Recombinant Pyriform Spider Silk Expression and Wet-Spinning
2022-03-08LIEBIEDIEVPavloLINYing
LIEBIEDIEV Pavlo, LIN Ying(林 瑛)
College of Chemistry, Chemical Engineering and Biotechnology, Donghua University, Shanghai 201620, China
Abstract: Spider silk is capturing the attention of scientists for its mechanical properties, biocompatibility, and biodegradability. Spiders can produce six types of silks, as well as special glue, which are used for survival and reproduction. During the last years of research, scientists deciphered gene sequences and expressed the most common types of spider silks. However, matching the mechanical properties of recombinant spider silks to native ones is still a big challenge. Moreover, in-depth studies are mostly focused on natural and recombinant ampullate silks, and only a few studies showed achievements on pyriform spidroin (PySp). In this study, repeatable parts of PySp were expressed, purified, and spun into fibers. Recombinase cloning strategy allowed to create highly-repetitive region parts clones efficiently in comparison to the traditional restriction enzyme cloning technique. A cost-effective high-yield purification strategy was used. This study provides strategies that can help to design recombinant spider silks with the same mechanical properties as native spider silks.
Key words: spidroin expression; protein purification; pyriform spidroin(PySp); recombinant spider silk; wet-spinning; mechanical property
Introduction
Orb web spinning spiders are producing a vast amount of valuable high-performance structural fibers for various applications[1-3]. The mechanical properties of those fibers are unmatched in the natural world and can surpass synthetic fibers created by modern technology[4].
Spider silks are made of spidroins secreted by special glands common to arthropods. Spidroin structure usually consists of multiple repeats of motifs that include an 8-10 residue poly-alanine block that formsβ-sheet crystals during the fiber spinning process, and a 24-35 residue long glycine-rich block. Later,β-sheet crystals crosslink proteins result in a polymer network of fibers with unmatched mechanical properties[2, 5]. Nonrepetitive serine-rich spacers are interrupting repetitive regions of spidroins in different places of the proteins. While the spacers sequences are different from repetitive regions, sequences of spacer regions themselves are almost identical to each other[6].
We associate different spider silks with distinct types of spidroins encoded by different members of a gene family. An individual spider can make up to seven task-specific silks, each with unique structural and functional differences[3]. The major ampullate silk, for example, is used by spiders to form an outer frame and spiral radii of the orb web. The toughness of it is comparable to the toughest human-made materials[2]. Flagelliform (Flag), which is also called capture silk, is used to capture rapidly moving prey while it contacts the web[7-8]. Aciniform silk’s purpose is to wrap prey and to form the inner liner of the egg case, while tubuliform silk is responsible for its outer shell[9-10]. Statistically, the difference among spider silks reaches up to 250% of performance[7].
To reach fascinating mechanical properties of silks, spiders are using a complex spinning process that involves a quick transformation of high molecular weight and water-soluble spidroins into solid fibers in natural conditions with ambient temperature and pressure, which leads to the creation of very safe, environmental-friendly fibers of high-performance properties[2]. The repetitive regions of spidroins have been linked to the exceptional mechanical properties of silk fibers and vary in sequence composition and repeat organization among spider silk protein types[2]. However, previously reported studies showed that mass production of strong spider silks is still a challenge due to low expression levels of large spidroins.
Compared to the rest of the spider silk types, pyriform spidroin (PySp) is unique because it combines a dry protein fiber and wet glue. The pyriform silk fiber is a component of the attachment disk, in opposite to other spider silks which are functioning as separate silk fibers[4]. PySp consists of N-terminal (NT) and C-terminal (CT) domains, and a core repetitive domain, similar to other spider silks. However, the repetitive domains of PySp have lengthy repeat units composed of segments that are Gln-, and Pro-rich[11]. This characteristic makes them different from other fibroins, allowing them to be spun into the aqueous matrix of the attachment disk[12]. Pyriform spider silk was fully discovered in the year of 2017 and since then only a few gene sequence identifications have been performed[13-15]. Considering the differences among PySp and other well-studied spider silks, further research can help to discover new properties and applications.
In this study, recombinant spidroins composed of 4 and 5 repeat units (R) of PySp ofAraneusventricosuswere spun into continuous fibers using a one-step wet-spinning process. Secondary structures and mechanical properties were studied and compared to previously published results[16]. This has allowed us to study the influence of NT and CT domains on final fibers and to reveal that not only repeatable regions but also terminal regions can have a direct impact on the mechanical properties of the fibers.
1 Experiments
1.1 Plasmid construction
Sequences encoding PySp repeat units were previously screened from theAraneusventricosusgenomic library[16]. Two primers were used for 4 repeat units(4R) and 5 repeat units(5R) construction:
R-FW(5′-TCAACTGGCAGCACCGGCTCCAGC ACCTGCACCTAGAC-3′) and R-RW(3′-GGCCGCG AATTGGCCCCCTCGAGTTACGGTGCTGCCAGTTGA GATAGAG-5′).
The cloning was performed by linearizing the cloning vector Blunt-zero in theBamHI restriction enzyme cutting site, and the rest of the procedure was performed according to the NovoRec plus One Step PCR Kit protocol. Overlapping DNA fragments are joined in a single isothermal reaction resulting in a fully ligated double-stranded DNA molecule, similar to the Gibson Assembly method[17]. After cloning, 4R and 5R parts were digested byBamHI andXhoI restriction enzymes and inserted in a pLX vector[18]for sequence length verification and expression.
1.2 Expression and purification of spider silk proteins
Escherichiacoli(E.coli) BL21 (DE3) (Tiangen Biotech, Beijing, China) containing 4R and 5R plasmids were incubated at 37 ℃ until an OD600reached 0.6-0.8, the cells were induced with isopropylβ-D-1-thiogalactopyranoside (IPTG) with a final concentration of 0.3 mmol/L, and the temperature decreased to 16 ℃ and incubated for 24 h.
Cells were harvested by centrifugation (25 min, 8 000 r/min, 4 ℃), the supernatant was discarded, and the cell pellet suspended in lysis buffer (20 mmol/L Tris, 100 mmol/L NaCl). Cell lysis was performed using a high-pressure homogenizer at 1 200 bar (JNBIO JN-3000, Guangzhou, China). Lysed cells were then centrifuged (4 000 g, 40 min, 4 ℃). The target proteins were present in the insoluble fraction. The pellets were resuspended in washing buffer (20 mmol/L Tris, 100 mmol/L NaCl, 3 mol/L Urea), sonicated for 30 min at 250 W in an ice bath, and centrifuged (4 000 g, 40 min, 4 ℃). Collected pellet resuspended in dissolving buffer (6 mol/L Guanidine-HCl (GuHCl)) and centrifuged to remove any insoluble material (1 000 r/min, 30 min, 25 ℃) and transferred to dialysis bags. Dialysis bags were sealed and put in dialysis buffer (ddH2O) for 78 h on a magnetic stirrer (dialysis buffer was changed every two hours). Then the content was centrifuged (8 000 r/min, 30 min, 4 ℃). The collected dialysate was freeze-dried using a FreeZone 6 Plus lyophilizer (48 h) (Labconco, USA).
1.3 Silk fibers production by wet-spinning
The spinning process is based on several patents[19-20]. Recombinant silk proteins are dissolved in 100% hexafluoroisopropanol (HFIP) and extruded through a thin needle/spinneret into an organic-based coagulation bath to form a solid fiber.
Here, the recombinant spider silk protein analogs are also first dissolved in 100% HFIP (silk-HFIP spinning dope), stirred at 20 r/min for 24 h (8%-10% volume fraction), and centrifuged at 25 ℃ for 30 min at 12 000 r/min to remove insoluble components. For the coagulation bath, 80% methanol/20% ddH2O was used.
Dope was transferred to a 1 mL syringe for extrusion using an LSP01-3A Syringe Pump (Longer Precision Pump Co., Ltd., Shanghai, China) through a 32 G syringe needle at a speed of 0.6 mL· h-1in a coagulation bath held at 20 ℃.
1.4 Fourier transform infrared (FTIR) spectroscopy
The secondary structure of recombinant spider silks was determined with an FTIR micro Nicoletln 10 MX spectrometer (Thermo Fisher Scientific, USA). The amide I band was decomposed into five Gaussian peaks centered at 1 626.5, 1 696.0, 1 646.5, 1 659.0, and 1 679.5 cm-1, forβ-sheet, random coil,α-helix, andβ-turn, and the five Gaussian peaks were used to calculate the fractions of secondary structures[16, 21-23].
1.5 Scanning electron microscopy (SEM)
The size and the morphology of silks were characterized with desktop SEM (FEI Quanta-250, USA). The silk fiber samples were gold coated for 30 s to give an estimated 10 nm Au layer on the samples. Then the samples were mounted on an aluminum stub with a conductive tape and observed with an acceleration voltage of 10 kV in Hi-Vac and a secondary electron (SE) mode at room temperature. The fracture morphology was characterized after breaking the fibers immersed in liquid nitrogen for 5 min with tweezers.
1.6 Mechanical testing of silk fibers
Each sample was prepared by placing fibers onto a paper frame and then the diameter of fibers was determined using a BX53 light microscope (Leica, Germany). A tensile test was performed for each fiber using a T150 universal test machine (UTM, KLA-Tencor Inc., OakRidge, USA) at a constant strain rate of 10-2s-1. Data were recorded and analyzed using the Nanosuite software.
2 Results and Discussion
2.1 Plasmid construction
AraneusventricosusPySp consists of 15 highly conserved 213-aa repeats and non-repetitive NT and CT domains[14]. Based on this sequence, the first experiments which showed the expression of smaller PySp fragments were reported[18]. Here we constructed plasmids composed of 4R (2 610 bp) and 5R (3 245 bp) which contain highly repetitive spider silk sequences fromAraneusventricosusPySp, respectively (shown in Fig. 1). Recombinase-based cloning strategy (shown in Fig. 2) was used in spider silk protein expression plasmid construction. In comparison with traditional molecular cloning techniques, it allows faster construction of clones consisting of multiple repeatable regions with a high success rate of transformation.

Fig. 1 Recombinant PySp 4R and PySp 5R construct elements: (a) schematic representation of recombinant spider silk proteins (PySp 4R, PySp 5R); (b) a detailed amino acid sequence of PySp repeatable region; (c) nucleic acid sequence of PySp repeatable region

Fig. 2 Schematic representation of single-tube cloning process
Compared to other well-described spider silk proteins, the PySp repeat unit has a unique amino acid composition with proline-rich (PX)nand glutamine-rich QQ(X)4QX motifs. It was indicated that the (PX)nmotifs form random coils and QQ(X)4QX motifs formα-helix orβ-sheet conformations[12-13, 24].
Multiple linear overlapping DNA fragments are joined in a single reaction. Adjacent segments created via polymerase chain reaction (PCR) contain identical sequences on the ends. PCR primers have a 5′ end that is identical to an adjacent segment and a 3′ end that anneals to the target sequence. Combined segments are mixed with NovoRec plus One Step Master Mix in assembly reaction and transformed into bacteria.
2.2 Protein expression and purification
4R and 5R recombinant PySp proteins were produced inE.colicells as inclusion body. Protein was purified using a cost-effective method that can be fully automatized for large-scale production. Purified protein was further subjected to sodium dodecyl sulphate-polyacrylamide gel electrophoresis (SDS-PAGE) which showed clear corresponding bands with different loading amounts (shown in Fig. 3). The total yield of purified protein was 83 mg/L for PySp 4R and 40 mg/L for PySp 5R. Image analysis showed that the purity was higher than 85%.

Fig. 3 SDS-PAGE analysis of purified recombinant PySp proteins: (a) purified PySp 4R; (b) purified PySp 5R
Purified protein sizes were analyzed using SDS-PAGE, followed by protein visualization via Coomassie Blue staining. For theE.coliexpression systems, yields of purified proteins have been generally in the 10 mg/L to 80 mg/L range. However, different spidroin types and lengths of final protein highly impact the yield of protein[25-27]. Data obtained in this study shows that compared to other spidroins, yield is high, but there are no recombinant PySp yield reports for comparison with results published here.
Yield and purity clearly show that this purification process is suitable for large-scale protein production. In comparison to other purification methods,e.g., affinity chromatography purification, the hereby presented method is more efficient and cost-effective.
2.3 Wet-spinning
Cells were harvested, lysed, purified, and screened for protein purity before the silk spinning process. For this experiment, a simple one step wet-spinning process was used due to its simplicity, which is important for large-scale production (shown in Fig. 4). Similar to the previous study[12], the ability for self-assembly and fiber formation of PySp spidroin was demonstrated due to the richness of Ser, Gln and Ala amino acids.

Fig. 4 One step wet-spinning of spider silk fiber process
Spinning dope (8%-10% volume fraction) is loaded to a 1 mL syringe for extrusion through a 32 G syringe needle at a constant speed of 0.6 mL· h-1. The coagulation bath was filled with 80% methanol/20% ddH2O and held at 20 ℃ throughout the spinning process.
All reported data in this paper is based on the usage of methanol as a coagulant. One step wet-spinning allowed to get continuous fibers with a consistent diameter, which is another benefit compared to the hand-drawn spinning technique (shown in Fig. 5).

Fig. 5 SEM images of PySp fibers from wet-spinning: (a) cross section of PySp 5R fiber(Φ12.0 μm, 5 000×); (b) cross section of PySp 5R fiber (Φ12.0 μm, 8 000×); (c) PySp 4R (Φ5.5 μm, 3 000×); (d) PySp 4R(Φ5.5 μm, 8 000×)
The extrusion rate of 0.6 mL·h-1allowed to spin fibers continuously. The length of fibers which could be spun was limited only by the amount of loaded spinning dope, as the spinning process was not interrupted by fiber’s breaks.
In future experiments, it is advisable to perform tests with the use of different coagulants. Other important conditions may include temperature, pH, and humidity, as they may also have a direct impact on final fiber mechanical properties[28].
2.4 Mechanical property testing
Sufficient amount of expressed protein that allowed wet-spinning was crucial for fibers’ mechanical property determination. After spinning, the mechanical properties of each fiber were tested. Larger PySp 5R spider silks showed a significant decrease in mechanical properties (shown in Fig. 6).

Fig. 6 Stress-strain curves of PySp fibers: (a) PySp 4R fibers; (b) PySp 5R fibers
Samples were placed onto a paper frame and tested at a constant strain rate of 10-2s-1. Data for each fiber were collected and analyzed using the Nanosuite software. Larger PySp 5R spidroin showed to be much weaker than PySp 4R in the same spinning conditions which might suggest protein instability due to lack of NT and CT domains.
A high-resolution nuclear magnetic resonance (NMR) recombinant MaSp study[29]demonstrated that CT domain forms a parallel-oriented dimeric 5-helix bundle with helix 4 constituting the main dimerization site. The presence of two highly conserved salt bridges (R43-D93 and R52-E101) that involve helices 1, 2, and 4 allows for critical intermolecular interactions responsible for maintaining correct folding, which serves silk protein storage and the inhibition of undesired aggregation[12, 29]. Based on obtained mechanical propertyresults, PySp might show similar behavior to previously studied major ampullate silk protein(MaSp) silks, as the absence of NT and CT domains has a direct influence on proper fiber formation and may be more obvious with large proteins.
Previously reported PySp 2R and 3R fibers showed that with an increase in size, the stress decreased by 15%[18]. Compared with another study, where only one repeatable unit of PySp was expressed, mechanical properties do not improve proportionally to the repeatable unit count (shown in Table 1). For each fiber, 10 samples were used to test the properties. With the presence of NT and CT domains, there has not been noticed a reduction of strength with protein size increase[16]. However, in another study[30]the increase of repeatable parts from 1 to 4 had a positive impact on mechanical properties, but the mechanical properties dropped significantly with a further increase to 8.
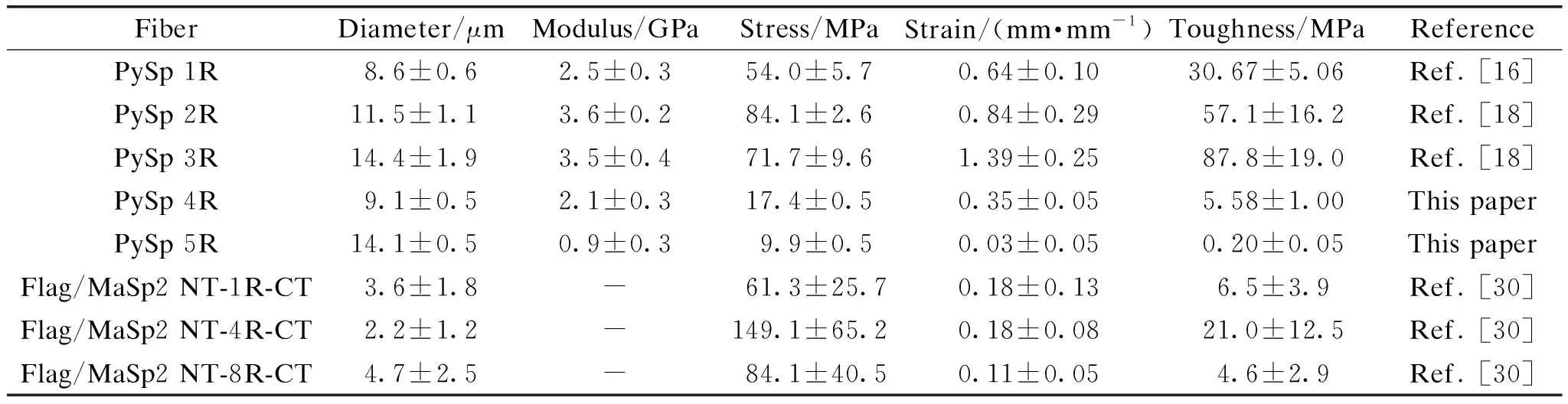
Table 1 Average mechanical property results
2.5 Secondary structural analysis
After evaluation of mechanical properties, the secondary structure contents of the spun fibers were tested (shown in Table 2). Due to the lack of NT and CT domains,β-sheet contents are higher (31%-33%) andα-helix content is lower, compared to those proteins which contain NT and CT domains[16]. The presence of random coil in those two particular proteins is higher than that in natural MaSp silk (about 12%) and synthetic aciniform fibers (about 12%)[31]. However,α-helical content is much lower, comparable to natural PySp silk[32], which can suggest protein instability during the spinning process. By increasing the number of repetitive domains, secondary structures only had very minor differences (shown in Fig. 7).

Table 2 Secondary structure contents of silk fibers

Fig. 7 FTIR spectra of PySp fibers: (a) PySp 4R fibers; (b) PySp 5R fibers
Highβ-sheet and lowα-helix content confirm the lack of NT and CT domains. The similarity between PySp 4R and PySp 5R FTIR spectra suggests that the number of repetitive domains does not impact the secondary structure content.
3 Conclusions
The use of recombinase cloning approach allows creating clones with highly repetitive sequences faster and more efficiently, compared to the traditional restriction enzyme cloning technique. The protein purification process tends to be more cost-effective and shows to have a high yield of protein, which means that the production can be scaled more easily. It allowed the time-efficient construction of multiple clones with different repeatable regions. The larger protein PySp 5R, which was spun in the same conditions and time as PySp 4R, had much weaker mechanical properties. Lack of NT and CT domains may suggest a significant impact on protein stability during the wet-spinning process.
杂志排行
Journal of Donghua University(English Edition)的其它文章
- Sentiment Lexicon Construction Based on Improved Left-Right Entropy Algorithm
- Expert Knowledge-Based Apparel Recommendation Question and Answer System
- Molecular Modulation of Structure and Ferroelectric Performance of Poly(vinylidene fluoride) Free Standing Films from Aspects of Molecular Weight and Crystallization Temperature
- Global Existence and Decay of Solution to Parabolic-Parabolic Keller-Segel Model in Rd
- Theoretical Calculation and Analysis of Muffler Based on Multilayer Sound Absorbing Material
- Preparation of Polyaniline/Cellulose Nanofiber Aerogel for Efficient Removal of Cr(VI) from Aqueous Solution
