Clinical significance of MLH1/MSH2 for stage ll/lll sporadic colorectal cancer
2019-12-14ShuiMingWangBinJiangYoupingDengShuLiangHuangMingZhiFangYuWang
Shui-Ming Wang,Bin Jiang,Youping Deng,Shu-Liang Huang,Ming-Zhi Fang,Yu Wang
Shui-Ming Wang,Bin Jiang,National Center of Colorectal Disease,Nanjing Hospital of Chinese Medicine Affiliated to Nanjing University of Chinese Medicine,Nanjing 210022,Jiangsu
Province,China
Youping Deng,Yu Wang,Bioinformatics Core,Department of Complementary and Integrative Medicine,University of Hawaii John A.Burns School of Medicine,Honolulu,HI 96813,United States
Shu-Liang Huang,Department of Pathology,Nanjing Hospital of Chinese Medicine Affiliated to Nanjing University of Chinese Medicine,Nanjing 210022,Jiangsu Province,China
Ming-Zhi Fang,Yu Wang,Department of Oncology,Nanjing Hospital of Chinese Medicine Affiliated to Nanjing University of Chinese Medicine,Nanjing 210022,Jiangsu Province,China
Abstract
Key words: Colorectal cancer; Mismatch repair gene; MLH1; MSH2; Microsatellite instability
INTRODUCTION
Colorectal cancer (CRC) is one of the most common malignancies of the digestive tract.In 2017,there were nearly 135430 newly diagnosed CRC cases with 50260 associated deaths in the United States[1].CRC is mainly associated with at least three distinct genetic pathways:Microsatellite instability (MSI),chromosomal instability(CIN),and CpG Island methylator phenotype (CIMP)[2].Most hereditary nonpolyposis CRC and 15%[3]of sporadic CRC are characterized by MSI[4,5].In contrast,85% of CRC develop from the CIN pathway and are characterized by aneuploidy,allelic losses,amplifications,and translocations[6].Meanwhile,many sporadic MSI CRC are also CIMP positive.These three pathways are not mutually exclusive,and most tumors are characterized by multiple pathways.The mismatch repair (MMR) gene[7]is a housekeeping gene that is highly conserved.MMRmaintains correct DNA replication and high fidelity by repairing DNA base mismatches,which allows for genomic stability and reduces spontaneous mutations[8].MSI is characterized by the deletion of DNA methylation orMMRcaused by genetic mutation,which leads to widespread alterations in the length of short repeated sequences[9,10].In China,the incidence of CRC has increased significantly in the last 10 years.While the mortality rate of male patients has increased annually,the mortality of female patients has tended to be relatively stable.CRC is the fifth leading cause of morbidity in men and fourth in women.The number of new cases of CRC in 2015 in China was 376300,including 215700 men and 160600 women.There were 191000 deaths due to CRC,including 111100 men and 80000 women[11].
Most cases of MSI appear to result fromMMRdeficiency.At least six of the genes involved inMMRhave now been identified,including MutL homolog 1 (MLH1),MutS homolog 2 (MSH2),MutS homolog 3 (MSH3),postmeiotic segregation increased 1(PMS1),postmeiotic segregation increased 2 (PMS2),and MutS homolog 6(MSH6)[12].MLH1is located on chromosome 3p21-23 and is connected with the creation of MLH3,PMS2,andPMS1.MSH2is located on chromosome 2p21 and has been shown to formMSH3andMSH6[13].Mutations in eitherMLH1orMSH2account for the majority of known germline mutations in CRC,and >90% of MMR deficiencies are deletions ofMLH1orMSH2that rarely appear in other genes,which are the major causes leading to the mutated phenotype[14,15].Mutations either inMLH1orMSH2or both of them are considered asMLH1/MSH2negative,and no mutations in either of them are considered asMLH1/MSH2positive.MLH1recruits and allocates other proteins to the mismatch repair system andMSH2can recognize any errors in DNA replication and replace the incorrect sequence using the parental strand sequence as the correct one[16].
The purpose of the current study was to evaluate the prognostic significance ofMLH1/MSH2status determined by immunohistochemical analysis in a large cohort of patients with stage II-III CRC.In particular,we sought to detect the relationship betweenMLH1/MSH2and overall survival (OS).
MATERIALS AND METHODS
Patients
Initially,we enrolled 836 consecutive patients who underwent curative-intent surgical resection between January 2013 and December 2016 at the Nanjing Hospital of Chinese Medicine Affiliated to Nanjing University of Chinese Medicine (Nanjing,China).One hundred and fifty-five patients were excluded due to loss to follow-up or because their specimens were not available for immunohistochemical analysis.There were at least 681 patients who had all data available at the time of follow-up (387 males and 294 females with a median age of 63 years; range 22-87 years).Diagnosis of CRC was confirmed according to World Health Organization (WHO) criteria and the TNM stage classification[17].Clinical data were obtained from hospital medical records and included details pertaining to patient gender and age; tumor differentiation,location,and size; and mucin,surgical margin,TNM stage,lymph node (LN)metastasis,and histopathological grade.Follow-up of all cases started from the postoperative period to December 2018.Of the 681 patients with CRC,300 underwent surgery only and 381 surgery plus adjuvant chemotherapy.One hundred and twentytwo (30.9%) of the 395 patients with stage II CRC and 259 (90.6%) of 286 with stage III CRC received postoperative adjuvant chemotherapy.Patients with stage II CRC were subdivided into high- and low-risk categories according to poor prognostic features.The National Comprehensive Cancer Network (NCCN) guidelines[18]consider the following high-risk factors for recurrence:Poorly differentiated histology [exclusive of those cancers that have MSI (MSI-H)] or undifferentiated tumors; pathological T4(pT4) disease; perineural invasion; bowel obstruction; indeterminate or positive margins or localized perforating tumors; and inadequate LN sampling (<12 LNs).
Treatment
Combining the NCCN guidelines with patients’ personal wishes,for stage II CRC,all patients with no high-risk factors,90% (36/40) of those with high-risk factors combined withMLH1/MSH2negativity,and 6.56% (12/183) of those with high-risk factors combined withMLH1/MSH2positivity received only regular follow-up.The remaining stage II patients received the CAPEOX regimen (oxaliplatin 130 mg/m2for 1 d,intravenous drip 2 h; capecitabine 1 g/m2/d for 14 d,oral administration,every 21 d for 6 cycles).For stage IIIA[19](T1-2N1M0 or T1N2M0) CRC,69.91%(79/113) of patients received the CAPEOX regimen,and for stage IIIB-IIIC (T3-4N1M0 or T1-4N2M0),80.92%(140/173) of patients with high-risk factors for recurrence received the FOLFOX6 regimen (oxaliplatin 85 mg/m2for 1 d,intravenous drip 2 h; calcium folinate 400 mg/m2for 1 d,intravenous drip; 5-fluorouracil (5-FU) 400 mg/m2for 1 d,intravenous injection; 5-FU 1.2 g/m2for 46 h,intravenous drip,every 14 d for 12 cycles),and the remaining ones received only regular follow-up.No patients received radiotherapy,neoadjuvant chemotherapy,or immunotherapy before surgery.Written informed consent was obtained from each patient.Ethical approval to perform this research was issued by the Human Research Ethics Committee of the Nanjing Hospital of Chinese Medicine affiliated to Nanjing University of Chinese Medicine.
Histopathological and immunohistochemical analyses
According to histopathological and immunohistochemical detection,tumor types were classified into adenocarcinoma with or without mucin production.Three hundred and ninety-five patients had stage II CRC and 286 had stage III on the basis of the WHO criteria.
The tumor tissue specimens were immersed in 4% paraformaldehyde for 4 h,and transferred to 70% ethanol.Individual lobes of tumor tissue biopsy material were placed in processing cassettes,dehydrated through a graded series of ethanol,and embedded in paraffin wax blocks.Before immunostaining,5-μm-thick colorectal tissue sections were dewaxed in xylene,rehydrated through decreasing concentrations of ethanol,washed in phosphate-buffered saline,and stained with hematoxylin and eosin.After staining,sections were dehydrated through increasing concentrations of ethanol and xylene.Staining was carried out using the BenchMark XT system (Roche,Shanghai,China).
The specimens were observed under a light microscope,with 10 fields with no fewer than 100 cells per field observed by a double-blind method.At least 5% of the tumor cells were stained positive,otherwise they were considered negative.The final result was reviewed by more than two senior pathologists.Complete loss ofMLH1orMSH2expression was classified asMLH1orMSH2negative,which formed theMLH1/MSH2-negative group.Normal expression ofMLH1andMSH2was classified asMLH1positive orMSH2positive,which formed theMLH1/MSH2-positive group[20].
Microsatellite analysis
One hundred and thirty-three fresh CRC tissues and matched tumor-adjacent normal tissues were collected,frozen in liquid nitrogen,and stored at -80 °C.DNA was extracted by a standard phenol-chloroform procedure.Before DNA extraction,frozen sections were cut from each tumor sample,which were stained with hematoxylin and eosin to verify by microscopic examination the presence of adequate neoplastic material (60%-70% of tumor cells).The primers,location,and sequence of MSI are listed in Table1.
In all 133 cases,MSI was evaluated at five microsatellite loci (BAT26,BAT25,D2S123,D5S346,andD17S250) using fluorescence-based polymerase chain reaction(PCR).Analysis of the PCR products was performed with an automated laseractivated fluorescent DNA sequencer using the Applied Biosystems 3130XL and analyzed with GenScan 3.1 software.MSI-H was defined as ≥2 mutation sites; low MSI (MSI-L) was defined as only one mutation site; and microsatellite stability (MSS)was defined as no mutations.
Statistical analysis
The relationship betweenMLH1/MSH2expression and clinicopathological factors was analyzed by theχ2test.Clinical factors that were analyzed included age,gender,tumor stage,differentiation,lymphocytic infiltration,tumor size,mucin,and tumor margin.Survival was estimated by the Kaplan-Meier method.Univariate and multivariate analyses were carried out using Cox’s proportional hazards regression models.P< 0.05 was defined as significant.The statistical analyses were performed using SPSS version 20.0.
RESULTS
MLH1/MSH2 expression and clinicopathological features
Among the 681 patients,131 (19.24%) wereMLH1/MSH2-negative and 550 (80.76%)wereMLH1/MSH2-positive.The relationship between clinicopathological characteristics andMLH1/MSH2expression is shown in Table2 and Figure1.MLH1/MSH2-negative CRC occurred more frequently in the right than in the left colon (27.88%vs17.86%,P= 0.029).It also occurred more frequently in the colon than in the rectum (22.82%vs15.80%,P= 0.025) and in poorly differentiated than well-moderately differentiated CRC (33.33%vs18.31%,P= 0.017).In addition,MLH1/MSH2-negative CRC was characterized by LN metastasis and mucinous tumor.MLH1/MSH2-negative tumors were more likely to contain mucin (P= 0.024).MLH1/MSH2expression was not associated with age,gender,tumor stage,tumor size,lymphocytic infiltration,or circumscribed margin (P> 0.05).
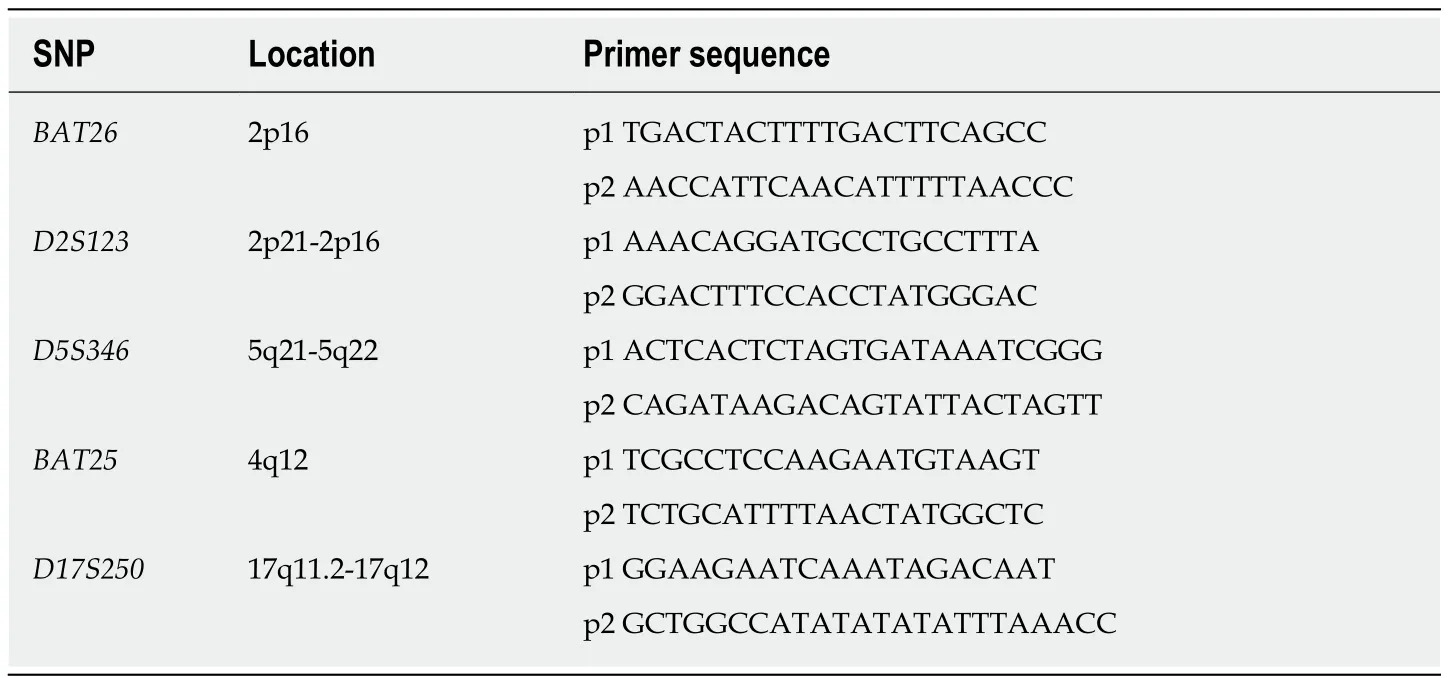
Table1 Name of primers,location,and the sequence of microsatellite instability
Survival analysis
With a median follow-up period of 56 mo (range 8.0-72.0 mo),36 of 395 (9.11%)patients with stage II CRC died and 42 (10.63%) had recurrence or metastasis during the study.For stage III CRC,90 of 286 (31.47%) patients died and 107 (37.41%) had recurrence or metastasis.Causes of death included cancer recurrence (n= 46),metastasis to other organs (n= 51),pulmonary infection (n= 15),heart disease (n=11),second primary cancer (n= 17),multiple organ failure (n= 7),and unspecified reasons (n= 8).Some patients had more than one cause of death.
Patients withMLH1/MSH2-negative stage II or III CRC showed a favorable trend for OS (68.62 ± 0.83vs62.11 ± 1.07 mo,P< 0.001).Stratified analyses showed that patients withMLH1/MSH2-negative stage II CRC had longer OS than those withMLH1/MSH2-positive CRC (70.67 ± 0.65vs66.02 ± 1.01 mo,P= 0.011).Patients withMLH1/MSH2-negative stage III CRC also had a longer OS than those withMLH1/MSH2-positive (66.05 ± 1.62vs63.40 ± 1.15 mo,P= 0.023).In patients who received adjuvant chemotherapy,those withMLH1/MSH2-negative CRC had an OS of 64.02 ± 1.61 mo compared with 62.11 ± 1.07 mo in those withMLH1/MSH2-positive CRC (P= 0.015).The 5-year survival rate for patients withMLH1/MSH2-negative CRC was 86.9%,compared with 59.1% for patients withMLH1/MSH2-positive CRC.The data for OS are listed in Figure2.
Univariate and multivariate analyses
In univariate analysis,patients withMLH1/MSH2-positive CRC had a significantly worse OS than those withMLH1/MSH2-negative CRC [P< 0.001,hazard ratio (HR) =3.799,95%CI:2.205-6.546].Several other factors were also associated with disease survival,such as age,tumor differentiation,tumor stage,lymphocytic infiltration,mucin,and circumscribed margin (P< 0.05).
In multivariate survival analysis incorporating status ofMLH1/MSH2,gender,age,tumor location,tumor differentiation,tumor stage,tumor size,lymphocytic infiltration,mucin,and tumor margin,the status ofMLH1/MSH2was an independent prognostic factor for OS (P< 0.001,HR = 4.064,95%CI:1.241-7.369).Besides that,age,tumor location,tumor stage,lymphocytic infiltration,mucin,and circumscribed margin were also independent prognostic factors for OS (P< 0.05).In the subgroup analysis of stage II CRC,patients withMLH1/MSH2-negative tumor demonstrated a better OS than those withMLH1/MSH2-positive tumor (multivariateP< 0.011,HR =5.583,95%CI:1.478-21.092).Patients with stage III disease had similar results butMLH1/MSH2status was less significant than in patients with stage II disease(multivariateP= 0.023,HR 2.289,95%CI:1.270-4.125) (Table3).
We observed no significant benefit from adjuvant chemotherapy for patients withMLH1/MSH2-negative (multivariateP= 0.147,HR = 1.563,95%CI:0.481-4.441) orMLH1/MSH2-positive (multivariateP= 0.070,HR = 1.267,95%CI:0.212-5.052) stage II CRC.However,a better survival was observed for patients withMLH1/MSH2-negative stage III CRC who received adjuvant chemotherapy (multivariateP< 0.001,HR = 7.660,95%CI:2.974-15.883).But a nonsignificant trend for survival benefit from adjuvant chemotherapy was observed in patients withMLH1/MSH2-positive stage III disease (multivariateP= 0.052,HR = 2.817,95%CI 0.223-6.671) (Table4).All findings are consistent for the OS end point.
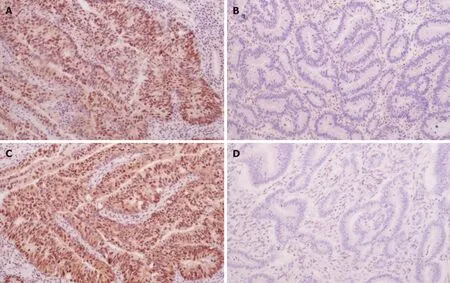
Figure1 Expression of MLH1 and MSH2 in colorectal cancer tissues detected by immunohistochemistry (DAB staining,×100).A:MLH1 positivity; B:MLH1 negativity; C:MSH2 positivity; D:MSH2 negativity.
MSI
Microsatellite analysis was performed in 133 CRC patients (71 stage II and 62 stage III)using GenScan,and 105 had MSS (78.95%) and 28 (21.05%) had MSI,including 18(13.53%) cases of MSI-H and 10 (7.52%) cases of MSI-L.All patients were detected by immunohistochemical analysis,which confirmed that patients with MSI-H carcinomas included 17 (94.4%) who were MLH1/MSH2-negative and one (5.6%)who wasMLH1/MSH2-positive (P< 0.001).Patients with MSS and MSI-L who hadMLH1orMSH2positivity were classified asMLH1/MSH2-positive.According to our data analysis,MSI-H was more frequent in patients aged > 50 years (P= 0.048),in the right colon (P< 0.001),in tumors with poor differentiation (P= 0.028),and in tumors with mucin (P= 0.037).The clinicopathological features of MSI are consistent with previous immunohistochemical results ofMLH1/MSH2expression.The analysis of MSI is shown in Figure3.
DISCUSSION
Defective DNA MMR is most often associated with loss ofMLH1andMSH2gene functions and results in MSI mutation.MLH1andMSH2promoter hypermethylation is an important DNA MMR pathway in sporadic proximal CRC[21].Abnormal methylation,causing alteration ofMLH1/MSH2,can form transcriptional target genes for silencing[22].Several studies[23-25]have revealed thatMLH1andMSH2play a critical role,and mutations in either gene result in complete loss of function,with tumor formation preferentially in the proximal colon,and this hypermethylation is significantly more common in sporadic than in hereditary MSI-positive tumors.Thibodeauet al[26,27]reported that in most colorectal carcinomas with MSI-H phenotype,91% of cases are confirmed asMLH1-negative.In another study,Hermanet al[28]reported that hypermethylation ofMLH1in sporadic CRC with MSI-H was as high as 84%.By contrast,Vasenet al[29]demonstrated thatMSH2mutation is associated with a higher risk of developing cancer thanMLH1mutation.MSH2generally forms a connection withMSH6orMSH3,so it can control most of the hypermethylation that occurs with different bound proteins.AndMLH1forms withPMS2,and goes through one pathway to combine with other proteins[30,31].Gene mutation is the key reason for the decrease ofMLH1andMSH2expression and both of them are the most dominant parts of the MMR system,so detection for those twogenes are important for discovering the pathogenesis of sporadic CRC.
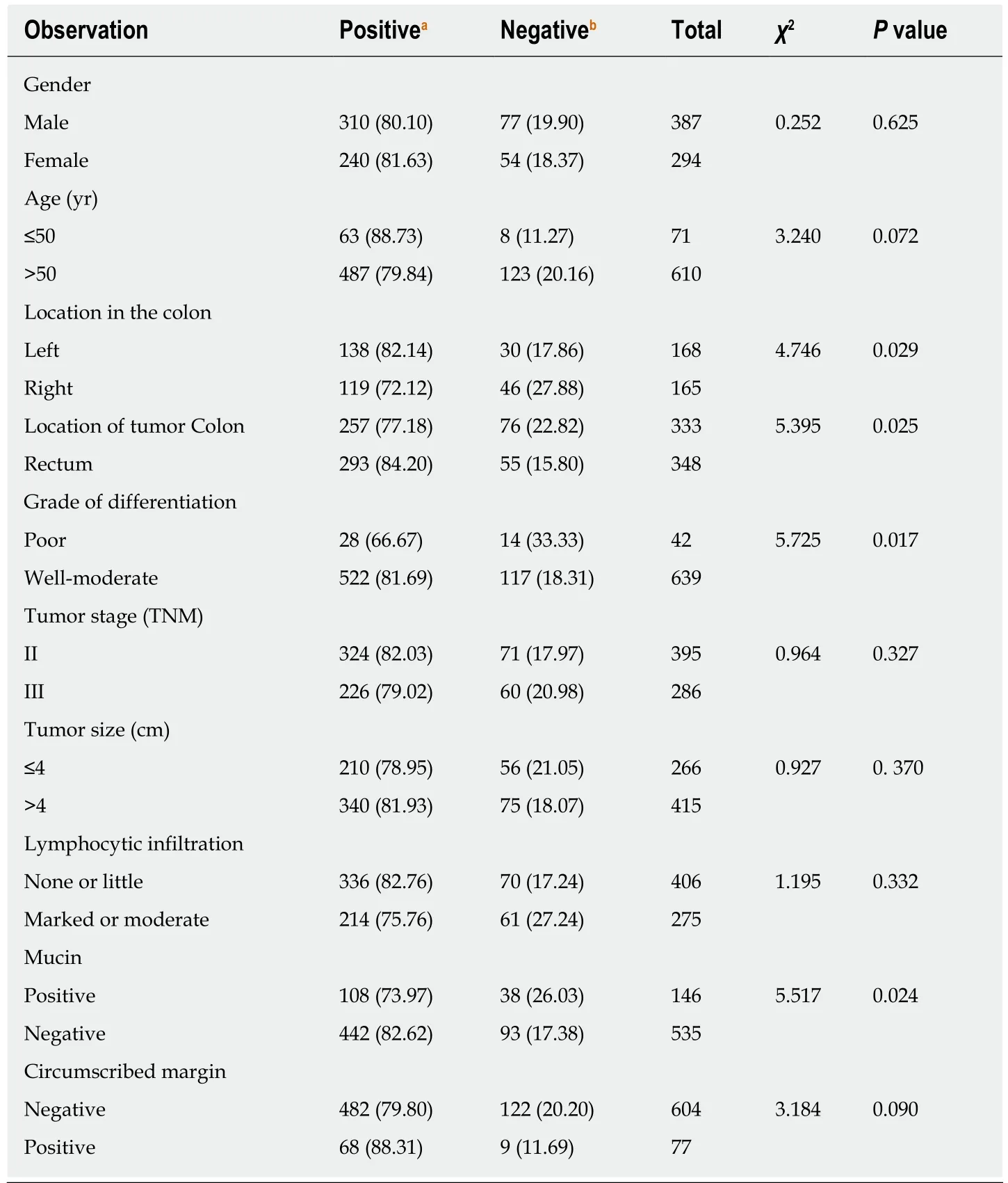
Table2 Relationship between clinicopathological features and MutL homolog1/MutS homolog2 expression in 681 patients,n (%)
In this study,we used immunohistochemistry to detectMLH1andMSH2expression in all postoperative patients because it is more accurate,rapid,and costeffective for assessment of MMR status than other methods[32,33].MLH1/MSH2status can verify MMR expression.In contrast,using GenScan to analyze MSI status can more accurately and directly demonstrate the difference between normal and abnormal loci,although the test is expensive and not easy to analyze in each postoperative patient[34].So,immunohistochemical staining for MMR is now performed as part of routine processing in the department of pathology in almost all hospitals after surgery[35-37].In our study,immunohistochemical analysis for MMR found thatMLH1/MSH2negativity was more frequent in the right colon,in tumors with poor differentiation,and in tumors with mucin production.With regard to MSI detection,MSI-H occurred mostly in patients aged > 50 years,in the right colon,in tumors with poor differentiation,and in tumors with mucin production.We confirmed that these two clinical assays have more consistency and accuracy than other detections.
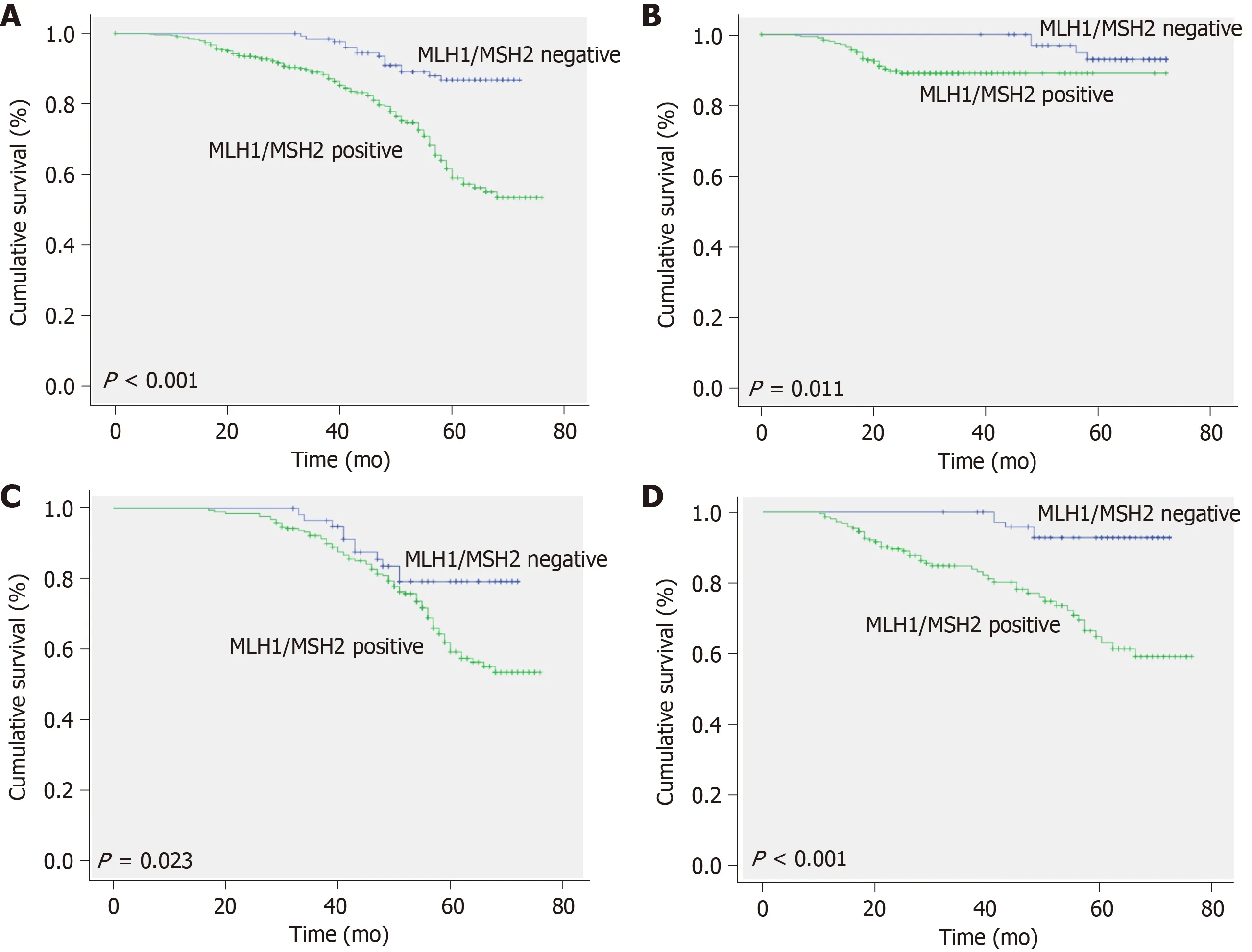
Figure2 Kaplan-Meier survival curves for patients with colorectal cancer,stratified according to expression of MLH1/MSH2.A:Overall survival of total colorectal cancer (CRC) patients classified as MLH1/MSH2-positive and MLH1/MSH2-negative subgroups; B:Overall survival of stage II CRC patients classified as MLH1/MSH2-positive and MLH1/MSH2-negative subgroups; C:Overall survival of stage III CRC patients classified as MLH1/MSH2-positive and MLH1/MSH2-negative subgroups; D:Overall survival of all adjuvant chemotherapy patients classified as MLH1/MSH2-positive and MLH1/MSH2-negative subgroups.
The result of our study clearly showed thatMLH1/MSH2-negative tumors were mostly in the right colon with poor differentiation and contained mucin.This conclusion is consistent with most of the published research.Benattiet al[38]reported that MSI-H occurred in 256 (20.3%) of 1263 patients,more frequently in tumors which were in the less advanced stage,right sided,poorly differentiated with mucinous phenotype,and had infiltrative growth than MSS[39].Numerous studies have a similar conclusion that most MSI-H tumors are mucinous adenocarcinoma,located in the right colon,and poorly differentiated (P< 0.05)[40,41].Though in the early studies,CRC was divided into colonic and rectal by anatomical site,they had some differences in specific treatments even if they have been treated as the same disease.And in recent years,with the deepening of understanding of this disease and the increase of evidence-based medical proof,CRC in different parts was considered to have distinct clinical pathological expression and prognosis.So the location of tumor is instructive for prognosis and treatment[42,43].More and more studies now tend to divide CRC into right site and left site according to the colonic splenic flexure[44].It is based on the right site originating from the midgut of embryo,while the left site originates from the hindgut of embryo.Anatomically,the right site was supplied by the superior mesenteric artery,and the left site was supplied by the inferior mesenteric artery[45,46].Therefore,the right-sided tumor has poorer differentiation,worse pathological stage,and earlier metastasis than the left-sided tumor[47].Since the rectal blood is supplied from the internal iliac artery,and rectal cancer is different from the colonic cancer in clinical treatment,in our study we divided CRC into the right colon,left colon,and rectum parts.As found in other studies[48,49],the frequency of mismatch repair deficiency (dMMR) in right-sided tumors with poor differentiation was significantly higher than that in tumors of the left colon and rectum in our study,indicating that MSI is mainly involved in the development of right colon cancer.Thus,the occurrence of CRC in different parts is not the same at the genetic level.
In terms of prognostic value of MMR phenotype in CRC,abundant studies[50-52]have acquired positive results.Gryfeet al[53]analyzed 607 patients with CRC and divided them into TNM stages I-IV.All patients with MSI-H had a survival advantage compared with MSS patients (P< 0.001,HR 0.42,95%CI 0.27-0.67).The incidences of distant metastasis (P= 0.02,HR 0.49) and regional LN metastasis (P< 0.001,HR 0.33)in MSI-H patients were lower than those in patients who had MSS tumors.Rothet al[54]investigated the effect of MMR at different stages.In 1404 patients with stage II or III CRC,the prognostic advantage conferred by MSI was more evident in stage II than in stage III (P= 0.04).In a large meta-analysis[55]of 12782 patients,there was a clear correlation between MSI-H tumors and improved OS.The data demonstrated that the OS of patients with MSI-H was significantly better than that of MSI-L and MSSpatients (P< 0.001),with an overall odds ratio of 0.6 (95%CI 0.53-0.69).In addition,disease-free survival (DFS) was also significantly different (P< 0.001).In our study,patients withMLH1/MSH2-negative stage II-III CRC had a better clinical outcome than those withMLH1/MSH2-positive CRC.Moreover,in multivariate analysis,the survival advantage forMLH1/MSH2-negative patients was independent from several other clinical and pathological parameters.These conclusions are consistent withalmost all related studies because of the difference in histology,anatomy,and accompanying degree of differentiation,histopathology in different locations of the intestine.
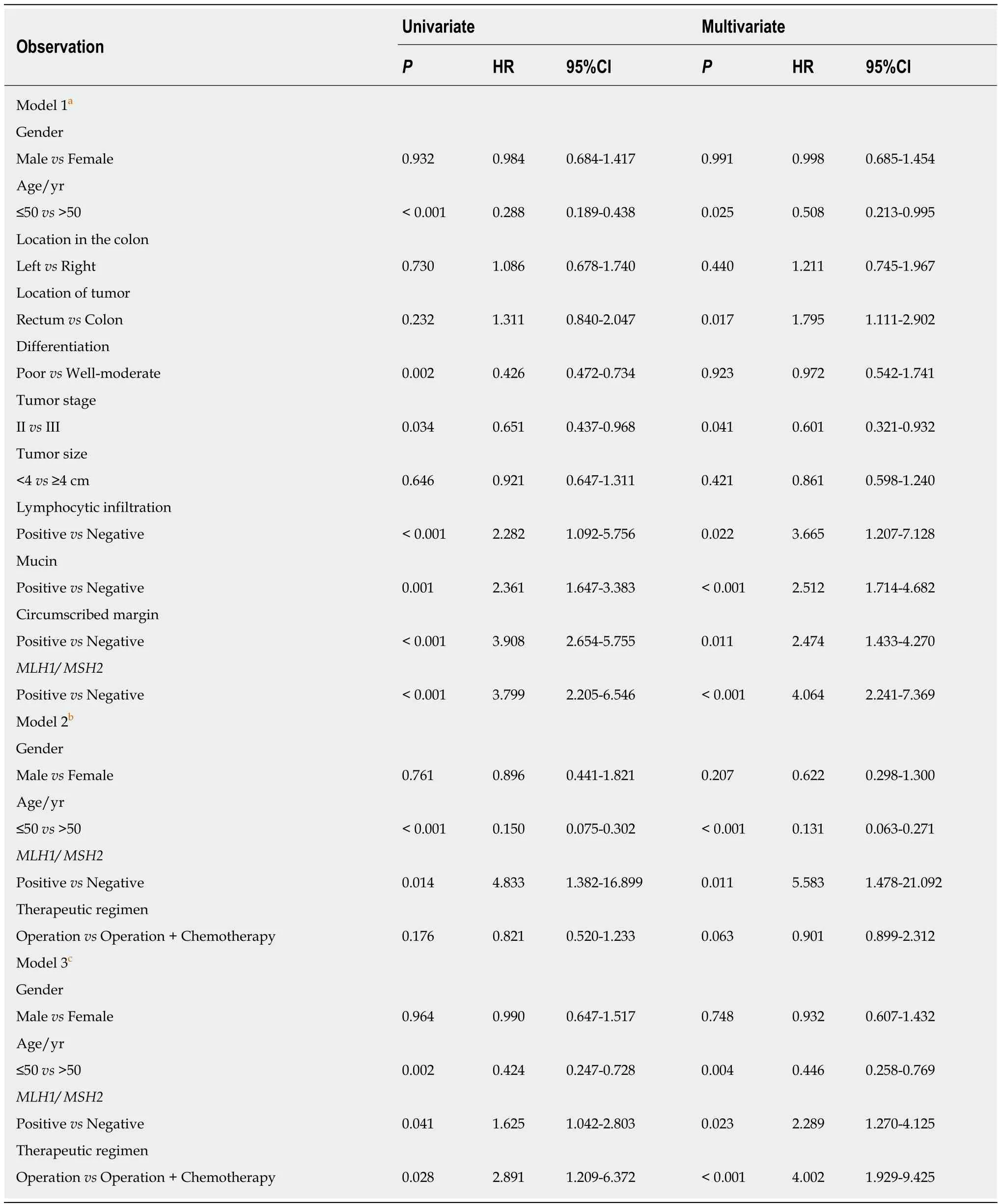
Table3 Prognostic factors for survival in univariate and multivariate analyses
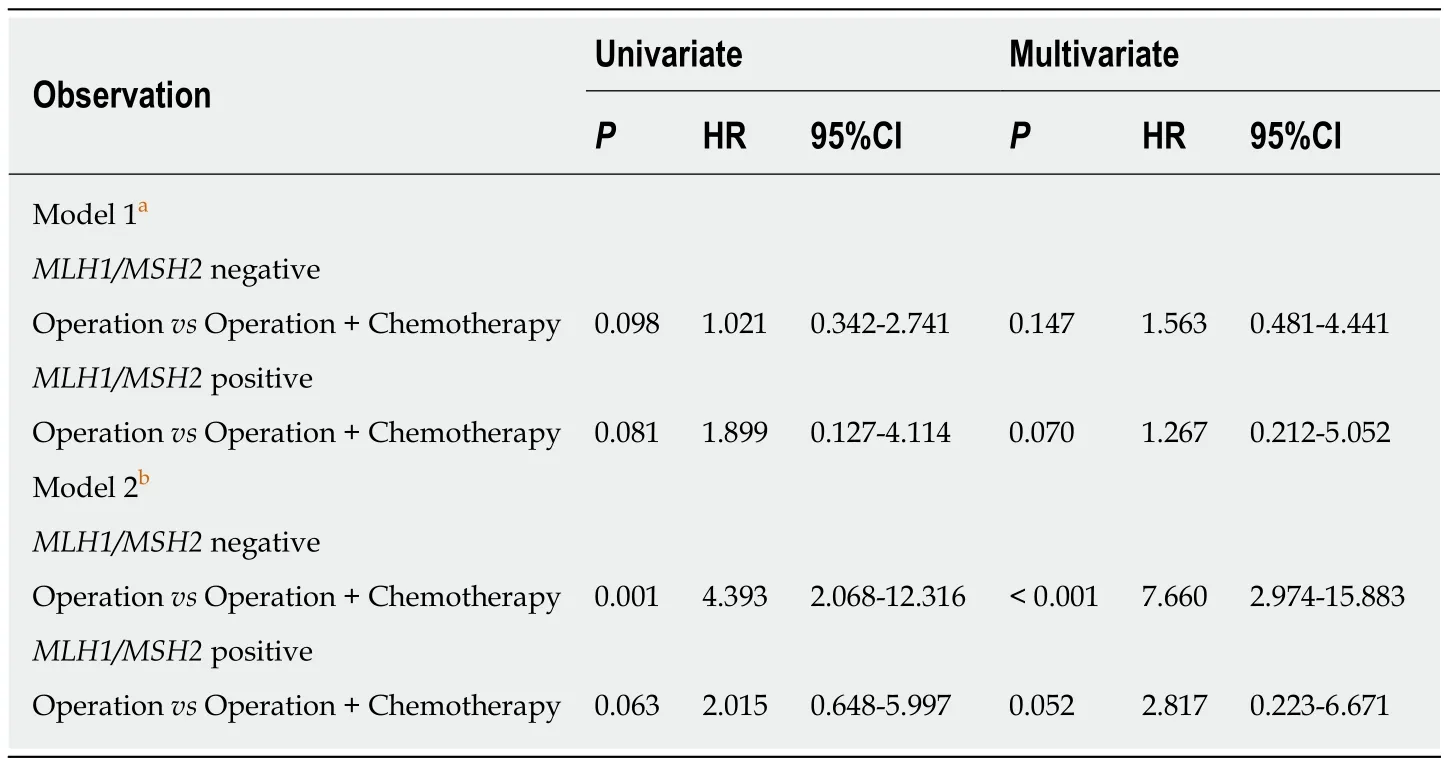
Table4 Predictive factors for survival in univariate and multivariate analyses
Adjuvant chemotherapy is considered as the gold standard for treatment of patients with stage III CRC.However,there was controversy in a previous study as to whether patients with stage II CRC should take adjuvant chemotherapy after surgery.The 2013 NCCN guidelines[18]suggested to test for MMR proteins for all patients < 50 years of age or with stage II disease.Stage II MSI-H patients may have a good prognosis and do not benefit from 5-FU adjuvant therapy[56].Our study followed this guideline and respected patients’ wishes to formulate their treatment schedule.We found that stage II-III CRC had different results on adjuvant chemotherapy.In stage II CRC,OS was not strongly associated with adjuvant chemotherapy either inMLH1/MSH2-negative (multivariateP= 0.147,HR = 1.563,95%CI:0.481-4.441) orMLH1/MSH2-positive patients (multivariateP= 0.070,HR = 1.267,95%CI:0.212-5.052).However,in stage III CRC,we found that OS of theMLH1/MSH2-negative patients (multivariateP< 0.001,HR = 7.660,95%CI:2.974-15.883) was more strongly associated with adjuvant chemotherapy than that ofMLH1/MSH2-positive patients (multivariateP= 0.052,HR = 2.817,95%CI:0.223-6.671).This is similar to the study by Elsalehet al[57]which revealed that patients with stage III colon cancer with MSI had improved survival when treated with 5-FU-based chemotherapy(fluorouracil and levamisole) compared with no chemotherapy (HR = 0.07,95%CI:0.01-0.53vsHR = 1.06,95%CI:0.65-1.72).In the study of Sinicropeet al[58](NCCTG N0147) of 2720 stage III colon cancer patients for 5-year disease-free survival,it was found that MMR proficient (pMMR) patients had statistically shorter survival time than dMMR patients (P< 0.0001).But in patients with stage III CRC,the predictive function of MMR for adjuvant chemotherapy remains controversial because there are some studies supporting the opposite conclusion.Ribicet al[59]showed no benefit from adjuvant chemotherapy in stage II-III patients with MSI.Sargentet al[60]enrolled 457 stage II-III CRC patients divided into 5-FU-based therapy (n= 229) and postsurgical treatment groups (n= 228).Patients with MSI who received 5-FU had no improvement in DFS (P= 0.85,HR 1.10; 95%CI 0.42-2.91) compared with the postsurgical treatment group.Joveret al[61]confirmed that patients with dMMR colon cancer do not benefit from adjuvant 5-FU/leucovorin.Many studies that analyzed the relationship between MMR and prognosis enrolled patients with all TNM stages of disease rather than stageII- III patients.Some studies included fewer patients with MSI-H,which made it difficult or inaccurate to evaluate 5-FU-based chemotherapy regimens.Those are the main reasons leading to the inconsistency of the final results.We encouraged to make risk-stratification for patients in evaluating the effect of adjuvant chemotherapy for patients with dMMR colon cancer.
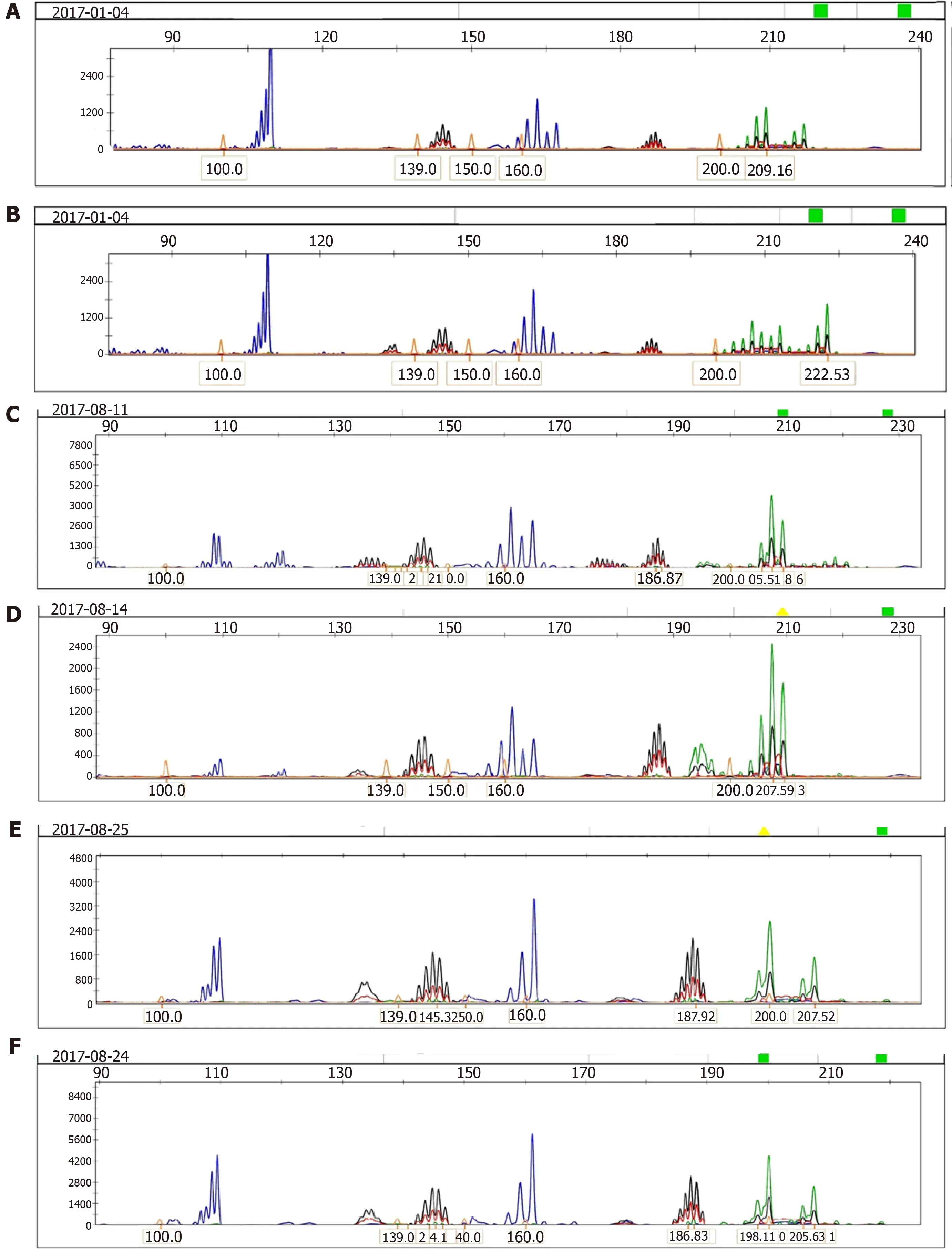
Figure3 Analysis of microsatellite instability in colorectal cancer tissues and corresponding normal mucosa using the five markers of the international workshop of Bethesda and fluorescence-based multiplex polymerase chain reaction.Fragment pattern of a high-frequency microsatellite instability tumor showing instability at all five loci examined is shown.A:Low microsatellite instability (MSI-L) of tumor tissue; B:MSI-L of corresponding normal tissue; C:High microsatellite instability (MSI-H) of tumor tissue; D:MSI-H of corresponding normal tissue; E:Microsatellite stability (MSS) of tumor tissue; F:MSS of corresponding normal tissue.
Tumor immunotherapy has greatly advanced in recent years,especially,PD-1/PDL1 blocking therapy has shown encouraging effects and become a major pillar of immunotherapy.Patients with metastatic colorectal cancer also gain new hopes from immunotherapy,particularly in dMMR patients for whom immune checkpoint inhibitor antibody can achieve a 40% objective response rate (ORR) and up to 78%clinical benefit rate[62].The clinical trial KEYNOTE-028[63]enrolled advanced colorectal adenocarcinoma patients who failed standard therapy and had PD-L1 expression in ≥1% of cells in tumor nests.The primary endpoints were ORR,safety,and tolerability.Patients received pembrolizumab 10 mg/kg every 2 wk and lasted more than 2 years or until confirmed unacceptable toxicity or progression.The results showed that in the 156 advanced CRC patients,23 were PD-L1 positive,1 gained complete remission(CR),and 1 experienced a partial response (ORR,4%; 95%CI:0-22%) who was confirmed as MSI-H.This trial revealed that PD-L1 expression cannot screen out the dominant population of anti-PD-1 immunotherapy.Based on the KEYNOTE-028 and several other clinical trial results,Leet al[64]designed a single-arm,phase II clinical study (NCT01876511) which aimed to explore the predictive value of MMR status in the treatment of PD-1.The investigator divided 41 patients with progressive metastatic carcinoma with or without dMMR into three groups,namely,11 with dMMR CRC,21 with pMMR CRC,and 9 with dMMR non-CRC (4 ampullary/cholangiocarcinoma,2 endometrial cancer,2 small intestine cancer,and 1 gastric cancer) and every patient was administered pembrolizumab 10 mg per kilogram of body weight every 14 d.The primary endpoints were ORR at 20 wk and progressionfree survival (PFS).The results showed that the ORR for the three groups were 40%,0,and 71%,respectively,and the PFS rates were 78%,11%,and 67%,respectively,at 20 wk.Interestingly,the investigators used whole-exome sequencing to check somatic mutations and found that the mutation rate was higher in dMMR than in pMMR (P=0.007).Moreover,the study demonstrated that high somatic mutation loads were associated with prolonged PMS (P= 0.02) and dMMR patients received clinical benefit of immune checkpoint blockade with pembrolizumab.
The data of these innovative single-arm clinical studies[65-67]led to accelerated approval of the United States FDA for pembrolizumab in patients with dMMR/MSIH solid tumors (including CRC) who failed previous treatment in May 2017[68].Subsequently,pembrolizumab and nivolumab were recommended for second-line or later treatment of dMMR/MSI-H CRC in the 2017 NCCN guidelines[69],but were approved in refractory or metastatic CRC,so there is just little evidence or trial using the new immunotherapeutic drugs,alone or in combination with chemotherapy,in patients with stage III CRC.Although checkpoint inhibitors (CPIs) have achieved remarkable efficacy in CRC,they still face the dilemma of limited effective drugs and limited access to dMMR patients.The proportion of dMMR with advanced CRC was less than 5%,so how to make the majority of pMMR patients benefit from immunotherapy in the future is an important problem to be solved.We may need to face the following questions:(A) How to screen out patients who may be effective in immunotherapy in a large pMMR population and then expand the indications for CPIs? (B) How to more optimize the treatment strategy to overcome the primary resistance of pMMR population to immunotherapy and improve the response to immunotherapy? (C) How to control the immune-related events more effectively? (D)Is there any other new targeted immune checkpoints?
In conclusion,this study demonstrated the function ofMLH1/MSH2expression in sporadic CRC,including its effect on prognostic and predictive factors,but we have few details about the correlation between MMR and tumorigenesis,loss of heterozygosity,and immunotherapy[70,71].Further studies should clarify the cause and mechanisms of hypermethylation inMLH1/MSH2and antineoplastic immunity.Data from the current study may be helpful to understand the roles ofMLH1/MSH2in the development and progression of CRC.They also suggest a new therapeutic strategy by regulating MMR expression to slow down the malignant progression of CRC and to improve the prognosis of CRC patients.
ARTICLE HIGHLIGHTS
Research background
Colorectal cancer (CRC) can arise through three distinct mutational pathways:Microsatellite instability,chromosomal instability,and CpG island methylator phenotype.We tested the hypothesis that CRC arising from the microsatellite-instability pathway through MLH1/MSH2-negative expression can lead a more favorable overall survival (OS) than MLH1/MSH2-positive patients.We also made an in-depth observation of the correlation between adjuvant chemotherapy and MLH1/MSH2 expression in different stages of CRC.
Research motivation
A larger sample size with a longer follow-up period was included to assess the effect of MLH1/MSH2 status on the prediction and prognosis of stage II-III CRC and its association with adjuvant chemotherapy.It is important for clinical doctors to choose optimal treatment regimen,especially adjuvant chemotherapy,for patients.
Research objectives
To evaluate the predictive and prognostic effects of MLH1/MSH2 status in stage II-III CRC patients and its significance in guiding adjuvant chemotherapy.
Research methods
We analyzed 681 postoperative patients with CRC with a median follow-up period of 56 mo(range,8.0-72.0 mo) between January 2013 and December 2016.The main outcome data includedMLH1/MSH2-positive rate,MLH1/MSH2-negative rate,and long-term follow-up outcomes.
Research results
The outcomes showed that 550 patients wereMLH1/MSH2-positive and 131 wereMLH1/MSH2-negative.MLH1/MSH2-positive tumors were significantly more frequent in the colon than in the rectum,and with poor differentiation and less mucin production (P< 0.05).Patients did not differ in terms of age,gender,tumor size,tumor stage,lymphocytic infiltration,or circumscribed margin.MLH1/MSH2-negative patients had a more favorable OS thanMLH1/MSH2-positive patients (P< 0.001).In both stages II and III,MLH1/MSH2expression was a strong prognostic factor in all patients [P< 0.001,hazard ratio (HR) = 4.064,95% confidence interval (CI):2.241-7.369].Adjuvant chemotherapy had a greater correlation with survival advantage inMLH1/MSH2-negative patients with stage III disease (P< 0.001,HR = 7.660,95%CI:2.974-15.883).Patients with stage II disease orMLH1/MSH2-positive stage III patients did not benefit from adjuvant chemotherapy.
Research conclusions
MLH1/MSH2phenotype constitutes a pathologically and clinically distinct subtype of sporadic CRC.MLH1/MSH2is an independent prognostic and predictive factor for outcome of stage II-III CRC.
Research perspectives
Our study demonstrated mismatch repair (MMR) is an important prognostic and predictive biomarker for stage II-III CRC,but we did not enroll the patients who used any PD-1/PD-L1 blocking therapy and had no data for survival improvement with different MMR statuses.In addition to detecting MMR status and tumor mutational burden,are there any indicators that are more sensitive to immunotherapy? The currently found immunologic drugs are only effective for high microsatellite instability (MSI-H)/dMMR population,but are ineffective for most patients with microsatellite stability (MSS).Would any new effective immune drugs be found for MSS patients? Is it possible to subdivide MSI-H (through the number of mutation sites) for enriching the dominant population in future? These issues will be the focal points and difficulty in our later research.
ACKNOWLEDGEMENTS
We thank Ya-Nan Shi,Xiao Chen,Xin-Yang Wu,Jing-Yu Shen,and Zong-Lan Zhang for data collection and Youping Deng’s team for excellent collaboration.We also thank Chris K Deng for his extensively revising the article.
杂志排行
World Journal of Gastrointestinal Oncology的其它文章
- New era for pancreatic endoscopic ultrasound:From imaging to molecular pathology of pancreatic cancer
- Endothelial cells in colorectal cancer
- Non-coding RNA in drug resistance of gastric cancer
- Calponin 3 promotes invasion and drug resistance of colon cancer cells
- Eight key long non-coding RNAs predict hepatitis virus positive hepatocellular carcinoma as prognostic targets
- Toll-like receptor 9 polymorphisms and Helicobacter pylori influence gene expression and risk of gastric carcinogenesis in the Brazilian population.
