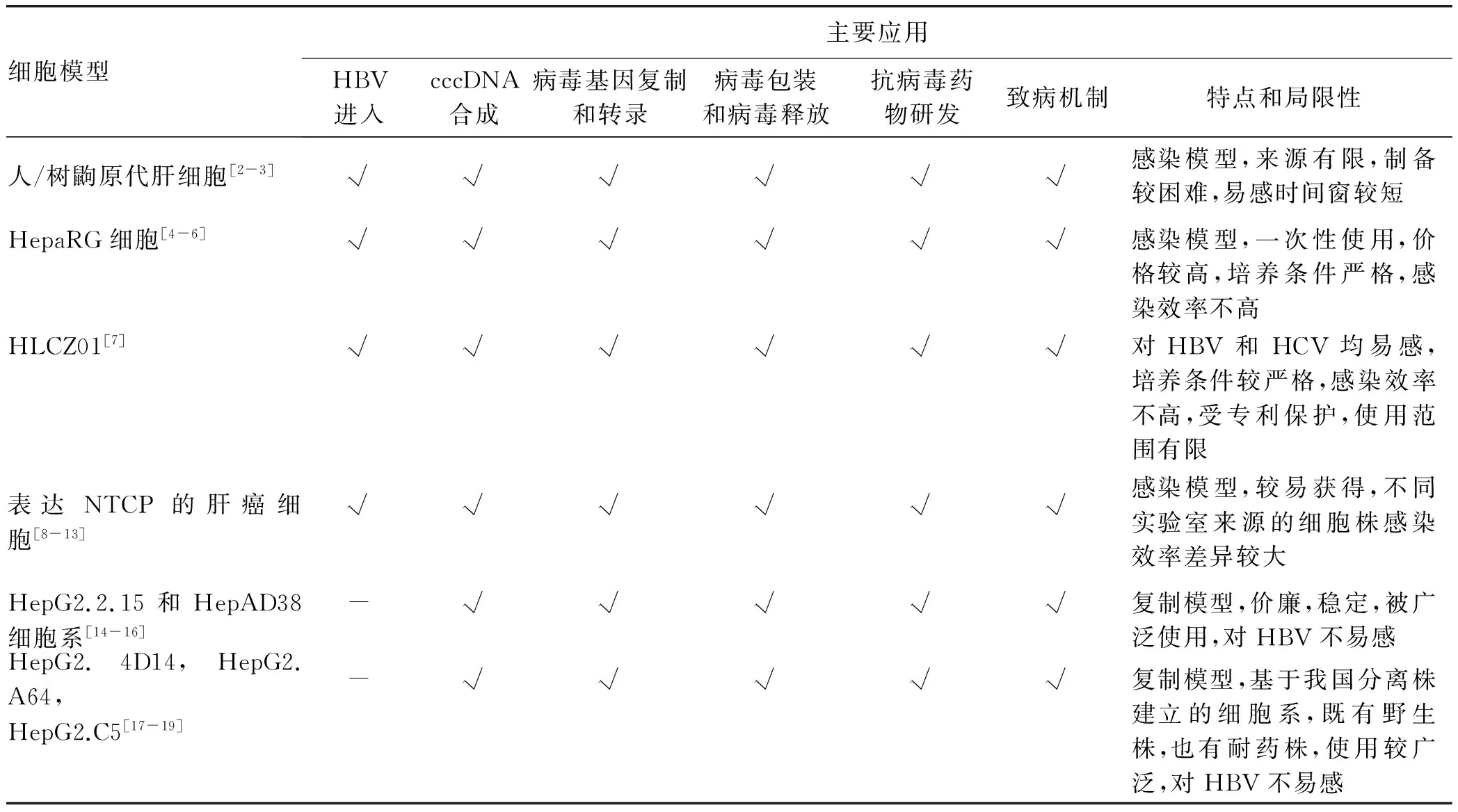
表1 HBV感染与复制细胞模型的比较
1.1 HBV感染的细胞模型
1.1.1 人/树鼩原代肝细胞 HBV能够感染高度分化的原代人肝细胞(primary human hepatocytes, PHH)。然而,PHH很难获得,且在体外培养条件下,会逐渐失去对HBV的易感性。原代树鼩肝细胞(primary tupaia hepatocytes, PTH)可作为体外研究HBV感染的替代模型。基于PTH模型的研究,李文辉等发现NTCP是HBV的受体。也有研究者发现,通过改进PHH的培养条件可以延长PHH HBV感染的时间窗,如胡康洪等通过三维培养技术将PHH对HBV易感的时间窗延长至3周(未发表数据)。为了克服PHH难以获得的问题,胡康洪等[2]还创新了胚胎肝细胞的冻存方法,并发现冻存的胚胎肝细胞与非实质性肝细胞同时培养可使胚胎肝细胞迅速成熟且对HBV的易感性持续至10周[3]。
1.1.2 HepaRG细胞和HLCZ01 HepaRG细胞是人肝脏前体细胞,来源于丙型肝炎肝癌患者的肝组织,保留了许多原代肝细胞的特征,包括关键代谢酶、药物转运蛋白以及核受体的表达[4]。HepaRG细胞在特定条件下培养几周后可以被HBV感染。尽管感染效率并不高,也未观察到病毒播散,但分化的HepaRG细胞(dHepaRG)可持续产生感染性HBV颗粒100 d以上[5]。HepaRG细胞HBV感染系统已成为一种公认的可用于抗病毒药物研发以及评估的有效工具[6]。朱海珍等[7]近年建立了一株来源于HCV相关肝癌样本的肝癌细胞系(HLCZ01),不仅可以被HBV感染,也对HCV易感,且无需分化诱导处理便可支持长达90 d的病毒感染。
1.1.3 表达NTCP的肝癌细胞 李文辉等发现NTCP是HBV进入的功能性受体。基于这一发现,有学者[8]构建了可组成性表达NTCP的肝癌细胞,并证实大部分转染了NTCP的肝癌细胞对HBV易感。应用NTCP稳定转染的HepG2细胞,Ko等[9]发现DEAD盒RNA解旋酶家族成员DDX3是影响cccDNA转录的宿主限制性因素。另外,最近的实验显示环孢素[10]及其衍生物[11]可以与NTCP直接作用并干扰HBV进入易感肝细胞。
有趣的是,表达NTCP的小鼠肝细胞可以支持HDV感染,但是却不能支持HBV感染[12]。进一步研究[13]显示,小鼠肝细胞对HBV感染的限制性也许发生在进入之后、病毒转录之前,敲除抗病毒信号通路中的几大类已知的分子对于这种限制性并无影响。
1.2 HBV复制细胞模型 通过将含有超长HBV基因组的重组质粒稳定转染至人肝癌细胞系所获得的能够支持HBV稳定复制的肝癌细胞系,如众所周知的HepG2.2.15和HepAD38细胞系,已被广泛用于筛选抗HBV药物[14]、制备HBV[15]、研究病毒宿主相互作用[16]。徐东平等建立了基于我国流行的C基因型野生株、阿德福韦酯(ADV)耐药株、恩替卡韦(ETV)耐药株、多重耐药株等一系列HBV稳定复制细胞系,上述细胞系均已申报专利并完成中国典型培养物保藏中心生物典藏。应用上述细胞系的研究发现CRISPR-Cas9可以清除细胞系中整合的HBV DNA[17]、多药耐药蛋白4可能影响核苷类药物的抗病毒效果[18]、HBV通过microRNA-15a-Smad7-TGFβ通路影响凋亡及肿瘤发生等[19]。
2 动物模型
由于HBV感染具有严格的种属限制性和组织亲嗜性,目前仅黑猩猩、树鼩和人源化小鼠肝脏可被HBV感染。鉴于嗜肝DNA病毒家族成员鸭乙型肝炎病毒(duck hepatitis B virus, DHBV)和土拨鼠肝炎病毒(woodchuck hepatitis virus, WHV) 与HBV高度同源,且能自然感染鸭和土拨鼠,因此,鸭和土拨鼠模型也属于感染动物模型。将HBV基因导入生殖细胞染色体建立的转基因小鼠或者通过尾静脉注射方式将HBV基因导入肝脏所建立的复制模型仅能重现部分HBV生命周期,对HBV并不易感。尽管如此,上述动物模型已被广泛应用于HBV研究的各个领域(表2)。
2.1 HBV感染的动物模型
2.1.1 黑猩猩模型 黑猩猩接种HBV血清后可发展为急、慢性HBV感染,同时伴随肝脏炎症及与HBV感染患者相似的免疫应答,是目前最理想的模拟HBV自然感染的动物模型[20]。已被用于研究HBV感染发病机制、评价抗病毒药物和治疗性疫苗效果[21-22]。但是,由于动物保护及费用等原因,黑猩猩模型无法广泛使用。Dupinay等[23]发现毛里求斯岛的食蟹猴自然持续感染可能来自人类的HBV,该动物是否能够作为HBV感染模型仍有待进一步研究。
2.1.2 树鼩模型 树鼩(Tupaiabelangeri),属树鼩科树鼩属,主要分布在我国西南省份和东南亚各国。其可作为HAV和HBV感染模型,在HBV感染的基础上加上黄曲霉毒素诱导的肝癌模型也被用于研究肝癌发病机制[4-26]。由于树鼩是野生动物,个体差异较大,成年动物人工感染HBV后多表现为急性自限性感染,较少形成慢性化。但是幼龄期感染HBV则较容易形成慢性感染,与人类感染相似[27]。近年来,我国学者建立了较大规模人工繁育树鼩种群,深入开展了遗传学和基因组学的系统研究,采用近亲繁殖和基因工程改造技术,有可能发展为可以广泛应用的实验室动物,用于HBV感染、糖尿病、非酒精性脂肪肝等疾病的研究[28-34]。

表2 HBV感染与复制动物模型的比较
2.1.3 人源化小鼠模型 最早的人肝嵌合小鼠模型使用的是尿激酶型纤溶酶原激活物(uPA)转基因的免疫缺陷(Rag2-/-,SCID、SCID/beige)小鼠。人肝细胞移植至uPA-SCID小鼠后可获得重建率较高的人源化肝脏模型,且能够支持HBV和HCV感染。随后在延胡索酰乙酰乙酸水解酶(Fah)缺陷的小鼠 (Fah-/-/Rag2-/-/IL2rg-/-,FRG)移植人肝细胞可使FRG小鼠中人肝细胞达到95%[35]。程通等[36]建立了基于FRG小鼠的HBV感染小鼠模型,优化模型制备流程,实现了规模化生产,并用于评价靶向HBsAg特殊表位的治疗性抗体的疗效。鉴于上述模型均缺乏人免疫细胞,因此不适用于研究免疫应答及免疫治疗策略。研究者通过多种策略构建了人免疫细胞和人肝细胞双嵌合小鼠模型,即人源化小鼠模型,如AFC8和A2/NSG小鼠模型。已被用来进行HCV感染、HBV感染以及肝脏炎症及纤维化机制研究[37-38]。
2.2 嗜肝DNA病毒感染的动物模型
2.2.1 DHBV感染的鸭模型 DHBV为嗜肝DNA病毒中的禽类嗜肝DNA病毒,可自然感染部分种类鸭子。郝友华等[39]发现不同鸭种DHBV的自然感染率不同,通过腹腔和静脉注射雏鸭DHBV感染率不同。DHBV持续感染的鸭模型被广泛用于评价抗HBV药物及联合治疗策略,如临床常用的核苷类药物ETV、核衣壳组装抑制剂等以及抗病毒联合免疫的治疗策略[40]。在发病机制研究方面,应用DHBV感染的鸭模型,发现早期天然免疫(非获得性免疫缺失)是导致雏鸭DHBV持续感染的原因[41],通过比较抗病毒治疗及未经治疗的鸭肝内cccDNA含量发现,cccDNA池并不会因HBV DNA复制水平而改变[42],持续的DNA疫苗联合IL-2和IFNγ质粒注射能够显著减少鸭肝内cccDNA,但仍无法实现彻底清除[43]。
2.2.2 WHV感染的土拨鼠模型 WHV是嗜肝DNA病毒中的正嗜肝DNA病毒。WHV不仅在病毒学特征方面与HBV高度近似,且其感染后的自然史与人感染HBV高度近似,因此被广泛用于研究HBV发病机制、评价抗病毒药物以及免疫预防和治疗策略[44-47]。笔者所在研究组长期从事土拨鼠模型研究,发现我国喜马拉雅旱獭(Marmotahimalayana)与土拨鼠是同属动物,对WHV高度易感[48]。已建立中国喜马拉雅旱獭养殖基地及种群,并开展了实验动物化的相关研究。比较了土拨鼠与旱獭肝脾组织的转录组学数据,发现土拨鼠和旱獭超过75%的不同功能分子的同源性高达90%以上;对20余种土拨鼠和旱獭免疫相关分子等进行了克隆、同源性分析、表达以及抗体制备;完善了WHV感染血清和组织病毒学指标检测技术以及免疫应答分析技术,为旱獭WHV感染模型的标准化奠定了基础;同时该模型也被用于抗病毒药物研发以及新的预防和治疗策略的研究,例如对基于核苷类药物的HBV暴露后预防替代策略进行评价,结果发现ETV单用或与核心蛋白DNA疫苗联用均能完全阻断WHV感染,并且ETV单用的部分动物及与DNA疫苗联用的所有动物均产生保护性免疫[49-62]。
2.3 HBV复制的动物模型
2.3.1 HBV转基因小鼠模型 HBV转基因小鼠已被广泛用于HBV发病机制和抗病毒药物研究。孔祥平等建立了3种特殊免疫遗传背景的HBV转基因小鼠——C57BL/6背景的HBV转基因小鼠、HLA-A2/HBV和Rag1-/-/HBV双转基因小鼠,并广泛提供其他研究者使用。任红教授将该模型用于评价GM-CSF和HBV S基因融合的DNA疫苗[63];田志刚教授亦应用该模型进行了肝细胞和血清蛋白组学[64]、自然杀伤细胞参与CCL4加速HBV转基因小鼠肝纤维化进程的相关研究[65]。WHV转基因小鼠也已用于抗HBV治疗新技术研究[66]。
2.3.2 HBV转染小鼠模型 静脉注射含HBV基因组的腺病毒载体(Ad-HBV),可以在小鼠体内建立HBV复制模型,改变注射剂量可以影响复制持续时间[67]。通过尾静脉在短时间内将大量含有裸DNA的液体注入小鼠体内的方法称为高压水注射,能够有效地将外源基因转运至肝细胞内。Chisari首次通过高压水注射方法将含有复制性HBV基因组的质粒转运至免疫功能正常的小鼠肝内,病毒血症可持续约1周。此后研究表明,质粒骨架、小鼠品系、性别以及注射剂量均可影响小鼠体内HBV的表达水平及复制持续时间。陈培哲等[68]采用pAAV/HBV1.2质粒和C57BL/6小鼠可使HBV复制长达6个月,而采用pAAV/HBV1.2质粒和C3H/HeN小鼠则可使HBV复制时间延长至46周。笔者课题组对该模型进行了深入研究,完善了制备技术并将其用于乙型肝炎发病机制和抗病毒治疗策略等研究,如发现了肝内的自然调节性T淋巴细胞虽然缺乏活化和增殖表型,但仍能抑制效应性T淋巴细胞应答;Poly(I∶C)通过IFN依赖的途径促进HBV清除;IFNα对不同基因型HBV的抗病毒效应存在差异;不同免疫抑制剂对HBV复制及肝内HBV特异性免疫应答的影响不同等[69-73]。
上海巴斯德研究所邓强研究团队[14]将一段含有重组位点的外源内含子序列插入单拷贝的HBV基因组中,构建出cccDNA前体质粒(prcccDNA)。prcccDNA在重组酶Cre表达的情况下,在肝细胞中可诱导HBV重组cccDNA(rcccDNA)并以微染色体的形式存在。通过尾静脉高压水注射方式,rcccDNA可在免疫健全小鼠的肝细胞中迅速积累,并诱导完整、有效的HBV复制。研究表明小鼠体内的T淋巴细胞免疫反应被特异性激活但并不完全。进一步减少DNA注射剂量,小鼠体内HBV复制则显著延长并伴随肝脏持续性炎症损伤。
3 小结
总之,近年来国内外在HBV复制/感染细胞和动物模型研究方面均取得了显著进展,并有力地促进了HBV相关研究。但是,现有的HBV复制/感染模型均存在一定的局限性,如何进一步优化和完善已有的HBV复制/感染模型,推进标准化和规模化,为我国病毒性肝炎基础研究、疫苗和新药研发等提供全方位的模型平台支持,是未来值得深入研究的重要方向。
[1] Chinese Society of Hepatology and Chinese Society of Infectious Diseases, Chinese Medical Association. The guideline of prevention and treatment for chronic hepatitis B: a 2015 update[J]. J Clin Hepatol, 2015, 31(12): 1941-1960. (in Chinese) 中华医学会肝病学分会, 中华医学会感染病学分会. 慢性乙型肝炎防治指南(2015年更新版)[J]. 临床肝胆病杂志, 2015, 31(12): 1941-1960.
[2] ZHOU M, HUANG Y, CHENG Z, et al. Revival, characterization, and hepatitis B virus infection of cryopreserved human fetal hepatocytes[J]. J Virol Methods, 2014, 207: 29-37.
[3] ZHOU M, ZHAO F, LI J, et al. Long-term maintenance of human fetal hepatocytes and prolonged susceptibility to HBV infection by co-culture with non-parenchymal cells[J]. J Virol Methods, 2014, 195: 185-193.
[4] VERRIER ER, COLPITTS CC, SCHUSTER C, et al. Cell culture models for the investigation of hepatitis B and D virus infection[J]. Viruses, 2016, 8(9): 261.
[5] HANTZ O, PARENT R, DURANTEL D, et al. Persistence of the hepatitis B virus covalently closed circular DNA in HepaRG human hepatocyte-like cells[J]. J Gen Virol, 2009, 90(Pt 1): 127-135.
[6] PHILLIPS S, CHOKSHI S, CHATTERJI U, et al. Alisporivir inhibition of hepatocyte cyclophilins reduces HBV replication and hepatitis B surface antigen production[J]. Gastroenterology, 2015, 148(2): 403-414.
[7] YANG D, ZUO C, WANG X, et al. Complete replication of hepatitis B virus and hepatitis C virus in a newly developed hepatoma cell line[J]. Proc Natl Acad Sci U S A, 2014, 111(13): e1264-e1273.
[8] YAN H, ZHONG G, XU G, et al. Sodium taurocholate cotransporting polypeptide is a functional receptor for human hepatitis B and D virus[J]. Elife, 2012, 1: e00049.[9] KO C, LEE S, WINDISCH MP, et al. DDX3 DEAD-box RNA helicase is a host factor that restricts hepatitis B virus replication at the transcriptional level[J]. J Virol, 2014, 88(23): 13689-13698.
[10] NKONGOLO S, NI Y, LEMPP FA, et al. Cyclosporin A inhibits hepatitis B and hepatitis D virus entry by cyclophilin-independent interference with the NTCP receptor[J]. J Hepatol, 2014, 60(4): 723-731.
[11] IWAMOTO M, WATASHI K, TSUKUDA S, et al. Evaluation and identification of hepatitis B virus entry inhibitors using HepG2 cells overexpressing a membrane transporter NTCP[J]. Biochem Biophys Res Commun, 2014, 443(3): 808-813.
[12] NI Y, LEMPP FA, MEHRLE S, et al. Hepatitis B and D viruses exploit sodium taurocholate co-transporting polypeptide for species-specific entry into hepatocytes[J]. Gastroenterology, 2014, 146(4): 1070-1083.
[13] LI H, ZHUANG Q, WANG Y, et al. HBV life cycle is restricted in mouse hepatocytes expressing human NTCP[J]. Cell Mol Immunol, 2014, 11(2): 175-183.
[14] van de KLUNDERT MA, ZAAIJER HL, KOOTSTRA NA. Identification of FDA-approved drugs that target hepatitis B virus transcription[J]. J Viral Hepat, 2016, 23(3): 191-201.
[15] LUCIFORA J, XIA Y, REISINGER F, et al. Specific and nonhepatotoxic degradation of nuclear hepatitis B virus cccDNA[J]. Science, 2014, 343(6176): 1221-1228.
[16] YAN R, ZHAO X, CAI D, et al. The interferon-inducible protein tetherin inhibits hepatitis B virus virion secretion[J]. J Virol, 2015, 89(18): 9200-9212.[17] LI H, SHENG C, WANG S, et al. Removal of integrated hepatitis B virus DNA using CRISPR-Cas9[J]. Front Cell Infect Microbiol, 2017, 7: 91.
[18] LIU W, SONG H, CHEN Q, et al. Multidrug resistance protein 4 is a critical protein associated with the antiviral efficacy of nucleos(t)ide analogues[J]. Liver Int, 2016, 36(9): 1284-1294.
[19] LIU N, JIAO T, HUANG Y, et al. Hepatitis B virus regulates apoptosis and tumorigenesis through the microRNA-15a-Smad7-transforming growth factor beta pathway[J]. J Virol, 2015, 89(5): 2739-2749.
[20] WIELAND SF. The chimpanzee model for hepatitis B virus infection[J]. Cold Spring Harb Perspect Med, 2015, 5(6): a021469.
[21] LANFORD RE, GUERRA B, CHAVEZ D, et al. GS-9620, an oral agonist of Toll-like receptor-7, induces prolonged suppression of hepatitis B virus in chronically infected chimpanzees[J]. Gastroenterology, 2013, 144(7): 1508-1517.
[22] ASABE S, WIELAND SF, CHATTOPADHYAY PK, et al. The size of the viral inoculum contributes to the outcome of hepatitis B virus infection[J]. J Virol, 2009, 83(19): 9652-9662.
[23] DUPINAY T, GHEIT T, ROQUES P, et al. Discovery of naturally occurring transmissible chronic hepatitis B virus infection among Macaca fascicularis from Mauritius Island[J]. Hepatology, 2013, 58(5): 1610-1620.
[24] XIAO J, LIU R, CHEN C. Tree shrew (Tupaia belangeri) as a novel non-human primate laboratory disease animal model[J]. Zool Res, 2017, 38(3): 127-137.
[25] YAO YG. Creating animal models, why not use the Chinese tree shrew (Tupaia belangeri chinensis)? [J]. Zool Res, 2017, 38(3): 118-126.
[26] TSUKIYAMA-KOHARA K, KOHARA M. Tupaia belangeri as an experimental animal model for viral infection[J]. Exp Anim, 2014, 63(4): 367-374.
[27] WANG Q, SCHWARZENBERGER P, YANG F, et al. Experimental chronic hepatitis B infection of neonatal tree shrews (Tupaia belangeri chinensis): a model to study molecular causes for susceptibility and disease progression to chronic hepatitis in humans[J]. Virol J, 2012, 9: 170.
[28] FENG Y, FENG YM, FENG Y, et al. Identification and characterization of liver microRNAs of the Chinese tree shrew via deep sequencing[J]. Hepat Mon, 2015, 15(10): e29053.
[29] FAN Y, YU D, YAO YG. Tree shrew database (TreeshrewDB): a genomic knowledge base for the Chinese tree shrew[J]. Sci Rep, 2014, 4: 7145.
[30] WU X, XU H, ZHANG Z, et al. Transcriptome profiles using next-generation sequencing reveal liver changes in the early stage of diabetes in tree shrew (Tupaia belangeri chinensis)[J]. J Diabetes Res, 2016, 2016: 6238526.
[31] YU W, YANG C, BI Y, et al. Characterization of hepatitis E virus infection in tree shrew (Tupaia belangeri chinensis)[J]. BMC Infect Dis, 2016, 16: 80.
[32] YE L, HE M, HUANG Y, et al. Tree shrew as a new animal model for the study of lung cancer[J]. Oncol Lett, 2016, 11(3): 2091-2095.
[33] RUAN P, YANG C, SU J, et al. Histopathological changes in the liver of tree shrew (Tupaia belangeri chinensis) persistently infected with hepatitis B virus[J]. Virol J, 2013, 10: 333.
[34] ZHAO F, GUO X, WANG Y, et al. Drug target mining and analysis of the Chinese tree shrew for pharmacological testing[J]. PLoS One, 2014, 9(8): e104191.
[35] BISSIG KD, WIELAND SF, TRAN P, et al. Human liver chimeric mice provide a model for hepatitis B and C virus infection and treatment[J]. J Clin Invest, 2010, 120(3): 924-930.
[36] ZHANG TY, YUAN Q, ZHAO JH, et al. Prolonged suppression of HBV in mice by a novel antibody that targets a unique epitope on hepatitis B surface antigen[J]. Gut, 2016, 65(4): 658-671.
[37] WASHBURN ML, BILITY MT, ZHANG L, et al. A humanized mouse model to study hepatitis C virus infection, immune response, and liver disease[J]. Gastroenterology, 2011, 140(4): 1334-1344.
[38] BILITY MT, CHENG L, ZHANG Z, et al. Hepatitis B virus infection and immunopathogenesis in a humanized mouse model: induction of human-specific liver fibrosis and M2-like macrophages[J]. PLoS Pathog, 2014, 10(3): e1004032.
[39] HAO YH, LI AY, DING HH, et al. Experimental study on duck hepatitis B virus infection model by different kinds of ducklings and anti-viral effect[J]. Chin J Comp Med, 2012, 22(11): 23-26. (in Chinese) 郝友华, 李安意, 丁红晖, 等. 不同种雏鸭建立鸭乙肝病毒感染模型及抗病毒效果的实验[J]. 中国比较医学杂志, 2012, 22(11): 23-26.
[40] CAMPAGNA MR, LIU F, MAO R, et al. Sulfamoylbenzamide derivatives inhibit the assembly of hepatitis B virus nucleocapsids[J]. J Virol, 2013, 87(12): 6931-6942.[41] TOHIDI-ESFAHANI R, VICKERY K, COSSART Y. The early host innate immune response to duck hepatitis B virus infection[J]. J Gen Virol, 2010, 91(Pt 2): 509-520.
[42] REAICHE GY, LE MIRE MF, MASON WS, et al. The persistence in the liver of residual duck hepatitis B virus covalently closed circular DNA is not dependent upon new viral DNA synthesis[J]. Virology, 2010, 406(2): 286-292.
[43] SAADE F, BURONFOSSE T, GUERRET S, et al. In vivo infectivity of liver extracts after resolution of hepadnaviral infection following therapy associating DNA vaccine and cytokine genes[J]. J Viral Hepat, 2013, 20(4): e56-e65.
[44] ROGGENDORF M, YANG D, LU M. The woodchuck: a model for therapeutic vaccination against hepadnaviral infection[J]. Pathol Biol (Paris), 2010, 58(4): 308-314.[45] MASON WS. Animal models and the molecular biology of hepadnavirus infection[J]. Cold Spring Harb Perspect Med, 2015, 5(4): a021352.[46] FLETCHER SP, CHIN DJ, CHENG DT, et al. Identification of an intrahepatic transcriptional signature associated with self-limiting infection in the woodchuck model of hepatitis B[J]. Hepatology, 2013, 57(1): 13-22.
[47] KOSINSKA AD, ZHANG E, JOHRDEN L, et al. Combination of DNA prime-adenovirus boost immunization with entecavir elicits sustained control of chronic hepatitis B in the woodchuck model[J]. PLoS Pathog, 2013, 9(6): e1003391.
[48] WANG BJ, TIAN YJ, MENG ZJ, et al. Establishing a new animal model for hepadnaviral infection: susceptibility of Chinese Marmota-species to woodchuck hepatitis virus infection[J]. J Gen Virol, 2011, 92(Pt 3): 681-691.
[49] LIU Y, WANG B, WANG L, et al. Transcriptome analysis and comparison of marmota monax and marmota himalayana[J]. PLoS One, 2016, 11(11): e165875.[50] FAN H, ZHU Z, WANG Y, et al. Molecular characterization of the type I IFN receptor in two woodchuck species and detection of its expression in liver samples from woodchucks infected with woodchuck hepatitis virus (WHV)[J]. Cytokine, 2012, 60(1): 179-185.
[51] YANG Y, ZHANG X, ZHANG C, et al. Molecular characterization of woodchuck CD4 (wCD4) and production of a depletion monoclonal antibody against wCD4[J]. Mol Immunol, 2013, 56(1-2): 64-71.
[52] YAN Q, LI M, LIU Q, et al. Molecular characterization of woodchuck IFI16 and AIM2 and their expression in woodchucks infected with woodchuck hepatitis virus (WHV)[J]. Sci Rep, 2016, 6: 28776.
[53] JIANG M, LIU J, ZHANG E, et al. Molecular characterization of woodchuck interleukin-10 receptor and enhanced function of specific T cells from chronically infected woodchucks following its blockade[J]. Comp Immunol Microbiol Infect Dis, 2012, 35(6): 563-573.
[54] WANG L, WANG J, LIU Y, et al. Molecular cloning, characterization and expression analysis of TGF-β and receptor genes in the woodchuck model[J]. Gene, 2016, 595(1): 1-8.
[55] ZHANG E, ZHANG X, LIU J, et al. The expression of PD-1 ligands and their involvement in regulation of T cell functions in acute and chronic woodchuck hepatitis virus infection[J]. PLoS One, 2011, 6(10): e26196.
[56] LU Y, WANG B, HUANG H, et al. The interferon-alpha gene family of Marmota himalayana, a Chinese marmot species with susceptibility to woodchuck hepatitis virus infection[J]. Dev Comp Immunol, 2008, 32(4): 445-457.
[57] WANG B, ZHU Z, ZHU B, et al. Nucleoside analogues alone or combined with vaccination prevent hepadnavirus viremia and induce protective immunity: alternative strategy for hepatitis B virus post-exposure prophylaxis[J]. Antiviral Res, 2014, 105: 118-125.
[58] LIU J, ZHANG E, MA Z, et al. Enhancing virus-specific immunity in vivo by combining therapeutic vaccination and PD-L1 blockade in chronic hepadnaviral infection[J]. PLoS Pathog, 2014, 10(1): e1003856.
[59] PAN D, LIN Y, WU W, et al. Persistence of the recombinant genomes of woodchuck hepatitis virus in the mouse model[J]. PLoS One, 2015, 10(5): e0125658.
[60] ZHANG E, KOSINSKA AD, MA Z, et al. Woodchuck hepatitis virus core antigen-based DNA and protein vaccines induce qualitatively different immune responses that affect T cell recall responses and antiviral effects[J]. Virology, 2015, 475: 56-65.
[61] MA Z, ZHANG E, YANG D, et al. Contribution of Toll-like receptors to the control of hepatitis B virus infection by initiating antiviral innate responses and promoting specific adaptive immune responses[J]. Cell Mol Immunol, 2015, 12(3): 273-282.
[62] MENG Z, ZHANG X, WU J, et al. RNAi induces innate immunity through multiple cellular signaling pathways[J]. PLoS One, 2013, 8(5): e64708.
[63] QING Y, CHEN M, ZHAO J, et al. Construction of an HBV DNA vaccine by fusion of the GM-CSF gene to the HBV-S gene and examination of its immune effects in normal and HBV-transgenic mice[J]. Vaccine, 2010, 28(26): 4301-4307.
[64] DING C, WEI H, SUN R, et al. Hepatocytes proteomic alteration and seroproteome analysis of HBV-transgenic mice[J]. Proteomics, 2009, 9(1): 87-105.
[65] JIN Z, SUN R, WEI H, et al. Accelerated liver fibrosis in hepatitis B virus transgenic mice: involvement of natural killer T cells[J]. Hepatology, 2011, 53(1): 219-229.
[66] MENG Z, MA Z, ZHANG E, et al. Novel Woodchuck Hepatitis Virus (WHV) transgene mouse models show sex-dependent WHV replicative activity and development of spontaneous immune responses to WHV proteins[J]. J Virol, 2014, 88(3): 1573-1581.
[67] HUANG LR, GBEL YA, GRAF S, et al. Transfer of HBV genomes using low doses of adenovirus vectors leads to persistent infection in immune competent mice[J]. Gastroenterology, 2012, 142(7): 1447-1450.
[68] PENG XH, REN XN, CHEN LX, et al. High persistence rate of hepatitis B virus in a hydrodynamic injection-based transfection model in C3H/HeN mice[J]. World J Gastroenterol, 2015, 21(12): 3527-3536.
[69] DIETZE KK, SCHIMMER S, KRETZMER F, et al. Characterization of the treg response in the hepatitis B virus hydrodynamic injection mouse model[J]. PLoS One, 2016, 11(3): e0151717.
[70] WU J, HUANG S, ZHAO X, et al. Poly(I∶C) treatment leads to interferon-dependent clearance of hepatitis B virus in a hydrodynamic injection mouse model[J]. J Virol, 2014, 88(18): 10421-10431.
[71] SONG J, ZHOU Y, LI S, et al. Susceptibility of different hepatitis B virus isolates to interferon-alpha in a mouse model based on hydrodynamic injection[J]. PLoS One, 2014, 9(3): e90977.
[72] WANG J, WANG B, HUANG S, et al. Immunosuppressive drugs modulate the replication of hepatitis B virus (HBV) in a hydrodynamic injection mouse model[J]. PLoS One, 2014, 9(1): e85832.
[73] LI L, SHEN H, LI A, et al. Inhibition of hepatitis B virus (HBV) gene expression and replication by HBx gene silencing in a hydrodynamic injection mouse model with a new clone of HBV genotype B[J]. Virol J, 2013, 10: 214.[74] QI Z, LI G, HU H, et al. Recombinant covalently closed circular hepatitis B virus DNA induces prolonged viral persistence in immunocompetent mice[J]. J Virol, 2014, 88(14): 8045-8056.
引证本文:WANG BJ, ZHU B, GUO WN, et al. Establishment and application of in vitro and in vivo models of hepatitis B virus infection[J]. J Clin Hepatol, 2017, 33(8): 1458-1464. (in Chinese) 王宝菊, 朱彬, 郭伟娜, 等. HBV感染与复制模型的建立及应用[J]. 临床肝胆病杂志, 2017, 33(8): 1458-1464.
(本文编辑:葛 俊)
Establishment and application of in vitro and in vivo models of hepatitis B virus infection
WANGBaoju,ZHUBin,GUOWeina,etal.
(DepartmentofInfectiousDiseases,UnionHospitalAffiliatedtoTongjiMedicalCollegeofHuazhongUniversityofScienceandTechnology,Wuhan430022,China)
Hepatitis B is still an important infectious disease which threatens human health, and current antiviral therapy, including interferon and nucleos(t)ide analogues, cannot cure chronic hepatitis B. Therefore, it is urgent to explore the detail mechanisms of HBV replication and pathogenesis, identify new therapeutic targets, and develop new drugs or treatment regimens, which relies on the development of suitable models for HBV infection and replication. Species restriction and tissue tropism of HBV have limited the development of models for HBV infection and replication. With the support by National Science and Technology Major Project for Infectious Diseases, the researchers in China have developed a series of cellular and animal models for HBV. This article reviews these models with reference to recent research advances in China and foreign countries.
hepatitis B virus; cell model; disease models, animal
10.3969/j.issn.1001-5256.2017.08.009
2017-06-30;
2017-07-12。
国家传染病科技重大专项(2008ZX10002011,2012ZX10004503);国家自然科学基金(81001313,81101248,81371828,81461130019);国家国际科技合作计划(2011DFA31030);国家科技支撑计划课题(2015BAI09B06);德国科学基金会德中跨学科重大合作项目(TRR60)
王宝菊(1973-),女,副教授,博士,主要从事乙型肝炎动物模型及发病机制的研究。
杨东亮,电子信箱:dlyang@hust.edu.cn。
R512.62
A
1001-5256(2017)08-1458-07
