Enzymatic Preparation of Peptone from Cold-Pressed Peanut Meal and Its Application in Microbial Culture
2020-01-07XIAOHuaiqiuLIYuzhenLINQinluLIUJunLANLixin
XIAO Huaiqiu, LI Yuzhen,*, LIN Qinlu, LIU Jun, LAN Lixin
(1. School of Pharmaceutical and Bioengineering, Hunan Chemical Vocational Technology College, Zhuzhou 412004, China;2. College of Food Science and Engineering, Central South University of Forestry and Technology, Changsha 410004, China)
Abstract: In this study, cold-pressed peanut meal hydrolysate (also named as cold-pressed peanut meal peptone, CPMP)was prepared by sequential enzymatic hydrolysis using pepsin and alkali proteinase. The molecular mass distribution,proximate analysis and amino acid composition of CPMP were determined. Furthermore, its potential as an alternative to the commercial peptone in microbial culture was evaluated. CPMP was mainly composed of < 5.0 kDa peptide fractions.CPMP was rich in Ala, Arg, Asp, and Cys, especially Glu. Escherichia coli grew better for 8 h in CPMP medium than in beef peptone yeast medium. Bacillus subtilis exhibited a longer lag phase in CPMP medium than in Luria-Bertani medium and possessed a better growth behavior after 8 h incubation. The pH of CPMP medium inoculated with E. coli or B. subtilis displayed no statistically signi ficant difference compared with the control medium (P > 0.05). Lactobacillus casei exhibited a different growth behavior in CPMP medium from that in MRS medium (P < 0.05). Staphylococcus aureus grew well in CPMP medium with no signi ficant difference from the control. Saccharomyces cerevisiae in CPMP showed similar growth behavior and pH trend to those in the control. Aspergillus oryzae grew well in CPMP, similar to that in Czapek-Dox medium. Our evidence demonstrated that CPMP has a good potential as a commercial peptone alternative for microbial growth.
Keywords: cold-pressed peanut meal; peptone alternative; synergistic hydrolysis; microbial culture
Peptones are defined as protein hydrolysates from proteinaceous materials that are soluble in water but not heat coagulable, and contain secondary protein derivatives such as polypeptides, dipeptides and amino acids. They are mainly used as assimilable nitrogen source in microbial media[1].Peptones are particularly suitable to use as the nitrogen source for microbial growth. It’s well-known that peptones are one of the most important constituents of culture media,and the costs of peptone often make up a major part of costs in the biopharmaceutical production and fermentation industry[2-3]. Hence, it is necessary and urgent to search for some new, reliable sources of high quality peptones that are less expensive than those currently available[4]. Currently,various proteinaceous materials from animals and plants,such as ram horn[5], fishery by-product[6], legumes[7], wool protein[8], chicken feathers[1], chicken bone[4], soybean[9]etc.have been studied for peptone preparation. Although the animal peptones are effective for cell cultivation, there is a potential risk of species cross contamination[10]. Animalderived ingredients have become increasingly banned from culture media for biopharmaceutical production[11-12].Recently, plant peptones that are manufactured by enzymatic hydrolysis of vegetal raw materials rich in proteins have been used as nitrogen sources as a peptone alternative in culture media for cell cultivation[7,13].
Chemical and enzymatic methods have been adopted for plant peptone production. Chemical methods involve alkali, such as NaOH and acid, such as HCl. Acid/alkaline hydrolysis allows high yields, however, this process results in high ash content in the final peptone products, as the neutralization step cannot be avoided. Enzymatic hydrolysis involves minimum side reactions and leads to limited hydrolysis of specific peptide bonds. Although enzymatic treatment generally has relative lower degree of hydrolysis(DH), the hydrolysis is more specific and can eliminate many of the problems associated with chemical hydrolysis[14].Therefore, enzymatic hydrolysis, which is moderately cheaper, more specific, faster, better controllable and less destructive than chemical hydrolysis[14], is widely applied to the hydrolysis of proteinaceous materials to the utilization of all valuable components[15].
Plant proteinaceous materials, especially oilseed protein sources, are regarded to play an important role in satisfying the nitrogen demand in microbial growth to replace the currently decreasing or economically inaccessible animal peptone sources, due to the renewability and availability of these sources[16]. Peanut (Arachis hypogaea L.) is an important oilseed crop and a major food legume that has been cultivated in over 100 tropical and subtropical countries,and is ranked the fifth largest source of vegetable oil in the world[17-18]. The main by-product for oil extraction processing is peanut meal, which contains 47%-55% proteins[19].Millions of tons of peanut meal are produced annually.Consequently, it is one of the most attractive and promising plant proteinaceous sources. Peanut meal can be classified into hot-pressed peanut meal (HPM) and cold-pressed peanut meal (CPM) depending on the processing methods[20].Although hot-pressed technique has a relatively higher oil yield than cold-pressed one, the remaining proteins in HPM are hard to utilize, since proteins in HPM are denatured. This results in a significant decrease in the solubility of HPM after hot pressing[17]. For these reasons, HPM has less value than CPM, and the majority of HPM are used as animal feed.Proteinaceous material determines not only the yield, but also the characteristics of peptones[14]. CPM keeps the natural protein spatial structure, therefore, it is of great significance to exploit an effective way to take advantage of CPM.
Currently, the utilization of peanut meal has attracted many interests and many investigations were performed,such as the preparation of peanut protein concentrate, peanut protein isolation, peanut peptide[21], peanut protein beverage,etc. At present, value-added utilization of CPM is still limited. There are rarely studies concerning the utilization of CPM as a nitrogen source for bacteria and fungi growth. In this paper, CPM was chosen as the proteinaceous resource for plant peptone preparation by sequential enzymatic hydrolysis,aiming to provide an alternative peptone to promote proliferation of typical industrial microbes.
1 Materials and Methods
1.1 Materials and reagents
CPM was purchased from Dezhou Hongxin Peanut Protein Food Co. Ltd. (Shandong, China); Pepsin and alkali proteinase 2709 were obtained from Guangxi Pangbo Biological Engineering Co. Ltd. (Guangxi, China). Beef extract and casein peptone (CP) were purchased from Shanghai Boao Biotechnology Co. Ltd. (Shanghai, China); Agar powder was bought from Tianjin Zhiyuan Chemical Reagent Co. Ltd.(Tianjin, China); All the other chemicals and reagents used were of analytical grade and obtained in China.
1.2 Instruments and equipment
DK-98-11A electric constant temperature water bath was produced by Tianjin Taisite Instrument Co. Ltd.; UV-2500 ultraviolet-visible spectrophotometer was manufactured by Shimadzu Corporation; DH-360 electrothermal constant temperature incubator was produced by Beijing Kewei Yongxing Instrument Co. Ltd. (Beijing, China); PHS-3C meter was produced by INESA Scientific Instrument Co. Ltd.(Shanghai, China); LDZX-50FAS vertical pressure steam sterilizer was manufactured by Shanghai Shen’an Medical Instrument Factory (Shanghai, China); KDN-08C Kjeldahl nitrogen determiner was produced by Hangzhou Top Instrument Co. Ltd. (Hangzhou, China); 835-50G automatic amino acid analyzer was manufactured by Hitachi Ltd. (Tokyo, Japan).
1.3 Methods
1.3.1 Sequential hydrolysis procedure
Pepsin-alkali proteinase sequential hydrolysis of CPM (protein content was determined by Kjeldahl assay with a conversion factor of 5.46[21], and protein content was (45.20 ± 1.73)% , n=3) was conducted in a hydrolysis reactor according to the method described by Megías et al.[22]with slight modification. Briefly, the CPM powder was dispersed into distilled water (1:50, V/V)and adjusted to pH 2.0 using 1.0 mol/L HCl, and then 5%(m/m) of pepsin (protein basis, 3 000 U/mg) was added into the reaction vessel and incubated at 37 ℃ in a water bath for 200 min. DH was monitored at different intervals.Subsequently, the pH of the resulting hydrolysate was raised to 8.0 with 1.0 mol/L NaOH solution. 2% (m/m) of alkali proteinase 2709 (m/m, protein basis, 50 000 U/g) was added and kept at 50 ℃ for 190 min. DH was also monitored at different intervals. All of the collected hydrolysates were placed in boiling water for 10 min for terminating the digesting procedure, the hydrolysates were cooled off to ambient temperature and centrifuged at 5 000 × g for 25 min at 4 ℃. The resulting supernatant were lyophilized and stored at 4 ℃ before use.
1.3.2 Determination of DH
DH is defined as the ratio of the number of peptide bonds cleaved expressed as hydrolysis equivalents (h), in relative to the total number of peptide bonds before hydrolysis (htot)[23].The htotis equivalent to the amino acid composition of the protein, calculated from amino acid analysis by summing the mmoles of each individual amino acid per gram of protein.The DH was measured by the pH-stat method[23]. The DH was calculated according to the equation (1):
Where B is the base consumption in mL; Nbis the normality of the base; Mpis the mass of protein being hydrolyzed; htotis the total number of peptide bonds in the protein substrate/(meqv/g); htotis 7.13 meqv/g for peanut protein[23]. The 1/α is the calibration factor for pH-stat, and is also the reciprocal of the degree of dissociation of the α-NH2groups, the α was calculated as equation (2):

Where pK is the average dissociation value for the α-amino groups, calculated according to the Gibbs-Helmholz equation[24], in this study, the average dissociation value pK can be calculated to be 7.4[23].
1.3.3 Molecular weight distribution
The molecular weight (mw) distribution of hydrolysate was determined by size exclusion chromatography (SEC) on a TSKGEL G3000PWxL (300 mm × 7.8 mm) with a HPLC system described by Zhao Mouming et al.[25]. The mobile phase used was 0.15 mol/L sodium phosphate buffer and was degassed and filtered through 0.2 μm membrane, and eluted at a fl ow rate of 0.5 mL/min at 25 ℃ and monitored at 214 nm. Standard molecular weight markers, including conalbumin (75 000 Da),oralbumin (43 000 Da), cytochrome C (12 384 Da), aprotinin(6 512 Da), VB12(1 855 Da) and glutathione (307 Da), were employed to calculate mw. A calibration curve was drawn from the lgmwof the protein markers vs their respective elution volume (y=-2.295x + 19.252, R2=0.996 4, x is the elution volume, and y is the lgmw). Cold-pressed peanut meal peptone (CPMP) with various DH were dispersed into the mobile phase to a final concentration of 3 mg/mL. After centrifugation (12 000 × g, 20 min, 4 ℃), the supernatants were filtered on a 0.2 μm PVDF membranes.
1.3.4 Proximate analysis and amino acid analysis
CPMPs were analyzed for protein, amino nitrogen, fat,ash, and moisture content. Nitrogen content was measured using a Micro-Kjeldahl assay, and total protein contents were calculated from the nitrogen value using a Kjeldahl foctor of 5.46[21]. GB 5009.6-2016Determination of fat in foods[26]shows that total lipid content was determined by the Soxhlet extraction method using diethyl ether as solvent. GB 5009.3-2016Determination of moisture in foods[27]showed that moisture content was determined by drying in an oven at 105 ℃until the constant weight. GB 5009.4-2016Determination of moisture in foods[28]showed that total sugar content was measured by the phenol-sulphuric acid method using glucose as a standard. Ash was determined by combusting a dry sample for 4 h in a muff l e furnace at (550 ± 25) ℃; Amino acids were analyzed using a Hitachi 835-50G automatic amino acid analyzer according the method described by Kain et al.[16]. Samples were placed in a sealed ampule for 24 h at 110 ℃ using 1 mL ofc(HCl)=6 mol/L in vacuum. Acid hydrolysates were evaporated and then the dried residue dissolved inc(HCl)=0.02 mol/L. Sample was filtered through a 0.45 µm nylon filter before being injected.
1.3.5 Tested microbes and corresponding medium ingredients
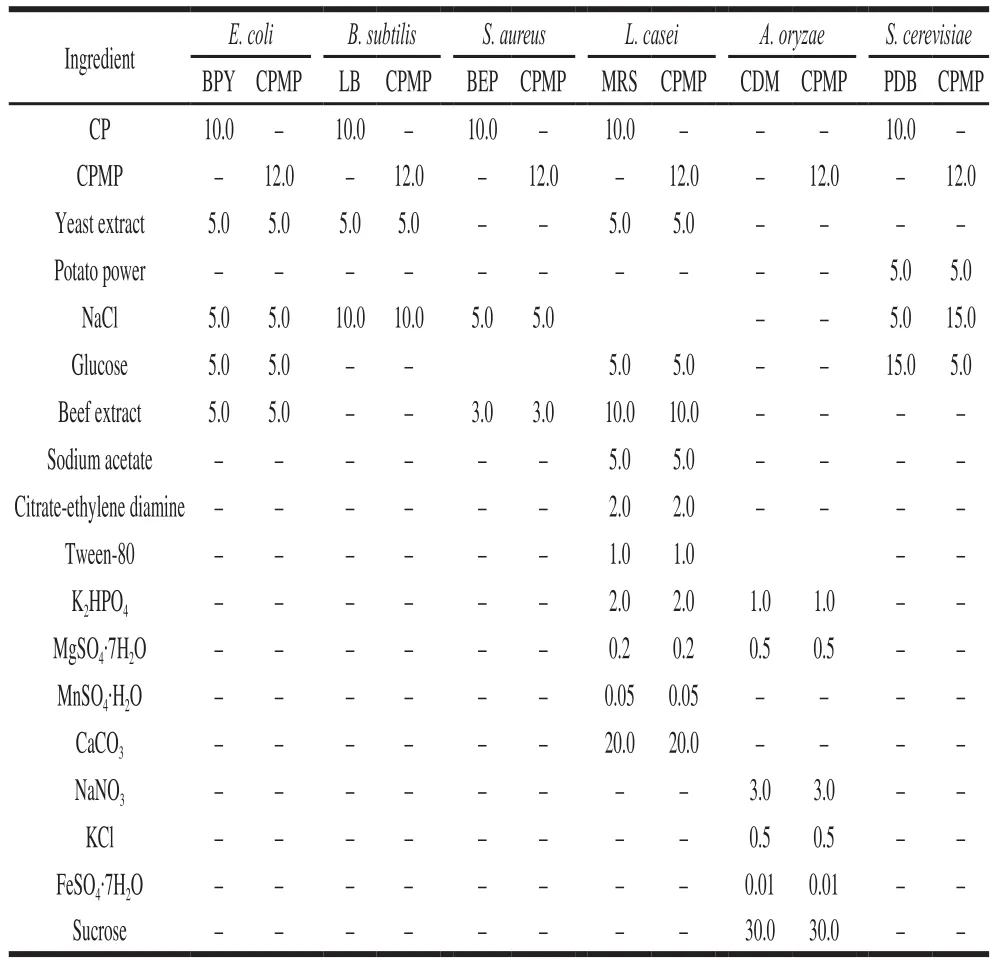
Table 1 Ingredients of media used for cultivation of tested microbes g/L
Four types of bacteria (Esherichia coli,Bacillus subtilis,Staphylococcus aureusandLactobacillus casei) and two types of fungi (a filamentous fungiAspergillus oryzae, and a yeastSaccharomyces cerevisiae) were adopted as a tested microbe. The tested microbes and corresponding medium ingredients were shown in Table 1.
To conveniently make a comparison of microbial growth behavior, CPMP was used in the medium to replace the CP with equivalent nitrogen content, and a standard medium was set up as a contrast for every tested microbe. In all cases, microbes were incubated in 250 mL Erlenmeyer flasks with 100 mL of medium with 150 r/min orbital shaking incubator, all the cultures were conducted independently in triplicates.
1.3.6 Determination of growth behavior
For bacteria (E. coli,B. subtilis,S. aureusandL. casei)and yeast (S. cerevisiae), absorbance at 600 nm (A600nm) and pH value were used as the appraisal indicators of growth behavior at 3 h intervals for up to 36 h. For the filamentous fungiA. aryzae, dry-matter weight of mycelium (biomass)was selected as an appraisal index of growth performance and the culture broths were taken out at different intervals(6 h intervals) up to 96 h. The procedures were as follows:50 mL of culture broths was centrifuged at 6 000 ×gfor 10 min, the mycelium was resuspended in 20 mL of sterile water, the suspension was centrifuged again under the same conditions, and the washed mycelium was then dried at 105 ℃ for obtaining a constant dry weight[8].
1.4 Statistical analyses
All the experiments were conducted independently in triplicates and the data were expressed as ±s. ANOVA was conducted to identify differences among means using SPSS 13.0 software. The differences between the means were determined by Duncan’s multiple test. Statistically significant differences between means were determined using Tukey’s multiplecomparison test at 99% con fidence level. Values withP< 0.01 were considered to be statistically signi ficant at 0.01 level.
2 Results and Analysis
2.1 DH changing tendency during sequential proteolysis
The most efficient way to produce protein hydrolysates that are used to as alternative peptone is sequential proteolysis of proteinaceous materials treated by proteinases with different specificity coupled with a post proteolysis[22]. CPM powder containing 200 mg of protein was hydrolyzed with 5%pepsin at 37 ℃, pH 2.0 up to 200 min, the DH was monitored at different intervals. DH changes during pepsin proteolysis were depicted in Part A of Fig. 1. The resulting hydrolysate continued to be digested by adding 2% alkali proteinase 2709 at 50 ℃, pH 8.0 for further 190 min, DH changes of post proteolysis was illustrated in Part B. Many studies indicated that the extent to a protein would be altered by hydrolysis was highly dependent on the degree to which the protein had been hydrolyzed[21]. And the functionality of the hydrolysates was closely related to the DH[29]. Hence, DH was used as a monitor indicators for protein hydrolysis[29].

Fig. 1 Change in degree of hydrolysis during sequential enzymatic hydrolysis of CPM
In the pepsin proteolysis (Part A), DH climbed rapidly in the first 60 min, the changing curve seems to be a straight line (y= 0.258 8x-0.289 3,R2= 0.992 2). After that, hydrolysis proceeded slowly. At the end of pepsin proteolysis, DH attained (23.95 ± 0.90)%. DH changes for subsequent post proteolysis treated by alkali proteinase were illustrated in Part B. It was observed that the synergistic effect of sequential proteolysis enhanced the proteolysis[22],at the end of the whole proteolysis, the final DH was(39.42 ± 1.84)%. Although CPM protein was hydrolyzed efficiently during the pepsin proteolysis, the proteolysis efficiency still can be enhanced with a post proteolysis,mainly owing to the alkali proteinase that had a broad specificity[22], which was accordance with the findings observed by Guerard[30]and Megías[22]et al. found that hydrolysis rate with pepsin climbed rapidly during the initial proteolysis, subsequently proceeded slowly and attained the plateau. However, the DH was increased rapidly with a post proteolysis[22]. During the pepsin proteolysis, the CPM protein was degraded into larger molecular weight peptide fractions,and may dissociate or even unfold in structure[22], the alkali proteinase treatment sequentially can dissociate them into smaller fragments[5]with an accumulation of shorter peptides and amino acids, which was beneficial for the microbial growth and water solubility. Hence, it had been proved that sequential hydrolysis with different hydrolysis specificities would be a better strategy for yielding plant peptone.
2.2 Molecular weight distribution
Peptide fractions with low molecular weight usually exhibited better functional properties and bioactivity[4,17].Peptide fractions derived from sequential proteolysis were separated and divided into three fractions (mw> 5 kDa,3-5 kDa and < 3 kDa), the quantitative changes of three peptide fractions were listed in Table 2.
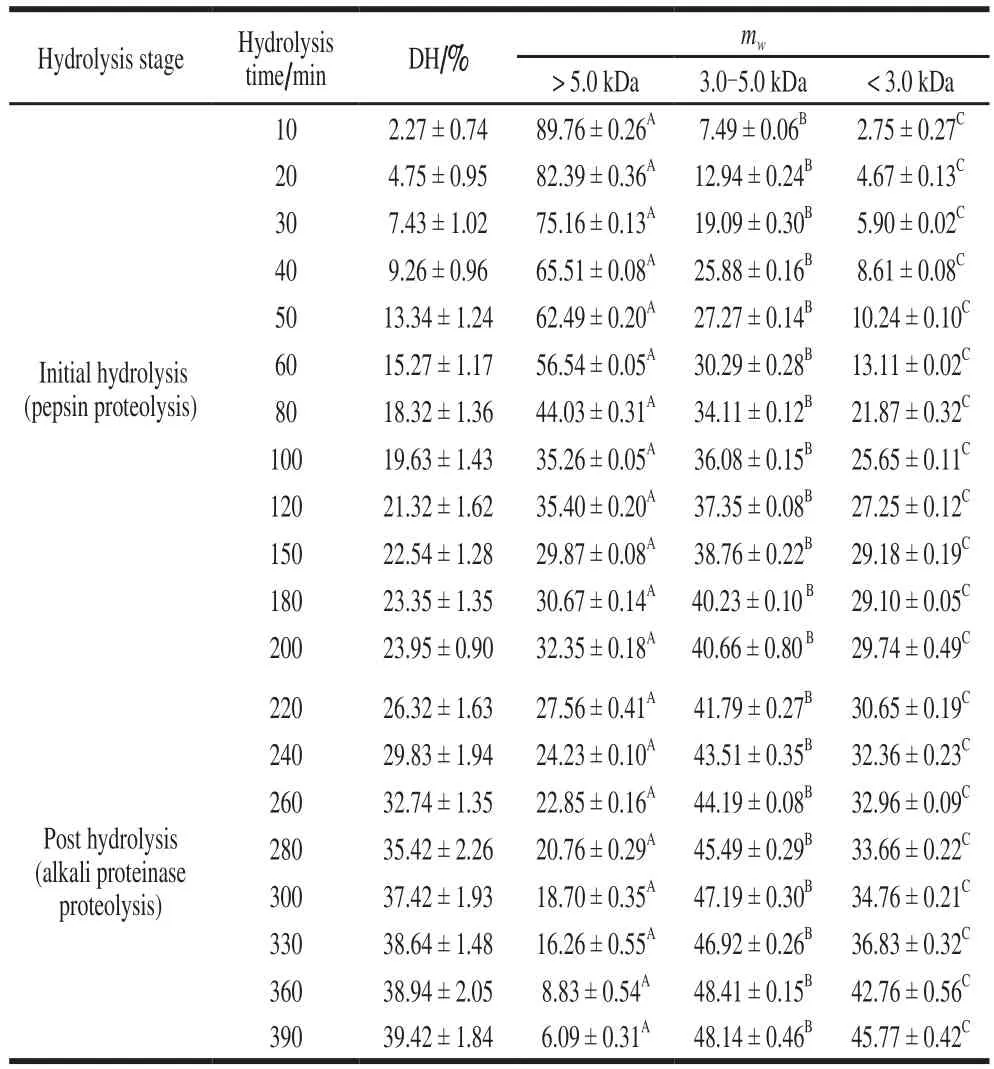
Table 2 Molecular mass distribution of CPMP
From Table 2, it can be concluded that themwdistribution of CPMP was noticeably distinct (P< 0.01),reflecting the differences in peptide chain lengths, which could greatly affect amino acid performances of peptide fractions. During 0-60 min pepsin proteolysis, hydrolysates were dominated by the fractions that were larger than 5.0 kDa, and the occupancy rate of larger than 5.0 kDa is(56.54 ± 0.05)% at the 60 min hydrolysis. The peptide fractions that between 3.0-5.0 kDa and < 3.0 kDa were(30.29 ± 0.28)% and (13.11 ± 0.02)% at this point,respectively. After sequential post proteolysis, the fractions larger than 5.0 kDa was drastically reduced to (6.09 ±0.31)%. Nevertheless, the fractions with molecular weights of 3.0-5.0 kDa and < 3.0 kDa were increased to (48.14 ±0.46)% and (45.77 ± 0.42)%, respectively, indicating that CPM protein is degraded into shorter peptide fractions,which is consistent with the findings observed by Agudelo et al.[31], who declared that large polypeptides were the main product of casein protein with initial hydrolysis. It can be concluded that sequential proteolysis can strengthen protein proteolysis, and the CPM was substantially fragmented into shorter peptide fractions[22]. The findings also confirmed that sequential proteolysis using pepsin plus alkali proteinase was prefer to pepsin proteolysis, yield higher DH and more shorter peptide fractions.
2.3 Proximate analysis
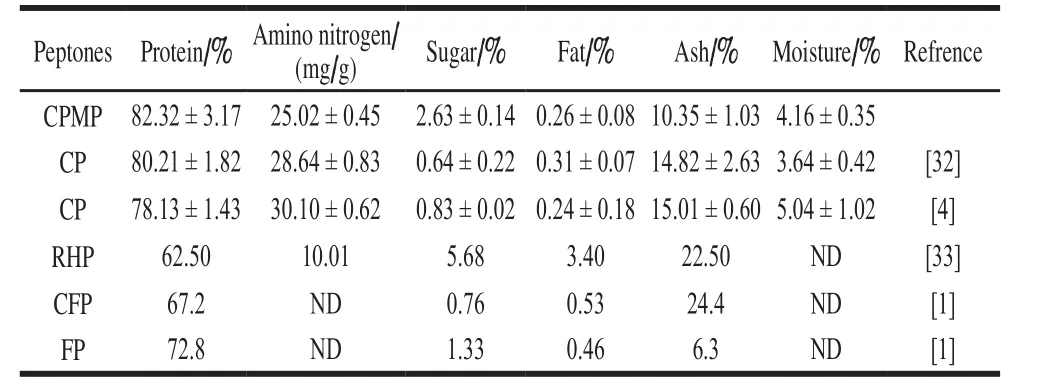
Table 3 Proximate analysis of CPMP in comparison with other peptones
From Table 3, it can be seen that CPMP contained some nutritional substances required for microbial growth.When compared with the other peptone (CP, ram horn peptone (RHP), chicken feather peptone (CFP), and fish peptone (FP)), it’s worthy to notice that the protein content of CPMP (82.32 ± 3.17)% was higher than other peptones,such as CP (80.21 ± 1.82)%[32]or (78.13 ± 1.43)%[4], RHP(62.50%)[33], CFP (67.2%)[1], and FP (72.8%)[1]. It also can be seen from table 3 that CPMP had relative higher amino nitrogen content (25.02 ± 0.45)%, and higher than that of RHP (1.001%), but lower than CP ((2.864 ± 0.83)%or (3.010 ± 0.62)%). Sugar content in CPMP was lower than RHP(5.68%), but higher than that of CP((0.64 ± 0.22)% or (0.83 ± 0.02)%), CFP (0.76%) and FP (1.33%), and this is mainly due to CPM riches in polysaccharide and carbon. Seifert[34]reported that peanut carbohydrate was comprised of 2%-5% sugar, 4% starch, 4%pectic material and 2% cellulose, this finding also indicated that CPMP was beneficial for microbial growth, since carbon source was a critical nutritional substance for microbial metabolisms. Fat content in CPMP was lower than all other contrast because of the application of skim material. Ash was a reflection of mineral levels in the peptones and served as the main source of mineral elements needed for microbial activities[8]. It should be realized that too high or low in mineral level was disadvantageous for microbe growth[35].Data indicated that the ash content of CMPH was close to that of CP, but lower than that of RHP, CFP, and this due to the RHP that was treated with acid (6N-HCl)[33]and CFP was derived from feathers using alkaline (KOH)[1], mainly due to the neutralization steps that were unavoidable during the preparation of RHP and CFP[4,32-33]. The moisture content for all peptones had insignificant difference.
2.4 Amino acid analysis
The amino acid content and composition of a peptone had a significant inf l uence on cell growth and product yield[2].The amino acid contents of CPMP, Bacto-tryptone (BTP),wool peptone (WP), RHP, CFP and FP were list in Table 4.

Table 4 Amino acid composition of CPMP and other peptones%
From Table 4, the contents of some amino acids, such as Ala, Arg, Asp, Gly, His, Phe, Trp, and Tyr, of CPMP,especially Arg, were higher than that of BTP. The Arg content in CPMP was nearly quadruple that of BTP. Arg was not observed in CFP[1]and FP[36]. Cys and Trp were not found in BTP[32], it is likely that the Cys and Trp lost during BTP preparation. Glu was the most abundant amino acid in CPMP(22.0%,m/m), followed by those of Arg (12.7%,m/m), and Asp (9.75%,m/m), etc. Trp was missing for WP and RHP may be due to its breakdown during chemical hydrolysis[33].It cannot be ignored that Trp, an essential amino acid, was destructed for BTP, WP, RHP, CFP and FP, except for CPMP. The Cys content of CPMP was lower than that of WP, CFP and FP, but higher than BTP and RHP[33]. It can be concluded that CPMP had a balanced amino acid composition and can provide more nutrition for microbial growth. Besides,all of eight essential amino acids (Met, Trp, Val, Lys, Ile,Leu, Phe, Thr and His) and two semi-essential amino acids(Cys and Tyr) for human beings were included. All evidences demonstrated that CPMP had a higher nutritional value that either for food additive, or as nitrogen source for microbial growth and a balanced amino acid composition and with utilization potentiality as the nitrogen source for microbial culture.
2.5 Performances of microbial growth performances
Peptone is one of the most important constituents of microbial medium, and often makes up the major part of the production costs of microbial cells and bio-products in the fermentation industry[4]. E. coli, B. subtilis, L. casei,A. aryzae and S. cerevisiae are the most important industrial microorganisms and are widely adopted as seeds for enzyme production, genetic engineering vector, type strains, etc.Although S. aureus is not always pathogenic, it is a common cause of skin infections and food poisoning. All above mentioned six strains were chosen to evaluate the potentiality and feasibility of CPMP to serve as the alternative peptone for microbial culture and biopharmaceutical production.
2.5.1 Bacterial culture
E. coli and B. subtilis are the most important microbes for servicing as cloning and over-expression of proteinaseencoding genes. B. subtilis is widely used in industry to yield alkali proteinase in Japan, China, and other countries,and applied in pharmaceuticals, detergent and food fields[37].E. coli is widely adopted as a gene manipulation systems host cell and as a strain for enzymes and many industrial products[38].
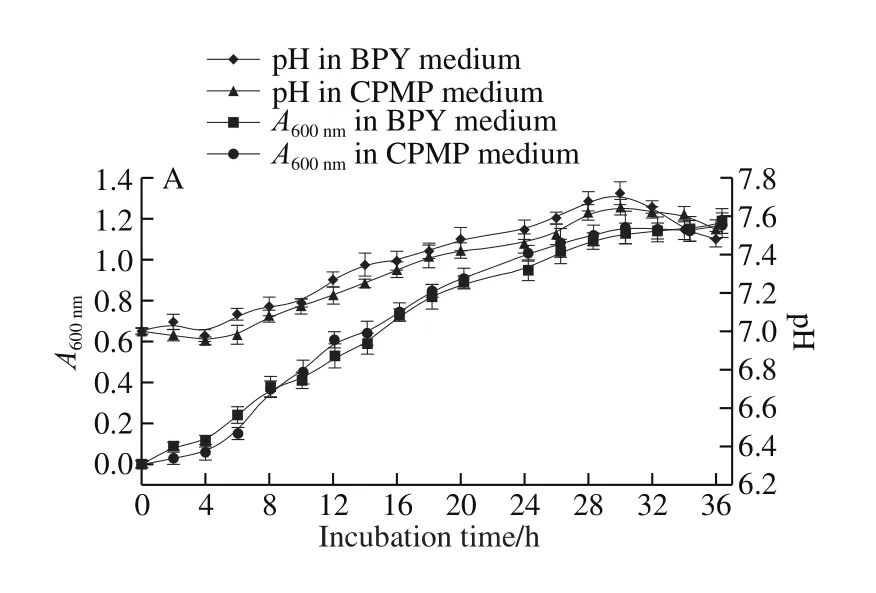
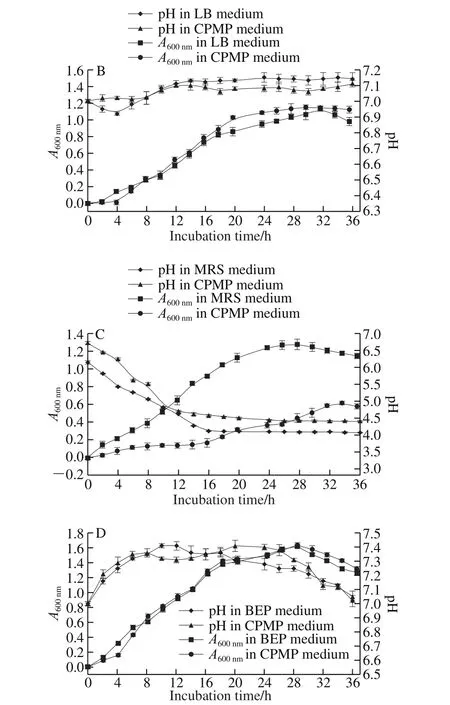
Fig. 2 Growth curves and pH changes of E. coli (A), B. subtilis (B),L. casei (C) and S. aureus (D) in CPMP and corresponding standard media
E. coli grew well in CPMP medium and BPY medium with a similar growth curve and pH change (Fig. 2A). It was observed that the A600nmof CPMP was slightly lower than that of BPY from 0-8 h, after that, the growth performance of E. coli incubated in CMPH was better than that of BPY medium. The difference of pH changes was not significant(P > 0.05). For B. subtilis, there was a relatively longer lag phase in CPMP (4 h) than in LB (2 h), as depicted in Fig. 2B.B. subtilis exhibited a better growth performance in CPMP medium than that in LB medium after 8 h incubation, A600nmwas increased by 1.026 in CPMP medium and 0.857 in LB medium, respectively, at 20 h incubation (P > 0.05). There was no statistically significant difference for pH value between incubated in CMPH and LB during the whole incubation, which was accordance with the findings observed by Kurbanoglu et al.[33], who found that the absorbance value of E. coli in RHP was similar to that of bacto peptone.Hence, a conclusion can be made that E. coli and B. subtilis can be cultured well in the medium containing CPMP as an alternative of CP, and the growth rate of the two tested microbes in CPMP could provide a relatively economical approach for such countries like China that need to import peptone from abroad.
L. casei displayed a noticeably distinct growth behavior in CPMP from that in MRS, as plotted in Fig. 2C. There was a longer lag-phase up to 16 h in CPMP medium, while that in standard MRS rapidly stepped into a logarithmic phase at 4 h incubation up to 24 h. Concurrently, the pH in CPMP kept on decreasing from 6.73 to nearby 4.41, while that of the MRS group decreased rapidly from 6.15 to 4.12 at 16 h incubation.L. casei was a fastidious food bacterium, which demands for rich in nutritional ingredients[39]. A conclusion can be drawn that L. casei could grow well in CPMP, but not as fast as in the MRS medium, a similar result was observed by Wang Jinzhi et al.[4]. A contradictory finding was also made by Safari et al.[40], who drawn a conclusion that lactic acid bacteria exhibited a better growth rates in the FP medium than meat and CP, this was mainly due to the ingredient difference of various peptones[37]. The growth behavior and pH changes of S. aureus were figured in Fig. 2D.S. aureus grew well in CPMP, which was equal to that in BEP(P > 0.05). The optimum growth time of S. aureus incubated in CMPM and BEP medium was regarded at the same incubation time, 28 h. Hence, it suggested that CPMP was equally suitable for the growth of S. aureus as the standard CP.
2.5.2 Yeast culture

Fig. 3 Growth curves and pH changes of S. cerevisiae in CPMP and PDB medium
S. cerevisiae in CPMP medium exerted an analogous growth behavior (Fig. 3) with a weaker growth performance between 0-10 h incubation as compared to that in PDB medium. A600nmof CPMP group was nearly equal to that in PDB after 10 h incubation. The pH in CPMP and PDB were slowly decreased to nearby 5.45 during 0-18 h incubation,and gradually increased to 6.82 and 6.87 respectively at 36 h incubation. Nitrogen source was the restrictive nutrition for S. cerevisiae growth[41], the maximum growth rate was closely related to the assimilable nitrogen content in medium. It can be concluded that the assimilable nitrogen and amino acids of CPMP peptone were equivalent for S. cerevisiae as that of CP.
2.5.3 Fungi culture
A. oryzae was an important industrial fungus for enzyme production, such as proteinase, amylase, glucoamylase,cellulose, etc. Growth performance of A. oryzae in CPMP medium was similar to that in CDM medium, which was depicted in Fig. 4, but grew faster than that in CDM medium.The higher sugar content (2.63 ± 0.14)% in CPMP medium probably was one of the primary causes accounting for the growth difference, because the sugar was essential and critical to fungal growth and could be used as carbon source[4].Similar phenomena were discovered in FP and RHP[5]. The biomass of A. oryzae reached the highest level (16.2 g/L) at 78 h incubation in CPMP medium, the biomass in CDM was only 13.0 g/L. Hence, CPMP can be used as an alternative peptone for A. oryzae growth.
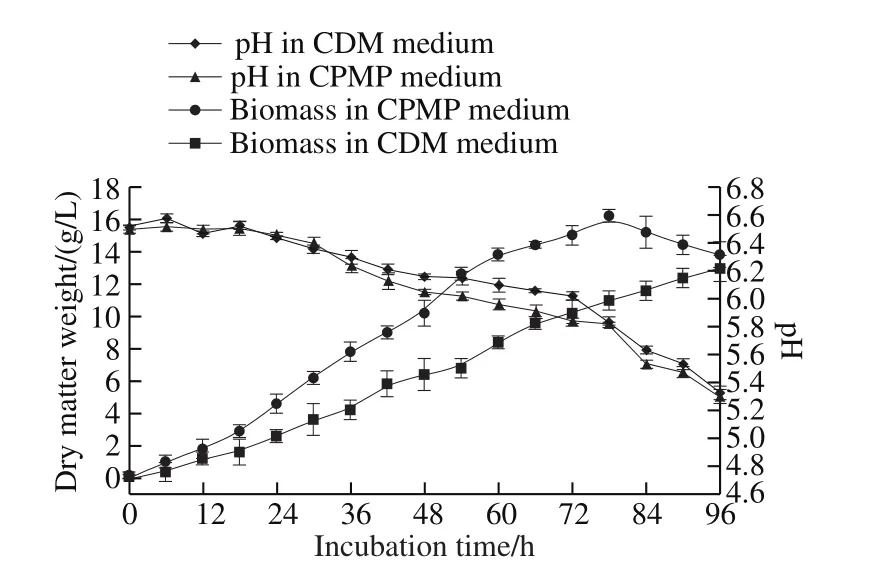
Fig. 4 Growth curves and pH changes of A. oryzae in CPMP and CDM medium
Many studies reported that peptones from different proteinaceous materials affect bacterial and fungal growth in spite of containing the same nitrogen content[4]. It was owing to the metabolic complexity and the nutrimental difference for various microbe demands. There were different growth behaviors in CPMP medium for the chosen microbes when compared to commercial peptone, which can be accounted for in the following assumptions[8,16,36]:
1) The amino acid content in CPMP medium was different from that of commercial peptone. CPMP possessed higher content of certain amino acids, such as Arg, Asp, Ala,Cys, etc. Some amino acids, such as Glu, Ile, Leu, Lys, etc.were slightly lower than that from CP. Therefore, CPMP contained a balanced amino acid composition like BTP,but with different amino acid formula. This might be an important reason for resulting in the growth difference. It is worthy of noticing that the differences in amino acid formulas for WP, RHP, CFP and FP among of the contrastive, which was mainly due to the different protein had various nitrogen content and amino acid compositions.
2) The nutrition required for tested microbes were different. Therefore, tested microbe exhibited different growth behavior in CPMP medium and corresponding standard medium.
3) CPMP had higher sugar content, resulting in different growth. A similar finding was also observed in peptones with higher sugar content and higher ash content, such as RHP[33].
4) The sequential hydrolysates were mainly composed of lower molecular weight peptide fractions (3-5 kDa and < 3.0 kDa).It is well known that lower molecular weight peptide fractions were more easily utilized by microbes than those with higher molecular weight[38]. The result shows that CPMP can be suitable for bacterial and fungal growth.
3 Conclusion
A simple and low-cost assay for effective proteolysis of CPM powder through sequential digested by pepsin plus alkali proteinase has been investigated, aiming to provide a way to convert CPM into CPMP. CPM was digested using 5% pepsin firstly, and successively digested by 2% alkali proteinase. The hydrolysate (also called CPMP) was mainly composed of lower molecular peptide fractions. At the end of the proteolysis, the fractions that between 3.0-5.0 kDa and < 3.0 kDa were (48.14 ± 0.46)% and (45.77 ± 0.42)%,respectively. Protein content of CPMP was significantly higher than CP, RHP, CFP, and FP. Sugar content in CPMP was higher than that of CP, CFP and FP. The ash content of CMPH was closer to that of CP, but lower than that of RHP, CFP. The moisture content for all peptones were not significant different. E. coli grew well in both CPMP medium and BPY medium with a similar growth curve and pH performance. B. subtilis had a longer lag phase in CPMP medium than in LB medium, but possessed a better growth behavior after 8 h incubation, L. casei displayed a noticeably distinct growth behavior in CPMP medium from that in MRS medium. S. aureus grew well in CPMP medium and BEP medium. S. cerevisiae in CPMP medium exerted an analogous growth curve compared with that in PDB medium.Growth behavior of A. oryzae in CPMP medium was similar to that in CDM medium. Currently, it’s an urgent demand to utilise plant peptone for replacement of animal peptone in industrial production of enzyme, biopharmaceutical production, animal or plant cell cultivation, etc. for avoiding species cross contamination and reducing production cost.This is the first investigation on the use of CPMP as a commercial peptone alternative for bacteria and fungi growth.It’s proved that CPMP was a good peptone alternative,especially suitable for B. subtilis, E. coli, A. oryzae and S. aureus, but not appropriated for L. casei growth. All evidences indicated that CPMP had a better potentiality for replacing commercial CP. Research indicated that the proposed process for inexpensive peptone production provided an effective way to utilize CPM.
