低介电常数聚喹啉衍生物薄膜的合成与表征
2010-12-11赵雄燕
赵雄燕
(河北科技大学材料科学与工程学院,石家庄 050018)
低介电常数聚喹啉衍生物薄膜的合成与表征
赵雄燕*
(河北科技大学材料科学与工程学院,石家庄 050018)
采用等离子体聚合技术合成了一种新型的低介电常数聚喹啉衍生物薄膜:聚3-氰基喹啉(PP3QCN)薄膜.借助于傅里叶变换红外光谱(FT-IR)、紫外-可见(UV-Vis)吸收光谱、X光电子能谱(XPS)和原子力显微镜(AFM)对薄膜结构进行了系统表征.结果表明,等离子体聚合条件对沉积膜的化学结构、表面组成、膜形态以及介电性能均有影响.在较低的等离子体放电功率(10 W)条件下,可得到具有较高芳环保留率和较大π-共轭体系的高质量聚3-氰基喹啉薄膜材料;而在较高功率(25 W)条件下,聚合过程中会出现比较严重的单体分子破碎,形成较多非π-共轭体系的聚合物,从而导致聚3-氰基喹啉的共轭度降低.聚3-氰基喹啉薄膜的介电性能测试结果表明,低放电功率(10 W)条件下制得的聚3-氰基喹啉薄膜具有比较低的介电常数值,仅为2.45.
等离子体聚合;低介电常数;3-氰基喹啉;集成电路
With the development of ultra-large scale integration(ULSI) circuit,the feature sizes of the integrated circuit have decreased to about 130 nm or even smaller[1-3].Consequently,reduction in the electrical resistance inducing by interaction between interconnect lines,time delay caused by wire capacitance,cross-talk, and the power dissipation has become the bottle-neck problem limiting the progress of the high speed,low power cost,and multi-functional integrated circuit[4-6].For this reason,three major technological advances have been made in recent years to meet these ever increasing requirements for electrical performance of on-chip wiring:(1)copper has replaced aluminum as the new interconnect metal of choice,with the introduction of the damascene processing;(2)simulations and optimizations of the effects of the dimensional sizes between layers and lines,technology and materials on the thermal and electrical properties have been made;(3)alternatives for SiO2with a lower dielectric constant(εr) are being developed and introduced as the dielectric between layers and lines[7-11].Among the broad variety of accessible alternatives,polymer has been one of the most widely studied materials due to its outstanding and promising mechanical and physicochemical properties.It takes advantage of the unique combination of excellent chemical inertness,good mechanical properties,thermal stability,electrical property,formability,and easy shaping.Thus,one of the exciting and promising developments in material science today is the design and synthesis of novel low dielectric constant polymer materials.Recently,a number of investigations have been devoted to polymerize dielectric materials,including polyimides[12-14],poly(nitrile)s[15-16], SiLK resin[17-19],benzocyclobutene resins[20-22],poly(binaphthylene ether)[23-24],polynorbornenes[25],organic-inorganic hybrid polymer[26-28].
Plasma polymerization is now becoming one of the fastest growing areas in the field of direct thin film deposition of polymeric materials,which are hard to obtain by the conventional polymerization methods.Moreover,the films obtained by plasma polymerization are generally of high quality,homogeneous, adherent,and thermally stable films.In the present work,the technique of plasma polymerization was used in the preparation of plasma-polymerized 3-cyanoquinoline(PP3QCN)thin films with the aims to develop a polyquinoline derivative thin film with a low εrvalue.This article will focus on preparation and analysis of structural and dielectric properties of plasma-polymerized 3-cyanoquinoline thin film.
1 Experimental
1.1 Materials
The substrates used were heavily doped,p-type,silicon(100) wafers,quartz glass and freshly pressed infrared-grade potassium bromide(KBr).The high purity argon was used for the substrate cleaning.The monomer,3-cyanoquinoline(98%)was obtained from the Aldrich Chemical Company and used without further purification.The chemical structure of monomer is shown in Scheme 1.
1.2 Substrate cleaning

Scheme 1 Chemical structure of monomer 3-cyanoquinoline
The quartz substrates used in plasma polymerization were cleaned consecutively three times.The substrates were first base cleaned for 15 min at 70℃on a hot plate in a mixture of 25 mL 30%NH4OH,25 mL 30%H2O2,and 125 mL deionized water. After rising with deionized water,the substrates were then acid cleaned for 15 min at 70℃on a hot plate in a mixture of 25 mL of HCl,25 mL of 30%H2O2,and 125 mL of deionized water. The cleaned substrates were next rinsed with deionized water and then were kept in the vacuum oven for 24 h at 120℃for drying.Finally,the substrates were put into plasma chamber and cleaned with argon plasma(80 W)for 3 min just prior to plasma polymerization.
1.3 Thin film preparation
Plasma polymerization of 3-cyanoquinoline was carried out using a radio frequency(13.56 MHz)capacitively coupled glow discharge system.A cylinder-shaped stainless steel plasma polymerization reactor was fitted with parallel plate electrodes.The substrates were centered on the bottom electrode,after evacuation and purging with high purity nitrogen three times,the radio frequency(RF)system was adjusted to the assigned power and a glow discharge was allowed to occur for fixed times.The pressure of the reaction chamber was maintained at about 5.3-6.0 Pa during the glow discharge.After the plasma was extinguished, the high purity nitrogen was induced to the chamber until the pressure of reactor became higher than 267 Pa,after which the reactor was brought to atmospheric pressure with air.The polymer thin films deposited directly onto quartz substrate were used to the characterization of UV-Vis,AFM,XPS as well as the measurements of film thickness,and while the plasma thin films deposited onto potassium bromide(KBr)pellet was used only for FT-IR analysis.The plasma-polymerized 3-cyanoquinoline thin films deposited at 10 and 25 W are abbreviated as PP3QCN10 and PP3QCN25,respectively.The thicknesses of PP3QCN10 and PP3QCN25 are 505.0 and 835.8 nm.
1.4 Characterization
The UV-Vis absorption spectra were recorded using a UV-Vis recording spectrophotometer(Shimadzu,Model UV-2501PC). Fourier transform infrared(FT-IR)spectra were measured on a Perkin-Elmer System 2000 FT-IR spectrometer.Each spectrum was collected by accumulating 64 scans at a resolution of 8 cm-1. The film thickness was measured using a surface profiler(Alpha-Step 500).An X-ray photoelectron spectrometer(XPS)(Kratos, Axis ULTRA)was used for XPS measurements with an Al KαX-ray source.The sensitivity factors used for C 1s,N 1s and O 1s are 0.287,0.477 and 0.780,respectively.The C 1s spectra were analyzed by fitting Gaussian peaks with a 20%Lorentzian contribution.The morphology of plasma film was characterized using an atomic force microscopy(AFM)in tapping mode under a constant force(Digital Instruments,NanoScope IIIa);the arithmetic mean of the surface roughness(Ra)was obtained from an area of 2 μm×2 μm.An HP 4284 A semiconductor parameter analyzer available from Hewlett-Packard Company was used to measure the capacitance of the PP3QCN thin films in the frequency range from 100 kHz to 1 MHz at the temperature of 303 K.The dielectric constant(εr)was calculated from the known values of capacitance(C),film thickness(d),the area of the aluminumcontactingdot(A)andthepermittivityoffreespace(ε0) using the following relation:εr=C·d/ε0·A.
2 Results and discussion
2.1 Film structural analysis
Fig.1 shows the UV-Vis absorption spectra of the monomer 3-cyanoquinoline and its corresponding two plasma-polymerized thin films deposited at different powers.The pure 3-cyanoquinoline shows an absorption band at 235 nm,which can be attributed to the π-π*transition of the aromatic 3-cyanoquinoline ring.In comparison with the monomer absorption band,the absorption bands of both PP3QCN thin films turn out to be stronger and extend to the visible region,the maximum absorption wavelength (λmax)of both PP3QCN thin films shows an obvious red shift,i.e., the maximum of red shift of more than 20 nm(257 nm vs 235 nm)is observed for PP3QCN10 thin film.It is well known that increasing the length of the conjugated π-system generally moves the absorption maximum to longer wavelengths[29].Appearing a red shift and wider peak width indicate that a larger conjugated π-system has been formed in PP3QCN10 film duration the polymerization of 3-cyanoquinoline.For the PP3QCN25 film deposited at 25 W,however,it was found that the absorption band of PP3QCN25 film shifts to lower wavelength as compared with the PP3QCN10 film and its absorption intensity become weaker than that of PP3QCN10 thin film.This indicates that the effective conjugation length of PP3QCN25 film has been diminished under the higher discharge power plasma polymerization.Since,as we reported before[30],when the plasma polymerization is carried out under a high discharge power,more aromatic rings of monomers are dissociated and fragmented, which results in more non-conjugated polymers being formed in the plasma polymerized films and this effect is became more prominent with further increasing discharge power.Hence,it is not difficult to understand that the high discharge power plasma of 25 W gave rise to a decrease in the effective conjugation length of plasma-polymerized 3-cyanoquinoline as indicated by the result shown in Fig.1.
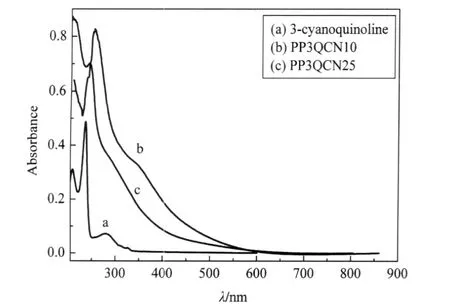
Fig.1 UV-Vis spectra of monomer and plasma-polymerized 3-cyanoquinoline
The FT-IR spectra of the plasma-polymerized thin films deposited at 10 and 25 W are illustrated in Fig.2.The absorption spectrum of monomer 3-cyanoquinoline is shown for the comparison.It is clear that there appeared a broader and considerably stronger absorption band at 1663 cm-1for both plasma PP3QCN films,attributed to the conjugated C=N stretching vibration as compared with the 3-cyanoquinoline monomer,which indicated that extensively conjugated C=N double bonds were formed during the plasma polymerization of monomer 3-cyanoquinoline.It is also clear that the characteristic absorptions at 3053 and 3020 cm-1for the aromatic C—H stretching vibration have been preserved to a large extent for the PP3QCN10 thin film,indicating greater retention of aromatic ring structure in the PP3QCN10 film.Moreover,the greater retention of aromatic ring structure in the PP3QCN10 thin film is also indicated by the prominent characteristic absorption bands at 1604,1555, 1501,and 1410 cm-1of the aromatic ring backbone stretching vibrations.Thus,the FT-IR results suggest that the plasma polymerization of 3-cyanoquinoline has proceeded mainly via the opening of π-bond of the C≡N functional groups under a low discharge power of 10 W.For PP3QCN25 film deposited at the discharge power of 25 W,however,a quite different FT-IR absorption spectrum was observed as compared with that of PP3QCN10 thin film.It can been seen from Fig.2c that the intensity of absorption bands corresponding to aromatic ring backbonestretching vibration hasdiminished significantly in PP3QCN25 film.This is indicative of increasing monomer(aromatic ring)fragmentation during the high power plasma polymerization of 25 W,which agrees well with the UV-Vis results. Furthermore,the new absorption bands appeared at 2930 cm-1(—CH3,νasstretchingvibration)and2863cm-1(—CH2,νsstretching vibration)in PP3QCN25 film further confirming the above deduction.
The survey XPS spectra of PP3QCN thin films shown in Fig.3 reveal that besides carbon and nitrogen,there are also significant amounts of oxygen in all the plasma-polymerized conjugated polynitrile films.The presence of oxygen is normally expected in the plasma-polymerized films owing to the existence of trace oxygen absorbed on the wall of the reaction chamber system and also due to the reaction of long-lived radicals with atmospheric oxygen after being exposed to the air.

Fig.2 FT-IR spectra of monomer 3-cyanoquinoline(a), PP3QCN10(b),and PP3QCN25(c)
In order to get the detailed surface stoichiometry of plasma conjugated polynitrile thin films,the high-resolution C 1s spectra of PP3QCN10 and PP3QCN25 films were evaluated and are shown in Fig.4.It has been reported that the full width at half maximum(FWHW)for plasma polymers is expected to be relatively large,about 2 eV due to their irregular molecular structure[31-32].Thus,the C 1s spectra for PP3QCN10 and PP3QCN25 films were curve-fitted using 2 eV as the FWHW.It should be noted that some fitting peaks significantly overlap due to the small difference in the binding energies associated with these peaks.The C 1s spectra of PP3QCN10 film can be deconvoluted into five component peaks as indicated in Fig.4.The binding energy for aromatic C=N(285.97 eV)and C=C(285.47 eV)was assigned in accordance with the results in the literature[33]and the peak positions for both aromatic components were fixed during the fit procedure.The other three chemically distinct C atoms are assigned as follows:the peak at 284.90 eV represents saturated hydrocarbon C—H;the peak at 286.75 eV represents the C—O group;and the peak at 288.04 eV represents C=O.In addition,a broad high binding energy peak centered at 292.88 eV, which assigned to the π-π*shake-up satellite arising from the aromatic rings is observed for PP3QCN10 thin film,indicating that the PP3QCN10 film contains an extended π-electron system.In other words,a higher retention of the aromatic ring structure in the PP3QCN10 film is obtained during the plasma polymerizationof3-cyanoquinolineunderthedischargeof10W.

Fig.3 XPS spectra of plasma-polymerized 3-cyanoquinoline film

Fig.4 C 1s spectra of PP3QCN10 and PP3QCN25
For sample PP3QCN25,however,besides the C=C,C=N, C—H,C—O,and C=O components,the O—C=O contribution was likewise detected in the plasma films.Also the amounts of the aromatic C=C and C=N component in PP3QCN25 film were found to decrease substantially comparing with that for the films deposited under the low power(10 W)used.Meanwhile, the relative amounts of C—H,C—O,and C=O components, formed during the plasma polymerization process,were found to increase,indicating more intensive monomer fragmentation process promoted at the high discharge power(25 W).Thus,it is clear that the aromatic ring structure in the PP3QCN10 films are preserved more effectively for deposition carried out at the lower 10 W plasma discharge.Furthermore,it is also clear from Fig.4 that the intensity of π-π*shake-up satellite for PP3QCN25 film is much weaker than that of the films deposited at a discharge power of 25 W,revealing less retention of the aromatic ring structure in the PP3QCN25 film at 25 W.The difference in structure of the PP3QCN films for high and low discharge power probably arises from the difference in bond scission mechanism during the plasma polymerization.It has been reported that at a low discharge power,the plasma polymerization mainly carries out in an energy-deficient state and the energy per unit mass of the molecule is very low.Thus,bond scission occurs mainly at the π-bond of the functional group C≡N,which has the lowest bond energy.Under these conditions,rearrangement of the active radicals leads to a plasma polymer having a similar chain structure to that of the conventional polymer.For a high discharge power,however,the plasma polymerization process transforms from an energy-deficient state to a more energetic state.More energy per unit mass of the monomer will result in more severe molecular fragmentation.So it is easy to understand that under a high discharge power,plasma polymerization of 3-cyanoquinoline will produce more active radicals,which come from scissions of not only the π-bond of the C≡N functional group but also the aromatic ring.The latter reactions help to account for the decrease in contents of the aromatic species C=C and C=N and an increase in content of the aliphatic CH,which results in more non-conjugated polymers being formed in the plasma polymerized films.
2.2 Growth of film and morphology
The thickness of PP3QCN thin film was measured as a function of deposition time at different discharge powers.As shown in Fig.5,a linear increase in film thickness with deposition time is observed for the two thin films deposited under different discharge powers.It should be pointed out that the plotted values are the average thickness taken from four positions of the bottom electrode.The slope of each linear curve can give the deposition rate of the thin films corresponding to different discharge powers.It can be seen that the deposition rate increased with increasing plasma discharge power.The deposition rate of PP3QCN25 film was as 1.7 times as that of PP3QCN10 film. The increase in the deposition rate with increasing plasma power can be attributed to a combination of two plasma processes. First,the increase of discharge power will result in an increased density of energetic electrons and ions,and thus,more functional group of C≡N triple bonds will be opened to form a C=N conjugated plasma polymer.Second,the higher density of energetic electrons and ions caused by increasing RF power will also bring about the enhancement of monomer molecule fragmentation and to form a non-conjugated polymer.Although both these plasma processes can increase the deposition rate,their contribution to the deposition rate is different,depending on the discharge power used in plasma polymerization.At a low plasma power,the formation of a C=N conjugated plasma polymer is the dominant plasma process,and the plasma polymerization proceeds mainly on the substrate surface.For a high discharge power,on the other hand,both plasma processes play an important role in increasing the deposition rate.In other words,plasma polymerization of monomer 3-cyanoquinoline at a higher power includes not only the formation of C=N conjugated polymer via the opening of C≡N triple bonds but also the formation of the non-conjugated polymer by the combination of a variety of reactive fragments.The high power plasma polymerization occurs predominantly in the gas phase,as confirmed by the results of bothstructuralandmorphologicalanalysis.Furthermore,itshould be mentioned that the plasma etching reaction also affects the thickness of plasma film,especially for higher discharge powers or a longer plasma duration time,in which ablation gives rise to the decrease in thickness of plasma film.

Fig.5 Thickness of films as a function of deposition time at different powers
Fig.6 shows the surface morphology of PP3QCN thin films deposited at different discharge powers.Clearly,a high-quality PP3QCN10 film(Fig.6(a))could be obtained under a discharge power of 10 W.It is transparent and homogeneous,quite suitable for the measurement of dielectric properties.The RMS roughness for PP3QCN10 film is only 0.553 nm.The smooth morphology suggests that the plasma polymerization has occurred predominantly on the substrate surface,instead of in the gas phase[32].Theplasmafilmgrowsthroughreactionwith monomeric species that reach the substrate surface.This plasma polymerization mechanism at low discharge power leads to a lower filmforming rate on the surface of substrate,as indicated in Fig.5. On the other hand,in the case of higher discharge power such as 25 W,the PP3QCN25 film(Fig.6(b))has a rougher surface with an RMS roughness of 2.352 nm,much larger than the value obtained at the low power,suggesting that the plasma polymerization might be carried out mainly in the gas phase.In other words,the particles might be formed in the gas phase and then deposited on the substrate[34],which results in a relatively rapid deposition rate,as shown in Fig.5.
2.3 Film dielectric properties
Fig.7 illustrates the capacitance-voltage(C-V)curves of the PP3QCN thin films measured at 303 K.The dielectric constants of PP3QCN10 and PP3QCN25 thin films,deduced from the CV curves,are also shown in Fig.7.It is clear that the PP3QCN10 film has a much lower dielectric constant(εr=2.45)as compared with that of PP3QCN25 film(εr=5.84).The significant difference in dielectric constant between PP3QCN10 film and PP3QCN25 film may be attributed to their different free radical concentrations.Plasma polymerization at high discharge power results in a high concentration of radicals in the deposited film,a high concentration of radicals in PP3QCN25 film can lead to a higherpolarizabity in PP3QCN25 film than PP3QCN10 film.Thus,it is not difficult to understand that PP3QCN10 film can exhibit a lower dielectric constant in comparison with PP3QCN25 film.

(a) (b)Fig.6 AFM images of PP3QCN10(a)and PP3QCN25(b)substrate:quartz;roughness:0.106 nm

Fig.7 C-V curves of PP3QCN thin films measured at 1 MHz at 303 K
The variation of dielectric constant with frequency in the range from 100 kHz to 1 MHz is shown in Fig.8.It can be seen that in the whole frequency range there is an increase in dielectric constant with a decrease in frequency.The observed frequency dependence of the dielectric constant might be attributed to combination of two factors.First,it has been reported[35-36]that there are some charges present in dielectric materials which can migrate some distance through the dielectric as an electric field is applied.Space charge and macroscopic field distortions are resulted when such carriers are impeded in their motion.As the charge carriers migrate under the influence of an electric field,it is possible that they get blocked at the electrode dielectric interface,which leads to interfacial polarization.Such distortion causes an increase in capacitance of PP3QCN thin films at low frequencies.Second,the plasma deposited film has a high concentration of radicals,as has been reported by our previous publication[37].The rotation of radicals in PP3QCN thin films may result in an increase in the polarization in the low frequency region.And thus,the corresponding dielectric constant for PP3QCN thin films will increase with a decrease in frequency.
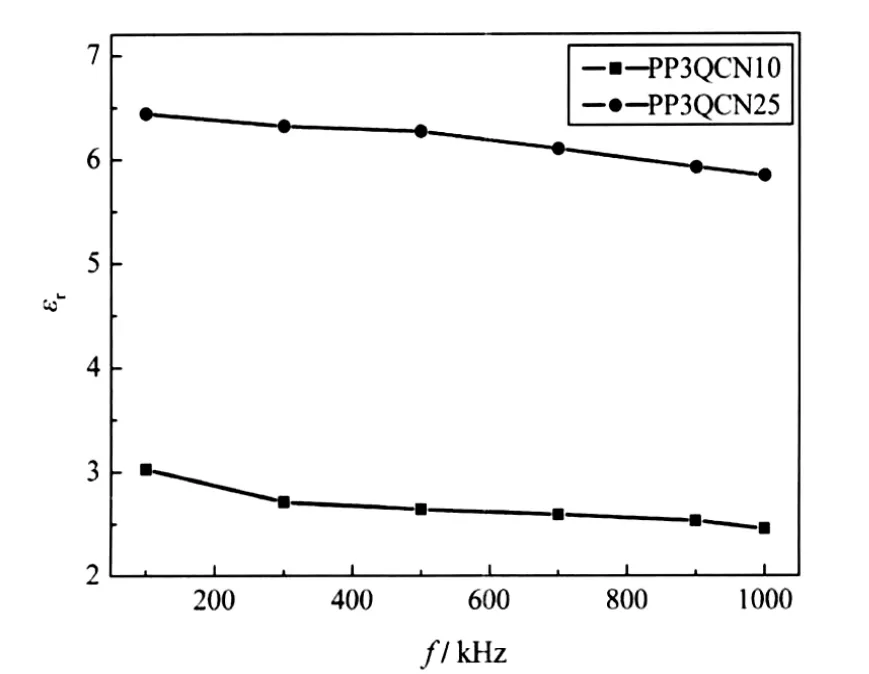
Fig.8 Variation of dielectric constant(εr)of PP3QCN thin films as a function of frequency
3 Conclusions
In this article,we report the synthesis and characterization of a novel low dielectric constant polyquinoline derivative thin film.The results show that a higher retention of the aromatic ring structure of the starting monomer in the deposited plasma films can be obtained at low plasma discharge power of 10 W, the obtained thin film is transparent,smooth and homogeneous (roughness of 0.553 nm).A high discharge power of 25 W brings about more severe monomer fragmentation and a significantly rougher surface(roughness of 2.352 nm).Moreover,PP3QCN10 thinfilmdepositedat10Wgivesamuchlower dielectric constant (εr=2.45)as compared with that of PP3QCN25 film deposited at 25 W.The significant difference in dielectric constant between PP3QCN10 film and PP3QCN25 film may be ascribable to their different film structures.The plasma-polymerized 3-cyanoquinoline might be a potential candidate to be used as intermetallic dielectrics in microelectronics.
1 Chiu,C.C.;Lee,C.C.;Chou,T.L.;Hsia,C.C.;Chiang,K.N. Microelectronic Engineering,2008,85:2150
2 Broussous,L.;Puyrenier,W.;Rebiscoul,D.;Rouessac,V.;Ayral, A.Microelectronic Engineering,2007,84:2600
3 Vilmay,M.;Roy,D.;Volpi,F.;Chaix,J.M.Microelectronic Engineering,2008,85:2075
4 Chapelon,L.L.;Petitprez,E.;Brun,P.;Farcy,A.;Torres,J. Microelectronic Engineering,2007,84:2624
5 Yu,M.B.;Ning,J.;Balakumar,S.;Bliznetsov,V.N.;Lo,G.Q.; Balasubramanian,N.;Kwong,D.L.Thin Solid Films,2006,504: 257
6 Chang,T.S.;Chang,T.C.;Liu,P.T.;Chiang,C.Y.;Chen,S.C.; Yeh,F.S.Thin Solid Films,2006,515:1117
7 Charles-Alfred,C.;Jousseaume,V.Surface and Coatings Technology,2007,201:9260
8 Lee,K.H.;Lee,K.;Suk,M.;Choi,J.M.;Im,S.;Jang,S.;Kim,E. Organic Electronics,2009,10:194
9 Yang,F.Y.;Chang,K.J.;Hsu,M.Y.;Liu,C.C.Organic Electronics,2008,9:925
10 Wang,R.C.J.;Chang-Liao,K.S.;Wang,T.K.;Chang,M.N.; Wang,C.S.;Lin,C.H.;Lee,C.C.;Chiu,C.C.;Wu,K.Thin Solid Films,2008,517:1230
11 Srikanth,N.;Tiong,L.C.;Vath,C.J.Microelectronic Engineering, 2008,85:440
13 Behniafar,H.;Habibian,S.Polymer International,2005,54:1134
14 Behniafar,H.;Haghighat,S.Polymer Advanced Technology,2008, 19:1040
15 Zhao,X.Y.;Wang,M.Z.;Zhang,B.Z.;Wang,Z.Thin Solid Films,2008,516:8272
16 Zhao,X.Y.;Wang,M.Z.;Wang,Z.Plasma Processes and Polymers,2007,4:840
17 Tseng,H.S.;Chiou,B.S.;Wu,W.F.;Ho,C.C.Microelectronic Engineering,2008,85:104
18 Yang,P.;Lu,D.;Kumar,R.;Moser,H.O.Nuclear Instruments and Methods in Physics Research Section B-Beam Interactions with Materials and Atoms,2005,238:310
19 Gonda,V.;Jansen,K.M.B.;Ernst,L.J.;Toonder,J.D.;Zhang,G. Q.Microelectronics Reliability,2007,47:248
20 Xu,T.T.;Liang,G.Z.;Lu,T.L.Polymer Bulletin,2006,11:69
21 Ouaknine,M.;Malik,I.J.;Odera,M.;Ishigaki,T.;Ueda,T.; Fukada,T.;Yoo,W.S.;Soussan,P.;Muller,P.Microelectronic Engineering,2007,74:2646
22 Liao,E.B.;The,W.H.;Teoh,K.W.;Tay,A.A.O.;Feng,H.H.; Kumar,R.Thin Solid Films,2006,504:252
23 Tsuchiya,K.;Ishii,H.;Shibasaki,Y.;Ando,S.;Ueda,M. Macromolecules,2004,37:4794
24 Matsumoto,K.;Shibasaki,Y.;Ando,S.;Ueda,M.Polymer,2006, 47:3043
25 Liu,B.Y.;Li,Y.;Mathews,A.S.;Wang,Y.G.;Yan,W.D.; Abraham,S.;Ha,C.S.;Park,D.W.;Kim,I.Reactive&Functional Polymers,2008,68:1619
26 Cho,S.J.;Bae,I.S.;Park,Y.S.;Hong,B.;Park,W.;Park,S.C.; Boo,J.H.Surface and Coatings Technology,2008,202:5654
27 Cho,S.J.;Bae,I.S.;Jeong,H.D.;Boo,J.H.Applied Surface Science,2008,254:7817
28 Bae,I.S.;Cho,S.J.;Choi,W.S.;Hong,B.Y.;Kim,Y.J.;Kim,Y. M.;Boo,J.H.Thin Solid Films,2008,516:3577
29 Lambert,J.H.;Lightner,D.A.;Shurvell,H.F.;Cooks,R.G. Introduction to organic spectroscopy.New York:MacMillan Publishing,1987
30 Zhao,X.Y.;Hu,X.;He,Y.K.;Chen,H.Y.Polym.Eng.Sci., 2000,40:2551
31 Sandrin,L.;Silverstein,M.S.;Sacher,E.Polymer,2001,42:3761
32 Silverstein,M.S.;Visoly-Fisher,I.Polymer,2002,43:11
33 Beamson,G.;Briggs,D.High resolution XPS of organic polymers. New York:Wiley,1992
34 Chen,R.;Silverstein,M.S.J.Polym.Sci.A-Polym.Chem.,1996, 34:207
35 Jayalekshmi,S.;Pillai,M.G.K.Thin Solid Films,1984,122:197
36 Sakthi K.D.;Yoshida,Y.Sur.Coat.Tech.,2003,169-170:600
37 Zhao,X.Y.;Hu,X.;He,Y.K.;Chen,H.Y.J.Mater.Sci.-Mater. Electron.,2001,12:557
November 9,2009;Revised:January 31,2010;Published on Web:March 2,2010.
Synthesis and Characterization of a Polyquinoline Derivative Thin Film with a Low Dielectric Constant
ZHAO Xiong-Yan*
(College of Material Science and Engineering,Hebei University of Science and Technology,Shijiazhuang 050018,P.R.China)
We prepared a novel plasma polyquinoline derivative thin film,plasma-polymerized 3-cyanoquinoline (PP3QCN).Fourier transform infrared spectroscopy(FT-IR),UV-visible(UV-Vis)absorption spectroscopy,X-ray photoelectron spectroscopy(XPS),and atomic force microscopy(AFM)characterization revealed that the plasma polymerization conditions affected the chemical structure,surface composition,morphology,and dielectric property of the plasma-deposited films.A smooth and homogenous PP3QCN film with a large π-conjugated system and a high retention of the aromatic ring structure of the monomer was obtained at a low discharge power of 10 W.At 25 W,more severe monomer molecular fragmentation was apparent during the plasma polymerization and thus the conjugation length of the PP3QCN films decreased because of the formation of a non-conjugated polymer.A low dielectric constant of 2.45 was obtained for the as-grown PP3QCN thin film deposited at 10 W.
Plasma polymerization; Low dielectric constant;3-Cyanoquinoline;Integrated circuit
*Corresponding author.Email:zhaoxy66@126.com;Tel:+86-311-88632425.
The project was supported by the Natural Science Foundation of Hebei Province,China(B2007000613)and Scientific Research Key Foundation for Returned Overseas Chinese Scholars,Ministry of Personnel of China.
河北省自然基金(B2007000613)和国家人事部回国人员重点资助项目
赵雄燕,1993年9月至1996年7月在北京大学化学系学习,获得理学博士学位.
O646
