腰椎间盘髓核组织HSPA8表达及对退变程度影响
2019-09-10王华聪陈伯华金沧海相宏飞刘勇
王华聪 陈伯华 金沧海 相宏飞 刘勇
[摘要]目的探讨70 kDa热休克蛋白8(HSPA8)在人腰椎间盘髓核组织中的表达及其与人椎间盘退变程度的关系。方法收集人腰椎间盘髓核组织50例,其中15例为正常对照组,35例为退变腰椎间盘髓核组织(实验组,根据术前MRI分为突出组12例,脱出组13例,游离组10例)。分别应用Western`-blot、苏木精`-伊红(HE)染色、免疫组织化学方法检测各组髓核组织中HSPA8的表达。结果3种方法检测结果均显示,对照组、突出组、脱出组、游离组腰椎间盘髓核组织中HSPA8表达依次降低,差异有显著性(F=153.52~1 662.43,P<0.001)。结论HSPA8在人椎间盘髓核组织中稳定表达,并且与椎间盘退变的程度相关。
[关键词]HSC70热休克蛋白质类;椎间盘退行性变;印迹法,蛋白质;苏木精;免疫组织化学
EXPRESSION OF HSPA8 IN THE NUCLEUS PULPOSUS TISSUE OF LUMBAR INTERVERTEBRAL DISCS AND ITS EFFECT ON DEGENERATION OF INTERVERTEBRAL DISCS WANG Huacong, CHEN Bohua, JIN Canghai, XIANG Hongfei, LIU Yong(Department of Spinal Surgery, The Afflicated Hospital of Qingdao University, Qingdao 266100, China)
[ABSTRACT]ObjectiveTo investigate the expression of the 70 kDa heat shock protein member 8 (HSPA8) in the nucleus pulposus tissue of human lumbar intervertebral discs (LIDs) and its correlation with the degree of degeneration of human intervertebral discs. MethodsThe nucleus pulposus tissue of human LIDs was collected from 50 cases, including 15 cases as control group and 35 cases with degenerated LIDs as experimental group. According to preoperative MRI results, the experimental group was subdivided into protrusion group (n=12), prolapse group (n=13), and sequestration group (n=10). The expression of HSPA8 in the nucleus pulposus tissue of each group was determined by Western blot, hematoxylin`-eosin staining, and immunohistochemistry. ResultsAll the results obtained from the three determination methods showed decreased expression of HSPA8 in the nucleus pulposus tissue of each group, with significant differences observed between groups (highest expression seen with the control group, successively followed by the protrusion group, the prolapse group, and the sequestration group) (F=153.52-1 662.43, all P<0.001). ConclusionHSPA8 is expressed stably in the nucleus pulposus tissue of human intervertebral discs, and its expression is correlated with the degree of degeneration of intervertebral discs.
[KEY WORDS]HSC70 heat`-shock proteins; intervertebral disc degeneration; blotting, Western; hematoxylin; immunohistochemistry
椎間盘退变是骨科常见的疾病,据统计,全球有70%~90%的人会受到其困扰[1`-3],腰腿痛是导致人类生活质量下降、劳动力丧失的最常见原因[4`-6]。腰椎管狭窄症、腰椎间盘突出症是导致腰腿痛最常见的疾病[7]。腰椎间盘退变与遗传和环境因素有关,其特征之一就是椎间盘髓核蛋白多糖和水分含量丧失[8`-10]。热休克蛋白是一种能够在细胞核和细胞质之间穿梭的多功能蛋白[11],参与多种细胞功能的调控,是分子伴侣系统重要的一部分[12`-13]。70 kDa热休克蛋白8(HSPA8)是HSC70家族中高度保守的成员,具有多效性,对细胞的存活起着至关重要的作用,在应激反应中发挥着不可或缺的作用[14`-16]。LOEFFLER等[17]研究显示,脑脊液中的HSPA8随着年龄的增长而降低,并且与氧化应激有关。YE等[18]首次报道HSPA8在退变椎间盘纤维环细胞中表达量降低。但是,HSPA8在人腰椎间盘髓核组织是否表达及其与椎间盘退变程度的关系尚不清楚。本文研究联合应用Western`-blot、苏木精`-伊红(HE)染色、免疫组织化学方法,对人腰椎间盘髓核组织中HSPA8表达及其与腰椎间盘退变程度的关系进行研究,为腰腿痛病人的治疗提供理论依据。
1资料与方法
1.1一般资料
收集2017年6月—2018年4月在青岛大学附属医院行腰椎手术病人的椎间盘髓核组织50例,其中15例取自椎体爆裂骨折的青少年髓核组织(对照组),35例取自腰椎间盘退变病人髓核组织(实验组)。根据SPENGLER[19]的方法及术前MRI将实验组分为突出组(12例)、脱出组(13例)、游离组(10例)。纳入标准:①入院后经MRI检查明确诊断;②符合腰椎间盘突出症的诊断标准;③病人或其家属自愿签署知情同意书。排除标准:①哺乳期、妊娠期妇女;②合并感染病人;③合并肿瘤病人;④合并后纵韧带骨化、黄韧带骨化病人;⑤合并免疫性疾病或其他全身疾病的病人。本文研究经青岛大学附属医院伦理学委员会审批通过。
1.2主要材料
聚氰基丙烯酸正丁酯(BCA)购自北京索莱宝科技有限公司;十二烷基磺酸钠`-聚丙烯酰胺凝胶电泳 (SDS`-PAGE)蛋白上样缓冲液(5×)购自上海碧云天生物技术有限公司;HSPA8一抗购自Abcam公司;内参一抗、二抗购自武汉伊莱瑞特生物科技股份有限公司;免疫组化试剂盒购自上海麦约尔生物科技有限公司。
1.3研究方法
1.3.1Western`-blot方法检测HSPA8表达剪取0.1 g椎间盘组织置于研磨碗中,再加入500 μL的RIPA裂解液提取蛋白。4 ℃条件下12 000 r/min离心10 min,吸取上层透亮液体加入上样缓冲液中。BCA法测定蛋白浓度。从-20 ℃冰箱中取出样品,按照4∶1比例加入buffer液体,95 ℃下煮沸15 min,从中取出100 μL混合液置入新的EP管中。SDS`-PAGE电泳:先恒压80 V电泳30 min,然后120 V电泳60 min。用湿转法将电泳完毕的胶转膜至聚偏二氟乙烯膜(PVDF膜)上,以恒电流300 mA转膜90 min,室温下封闭1 h。然后加入HSPA8单克隆一抗(含有10 μL鼠抗HSPA8一抗及10 mL一抗稀释液)、兔抗人GAPDH单克隆一抗(5 μL GAPDH一抗+20 μL含1.5 g/L脱脂奶粉的TBST溶液),置4 ℃条件下孵育过夜。第2天回收抗体,加入羊抗兔二抗(按照1∶4 000比例与二抗稀释液混合),孵育1 h后,用显影液显影成像,获得条带图。以GAPDH作为内参,应用Image J软件测量各条带的灰度值,以其表示HSPA8表达量。实验重复3次。
1.3.2HE染色检测HSPA8表达将人腰椎间盘髓核组织用生理盐水冲洗干净,放入40 g/L多聚甲醛溶液中固定,使组织蛋白变性凝固;流水冲洗组织,乙醇脱水,二甲苯脱乙醇,置于装满石蜡的金属盒中包埋,切片,二甲苯脱蜡处理,乙醇水化,HE染色,并滴加适量的中性树胶封片,晾干后显微镜下观察HSPA8染色情况。采用Image`-Pro Plus 6.0图像分析软件对HE染色结果进行分析,以染色面积表示HSPA8表达量。实验重复3次。
1.3.3免疫组织化学法检测HSPA8表达制备人腰椎间盘髓核组织4 μm厚切片,烘烤2 h,脱蜡、水化,将体积分数0.03甲醇`-过氧化氢加到切片上,高压抗原修复,放入PBS溶液中冲洗。切片上滴加50 μL一抗(一抗HSPA8/HSC71稀释比例为1∶50,GAPDH稀释比例为1∶50),4 ℃过夜,次日取出,待切片恢复至室温后,放入PBS洗3次,每次5 min。滴加50 μL二抗,DAB染液显色5~10 s,苏木精复染,梯度乙醇脱水,二甲苯透明,封片,显微镜下照相。采用Image`-Pro Plus 6.0软件计算平均光密度值,以其表示髓核组织中HSPA8的表达。实验重复3次。
1.4统计学分析
应用SPSS 3.0软件进行统计学分析,结果以±s形式表示,组间比较采用方差分析、ANOVA分析。P<0.05为差异有统计学意义。
2结果
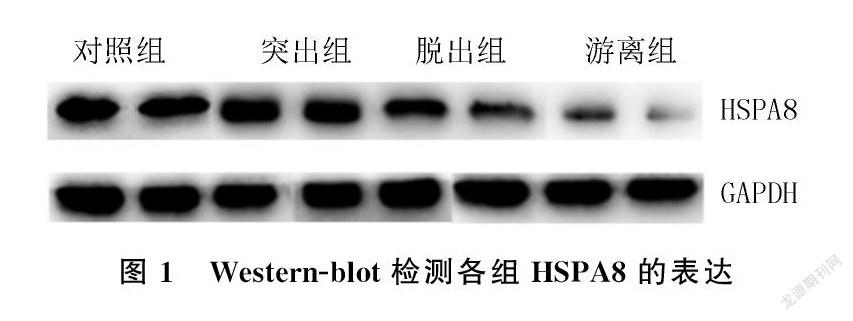
2.1各组Western`-blot结果比较
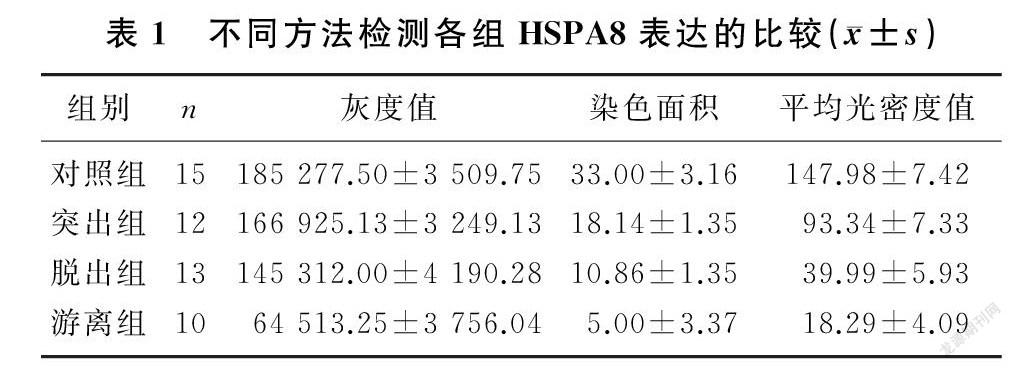
对照组、突出组、脱出组、游离组HSPA8表达(灰度值)依次降低,差异有显著性(F=1 662.43,P<0.001)。见图1、表1。
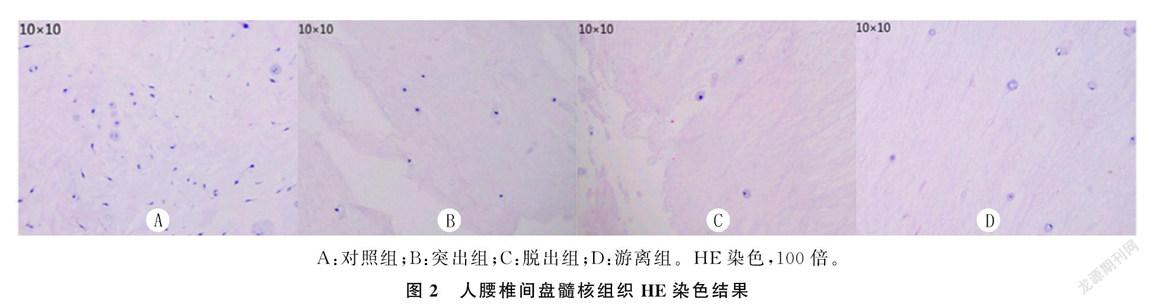
2.2各组HE染色结果比较
对照组:髓核组织结构疏松,髓核细胞形态较规则,基质成分完整,细胞数量较多,呈短梭形,分布均匀,轮廓清晰,胞膜完整,胞浆均匀,细胞核呈蓝紫色;退变组:髓核组织水分较少,髓核细胞形态不规则,基质成分减少,细胞数目较少,呈长梭形,分布不均匀,轮廓不清,胞内聚集大量溶酶体,呈空泡样(图2)。各组染色面积比较差异有显著性(F=153.52,P<0.001)。见表1。
2期王华聪,等. 腰椎间盘髓核组织HSPA8表达及对退变程度影响135
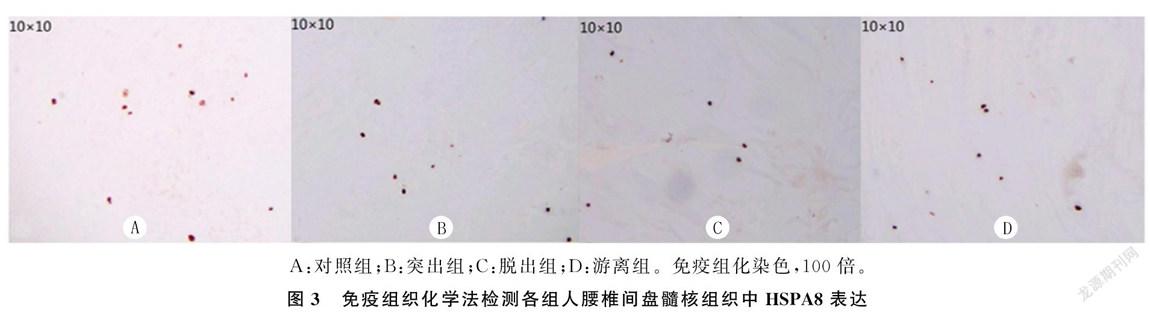
2.3各组免疫组织化学法检测结果比较
对照组、突出組、脱出组、游离组中HSPA8的表达(平均光密度值)依次降低, 差异有统计学意义(F=633.28,P<0.001)。见图3、表1。
3讨论
腰椎退行性病变是脊柱外科常见的疾病,是随着年龄的增长自然发生的病理过程,其症状严重影响病人的日常生活,明显降低病人的生活质量[20`-22]。本文研究以腰椎间盘退变为切入点,联合应用HE染色、Western`-blot方法、免疫组织化学法检测不同退变程度的人腰椎间盘髓核组织中HSPA8的表达情况。研究结果显示,对照组腰椎间盘髓核组织中HSPA8强阳性表达,突出组中HSPA8阳性表达,脱出组中HSPA8呈弱阳性表达,游离组中HSPA8弱阳性表达或表达极少,提示HSPA8表达与人类腰椎间盘退变相关,其表达随着腰椎间盘退变的程度的加重而减少。HSPA8具有多效性,对细胞的存活起着重要的作用,尤其是参与新合成蛋白的折叠[15,23]。GOLDFA等[24]研究结果表明,HSPA8和HSP70对调节小鼠上皮钠通道起着相反的作用。与HSP70相比,HSPA8在促进膜蛋白折叠、脂质体介导的钙离子多肽易位方面更具有优势。影响HSPA8表达的因素有很多,如有机氯、镉等[15,25`-26]。YE等[18]研究结果显示,纤维环组织中HSPA8表达量减少,HSPA8等一些差异蛋白可以作为诊断腰椎间盘退变的生物学标志物。但是,目前尚无HSPA8是否在腰椎间盘髓核组织中表达及其表达量是否与腰椎间盘退变程度有关的报道。本文研究立足于腰椎间盘纤维环与HSPA8有密切联系,探讨HSPA8在腰椎间盘髓核组织中的表达及其与椎间盘退变的关系。HSPA8是新发现的与腰椎间盘退变髓核组织有关的蛋白,其特点在于表达恒定。MAKINO等[27]研究了抑制c`-Fos/AP`-1对椎间盘退行性变及相关疼痛的影响,结果显示,金属蛋白酶通过增加c`-Fos的表达来降解椎间盘中的细胞外基质,抑制细胞外组分的表达,选择性c`-Fos/AP`-1抑制剂T`-5224似乎适合作为一种治疗人类IVD变性的药物,对腰椎间盘退变的治疗起到指导性作用。姜棚菲等[28]研究显示,新型金属蛋白酶(ADAMTS`-7)、TNF`-α在腰椎间盘退行性疾病中表达量随着退变程度的加重而增加。NIKOLAOU等[29]研究显示,椎间盘退变程度的增加与人椎间盘细胞中核蛋白和胞质蛋白糖基化修饰的增加有关。JOHNSON等[30]研究结果显示,在人类退变的椎间盘组织中,水通道蛋白1和水通道蛋白5的反应较灵敏,低氧诱导因子`-1α在髓核细胞中可以维持水通道蛋白的表达,在退行性变过程中,低氧诱导因子`-1α活性降低可能抑制髓核细胞中水通道蛋白的水平,损害其对细胞外渗透压变化的反应能力。WANG等[31]研究结果显示,椎间盘退变病人髓核细胞中的miR`-154表达升高,抑制miR`-154可增加Ⅱ型胶原纤维和聚集蛋白聚糖表达,金属蛋白酶`-13和蛋白聚糖酶`-1表达下调;而过表达的miR`-154可以逆转髓核细胞的这种效应,推动椎间盘退变的进展,miR`-154可能为椎间盘退变的治疗靶点。但是HSPA8与椎间盘退变关系的研究不多,本文主要研究了HSPA8与腰椎间盘髓核组织退变程度的关系,结果显示HSPA8表达量随着椎间盘退变程度的加重而降低,可能为腰椎间盘退变疾病术前诊断及治疗提供新的方向。
综上所述,HSPA8在髓核组织中表达,且随着腰椎间盘退变程度的加重表达逐渐降低,HSPA8可能成为腰椎间盘退变严重程度的生物学检测指标之一。但是,在腰椎间盘退变过程中,究竟是通过哪条信号通路调节HSPA8的表达,有待进一步研究。
[参考文献]
[1]CHEN Fengfeng, HUAN Liu, YANG Yang. Growth and differentiation factor`-5 contributes to the structural and functio`-nal maintenance of the intervertebral disc[J]. Cellular Physio`-logy and Biochemistry: International Journal of Experimental Cellular Physiology, Biochemistry, and Pharmacology, 2015,35(1):1`-16.
[2]BLANQUER S B, GRIJPMA D W, POOT A A. Delivery systems for the treatment of degenerated intervertebral discs[J]. Advanced Drug Delivery Reviews, 2015,84:172`-187.
[3]马春杰,罗珊珊,李建设. 低氧诱导因子在腰椎间盘突出中的病理机制研究[J]. 安徽医药, 2017,21(7):1306`-1310.
[4]MASAYUKI O, WATANABE K, HIRANO T, et al. Predicting factors at skeletal maturity for curve progression and low back pain in adult patients treated nonoperatively for adolescent idiopathic scoliosis with thoracolumbar/lumbar curves: a mean 25`-year follow`-up[J]. Spine, 2018,43(23): E1403`-E1411.
[5]MATSUI H, KANAMORI M, Ishihara H, et al. Familial predisposition for lumbar degenerative disc disease. A case`-control study[J]. Spine, 1998,23(9):1029`-1034.
[6]ALA`-KOKKO L. Genetic risk factors for lumbar disc disease[J]. Annals of Medicine, 2002,34(1):42`-476.
[7]SCHISTAD E I, BJORLAND S, ROE C, et al. Five`-year development of lumbar disc degeneration-a prospective study[J]. Skeletal Radiol, 2018. doi:10.1007/s00256`-018`-3062`-x.
[8]SONG Y Q, KARASUGI T, CHEUNG K M, et al. Lumbar disc degeneration is linked to a carbohydrate sulfotransferase 3 variant[J]. The Journal of Clinical Investigation, 2013,123(11):4909`-4917.
[9]張文庆,姜杰,庞晓东,等. 腰椎间盘退变的机制[J]. 中国矫形外科杂志, 2008,16(21):1635`-1636.
[10]YALTIRIK C K, TIMIRCI`-KAHRAMAN O, GULEC`-YILMAZ S, et al. The Evaluation of proteoglycan levels and the possible role of gene (c.6423T>C) variant in patients with lumbar disc degeneration disease[J]. In Vivo (Athens, Greece), 2019,33(2):413`-417.
[11]LAMIAN V, SMALL G M, FELDHERR C M. Evidence for the existence of a novel mechanism for the nuclear import of Hsc70[J]. Experimental Cell Research, 1996,228(1):84`-91.
[12]MARCINIAK J, LOSSDORFER S, KIRSCHNECK C, et al. Heat shock protein 70 dampens the inflammatory response of human PDL cells to mechanical loading in vitro[J]. J Periodontal Res, 2019. doi:10.1111/jre.12648.
[13]BI Xiaowen, XU Miao, LI Jinfei, et al. Heat shock protein 27 inhibits HMGB1 translocation by regulating CBP acetyltransferase activity and ubiquitination[J]. Mol Immunol, 2019,108:45`-55.
[14]HUNT C, MORIMOTO R I. Conserved features of eukaryotic hsp70 genes revealed by comparison with the nucleotide sequence of human hsp70[J]. Proceedings of the National Academy of Sciences of the United States of America, 1985,82(19):6455`-6459.
[15]LIU T, DANIELS C K, CAO S. Comprehensive review on the HSC70 functions, interactions with related molecules and involvement in clinical diseases and therapeutic potential[J]. Pharmacology &Therapeutics, 2012,136(3):354`-374.
[16]KITYK R, KOPP J, SINNING I, et al. Structure and dyna`-mics of the ATP`-bound open conformation of Hsp70 chaperones[J]. Molecular Cell, 2012,48(6):863`-874.
[17]LOEFFLER D A, KIAVER A C, COFFEY M P, et al. Age`-related decrease in heat shock 70 kDa protein 8 in cerebrospinal fluid Is associated with increased oxidative stress[J]. Front Aging Neurosci, 2016,8:178.
[18]YE Dongping, LIANG Weiguo, DAI Libing, et al. Comparative and quantitative proteomic analysis of normal and degene`-rated human annulus fibrosus cells[J]. Clinical and Experimental Pharmacology &;Physiology, 2015,42(5):530`-536.
[19]SPENGLER D M. Lumbar discectomy. Results with limited disc excision and selective foraminotomy[J]. Spine, 1982,7(6):604`-607.
[20]VADALA G, RUSSO F, Di MARTINO A, et al. Intervertebral disc regeneration: from the degenerative cascade to mole`-cular therapy and tissue engineering[J]. Journal of Tissue Engineering and Regenerative Medicine, 2015,9(6):679`-690.
[21]GORNET M G, PEACOCK J, CIAUDE J, et al. Magnetic resonance spectroscopy (MRS) can identify painful lumbar discs and may facilitate improved clinical outcomes of lumbar surgeries for discogenic pain[J]. Eur Spine J, 2019. doi:10.1007/s00586`-018`-05873`-3.
[22]ZEHRA U, CHEUNG J P Y, BOW C, et al. Multidimensio`-nal vertebral endplate defects are associated with disc degeneration, Modic changes, facet joint abnormalities and pain[J]. J Orthop Res, 2018. doi:10.1002/jor.24195.
[23]STRICHERR F, MACRI C, RUFF M, et al. HSPA8/HSC70 chaperone protein: structure, function, and chemical targeting[J]. Autophagy, 2013,9(12):1937`-1954.
[24]GOLDFA R B, KASHLAN O B, WATKINS J N, et al. Diffe`-rential effects of Hsc70 and Hsp70 on the intracellular trafficking and functional expression of epithelial sodium channels[J]. Proceedings of the National Academy of Sciences of the United States of America, 2006,103(15):5817`-5822.
[25]DEANE E E, WOO N Y. Impact of heavy metals and organochlorines on Hsp70 and Hsc70 gene expression in black sea bream fibroblasts[J]. Aquatic Toxicology (Amsterdam, Ne`-therlands), 2006,79(1):9`-15.
[26]GERACI F, PINSINO A, TURTURICI G, et al. Nickel, lead, and cadmium induce differential cellular responses in sea urchin embryos by activating the synthesis of different HSP70s[J]. Biochemical and Biophysical Research Communications, 2004,322(3):873`-877.
[27]MAKINO H, SEKI S, YAHARA Y, et al. A selective inhibition of c`-Fos/activator protein`-1 as a potential therapeutic target for intervertebral disc degeneration and associated pain[J]. Scientific Reports, 2017,7(1):16983.
[28]姜棚菲,馬张稳,张民泽,等. 腰椎间盘退行性改变患者ADAMTS`-7表达及其机制研究[J]. 实用医院临床杂志, 2017,14(6):101`-104.
[29]NIKOLAOU G, ZIBIS A H, FYIIOS A H, et al. Detection of O`-Linked`-N`-Acetylglucosamine modification and its associated enzymes in human degenerated intervertebral discs[J]. Asian Spine J, 2017,11(6):863`-869.
[30]JOHNSON Z I, GOGATE S S, DAY R, et al. Aquaporin 1 and 5 expression decreases during human intervertebral disc degeneration: novel HIF`-1`-mediated regulation of aquaporins in NP cells[J]. Oncotarget, 2015,6(14):11945`-11958.
[31]WANG Jingjie, LIU Xiaoyan, SUN Bing, et al. Upregulated miR`-154 promotes ECM degradation in intervertebral disc degeneration[J]. J Cell Biochem, 2019. doi:10.1002/jcb.2847.
