Yajieshaba prevents lipopolysaccharide-induced intestinal barrier injury via anti-inflammatory and anti-apoptosis
2022-10-14YANGLipingYUXinglinZHANGChaoCHENPuDUANXiaohua
YANG Liping,YU Xinglin,ZHANG Chao,CHEN Pu,DUAN Xiaohua
YANG Liping,YU Xinglin,ZHANG Chao,CHEN Pu,DUAN Xiaohua,Yunnan Key Laboratory of Dai and Yi Medicines,Yunnan University of Chinese Medicine,Kunming 650500,China
Abstract OBJECTIVE: To investigate the protective effect of Yajieshaba (YJSB) on the intestinal barrier dysfunction induced by lipopolysaccharide (LPS).METHODS: C57BL/6 mice and rat intestinal epithelial cells were treated with LPS.Thiazolyl Blue Tetrazolium Bromide assay were used to detect cell viability.DLactate,diamine oxidase and myeloperoxidase and cytokines were determined by enzyme-linked immunosorbent assay.Western blot was used to detect apoptosis-related proteins and tight junction (TJ) proteins.Real-time quantitative polymerase chain reaction was used to quantify the levels of mRNA expression of cytokines.Histological analysis was performed by hematoxylin and eosin staining.An immunofluorescence staining assay was performed to determine the expression level of TJ protein.RESULTS: YJSB increased cell viability and decreased apoptosis,maintained intestinal permeability after LPSinduced.YJSB inhibited LPS-induced decrease of TJ protein expression,pro-inflammatory cytokine levels and neutrophil infiltration.CONCLUSION: YJSB protect against LPS-induced intestinal barrier dysfunction via anti-inflammatory and anti-apoptosis,suggesting its therapeutic potentialagainst intestinal barrier injury-related diseases.
Keywords: inflammation;apoptosis;lipopolysaccharides;Yajieshaba;intestinal barrier function
1.INTRODUCTION
The intestinal barrier primarily comprise of epithelial cells and intercellular tight junction (TJ).The function of the intestinal barrier,the most crucial physical barrier against endogenous and exogenous noxious antigens and pathogens,mainly,is dependent on the intestinal epithelium integrity.1Studies have revealed that intestinal barrier dysfunction plays a pivotal role in the pathogenesis of gastrointestinal diseases,for instance,by increasing intestinal permeability,subsequent translocation of bacteria from the gut,systemic inflammatory response,and septic multiple organ dysfunctions,all of which,are implicated in the pathogenesis of various intestinal diseases.2-5Cytokines and inflammatory mediators,such as tumor necrosis factor (TNF)-α,interleukin (IL)-1β,and interleukin (IL)-6 influence the functions of the epithelial barrier and TJ.6Mounting evidence indicates that systemic inflammations,including sepsis,play a crucial role in intestinal barrier functionsviadifferent mechanisms,for example,induction of gross lesions,intestinal epithelial cells apoptosis and shedding,and reduction in TJ,subsequently causing intestinal barrier dysfunction.4,7,8Studies have shown that lipopolysaccharide (LPS),the major cell wall component of Gram-negative bacteria,may impair the mucosal barrier integrity and cause different pathological changes.9,10LPS-mediated disruption of the TJ structure was shown to cause mucosal hyper-permeability,inducing both local and systematic inflammatory responses.11-13
Traditional Dai medicine,one of China's four important systems of ethnomedicine,has a history of 2500 years.Yajieshaba (YJSB) is a classic traditional Dai medicine prepared by Xishuangbanna Hospital of Dai Indigenous Medicine.Its major constituents include five herbs:Kuteng (Herba Dregeae Sinensis),Zhuyelan (Herba seu Rhizoma Arundinae Graminifoliae),Tianxianteng(Caulis Aristolochiae Debilis),Tianguotenggen (RadixMappianthi Iodoidis) and Choumoli (Folium seu Radix Clerodendri Simplicis) is named Dai Bai Jie,Bai Yang,Hei Tao Han,Deng Hei Hand,and Bin Hao in Dai medicine.In traditional Dai hospitals,YJSB has proved to be effective in managing different types of gastrointestinal disorders,including inflammatory bowel disease,irritable bowel syndrome,stomach ulcers,and diarrhea.14-17In our previous work,we found that palmatine and berberine,the active components of YJSB,potentially alleviated food allergy symptoms in ovalbumin-sensitized mice.18These data suggest that YJSB may protect the intestinal barrier against pathogen invasion from the external environment.However,it remains elusive as to how YJSB influences the intestinal barrier function.
Herein,we investigate the potential regulatory role of YJSB on LPS-induced intestinal barrier injury,and explore the potential mechanisms.This study,for the first time,provides evidence for the efficacy of YJSB in preventing LPS-induced intestinal barrier dysfunction,which may enrich our understanding of the mechanisms of YJSB in managing gastrointestinal diseases.
2.MATERIALS AND METHODS
2.1.Animals
Male C57BL/6 mice (weight,18-22 g) were obtained from the Institute of Laboratory Animal Sciences(Sichuan,China).The Animal Ethics Committee of Yunnan University of Traditional Chinese Medicine approved all procedures,performed under the National Institutes of Health guidelines for the care and use of laboratory animals.
2.2.Treatment groups and medication procedure
We constructed the animal model using a method previously described.19Following 3 d of habituation,mice were divided into 4 groups (n=12 per group),and orally administered with saline or YJSB (1.17,3.51 mg/kg) for 7 d.The control group received intraperitoneal injection of saline on day 7,while the remaining three groups underwent intraperitoneal injection with LPS (5 mg/kg) (Sigma,St.Louis,MO,USA) 1 h after YJSB or saline treatment.All mice were sacrificed 5h after intraperitoneal injection of LPS,and blood was collectedviaretro-orbital puncture.We removed the whole small intestine and jejunum isolated for subsequent analysis.
2.3.Histopathologic evaluation
The jejunum was fixed in 10% formaldehyde and embedded in paraffin for section.The tissue was stained with hematoxylin and eosin (HE) to evaluate the intestinal histopathological injury.Images were captured using an optical microscope (Olympus,Tokyo,Japan)and was followed by pathological analysis.The villi height and crypt depth were measured by ImageJ using the segmented line tool.
2.4.YJSB extraction
YJSB was obtained from Xishuangbanna Dai Autonomous Prefecture People's Hospital (Xishuangbanna,China) and Professor Chao Zhang from Yunnan University of Traditional Chinese Medicine,Kunming,China assisted in the identification.YJSB was powdered and extracted thrice with 70% alcohol for 3 h.Subsequently,extracts were combined and concentrated using a vacuum rotary evaporator and lyophilized.The dried powder was dissolved in distilled water,filtered and stored at-20 ℃.
2.5.Cell culture and treatment
Intestinal epithelial cells (IEC-6) cells were obtained from Conservation Genetics CAS Kunming Cell Bank and cultured in DMEM (Biological Industries Israel Beit Haemek Ltd.,Kibbutz Beit Haemek,Israel)supplemented with 10% fetal bovine serum (Biological Industries Israel Beit Haemek Ltd.,Kibbutz Beit Haemek,Israel),100 U/mL penicillin and 100 mg/mL streptomycin (Biological Industries Israel Beit Haemek Ltd.,Kibbutz Beit Haemek,Israel) in a humidified incubator (5% CO2at 37 ℃).After 24 h of incubation,cells were plated and treated,with YJSB extract for 1h and then co-exposed to LPS for another 24 h.We used the supernatant was used to detect the contents of TNFα,IL-1β and IL-6.
2.6.Cell viability
Thiazolyl Blue Tetrazolium Bromide (MTT) assay was employed for cell viability.IEC-6 cells (1 × 105) were seeded in 96-well culture plates and left for 24 h.Next,the cells were treated with YJSB or LPS.5 mg/mL of MTT (Sigma-Aldrich,St.Louis,MO,USA) incubated for 4 h,and then lysed.The dark blue crystals were solubilized with dimethylsulfoxide (DMSO).Absorbance measurement was taken at 490 nm using a microplate reader (ThermoFisher Scientific,Waltham.MA,USA).
2.7.Enzyme-linked immunosorbent assay (ELISA)
The levels of TNF-α,IL-1β,IL-6,D-Lactate,myeloperoxidase (MPO),and diamine oxidase (DAO)were measured by ELISA kit following the manufacturer’s instructions.IL-1β,TNF-α and IL-6 assay kits were purchased from ProteinTech (Chicago,IL,USA).D-Lactate assay kit was purchased from Abcam (Cambridge,UK).MPO assay kit and DAO assay kit were purchased from CUSABIO (Wuhan,China).
2.8.Immunofluorescence staining
Cells seeded on the coverslips were fixed,permeabilized and incubated with the primary antibody overnight at 4 ℃.This was followed by a 1h-incubation with a secondary antibody and counterstained with Hoechst.Images were captured by a fluorescence microscope(Nikon,Tokyo,Japan).
2.9.Real-time quantitative polymerase chain reaction(RT-qPCR)
Total RNA of mice jejunum was isolated with Trizol(Takara,Kusatsu,Japan) following manufacture’s protocol and reversed transcribed to cDNA by PrimeScript RT reagent Kit (ABI,Foster City,CA,USA).A real-time PCR system (Applied Biosystems,CA,USA)was employed for PCR amplification of the cDNA using the SYBR Green PCR Master Mix (ABI,Foster City,CA,USA).Relative changes in gene expression levels were analyzed via the 2-ΔΔctmethod.Primers were designed using Primer3 and synthesized by Sangon Technologies(Shanghai,China).PCR reactions were performed as follows: one cycle of 5 min at 95 ℃ and 40 cycles of 15 s at 95 ℃ and 30 s at 60 ℃.The primers were: TNF-α forward 5’-TCAACCTCCTCTCTGCCATC-3’ and reverse 5’-CCAAAGTAGACCTGCCCAGA-3’;IL-1β forward 5’-ACAGATGAAGTGC-TCCTTCCA-3’ and reverse 5’-GTCGGAGATTCGTAGCTGGAT-3’;IL-6 forward 5’-TTCCAGCCAGTTGCCTTCTT-3’ and reverse 5’-AAGCCTCCGA-CTTGTGAAGTG-3’;βactin forward 5’-AGCCTCAAGATCATCAGCAATG-3’and reverse 5’-TGTGGTCATGAGTCCTTCCACG-3’.
2.10.Western blotting
Total protein extracts of jejunum were prepared in lysis buffer (Meilunbio,Dalian,China).The BCA assay(Beyotime,China) was applied to determine protein concentrations.Equal amounts of protein were subjected to sodium dodecyl sulfate polyacrylamide gel electrophoresis gel and then transferred to polyvinylidene fluoride membranes.Membranes were immunoblotted with the primary antibodies (ProteinTech,Chicago,IL,USA).The membranes were incubated with secondary antibodies after extensive washing.We used the enhanced chemiluminescence method to detect protein bands and an Automatic Gel Imaging and Analysis System (Beijing,China) for analysis.
2.11.Statistical analysis
Data were analyzed with SPSS 17.0 statistical software(SPSS Inc.,Chicago,IL,USA),and presented as means± standard deviation.Differences between groups were analyzed by a one-way analysis of variance.P< 0.05 denoted statistically significant.
3.RESULTS
3.1.YJSB attenuates LPS-mediated IEC-6 cell cytotoxicity
The MTT assay results showed that LPS had a dosedependently toxic effect on IEC-6 cells (Figure 1A).LPS(100 μg/mL) exerted a significant toxic effect on cells;therefore was used in subsequent studies.YJSB alone did not affect cell viability,even at a relatively high concentration of 480 μg/mL (Figure 1B).Lastly,IEC-6 cell viability was partly improved by YJSB when exposed to LPS (Figure 1C).
3.2.YJSB alleviates LPS-induced decrease of TJ protein
We investigated whether YJSB could maintain barrier function by tuning the LPS-induced expression of TJ proteins.Results demonstrated that LPS at 100 μg/mL altered the distribution of TJ proteins at the cellular borders,providing evidence of TJ dysfunction.This effect was suppressed significantly by YJSB (Figure 2A).Evaluation of the expression of intestinal TJ proteins in mice yielded similar results that YJSB alleviated LPSinduced decrease of TJ proteins (Figure 2B).
3.3.YJSB promotes the recovery of intestinal barrier dysfunction in mice
Intestinal barrier dysfunction potentially increases intestinal permeability accompanied by enhanced levels of D-Lactate and DAO in the serum.20,21To examined the effect of YJSB on intestinal permeability and absorption ability,we evaluated changes in the levels of D-Lactate and DAO in mice serum.Results demonstrated that,as compared with the LPS-treated group,mice treated with 3.51 g/kg of YJSB after LPS injection exhibited significantly lower serum D-Lactate concentrations(Figure 3A).Serum DAO levels in YJSB-treated mice were notably lower than those in the LPS-treated group(Figure 3B).H&E staining of jejunum tissue revealed that LPS-induced intestinal pathological injury was markedly attenuated after pretreatment with YJSB(Figure 3D,G).These results provide insights into the potential role of YJSB in LPS-induced intestinal injury.

Figure 1 YJSB attenuates LPS-mediated IEC-6 cell cytotoxicity
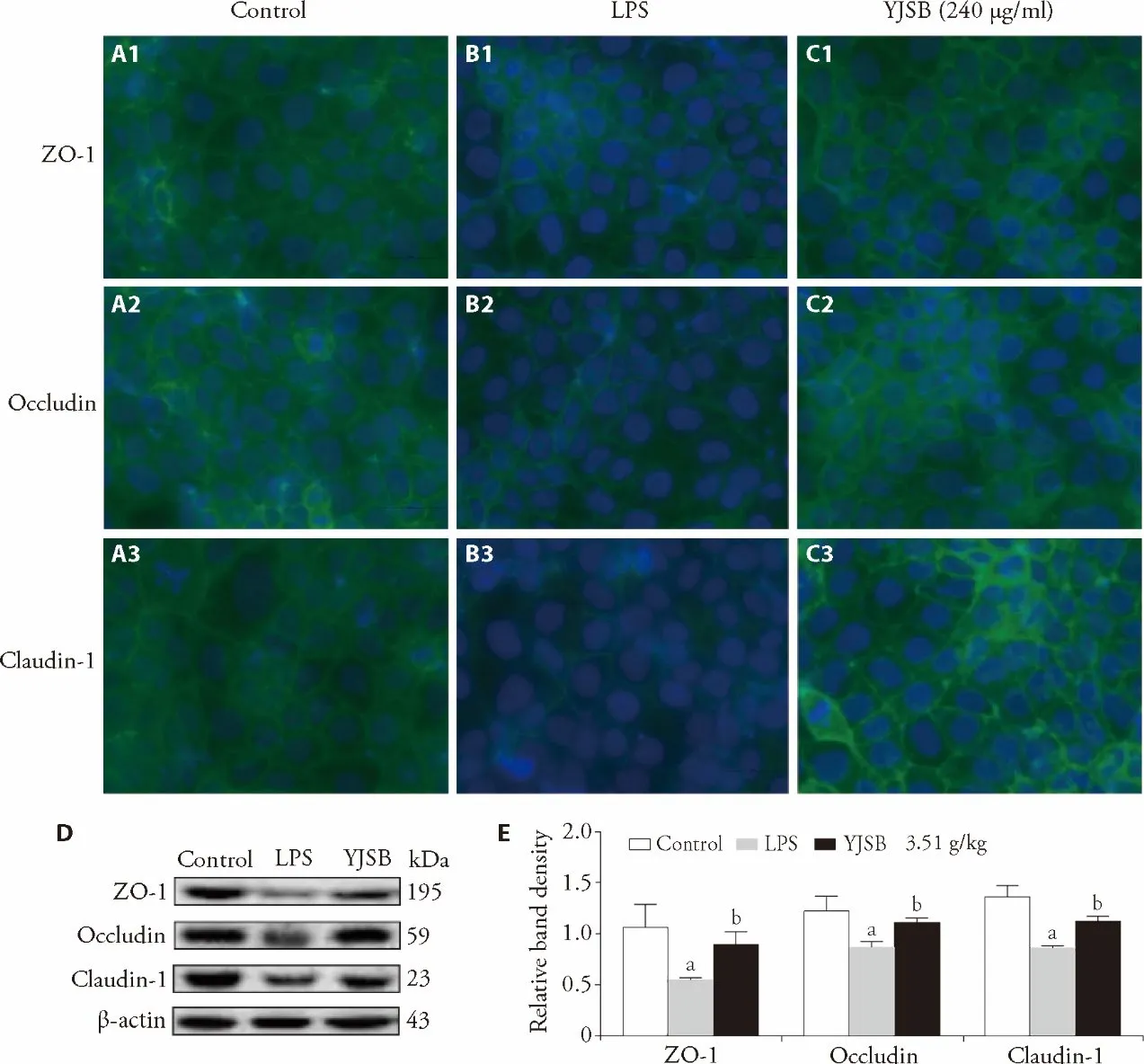
Figure 2 Effect of YJSB on the expression of TJ proteins
3.4.YJSB attenuates LPS-induced cell apoptosis
By examining the effect of YJSB on the expression levels of B-cell lymphoma-2 (Bcl-2) and Bcl-2 assaciated X protein (Bax) in the intestine.LPS was revealed to inhibit Bcl-2 expression but promoted the expression of Bax (Figure 4).However,YJSB abolished the effect of LPS on the expression of Bcl-2 and Bax.The findings demonstrated that YJSB inhibits LPS-induced apoptosis.
3.5.YJSB ameliorates LPS-induced inflammatory response in vitro and in vivo
We reported significantly higher mRNA expression of TNF-α,IL-1β,and IL-6 and MPO activity in the LPStreated group;however,YJSB significantly decreased both the pro-inflammatory cytokines and MPO activity(Figure 5).Similarly,ELISA results showed that the LPS-induced secretion of TNF-α,IL-1β and IL-6 was significantly decreased in the YJSB-treated IEC-6 cells.
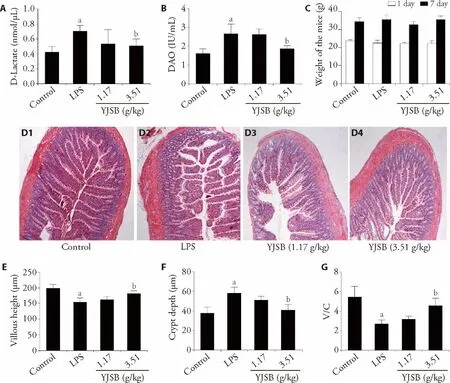
Figure 3 Effect of YJSB on intestinal histological appearance and permeability

Figure 4 Effect of YJSB on LPS-induced apoptosis

Figure 5 YJSB ameliorates LPS-induced inflammatory response

Figure 6 Mechanism of YJSB in protecting the intestinal barrier function
4.DISCUSSION
The homeostatic balance between proliferation and apoptosis in the intestinal epithelial cells plays a pivotal for the intestinal epithelium to function as a structural barrier.22This is why intestinal epithelial barrier dysfunction may cause inflammatory intestinal diseases.23There is evidence that LPS can disrupt intestinal function.24Our previous study on acute toxicity assay demonstrated that mice receiving YJSB at a concentration of 88 g/kg (body weight) did not have any toxic effects.In this study,we report the optimal concentration of YJSB for IEC-6 cells at a ranged of 30-480 μg/mL,demonstrating that YJSB has no overall significant toxicity.In this view,we investigated the protective effect of YJSB on intestinal barrier dysfunction was utilizing IEC-6 cells and an LPSinduced mouse model,but without a positive control because YJSB is a compound prescription yet no wellrecognized medicine is available for intestinal barrier injury treatment.Our analysis revealed that YJSB attenuates LPS-induced intestinal barrier injury of IEC-6 cells and intestinal barrier dysfunction in mice by inhibiting inflammation and apoptosis and maintaining TJ integrity.
A combinative interaction between epithelial cells to form TJs is the gold standard for the maintenance of intestinal barrier integrity.25The role of TJ protein in epithelial barrier function is precise.A study demonstrated that LPS-induced endothelial hyper-permeability was associated with a decrease in TJ-related protein expression.26The proteins,ZO-1,occludin and claudin-1 are crucial in the formation of TJ.In most cases,an increased abundance of TJ proteins is correlated with an improved epithelial barrier function.There is evidence that intestinal barrier dysfunction increases intestinal permeability,accompanied by elevated serum levels of D-Lactate and DAO.Here,we found that LPS effecttuated a dramatic decrease in the expression of ZO-1,occludin and claudin-1,which consequently,impaired intestinal barrier function and increased intestinal permeability.However,YJSB restored the reduction in the abundance of ZO-1,occludin and claudin-1,induced by the LPS challenge.Notably,this effect was evident in bothin vitroandin vivoexperiments.The serum DLactate level is a useful marker for intestinal injury assessment as it mirrors the change of intestinal permeability.20Many scholars have employed DAO to evaluate intestinal mucosal structure and function,by releasing it into the blood during intestinal mucosal injury and necrotization.21Ourin vivoinvestigation revealed significantly lower serum levels of DAO and DLactate in YJSB treated-mice compared to those in the LPS-induced group.These findings suggest that YJSB potentially promotes the recovery of LPS-induced intestinal mucosal barrier damage by strengthening intercellular TJ and reducing intestinal permeability.We also examined the effect of YJSB on intestinal morphology,using the villus height and crypt depth as a criterion to reflect intestinal function.27In the normal group,the colonic mucosa was intact,with a neat arrangement of the intestinal epithelial cells and glands.Moreover,LPS initiated intestinal morphologic changes,among them,epithelium sloughing,villous atrophy and reduced villus height and villus height to crypt depth ratio.YJSB treatment mitigated the intestinal morphologic damage and increased the villus height and villus height to crypt depth ratio.These events,protected and maintained the integrity of intestinal mucosa,supporting the view that YJSB could be critical for maintaining the gut barrier function.
Previous evidence had suggested that increased apoptosis and decreased proliferation are potentially the main mechanisms underlying epithelial barrier dysfunction and that epithelial cell apoptosis can increase the permeability of the epithelial barrier.7,28,29Elevated levels of apoptosis have been observed in the intestinal epithelium of patients with inflammatory bowel disease,providing convincing evidence that increased epithelial cell proliferation or decreased cell apoptosis may contribute to intestinal barrier restoration.30We examined the protein expression of Bcl-2 (anti-apoptotic proteins) and Bax (pro-apoptotic protein) to gather more evidence on whether YJSB treatment potentially mediates epithelial cell apoptosis.Intriguingly,YJSB increased and decreased the levels of Bax and Bcl-2,respectively.The finding suggests a role for YJSB in antagonizing LPS-induced apoptosis by regulating the expression of apoptosis-related proteins.Intestinal epithelial barrier dysfunction may also trigger or deteriorate intestinal inflammatory disorder.31Studies show that inflammatory cytokines release and neutrophil infiltration are significant in the pathogenesis of gastrointestinal diseases and that intestinal barrier injury causes mucosal hyper-permeability,noxious substances through the intestinal mucosa.32Intestinal inflammation is characterized by the excessive release of inflammatory cytokines which are known to aggravate intestinal damage and gut inflammation,inducing epithelial cell apoptosis and barrier permeability.33Studies have demonstrated significantly increased release of critical pro-inflammatory cytokines,TNF-α,IL-1β,and IL-6,in intestinal inflammation-related diseases.34Similarly,we,herein,reported that an increase in the production of TNF-α,IL-1β and IL-6 preceded the intestinal barrier dysfunction in both LPS-induced IEC-6 cells and the mouse model.However,there was a notable YJSBinduced suppression in the production of the cytokines in IEC-6 cells and mRNA expression of TNF-α,IL-1β,and IL-6 in the mouse model.LPS treatment markedly increased the MPO activity in mice serum,which is an index of neutrophil infiltration and inflammation.Contrarily,YJSB pre-treated mice showed significantly decreased MPO activity,further validating the antiinflammatory activity of YJSB,which is crucial in preventing LPS-induced alteration of the intestinal barrier function.
In conclusion,YJSB treatment improves LPS-induced intestinal barrier dysfunction by blocking the release of pro-inflammatory cytokines and apoptosis,in vivoandin vitro.Therefore,YJSB has potential clinical implications to ameliorate intestinal barrier dysfunction.
杂志排行
Journal of Traditional Chinese Medicine的其它文章
- Effectiveness and safety of tripterygium glycosides tablet (雷公藤多苷片) for lupus nephritis: a systematic review and Meta-analysis
- Efficacy of green tea extract on PC3 prostate cancer cells through upregulation of miR-195 expression and suppression of epithelial to mesenchymal transition
- Qilan preparation (芪蓝颗粒) inhibits proliferation and induces apoptosis by down-regulating microRNA-21 in human Tca8113 tongue squamous cell carcinoma cells
- Tenglong Buzhong granules (藤龙补中颗粒) inhibits the growth of SW620 human colon cancer in vivo
- Antihepatofibrotic effect of Guizhifuling pill (桂枝茯苓丸) on carbon tetrachloride-induced liver fibrosis in mice
- Huangqi decoction (黄芪汤) attenuates renal interstitial fibrosis via transforming growth factor-β1/mitogen-activated protein kinase signaling pathways in 5/6 nephrectomy mice
