Effects of silkworm pupa protein on apoptosis and energy metabolism in human colon cancer DLD-1 cells
2022-06-23XiojioJiJingWngAijinDuoFengYueHeWenjieYn
Xiojio Ji, Jing Wng, Aijin M, Duo Feng, Yue He, Wenjie Yn,*
a College of Biochemical Engineering, Beijing Union University, Beijing 100101, China
b Institute of Food and Nutrition Development, Ministry of Agriculture and Rural Affairs, Beijing 100125, China
c College of Food and Health, Beijing Technology and Business University, Beijing 100048, China
Keywords:
Colon cancer DLD-1 cells
Silkworm pupa protein
Apoptosis
Mitochondria
Glycolysis
A B S T R A C T
Objective: To investigate the effects of silkworm pupa (Bombyx mori) protein (SPP) on cell proliferation,apoptosis and energy metabolism in human colon cancer cells DLD-1. Methods: CCK-8 was used to detect cell proliferation rate after 72 h of cell culture for the control group (normal cultured DLD-1 cells) and SPP dose groups; Annexin-V/PI was applied to observe cell apoptosis; XFe24 Extracellular Flux Analyzer was used to detect cell mitochondrial respiratory function and glycolytic function. Results: Comparing with the control, SPP significantly inhibited the proliferation of DLD-1 cells with all the dosage tested (P < 0.01); flow cytometry showed that SPP significantly promoted apoptosis (P < 0.05). Additionally, SPP could significantly inhibited mitochondrial metabolism and glycolysis of DLD-1 cells and decreased cell energy metabolism in all groups treated with different doses. Conclusion: SPP can cause oxidative damage, promote apoptosis, and reduce mitochondrial respiratory and glycolysis rate in colon cancer DLD-1 cells, which reveals that SPP has the potential to serve as the anti-cancer drugs in the future, but further experimental evidence is needed.
1. Introduction
Malignant tumor is one of the severe public health problems that seriously threaten human health. It has the characteristics of diversity,complexity and high mortality, and are showing an increasing trend year by year according the global cancer incidence and mortality.The World Health Organization (WHO) statistics that 70% of cancer deaths occur in low- and middle-income countries, and it is estimated that cancer patients worldwide will continue to rise to 11 million in 2030 [1]. Colon cancer (CRC) is one of the most common malignant tumors in the world. In the United States, 95 270 new cases of CRC are expected and 43 030 new cases of rectal cancer in 2018, and 50 630 people are expected to die from colorectal cancer in the same year [2]. In China, CRC is also one of the most common malignant tumors, and its incidence ranks third after lung cancer and gastric cancer, with 388 000 new cases and 87 000 deaths [3]. In the past 10 years, the increase speed of colorectal cancer have been basically stable according to global incidence and mortality, but the proportion of malignant tumors has increased. Colorectal cancer has clear geographical distribution differences worldwide, and the incidence of colorectal cancer in developed areas is higher than that in underdeveloped areas [4]. Currently, natural products have been recognized for their tumor-preventive effects and have the potential of becoming chemopreventive and anti-cancer drug candidates.Yang et al. [5]found that isolated peptides in wild silk moth larvae inhibit the proliferation of rat liver cancer cells by inducing cell cycle arrest. Wang et al. [6]found that melittin induces apoptosis of liver cancer cells by activating the TAK1-JNK/p38 and the IκBα kinase-NF-κB pathway. Bincoletto et al. [7]found that royal jelly protein can prevent bone marrow suppression caused by tumor cell proliferation, and can reduce the level of prostaglandin E2 (PGE2)and improve the survival rate of EAT mice .
Silkworm pupa is a biological resource with great development potential and market value. It is rich in resources and has extremely high nutritional value and health care effects, it can enhance immunity, lower blood sugar, delay aging [8-11], etc. It can significantly promote fat metabolism and neuroprotective function by reducing oxidation [12,13].Silkworm pupa (45%-50% of dried pupa quality), rich in amino acids, well balanced, in line with the FAO/WHO recommendations of the essential amino acid model, is a natural high quality protein, with great economic value [14-16]. Lee et al. [17]has shown that it can prevent the formation of preadipocytes and can be used to treat dietary obesity in the future. Japan, Korea and other countries have listed silkworm pupa as a nutritional food, which has a broad market development prospect in the food field [18].
Mitochondria are important subcellular sites for energy metabolism in eukaryotic cells to generate adenosine triphosphate (ATP). Excessive production of reactive oxygen species from cellular oxidative damage can impair mitochondrial function and lead to abnormal energy metabolism. In addition, the glycolytic pathway also provides some energy to the cell. Due to their rapid growth, tumor cells have a higher demand for energy and intermediate metabolites than normal cells, so their metabolism of substances and energy is relatively more active.
Whether silkworm pupa (Bombyx mori) protein (SPP) affects energy metabolism in tumor cells has not been investigated to date. In this experiment, we investigated the effect of SPP on mitochondrial respiration and glycolysis in human colon cancer DLD-1 cells at the cellular level, and the relationship between energy metabolism,oxidative damage, and cell viability for possible mechanism of SPP toxicity to tumor cells were also investigated.
2. Materials and methods
2.1 About SPP
SPP (purity > 80%) was purchased from Nantong Fuer Biological Products Co., Ltd. (China). After chromatographic detection, its molecular weight is basically between 200-5 000 U, and most of the molecular weights are between 200-400 U, which should be dipeptides or tripeptides Ingredients, where favorable amino acids are present in the highly polar parts [19,20].
2.2 Cultured cell conditions
DLD-1 cells (provided by the Cell Bank of Type Culture Collection of Chinese Academy of Sciences) were cultured in RPMI-1640 medium with 10% fetal bovine serum and 1% antibodies(100 U/mL penicillin, 100 mg/mL streptomycin) at 37 °C in a 5% CO2saturated humidified incubator, and the cells were digested with 0.25% trypsin to reach a confluence of 80%-90%, and the cells in the logarithmic growth phase were used for subsequent experiments.
2.3 CCK-8 assay
Cells at a density of 5 000 cells/100 μL in a 96-well cell culture plate were incubate in a 5% CO2incubator at 37 °C for 24 h. 100 μL of SPP (0, 0.8, 1.6, 2.4, 3.2, 4.0 mg/mL) were added to each well, and 6 replicate wells for each group were incubated at 37 °C and 5% CO2incubator for 24 h. After 24 h incubation under saturated humidity,100 μL of medium containing 10% CCK-8 were added to each well,and the cells were further incubated for additional 1 h, and absorbance of 450 nm were recorded [21].
2.4 Apoptosis assay
Two milliliters (1 × 105cells/mL) of cells in the logarithmic growth phase were seeded into 6-well plates and incubated at 37 °C and 5% CO2in an incubator with saturated humidity for 24 h. After the cells had grown to a fused state, the culture medium was discarded and the groups were treated with 0, 0.8, 1.6, 2.4 and 3.2 mg/mL of SPP, respectively. The cell culture incubator was incubated under 5% CO2at 37 °C in a humidified atmosphere for 24 h. Then the apoptosis rate of DLD-1 cells was measured by flow cytometry (Becton,Dickinson and Company, USA) [19].
2.5 Metabolic assays
Optimization of experimental conditions: (A) Optimize cell number. DLD-1 cells at log growth phase were collected after trypsin digestion, centrifuged, and resuspended. 250 μL of cell suspension were added to each well of Seahorse XFe24 Extracellular Flux Analyzer(Agilent Biosciences, Santa Clara, CA, USA), cell culture plate to reach cell numbers of 1 × 104, 2 × 104, 4 × 104, 8 × 104(5 replicates).(B) Carbonylcyanidep-trifluoro methoxyphenylhydrazone (FCCP).After determination of the cell numbers, the assay was run by configuring different concentrations of FCCP. (C) Mitochondrial stress and glycolysis stress test. Oxygen consumption rate (OCR) was measured by adding oligomycin, FCCP and rotenone/antimycin A.OCR values were determined at different time points, thus reflecting the level of cellular oxidative phosphorylation. The cellular acid production rate reflects the rate of glycolysis of the cells, and changes in the rate of cellular glycolysis were determined by adding glucose,oligomycin, and 2-deoxyD-glucose (2-DG) and the acid production rate were measured at different time points [22-24].
3. Results
3.1 SPP inhibited the aggressive behavior of DLD-1 cells
After 24 h of treatment with different concentrations of SPP,DLD-1 cells growth was inhibited to different degrees, and cell viability was gradually decreased with increasing concentrations of SSP, in a dose-dependent manner (Fig. 1). Compared with the control group, the cell viability of each dose group was significantly different (P< 0.01). For subsequent experiments, the cell survival rate is too low if the SPP concentration is too high. When the cells are tested by flow cytometry and energy metabolism, the 4 mg/mL dose group is removed in the subsequent experiments since the influence of cell debries.
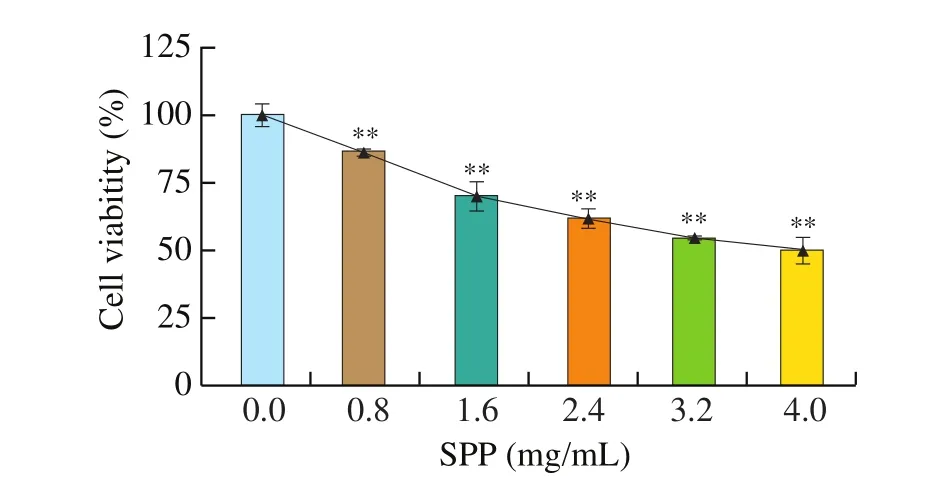
Fig. 1 CCK-8 analysis to assess cell proliferation. ** P < 0.01 versus control.
3.2 SPP induces apoptosis in DLD-1 cell
To further determine the apoptosis-inducing effect of SPP on DLD-1 cells, Annexin V-FITC/PI was used to stain DLD-1 cells, and it was found that the number of apoptotic cells increased significantly with the increasing of SPP concentration (Fig. 2A). The percentages of early and late apoptosis were increased in a dose-dependent manner,from 0.08% to 7.37% and from 2.83% to 23.45%, respectively (Fig. 2B).This result suggests that SPP may induce apoptosis in DLD-1 cells by disrupting the plasma membrane integrity.
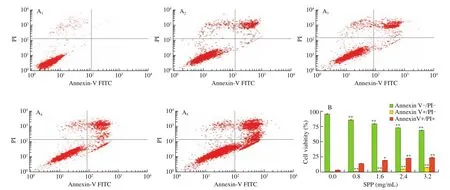
Fig. 2 LD-1 cells apoptosis changes. (A) DLD-1 cells were stained with propidium iodide (PI) and subjected to flow cytometric assay; (A1-A5) 0, 0.8, 1.6, 2.4 and 3.2 mg/mL of SPP, respectively; (B) Quantitative analysis for the cell apoptosis. ** P < 0.01 versus control, * P < 0.05 versus control.
3.3 Changes in energy metabolism of DLD-1 cells
When performing mitochondrial stress tests, cell density has a large impact on the determination of cellular OCR values. In this paper, it is found that the OCR value increases with the addition of different numbers of cells. In order to ensure the accuracy and sensitivity of the test signal, and according to the Seahorse XFe24 Extracellular Flux Analyzer instructions, when using the 24-well bioenergetics analyzer to determine cell OCR, the OCR of cells in the normal growth state should be in the range of 50-400 pmol/min,and the cells should be seeded with the same number of cells as in the 24-well bioenergetics analyzer. The density should be between 1 × 104-8 × 104cells/well. Therefore, it is appropriate to maintain the cells at 8 × 104(Figs. 3A-B).

Fig. 3 The number of DLD-1 cells and the effect of gradient concentration of FCCP on OCR. (A) OCR of DLD-1 cells in diverses cell density; (B) basal respiration rate in diverse cell density; (C) OCR of DLD-1 cells which were treated with a gradient concentration of FCCP (0, 0.125, 0.25, 0.5, 1, 2 μmol/L);(D) maximal respiration rate of the cells which were treated with gradient concentration of FCCP. Different letters indicate significant differences (P < 0.01).
FCCP is an uncoupling agent that attenuates cellular oxygen consumption and ATP synthase coupling, which increases oxygen consumption, and acts as a proton carrier. Thus it can reflux a large number of protons, leading to an increase in cellular oxygen consumption. Due to electron transfer and oxidative phosphorylation decoupling, high concentration of FCCP will be toxic to cells and can eventually cause cell death, so it is important to choose the right working concentration. In this study, after treatment of cells with oligomycin, an inhibitor of oxidative phosphorylation resulted in a decrease in OCR, different concentrations of FCCP were added in the cells mixture to examine the changes in OCR. The results showed that as the FCCP concentration increased the cellular OCR showed an increasing trend and reached a maximum value at 1 μmol/L,whereas when the FCCP concentration increased to 2 μmol/L, it did not significantly increase the maximum oxygen consumption rate level of the cells (Figs. 3C-D), indicating that the optimal working concentration of FCCP is 1 μmol/L.
It was proposed that SPP can inhibit the proliferation of colon cancer DLD-1 cells and promote apoptosis. In this study, the OCR of SPP-treated cells was measured by Bioenergetics Analyzer,and the relevant parameters were analyzed to determine the effect of SPP on the oxidative phosphorylation process of cells. As the concentration of SPP treatment increased, there was a significant decrease in the overall cellular OCR, especially in the growth phase when the cellular OCR decreased significantly after treatment with FCCP (Fig. 4A). The basal respiration and the ATP production of DLD-1 cells were gradually decreased (P< 0.01) by the above 4 oxidative phosphorylation and electron transfer chain regulator treatments. Compared with the control group, the basal respiration of SPP-treated cells decreased by 22.66%, 39.38%, 61.27%, and 65.77%,respectively (Fig. 4B), and ATP production decreased by 20.88%,37.73%, 45.91%, and 50.28%, respectively (Fig. 4C). The maximal respiration and spare respiration of DLD-1 cells decreased gradually with the increasing of SPP concentration (P< 0.01). Compared with the control group, the maximal respiration of SPP-treated cells decreased by 56.81%, 87.27%, 154.69%, and 170.70%, respectively(Fig. 4D), and spare respiration decreased by 34.15%, 47.89%,88.92%, and 109.40%, respectively (Fig. 4E). This indicated that SPP largely inhibited the mitochondrial energy metabolism process of the cell.

Fig. 4 Mitochondrial respiration changes in DLD-1 cells. (A) Overall curve of cellular OCR in response to SPP (0, 0.8, 1.6, 2.4, 3.2 mg/mL) treatment;(B) cellular basal respiration of cells subjected to SPP treatment; (C) alteration of cellular ATP production in response to SPP treatment; (D) maximal respiration of the control and SPP pre-treated cells; (E) cellular spare respiration variation of the control and SPP pre-treated cells. **P < 0.01 versus control.
Warburg et al. [25]found that tumor cells adapt to a hypoxic environment by switching metabolic pathways during growth, as evidenced by an increase in the rate of glycolysis in tumor cells, and that even after returning to normoxic conditions, tumor cells continue to rely on glycolysis for energy production. This metabolic process is defined as aerobic glycolysis. Bioenergy analyzers are capable of studying the oxygen consumption rate of cells and can be applied to the study of cellular glycolysis. In this study, Bioenergy Analyzer was adopted to determine the changes in cellular glycolysis after SPP treatment and to investigate the effect of SPP on cellular glycolysis.As the concentration of SPP increased, the cellular glycolysis process was significantly inhibited, and the overall level of extracelluar acidification rate (ECAR) decreased significantly (Figs. 5A–B),which was reflected in the gradual decrease of basal glycolysis rate, cellular glucose metabolism rate, maximal glycolysis rate and spare glycolysis capacity. Compared with the control group, the basal glycolysis rate of SPP treatment group was reduced by 4.58%,5.70%, 7.68%, and 8.01%, respectively (Fig. 5C), cellular glucose metabolism rate decreased by 20.19%, 30.37%, 39.21%, 72.72%,respectively (Fig. 5D), maximal glycolysis decreased by 26.16%,38.40%, 40.79%, 62.46%, respectively (Fig. 5E), and spare glycolysis capacity decreased by 20.11%, 28.17%, 63.50%, and 68.77%, respectively (Fig. 5F), and the differences were statistically significant (P< 0.01). The above results indicate that SPP can largely reduce the level of cellular glycolysis.

Fig. 5 Changes in regulating aerobic glycolysis in DLD-1 cells. (A) Overall ECAR curve of SPP pre-treated cells; (B) zoom in for 46.5-73.0 min in A;(C) basal glycolytic rate of control and SPP pre-treated cells; (D) cellular glycolytic capacity of convert in DLD-1 cells with or without SPP treatment; (E) maximal glycolytic rate of the control and SPP pre-treated, (F) spare glycolytic capacity alteration of DLD-1 cells due to SPP treatment. **P < 0.01 versus control.
4. Discussion
CRC is the 3rdmost common malignancy with the highest incidence rate (10.2%) worldwide [26], and its incidence and mortality rates vary greatly between developed countries and countries with economies in transition. According to the latest data published by the American Institute for Cancer Research (AICR)and the WHO International Agency for Research on Cancer (IARC),CRC is the 3rdmost common cancer in men and the 2ndmost common in women, with more than 1.8 million new cases worldwide in 2018, and the global burden of CRC is expected to increase by 60% by 2030 [26,27]. There are generally accepted potential or serious toxic side effects after cancer treatments such as chemotherapy,surgery, and radiation therapy. Therefore, the development of new effective and non-toxic antineoplastic drugs is urgently needed. In recent years, natural drugs have been playing the important role in cancer treatment. Various proteins or peptides with antitumor activity have been investigated for their antitumor effects in combinations with chemotherapeutic agents. Compared with anti-cancer drugs,biologically active proteins or peptides are preferred, since they don’t have teratogenic effect, accumulated toxicity, and are not easy to lead to drug resistance [28]. In this study, a range of concentrations of SPP could significantly inhibit the growth and proliferation of DLD-1 cells in a dose-dependent manner.
Apoptosis, a programmed cell death is a defense mechanism for normal organisms to remove senescent and incompetent cells maintaining the metabolic balance of normal cells and plays an important role in a variety of cellular functions [29]. The typical morphological characteristics of apoptosis are: the cellular morphology becomes round after the initiation of apoptosis, then the cellular adhesion disappears, the intercellular connections are disconnected,the cytoplasm becomes concentrated, the nuclear chromatin becomes concentrated and highly dense, the nucleus lyses, the typical vacuolar structure appears in the nucleus, the nuclear membrane becomes involuted, and the apoptotic body divides into multiple apoptotic cells at a later stage [30]. In addition to apoptosis, which occurs as a normal physiological process, there are alsoin vitroenvironmental factors that induce apoptosis. Apoptosis plays a role in the development and progression of many diseases including cancer, in which case the insufficient apoptosis and excessive cell proliferation happens due to the shutdown of this regulatory function of intracellular mitochondria.When apoptosis is blocked, leading to the disruption of equilibrium,which in turn lays the foundation for tumor formation [31]. The results showed that SPP could induce apoptosis and promote cell death in colon cancer DLD-1 cells.
The rapid proliferation of tumor cells will couple the greater demand for energy, therefore, targeting the bioenergetic metabolism of tumor has become a new and effective prevention approach for cancer therapy, and the efficient and complete study of the effect of molecules or drugs on the energy metabolism of tumor cells has become the main issue. The Bioenergy Analyzer uses a solid-state probe and a patented instantaneous microchamber technology to detect the cellular energy metabolic state in a real-time, sensitive,accurate and non-destructive manner, and can simultaneously measure the two main energy metabolic pathways in cells-mitochondrial respiration and glycolysis. The main indicators of cellular energy metabolism, such as basal respiration, ATP production, maximal respiration, basal glycolytic rate, maximal glycolytic rate, and spare glycolytic capacity, provide a new, convenient and rapid method for the determination of cellular energy metabolism.
In the present study, SPP showed a dose-dependent inhibition for basal respiration, maximal respiration, reserve respiration and ATP conversion, but the dose-effect relationship may be non-linear. The decrease may be due to the apoptotic state of SPP-treated cells and increased levels of reactive oxygen species (ROS)likely causing oxidative damage to cells and in turn to reduce the activity of mitochondrial respiratory chain complexes, and affect mitochondrial ATP synthesis capacity, and a high dose (3.2 mg/mL)of SPP on cellular mitochondrial aerobic/oxidative activity. This may be directly related to the mitochondrial energy metabolism disorder,and the detailed mechanism needs to be further investigated.
Glycolysis can rapidly supply energy to cells under hypoxic conditions. The results showed that SPP could reduce glycolysis and affect the glycolytic function of DLD-1 cells, but the degree of damage was lower than the decrease of basal oxygen consumption rate and aerobic respiratory maxima, suggesting that the main effect of SPP on cellular energy metabolism was due to the damage of mitochondrial function.
In summary, this article mainly studied the effect of SPP on energy metabolism of human colon cancer DLD-1 cells under different doses. The mechanism may be related to its inhibition of cell proliferation and promotion of cell apoptosis. Seahorse XFe24 cellular energy metabolism analyzer can sensitively detect changes in indicators such as basic aerobic respiration rate, ATP coupled aerobic respiration rate, etc. It provides the feasibility for SPP to induce tumor cell necrosis, but whether it can be applied to clinical treatment needs further exploration.
conflict of interest
The authors declare no potential conflicts of interest.
杂志排行
食品科学与人类健康(英文)的其它文章
- Hypoglycemic natural products with in vivo activities and their mechanisms: a review
- Bacteroides utilization for dietary polysaccharides and their beneficial effects on gut health
- Capsular polysaccarides of probiotics and their immunomodulatory roles
- Natural compounds may contribute in preventing SARS-CoV-2 infection: a narrative review
- A comprehensive review on the effects of green tea and its components on the immune function
- A review on current and future advancements for commercialized microalgae species
