Molecular Characterization and Expression Analysis of SKIV Infection of Interferon-Induced Protein with Tetratricopeptide Repeats 1 (IFIT1) in Epinephelus lanceolatus
2021-03-06WANGLeiMATengXUWentengCHENZhangfanZHOUQianZHENGGuiliangandCHENSonglin
WANG Lei, MA Teng, 2), XU Wenteng, CHEN Zhangfan, ZHOU Qian,ZHENG Guiliang, 3), and CHEN Songlin, *
Molecular Characterization and Expression Analysis of SKIV Infection of Interferon-Induced Protein with Tetratricopeptide Repeats 1 () in
WANG Lei1), MA Teng1), 2), XU Wenteng1), CHEN Zhangfan1), ZHOU Qian1),ZHENG Guiliang1), 3), and CHEN Songlin1), *
1),,,,266071,2),,201306,3),,266237,
Interferon-induced protein with tetratricopeptide repeats 1 (), also known as interferon-induced protein 56 () or Interferon-stimulated protein 56 (), was originally identified as a protein induced upon treatment with interferon and inhibited by viral replication and translational initiation. In this study,() gene was cloned for the first time. The complete cDNA ofgene includes 2921 nucleotides,and encodes a 437-amino acid (AA) protein. The putative ELIFIT1 protein has 9 TRP domains and is highly similar with IFIT1 proteins in other teleosts. In healthy fish,gene was highly expressed in the blood, which indicate its specific function in the peripheral immune system. Its expression was also observed in various immunity-related tissues including spleen, intestine, and kidney, Inducted with spotted knifejaw iridovirus (SKIV),gene ex- pression was upregulated in the spleen, kidney, and liver 24h after induction and reached its peak at 72h, indicating thatmay play an important role in antivirus. These findings contribute to the understanding of the antiviral regulation ofgene in teleost.
interferon-induced protein with tetratricopeptide repeats 1 ();; tetratricopeptide repeats (TPR) motif; expression pattern; antiviral function
1 Introduction
Interferons (IFNs) are enormous family of cytokines that play vital roles in defense, immune activation, and regulation of cell growth in vertebrates (Samuel, 2001). The IFN type I and II systems are pivotal in both innate and adaptive immunity against viral infection in teleost (Zou and Secombes, 2011). Viral dsRNA triggers IFNs to bind their receptors in virus-infected cells and subsequently ini- tiate the JAK/STAT signaling pathway, which induces the transcription of interferon-stimulated genes (ISGs) (Robert- sen, 2006). ISGs form the molecular mainstay of the innate immune system and limit the spread of virus as well as intra- and inter-cellular viral replication. Therefore, ISGs network is a critical therapeutic target against viral infections (Hubel., 2019).
Interferon-induced protein withetratricopeptide repeats 1 (), also called IFN-induced protein 56kDa () or Interferon-stimulated protein 56 (), was initially cloned from human fibroblastoid cells treated with IFN- beta (Chebath., 1983; Wathelet., 1986). IFIT1/ ISG56 and other three IFIT family members including IFIT2/ISG54, IFIT3/ISG60, and IFIT5/ISG58 have been characterized in humans and found to be localized in a cluster on chromosome 10q23 (Lafage., 1992; Fensterl and Sen, 2011). Different IFIT family members contain distinct numbers and arrangements of tetratricopeptide repeats (TPR) motif which mediate protein-protein interactions, thus substantiate their multifunctional role in cell proliferation and migration, translation initiation, dou- ble-stranded RNA signal recognition, and inhibition of vi- rus replication (D’Andrea and Regan, 2003; Fensterl and Sen, 2011; Diamond, 2014). Recent reports have demonstrated that IFIT1 can detect viral RNA as a sensor molecular and subsequently inhibit viral translation or seques- ter them for active replication function as effector molecules (Daffis., 2010; Kimura., 2013; Diamond, 2014). Meanwhile, human immunodeficiency virus type 1 (HIV-1) viral protein R (Vpr) upregulates IFIT1 gene ex- pression in human monocyte-derived macrophages anddendritic cells, indicating its function in fighting HIV (Za- hoor., 2014; Zahoor., 2015). IFIT proteins have broad-spectrum antiviral functions.They can fight against certain viruses by binding to the subunits of eukaryotic ini- tiation factor (eIF3) translation initiation complex and in- hibiting protein translation (Hui., 2003). Human IFIT1 and IFIT2 bind to eIF3E, while human IFIT2, mouse IFIT1, and mouse IFIT2 bind to eIF3C (Hui., 2005).
gene is conservatively found in mammalian species, birds, and amphibians. According to gene scan and se- quence analysis, only a fewhomologs have been re- ported in fishesincluding large yellow croaker (), zebrafish (), crucian carp (L.), and olive flounder (). From the previous results,gene expression is upregulated by fish IFNs, Poly (I:C) or viral hemorrhagic septicemia virus (VHSV) in zebrafish or olive flounder, in- dicating the important role of IFIT1 in response against viral infection (Zhang and Gui, 2004; Wan and Chen, 2008; Liu., 2013; Varela., 2014; Hwang., 2017). Giant grouper() is a commercially important marine fish, and is the largest grouper type in the world (Williams, 2009). Because of its fast growth performance,is preferably selected as the male parent in order to produce the hybrid grouper with,, andand so on(Kiriyakit., 2011; Chen and Long, 2018;Chen., 2018). The chromosome-level genome assem- bly of giant grouper has been well-documented in 2019 and it provides the characteristics of innate immune response and rapid growth (Zhou., 2019). Many grouper farms have suffered from financial losses induced by viral infection. In the present study, we cloned the full length ofgene in giant grouper and characterized its expression pattern in different tissues. Furthermore, we stu- died the expression level ofafter stimulation with SKIV. Our results will contribute to the knowledge of
2 Materials and Methods
2.1 Ethical Statement
Examinations and investigations were performed according to the guidelines and ethical standards of Regulations for the Administration of Affairs Concerning Experi- mental Animals of the State Science and Technology Com- mission of Shandong Province. This study was approved by the Ethics Committee of Yellow Sea Fisheries Research Institute, Chinese Academy of Fishery Sciences.
2.2 Fish, Virus Infection, and Sample Collection
Thirty-five healthy giant grouper (body weight: 700g±25g; body length: 30cm±3cm) were purchased from Chen- hai Aquaculture Company (Hainan, China) and cultured incirculating seawater at 25℃. Ten tissues (liver, spleen, kid- ney, intestine, gill, skin, muscle, brain, heart, and blood) were collected from five normal fish and transferred im- mediately into liquid nitrogen and stored at −80℃. Twen- ty-five fish were intraperitoneally injected with 106TCID50/fish spotted knifejaw iridovirus (SKIV) and five control fish were injected with sterilized phosphate-buffered saline (PBS, pH7.4) at a dose of 0.5mL per 200g fish. Spleen and kidney were collected from each group (5 fish each) at 0h, 12h, 24h, 72h, and 96h after the infection.
2.3 Cloning of ELIFIT1 cDNA Sequence and Multiple Alignment
Based on the partial sequence ofgene from the genome database (Zhou., 2019), we cloned the full length open reading frame (ORF) ofusing the primers (ELIFIT1-F1 and ELIFIT1-R1) listed in Table 1 through polymerase chain reaction (PCR) assay. Afterward, we obtained the 5’ and 3’ untranslated regions (UTR) ofby rapid amplification of cDNA ends (RACE) PCR using SMART RACE 5’/3’ Kit (Clontech) with the primers listed in Table 1. The cDNA sequence ofwas linked by DNAstar software and conserved domains of the predicted protein were analyzed using SMART pro- gram (http://smart.embl-heidelberg.de/). ProtScale (http:// web.expasy.org/protscale/) was used for hydropathicity/ hydrophobicity analysis of the protein and TMHMM v2.0 (http://www.cbs.dtu.dk/services/TMHMM/) was used to predict the transmembrane domain.

Table 1 Primers used in this study
2.4 Sequence, Homology, and Phylogeny Analysis
The predicted protein was analyzed by BLAST program (http://www.ncbi.nlm.nih.gov/blast). Putative TPR motifs was identified using TPRpred (http://toolkit.tuebingen.mpg.de/tools/tprpred/) (Karpenahalli and Soding, 2007) by com- paring it withIFIT1 (HsIFIT1),IFIT1 (MmIFIT1) andIFIT1 (EcIFIT1), and the E-value inclusion TRP&SEL is 1e-2. Putative amino acidsequence alignment was performed us-ing ClustalX and ESPript 3.0 (http://espript.ibcp.fr/ES- Pript/cgi-bin/ESPript.cgi) (Robert and Gouet, 2014). A phy- logenetic tree was constructed with the sequences listed in Table 2 using neighbor-joining algorithm. Reliability of the branching was tested through bootstrap resampling with 1000 replications.
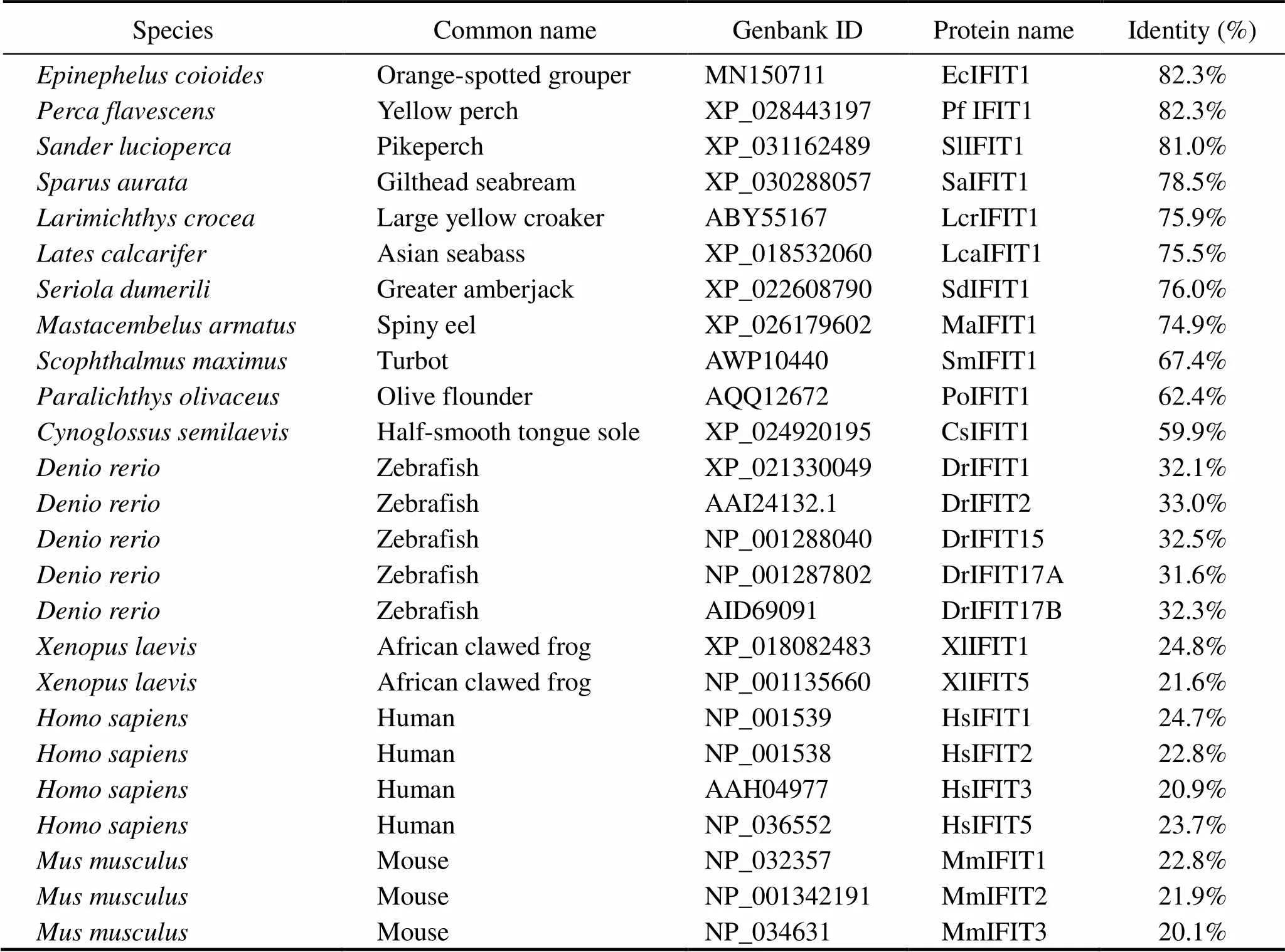
Table 2 Information of IFIT protein family in various species
2.5 Expression of ELIFIT1 Gene in Various Tissues of E. lanceolatus
Total RNA was extracted using Trizol Reagent (Takara, Dalian, China) from various tissues, respectively. The first strand cDNA was synthesized using PrimeScript RT Reverse kit (Takara, Dalian, China). Real-time quantitative PCR (RT-qPCR) using ELIFIT1-RT-F and ELIFIT1-RT-R was carried out according to the instructions for SYBR® Green I Premix Ex Taq™ (Takara, Japan) in a 7500 fast real-time PCR system (Applied Biosystems, USA). The as- say condition entails initial denaturation at 95℃ for 5min, followed by 40 cycles of 95℃ for 10s and 60℃ for 34s. Genewas used as an internal reference and therelative abundance of transcripts was analyzed by 2ΔΔCtmethod. The primers are listed in Table 1.
2.6 Expression of ELIFIT1 Gene in Immune Tissues of E. lanceolatus Under Infection with SKIV
Three immunity-related tissues including spleen, kidney, and liver were collected from the experimental groups and PBS group at 0, 12, 24, 72, and 96h after infection and five fish were dissected at each time point. RNA extraction and RT-qPCR were performed in triplicate as de- scribed previously.
2.7 Statistical Analysis
Data was expressed as mean±standard error (SE) of five repetitions for each experimental group. Data were analyzed by one-way ANOVA andtest using SPSS version 19.0 software package (SPSS Inc., Chicago, IL, USA).0.05 was considered as significant and0.01 was con- sidered as extremely significant.
3 Results
3.1 ELIFIT1 cDNA and AA Sequence Analysis
The full length sequence ofcDNA was obtained from the spleen ofby normal PCR and RACE. Completegene was 2921bp, with an ORF of 1314bp, which encodes a 437-AA peptide. The 5’ UTR is 99bp and the 3’ UTR is 1508bp, with a poly (A) tail (Fig.1). Molecular weight of the ELIFIT1 protein is 50.64kDa, and the isoelectric point is 6.353. The ELIFIT1 protein is a tetratricopeptide repeat (TPR) protein, which contains multiple tetratricopeptide repeat domains (domain architecture ID 12138572), according to the conserved domain analysis. As shown in Fig.1, 9 TPR motifs are located in position 52−85, 95–128, 140–173, 179–213, 214–247, 250–283, 293–326, 330–363, and 369–402. In architecture analysis, a low complexity region (266IDEAI- DLAEEALE278) was found in the ELIFIT1 protein (Fig.1). There are 147 hydrophobic AAs and 290 hydrophilic AAs in the ELIFIT1 protein and most of the AAs have values below zero in the hydrophilic/hydrophobicity analysis (Fig.2). ELIFIT1 protein is a secreted protein and has no transmembrane domain with TMHMM analysis.
3.2 Alignment and Phylogenetic Analysis
A Blast-protein search revealed that ELIFIT1 protein is significantly homologous with IFIT1 members in bony fish, which contain multiple TPR motifs. The TPR motif numbers of HsIFIT1 and MmIFIT1 are 10 and 11 in TPR- pred, and the TPR motif numbers of EcIFIT1 and ELIFIT1 are 6 and 4 (Fig.3). The first TPR motif in mouse was identified in 13aa–51aa. Some TPR motifs were identified to SEL-1 motif in EcIFIT1 and ELIFIT1, while the pro- bability is only 0.09% and 0.37%, separately. Based the re-sults of alignment, the number of TPR motifs were confirm-ed to be 9 in ELIFIT1. The entire amino acid sequences of fish IFIT1s and mammal IFIT1s were aligned using the ClustalX program and the TPRs were annotation in Fig.4.

Fig.1 Nucleotides and deduced amino acid sequences of ELIFIT1 cDNA.The TAG stop codon is indicated with an asterisk. Nine TPR motifs located in position 52–85, 95–128, 140–173, 179–213, 214–247, 250–283, 293–326, 330–363, and 369–402 are single-underlined and low complexity regions are double-underlined.

Fig.2 Hydrophilic/hydrophobic analysis of ELIFIT1 protein.Hydrophobic segments have values above zero in the Y-axis, while hydrophilic segments have values below zero in the Y-axis.

Fig.3 TPR predicted results of HsIFIT1, MmIFIT1, EcIFIT1 and ELIFIT1 in TPRpred.

Fig.4 Multiple sequence alignment of IFIT1 proteins from 11 fishes and two mammals. The amino acid numbers are shown on the top of the figure. Conservative amino acids have a red background and similar amino acids have a yellow background. TRP motifs are marked in blank wireframe and numbers of TRPs are covered with blue shadow.
The deduced ELIFIT1 protein shares >80% identity withthe sequences found in orange-spotted grouper () and two kinds of perch (,). The identity between ELIFIT1 and three flatfish IFIT1 (,,) are 67.4%, 62.4%, and 59.9%, respectively. The identity between ELIFIT1 and two mammals (,) are 24.7% and 22.8%, respectively. The phylogenic analysis showed that IFIT1 members from 12 marine fishes form an independent clad and five IFIT1 members ofform another independent clad. The IFIT1 family of(African clawed frog) and mammals form a unitary clad, which reflect that the distance of the evolutionary relationship was relatively far away between fish and mammalian IFITs (Fig.5).
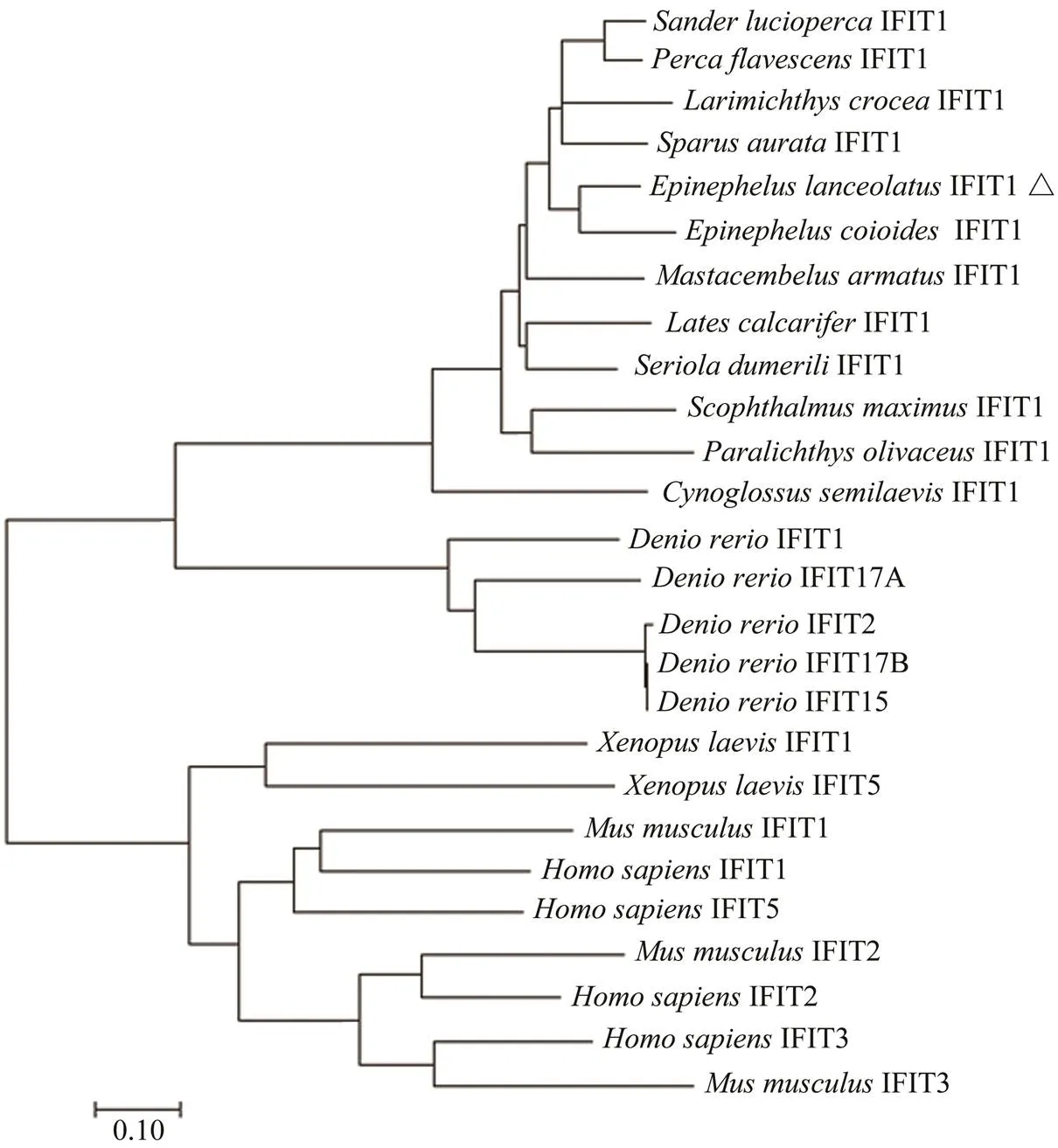
Fig.5 Phylogenetic relationship of IFIT protein family. The tree was constructed using Mega 7.0 by neighbor-joining me- thod. The bootstrap confidence values are based on 1000 replications.
3.3 ELIFIT1 mRNA Expression Pattern in Healthy Fish
RT-qPCR analysis was performed to investigate the ex- pression pattern ofmRNA in healthy fish (Fig.6). ThemRNA was mainly expressed in blood, and the higher mRNA levels were detected in spleen, kidney, and li- ver, followed by gill, heart, brain, skin, intestine, and muscle.

Fig.6 The expression levels of ELIFIT1 mRNA in different tissues from healthy fish. Geneβ-actin was used as an internal control of the RT-qPCR. The values are presented as means±SE (n=5) and letters represent significant difference in gene expression among tissues according to Duncan test in the statistical analysis.
3.4 Expression of ELIFIT1 in the Spleen and Kidney After SKIV Infection
To investigate whetherparticipates in immune response, especially during virus infection, RT-qPCR was used to analyze the expression pattern ofmRNAduring SKIV infection. As shown in Fig.7A, compared to 0h group and PBS group, the transcript levels ofmRNA were significantly upregulated by 5-fold at 72h and downregulated by 4-fold at 96h after infection in the spleen. The change in expression ofmRNA was even more drastic in the kidney and liver than spleen, and transcript levels ofmRNA was upregulated approximately by 30-fold at 72h and downregulated by 5- fold at 96h after infection (Figs.7B, C).
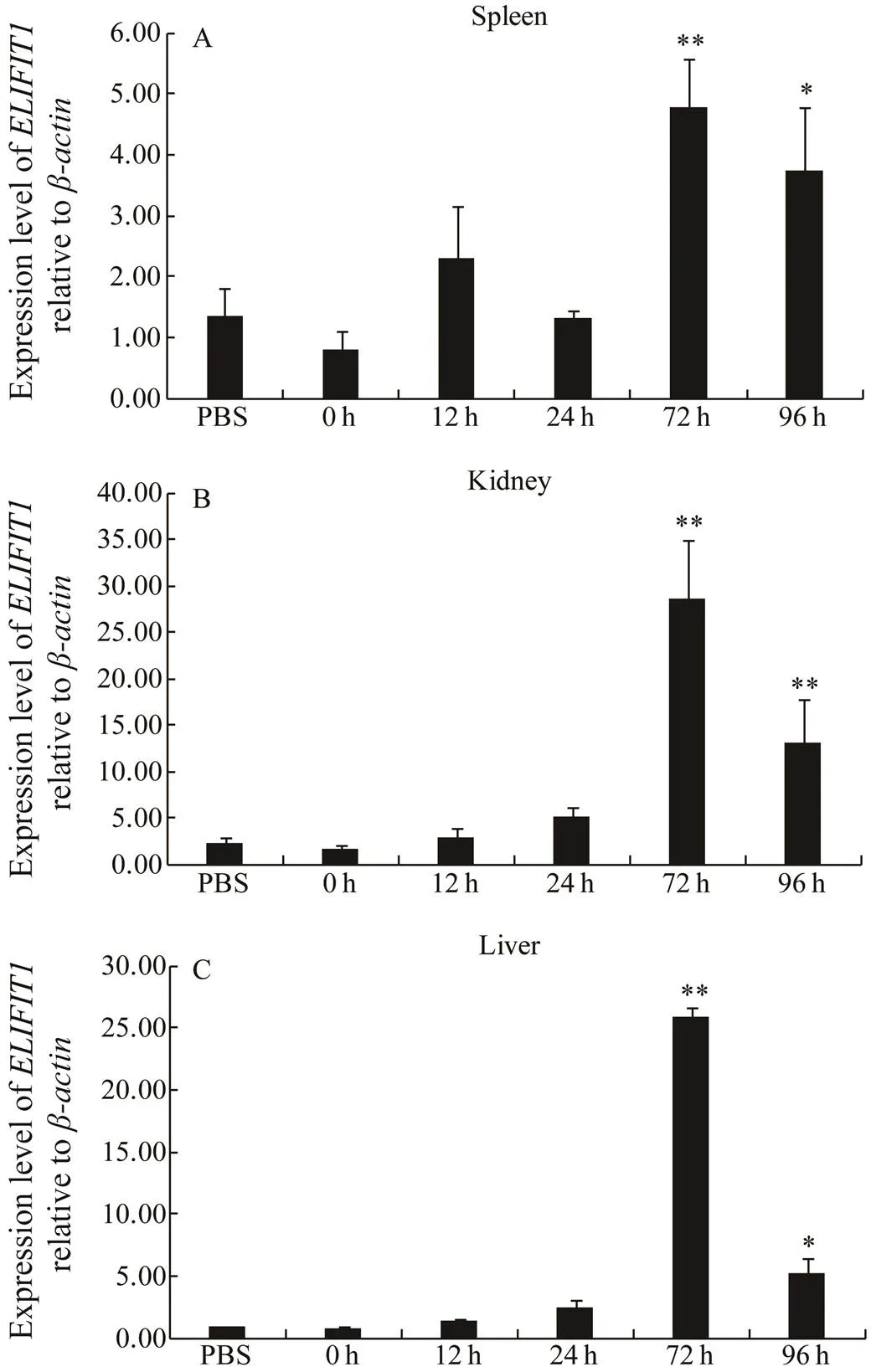
Fig.7 ELIFIT1 expression during SKIV infection. Expression of ELIFIT1 mRNA in the spleen (A), kidney (B), and liver (C) of control group (PBS) and SKIV-infected groups. The ELIFIT1 mRNA levels were determined by qRT-PCR at various time points. Values are presented as means±SE (n=5). *P<0.05, **P<0.01 as compared with 0h time point.
4 Discussion
In this study, the biological property ofwasanalyzed. Sequence analysis showed thatencodes a 437-amino-acid polypeptide, which share highest identity (82.3%) withIFIT1protein. As an impor- tant component of the IFIT1 family, IFIT1 proteins are con- served in various species, indicating their conservative func- tion. TPR motif was a classical protein structure which com- posed of 34 amino acids, and scaffolds formed among tandem TPR motifs of IFIT1 mediate protein-protein interactions (Blatch and Lassle, 1999). The number of TPR motifs was 10 in HuIFIT1 (NP_001539) and MuIFIT1 (NP_032357), while it is inconclusive in fish. In our study, the protein sequence of ELIFIT1 shows the highest similarity with EcIFIT1. Zhang. (2019) found only three TPR motifs are formed by residues 53–86, 96–129, and 139–174 in EcIFIT1. We retrieved the TPR motifs of EcIFIT1 using TPRpred and found 6 TPR motifs including TPR1 (53–86), TPR2 (96–129), TPR3 (141–174), TPR6 (251–284), TPR7 (294–327), and TPR9 (368–401). Inside the sequence, residues 196 to 231 was predicted to be SEL1 motif with a-value of 7.7e−04, and the probabi- lity for been SEL1 is 0.09%, so that we concluded that it was a wrong annotate (Fig.3). Hwang. (2017) predicted that there are 10 TPR motifs in PoIFIT1 through TPRpred and sequences alignment; however, we found that ‘TPR10s’ in fish is less than 34 amino acid, and cannot form a complete TPR motif. The similar IFIT1 without TPR10 was observed in large yellow croaker (), Crucian carp (), and pufferfish () (Zhang and Gui, 2004; Wan and Chen, 2008). Using both TPRpred and multiple se- quence alignment, we predicted that the number of TPR motifs is 9 in the ELIFIT1, as annotated in Fig.2. Phylogenic analysis showed that marine fish IFIT1 proteins form a distinct clad with the mammalian IFIT1 in Fig.5, suggesting that the mammalian IFIT1 might undergo amplification to 10 TPR motifs in some emergencies with evolution.
Previous studies have shown that the expression level of IFIT1 are higher in immunity-related tissues such as spleen, kidney, and liver in healthy fish (Wan and Chen, 2008; Long and Sun, 2014; Zhang., 2019). In our study, it is worth mentioning that the expression level ofmRNA in the blood was much higher than in visceral tis- sue, which suggests thatmay function as a safe guarder in the blood for protection against viral infection.
Mammals IFIT family members regulate immune re- sponse and restrict viral infections through blocking viral RNA translation.Moreover, IFIT1 regulates both viral and cellular functions. IFIT1 expression was obviously upre- gulated in other teleosts infected by DNA viruses such as SKIV and megalocytivirus (Long and Sun, 2014; Zhang., 2019), or RNA virus such as VHSV, red-spotted grouper nervous necrosis virus, and grass carp hemorr- hage virus (GCHV) (Zhang and Gui, 2004; Hwang., 2017; Zhang., 2019). The results indicated thatwas significantly increased in the spleen, kidney, and liver after SKIV infection, which is consistant with pre- vious studies. Previous studies have confirmed that IFIT1 is an antiviral protein that binds to free 5’-triphosphate vi- rus RNA or 5’ Capped 2’-O unmethylated RNA (Pichl- mair., 2011; Kimura., 2013). It is necessary to investigate the anti-SKIV function of ELIFIT1 in future re- search.
The presence of interferon motifs functioning as stimu- latory response element in the promoters of fishandhave been confirmed (Liu, 2002; He., 2017). In our future plan, we will study the transcriptional regulation mechanism of potential response elements in promoter of ELIFIT1. Xie. (2016) confirmed that poly- morph rs303218 in humangene can predict theIFNα treatment efficiency for Chinese hepatitis B virus infection. Our findings provide a new insight in the te- leost fish, while molecular polymorphism still needs fur- ther validation.
5 Conclusions
In this study, thegene fromwas identified for the first time.The predicted ELIFIT1 protein has 9 TRP domains. The multiple sequence alignment and phylogenetic analysis revealed that ElIFIT1 has high similarity with other IFIT1 proteins in teleosts. The expres- sion analysis indicated the important immunity function ofindicated its potential antiviral function, which will contribute to the functional research on IFIT1 proteins in the future.
Acknowledgements
This work was supported by the Shandong Breeding Project (No. 2016LZGC009), the Projects from Laboratory for Marine Fisheries Science and Food Production Processes, Pilot National Laboratory for Marine Science and Technology (Qingdao) (Nos. 2018-MFS-T08, 2017A STCP-OS15), the Central Public-interest Scientific Institution Basal Research Fund, CAFS (No. 2020TD20), and the Central Public-Interest Scientific Institution Basal Re- search Fund, YSFRI, CAFS (No. 20603022018026).
Blatch, G. L., and Lassle, M., 1999. The tetratricopeptide repeat: A structural motif mediating protein-protein interactions.,21 (11): 932-939, https://doi.org/10.1002/(SICI)1521- 1878(199911)21:11<932::AID-BIES5>3.0.CO;2-N.
Chebath, J., Merlin, G, Metz, R., Benech, P., and Revel, M., 1983. Interferon-induced 56,000 Mr protein and its mRNA in human cells: Molecular cloning and partial sequence of the cDNA.,11 (5): 1213-1226, https://doi.org/10. 1093/nar/11.5.1213.
Chen, X. Y., Shao, T. Y., and Long, X. H., 2018. Evaluation of the effects of different stocking densities on the sediment microbial community of juvenile hybrid grouper (female symbol×male symbol) in recirculating aquaculture systems.,13 (12): e208544, https://doi.org/10.1371/journal.pone.0208544.
Chen, Z. F., Tian, Y. S., Wang, P. F., Tang, J., Liu, J. C, Ma, W. H., Li, W. S., Wang, X. M., and Zhai, J. M., 2018. Embryonic and larval development of a hybrid between kelp grouper♀×giant grouper♂ using cry- opreserved sperm.,49 (4): 1407-1413, https://doi.org/10.1111/are.13591.
Daffis, S., Szretter, K. J., Schriewer, J., Li, J., Youn, S., Errett, J., Lin, T. Y., Schneller, S., Zust, R., Dong, H., Thiel, V., Sen, G. C., Fensterl, V., Klimstra, W. B., Pierson, T. C., Buller, R. M., Gale Jr., M., Shi, P. Y., and Diamond, M. S., 2010. 2’-O methylation of the viral mRNA cap evades host restriction by IFIT family members.,468 (7322): 452-456, https:// doi.org/10.1038/nature09489.
D’Andrea, L. D., and Regan, L., 2003. TPR proteins: The versatile helix.,28 (12): 655-662, https://doi.org/10.1016/j.tibs.2003.10.007.
Diamond, M. S., 2014. IFIT1: A dual sensor and effector molecule that detects non-2’-O methylated viral RNA and inhibits its translation.,25 (5): 543- 550, https://doi.org/10.1016/j.cytogfr.2014.05.002.
Fensterl, V., and Sen, G. C., 2011. The ISG56/IFIT1 gene family.,31 (1): 71-78, https://doi.org/10.1089/jir.2010.0101.
He, X. M., Du, X., Zhuo, J. S., Jing, X. Y., Yang, X. Q., and Liu, D. K., 2017. Promoter identification and effect on activation of NF-κB of porcine ISG58.,67: 1-6, https://doi.org/10.1080/ 09064702.2017.1341952.
Hubel, P., Urban, C., Bergant, V., Schneider, W. M., Knauer, B., Stukalov, A., Scaturro, P., Mann, A., Brunotte, L., Hoffmann, H. H., Schoggins, J. W., Schwemmle, M., Mann, M., Rice, C. M., and Pichlmair, A., 2019. A protein-interaction network of interferon-stimulated genes extends the innate immune system landscape.,20 (4): 493-502, https:// doi.org/10.1038/s41590-019-0323-3.
Hui, D. J., Bhasker, C. R., Merrick, W. C., and Sen, G. C., 2003. Viral stress-inducible protein p56 inhibits translation by block- ing the interaction of eIF3 with the ternary complex eIF2. GTP.Met-tRNAi., 278: 39477- 39482, https://doi.org/10.1074/jbc.M305038200.
Hui, D. J., Terenzi, F., Merrick, W. C., and Sen, G. C., 2005, Mouse p56 blocks a distinct function of eukaryotic initiation factor 3 in translation initiation., 280: 3433-3440.
Hwang, J. Y., Ahn, S. J., Kwon, M. G., Seo, J. S., Hwang, S. D., and Son, M. H., 2017. Interferon-induced protein 56 () is induced by VHSV infection but not by bacterial infection in olive flounder ().,66: 382-389, https://doi.org/10.1016/j.fsi.2017. 05.027.
Karpenahalli, M. R., Lupas, A. N., and Soding, J., 2007. TPRpred: A tool for prediction of TPR-, PPR- and SEL1-like repeats from protein sequences.,8: 2, https://doi.org/ 10.1186/1471-2105-8-2.
Kimura, T., Katoh, H., Kayama, H., Saiga, H., Okuyama, M., Oka-moto, T., Umemoto, E., Matsuura, Y., Yamamoto, M., and Ta- keda, K., 2013. Ifit1 inhibits Japanese encephalitis virus replication through binding to 5’ capped 2’-O unmethylated RNA., 87 (18): 9997-10003, https://doi.org/10. 1128/JVI.00883-13.
Kiriyakit, A., Gallardo, W. G., and Bart, A. N.,, 2011. Successful hybridization of groupers (×) using cryopreserved sperm.,320 (1): 106-112, https://doi.org/10.1016/j.aquaculture.2011.05.012.
Lafage, M., Clauss, I., Couez, D., Simonetti, J., Wathelet, M. G., and Huez, G., 1992. The interferon- and virus-inducible IFI- 56K and IFI-54K genes are located on human chromosome 10 at bands q23-q24.,13 (2): 458-460, https://doi. org/10.1016/0888-7543(92)90272-T.
Liu, M., Reimschuessel, R., and Hassel, B. A., 2002. Molecular cloning of the fish interferon stimulated gene, 15 kDa (ISG15) orthologue: A ubiquitin-like gene induced by nephrotoxic da- mage.,298 (2): 129-139, https://doi.org/10.1016/S0378- 1119(02)00932-0.
Liu, Y., Zhang, Y. B., Liu, T. K., and Gui, J. F., 2013. Lineage- specific expansion of IFIT gene family: An insight into coevolution with IFN gene family.,8 (6): e66859, https://doi.org/10.1371/journal.pone.0066859.
Long, H., and Sun, L., 2014. CsIFIT1, an interferon-induced pro- tein with tetratricopeptide repeat, inhibits viral infection in ton- gue sole ().,41 (2): 231-237, https://doi.org/10.1016/j.fsi.2014.09. 006.
Pichlmair, A., Lassnig, C., Eberle, C. A., Gorna, M. W., Bau-mann, C. L., Burkard, T. R., Bürckstümmer, T., Stefanovic, A., Krieger, S., Bennett, K. L., Rülicke, T., Weber, F., Colinge, J., Müller, M., and Superti-Furga, G., 2011. IFIT1 is an antiviral protein that recognizes 5’-triphosphate RNA., 12: 624-630.
Robert, X., and Gouet, P., 2014. Deciphering key features in protein structures with the new ENDscript server.,42 (Web Server issue): W320-W324, https://doi.org/ 10.1093/nar/gku316.
Robertsen, B., 2006. The interferon system of teleost fish.,20 (2): 172-191, https://doi.org/10. 1016/j.fsi.2005.01.010.
Samuel, C. E., 2001. Antiviral actions of interferons.,14 (4): 778-809, https://doi.org/10.1128/ CMR.14.4.778-809.2001.
Varela, M., Diaz-Rosales, P., Pereiro, P., Forn-Cuni, G., Costa, M. M., Dios, S., Romero, A., Figueras, A., and Novoa, B., 2014. Interferon-induced genes of the expanded IFIT family show conserved antiviral activities in non-mammalian species.,9 (6): e100015, https://doi.org/10.1371/journal.pone. 0100015.
Wan, X., and Chen, X. H., 2008. Molecular characterization and expression analysis of interferon-inducible protein 56 gene in large yellow croaker.,364 (2): 91-98, https:// doi.org/10.1016/j.jembe.2008.07.027.
Wathelet, M., Moutschen, S., Defilippi, P., Cravador, A., Collet, M., Huez, G., and Content, J., 1986. Molecular cloning, full- length sequence and preliminary characterization of a 56-kDa protein induced by human interferons.,155 (1): 11-17, https://doi.org/10.1111/j.1432- 1033.1986.tb09452.x.
Williams, K. C., 2009. A review of feeding practices and nutritional requirements of postlarval groupers.,292 (3): 141-152, https://doi.org/10.1016/j.aquaculture.2009.04.026.
Xie, D. Y., Wang, S. M., Yang, J. M., Wang, L. H., Chen, H. Y., Huai, C., Shang, J., Mao, Q., Lei, C. L., Luo, G. H., Qian, J., and Lu, D. R., 2016. IFIT1 polymorphisms predict interfe- ron-alpha treatment efficiency for hepatitis B virus infection.,22 (44): 9813-9821, https:// doi.org/10.3748/wjg.v22.i44.9813.
Zahoor, M. A., Xue, G., Sato, H., Murakami, T., Takeshima, S. N., and Aida, Y., 2014. HIV-1 Vpr induces interferon-stimulated genes in human monocyte-derived macrophages.,9 (8): e106418, https://doi.org/10.1371/journal.pone.0106418.
Zahoor, M. A., Xue, G., Sato, H., Murakami, T., Takeshima, S. N., and Aida, Y., 2015. Genome-wide transcriptional profiling re- veals that HIV-1 Vpr differentially regulates interferon-stimu- lated genes in human monocyte-derived dendritic cells., 208: 156-163, https://doi.org/10.1016/j.virusres.2015. 06.017.
Zhang, Y. B., and Gui, J. F., 2004. Identification and expression analysis of two IFN-inducible genes in crucian carp (L.).,325: 43-51, https://doi.org/10.1016/j. gene.2003.09.039.
Zhang, Y., Wang, Y. X., Liu, Z. T., Zheng, J. Y., Huang, Y. H., Huang, X. H., and Qin, Q. W., 2019. Grouper IFIT1 inhibits iridovirus and nodavirus infection by positively regulating in- terferon response.&,94: 81-89, https://doi.org/10.1016/j.fsi.2019.08.075.
Zhou, Q., Gao, H. Y., Zhang, Y., Fan, G. Y., Xu, H., Zhai, J. M., Xu, W. T., Chen, Z. F., Zhang, H., Liu, S. S., Niu, Y. P., Li, W. S., Li, W. M., Lin, H. R., and Chen, S. L., 2019. A chromosome-level genome assembly of the giant grouper () provides insights into its innate immunity and rapid growth.,19 (5): 1322-1332, https://doi.org/10.1111/1755-0998.13048.
Zou, J., and Secombes, C. J., 2011. Teleost fish interferons and their role in immunity.,35 (12): 1376-1387,https://doi.org/10.1016/j.dci. 2011.07.001.
March 2, 2020;
April 29, 2020;
October 23, 2020
© Ocean University of China, Science Press and Springer-Verlag GmbH Germany 2021
. Tel: 0086-532-85844606
E-mail: chensl@ysfri.ac.cn
(Edited by Qiu Yantao)
杂志排行
Journal of Ocean University of China的其它文章
- Corrosion Mechanism of 5083 Aluminum Alloy in Seawater Containing Phosphate
- Molecular Cloning, Expression and Characterization of Peroxisome Proliferators-Activated Receptors Gamma in the Sea Urchin (Strongylocentrotus intermedius)
- Otolith Shape Analysis as a Tool to Identify Two Pacific Saury (Cololabis saira) Groups from a Mixed Stock in the High-Seas Fishing Ground
- Characteristics and Influencing Factors of the Microbial Concentration and Activity in Atmospheric Aerosols over the South China Sea
- Characteristics of Atmospheric Rivers over the East Asia in Middle Summers from 2001 to 2016
- In vitro Antioxidant Effects of Porphyra haitanensis Peptides on H2O2-Induced Damage in HepG2 Cells
