甘肃省白菜死棵病病原菌鉴定及其融合群检测技术
2019-08-27王朵谢学文柴阿丽石延霞李宝聚
王朵,谢学文,柴阿丽,石延霞,李宝聚
甘肃省白菜死棵病病原菌鉴定及其融合群检测技术
王朵,谢学文,柴阿丽,石延霞,李宝聚
(中国农业科学院蔬菜花卉研究所,北京 100081)
【】明确引起甘肃省白菜死棵病的病原菌种类,并建立该病原菌的分子生物学检测方法,为白菜死棵病的预防及有效防治提供参考依据。【】对采自甘肃省的白菜死棵病菌分离、纯化培养,观察菌落形态,初步明确其病原菌为立枯丝核菌(),为准确鉴定,从分离菌株中选取部分菌株利用通用引物rDNA-ITS和TEF-1(translation elongation factor,1-alpha)进行PCR扩增、测序,采用Mega 7.0中最大似然法(maximum likelihood,ML)构建系统发育树,同时用特异性引物对F-RS/R-RS进行融合群鉴定;采用叶面喷雾和茎基部灌根法对20个白菜品种进行致病力测定。根据立枯丝核菌rDNA-ITS基因保守区域序列,运用Primer 5.0设计1对特异性检测引物,建立实时荧光定量PCR(real-time fluorescent quantitative PCR,real-time PCR)检测该病原菌的方法。利用立枯丝核菌融合群(anastomosis groups,AGs)AG3—AG11、双核丝核菌AG-A(binulceate Rhizoctonia AG-A)、水稻纹枯病菌(AG-1-IA)、镰孢菌(spp.)、腐霉菌(spp.)等常见病原菌的菌丝DNA进行常规PCR和real-time PCR检测,对特异性、灵敏度和可重复性进行评价。利用该体系对人工接种不同天数和田间采集的白菜病株及其根际土壤进行检测。【】共分离得到86株立枯丝核菌,经常规PCR特异性检测,结果表明大多数菌株属于立枯丝核菌融合群AG-2-1(68/86),少数为AG-1-IB(18/86);选取50个代表菌株进行ITS和TEF-1基因测序和系统发育分析,AG-2-1和AG-1-IB分别与相应融合群参考序列聚在一支,且亲缘关系置信度为100%;致病力测定结果表明,两个融合群代表菌株对20个白菜品种的茎基部均可致病,AG-2-1对金娃娃、小皇宝叶部无致病力,且AG-1-IB的致病力稍强于AG-2-1。设计的引物特异性强,常规PCR检测结果表明仅白菜死棵病菌有扩增条带。Real-time PCR检测结果表明引物F-RS/R-RS特异性强,对白菜上立枯丝核菌有唯一的产物吸收峰,对其余对照菌株均未检测到荧光信号。常规PCR检测的灵敏度为8.41×105copies/μL,real-time PCR的灵敏度可达到8.41×103copies/μL,提高了2个数量级。以携带目的基因片段的重组质粒为标准品,构建的real-time PCR标准曲线循环阈值与模板浓度呈良好的线性关系,熔解曲线的吸收峰单一,相关系数为0.9983,扩增效率为91%。该方法能成功检测出田间采集的白菜病株及根际土壤中的立枯丝核菌,对人工接种后不同发病天数的盆栽白菜进行real-time PCR检测,结果表明接种后第5天植株及土壤带菌量最大。【】通过对白菜死棵病菌进行分子生物学鉴定和致病力测定,明确了甘肃省白菜死棵病是由立枯丝核菌AG-2-1和AG-1-IB两个融合群侵染所致。建立的real-time PCR测定立枯丝核菌的方法特异性强、灵敏度高,可实现植株和土壤的带菌检测及监测,可为病害早期预警及管理提供技术支持。
白菜;立枯丝核菌融合群;实时荧光定量PCR;检测
0 引言
【研究意义】由立枯丝核菌()引起的十字花科蔬菜死棵病是一种重要的土传病害。甘肃武威、兰州等地,地势较低,气候冷凉,主要种植喜凉蔬菜如白菜,近年来由于土壤连作以及栽培管理不当等原因,死棵现象频发。由于立枯丝核菌寄主范围广和融合群(anastomosis groups,AGs)间关系的复杂性,在病害早期难以判断和准确量化[1-2],因此明确引起白菜死棵病的主要致病群体,以及建立一种能够准确、灵敏及快速检测田间样品的方法,对于该病害的有效防控具有重要意义。【前人研究进展】基于菌丝融合反应,立枯丝核菌可划分为多个AGs;根据致病力、寄主范围、形态变异和遗传特征等,一些AGs又可划分为多个融合亚群[3-6]。十字花科蔬菜丝核菌病害主要由AG-2-1引起,导致植株茎部溢缩,直至腐烂、倒伏,最终死亡,造成严重的经济损失[7-9];AG-1-IB常引起莴苣叶腐病,同时也对白菜等十字花科蔬菜有致病力[10-11]。前人对融合群的鉴定多基于菌丝融合反应,但2008年Sharon等[12]利用菌丝融合鉴定融合群时,发现该过程易受环境因素等影响,重复性较差,结果不可靠。目前有关融合群的分子生物学检测技术主要有基于SYBR Green I和TaqMan探针的实时荧光定量PCR(real-time PCR)技术,这两种方法均能特异性检测融合群,准确度好、灵敏度高,常用来对引起某种寄主病害的一个或多个融合群进行检测,如利用TaqMan探针检测马铃薯黑痣病菌AG-3[13]以及定量检测其菌核[14]、水稻纹枯病菌AG-1-IA[15]及毒素[16]和油菜根腐病菌AG-2-1[17],以及基于SYBR Green和TaqMan检测小麦根部病菌[18-19]等。【本研究切入点】目前尚未对甘肃省白菜死棵病原菌进行系统报道,笔者项目组于2015—2018年在甘肃地区采集疑似白菜死棵病株作为样本,进行病原菌融合群鉴定和检测技术的建立。【拟解决的关键问题】采用ITS和TEF通用引物对白菜死棵病菌进行PCR扩增和测序分析,明确其具体种类,并建立real-time PCR检测体系,实现田间植株和土壤样品的准确检测,为病害预警和提早防控提供技术支持。
1 材料与方法
试验于2018年在中国农业科学院蔬菜花卉研究所完成。
1.1 病样的采集与病原菌分离、保存
笔者课题组于2015—2018年共采集白菜死棵病样100份,品种为韩国介实金悦白菜。采用水琼脂分离法[20]进行病原菌快速分离鉴定,挑取菌丝边缘于PDA培养基上,25℃黑暗培养3 d后,记录菌落形态学特征,对病原菌进行显微形态观测、拍照,之后转移至灭菌麦粒上,待菌丝长满麦粒后保存于-20℃冰箱[21],共保存立枯丝核菌86株。
1.2 主要试剂及仪器
植物基因组DNA提取试剂盒(天根生化科技有限公司)、胶回收试剂盒(TaKaRa Mini BEST Agarose GEL DNA Extraction Kit)、质粒提取试剂盒(TIANprep Mini Plasmid Kit,天根生化科技有限公司)、pMDTM19-T Vector Cloning Kit(TaKaRa公司)、感受态细胞(Trans 5Competent Cells,TaKaRa公司)、DNase/RNase free water(天根生化科技有限公司)、FastDNA Spin Kit for Soil(MP Bio-medical,USA)、实时荧光定量PCR仪(ABI 7500美国ABI公司)。
1.3 病原菌的鉴定
1.3.1 病原菌的分子生物学鉴定 采用试剂盒法提取病原菌基因组DNA。采用ITS通用引物对[22]及立枯丝核菌翻译延伸因子引物对RSF/RSR[23]对各菌株进行PCR扩增(表1),反应结束后,取5 µL PCR产物用1.0%的琼脂糖凝胶电泳进行检测。将含有目的条带的PCR产物交由北京博迈德基因技术有限公司分别用各基因相对应的PCR扩增引物进行双向测序,将得到双向序列拼接,并将拼接后得到的序列提交到 GenBank(表2),然后用MEGA 7.0软件根据各菌株ITS和TEF-1基因序列进行系统发育进化分析。采用最大似然法(maximum likelihood,ML)构建系统发育进化树。同时采用Matsumoto[24]根据立枯丝核菌不同融合群的核糖体DNA序列差异设计的两对特异性引物(表1)对分离菌株进行PCR特异性扩增,用于鉴定分离菌株。
1.3.2 分离菌株对不同白菜品种致病力测定 为比较不同融合群菌株的致病力差异,选取两个融合群代表菌株WWC18081640(AG-2-1)和WWC18081618(AG-1-IB),在日光温室中对20个白菜品种进行致病力测定。采用叶部喷雾和茎基部灌根法,用无菌纱布过滤菌悬液,将菌丝打碎并用无菌水稀释到一定浓度接种[25]。对照处理喷无菌水,每处理10株苗,重复3次,将各种处理的幼苗置于25℃恒温保湿柜中,每天观察并记录发病情况,参照分级标准[26],记录接种植株的病级数,并计算病情指数(DI)。
1.4 Real-time PCR引物的设计与合成
根据NCBI数据库中立枯丝核菌ITS基因的保守序列,运用Primer 5.0软件设计real-time PCR检测特异性引物,并通过NCBI中的Primer-BLAST进行比对,保证所设计引物的特异性。上游引物F-RS:CAATACAAAGGCAATAGG;下游引物R-RS:GTCCTCAGCGATAGATAA。引物由博迈德基因技术有限公司进行合成。

表1 本研究中所用融合群鉴定的引物序列及其来源

表2 本研究中所用菌株及序列
1.5 质粒标准品的制备
以保存的DNA为模板进行PCR扩增,总反应体系为50 μL。PCR产物回收后参考丁天波等[27]的方法对回收产物进行连接、转化,并筛选出阳性质粒,计算质粒浓度拷贝数。
1.6 反应条件的优化及标准曲线的建立
质粒标准品按10倍梯度进行稀释,获得终浓度为8.41×109—8.41×103copies/μL的7个质粒样品作为模板进行检测。反应体系为20 μL:SYBR Green PremixPlus 10 μL,Rox Reference 0.4 μL,上下游引物各0.3 μL,模板1 μL,ddH2O补足。
1.7 特异性检测
用优化后的real-time PCR反应条件分别对其他病原菌,如茄镰孢(Fusarium solani)、尖镰孢(F. oxysporum)、芸薹生链格孢(Alternaria brassicicola)、胶孢炭疽菌(Colletotrichum gloeosporioides)、瓜果腐霉(Pythium aphanidermatum)、终极腐霉(P. ultimum)、水稻纹枯病菌(R. solani AG-1-IA)、马铃薯黑痣病菌(R. solani AG-3)、小麦纹枯病菌(R. cerealis)等进行特异性检测。
1.8 Real-time PCR的灵敏度检测
将质粒标准品按照10倍梯度稀释成8.41×109—8.41×103copies/μL的7个样品,并以此作为模板分别进行常规PCR和real-time PCR检测,比较这两种方法的检测灵敏度。
1.9 白菜病株及根际土壤中立枯丝核菌的检测
分别对1.3.2中接种后1、5、10、15、20 d的白菜病株及根际土壤取样,提取植物组织和土壤总DNA,通过real-time PCR检测立枯丝核菌(每个样品取3次重复,检测结果取平均值),根据扩增曲线、熔解曲线和Ct值确定其带菌情况。同时对田间采集的17份疑似白菜病株及0—10 cm深度根际土壤[19]进行总DNA提取,通过real-time PCR进行检测,根据扩增曲线、熔解曲线和Ct值确定其带菌情况。
2 结果
2.1 序列测定和系统发育分析
采用ITS1/ITS4和RSF/RSR对50个分离菌株进行PCR扩增,均获得了约650 bp的序列(图1),将测序结果提交至Genbank,获得登录号(表2)。将获得的序列进行比较并构建系统发育树,可看出32条序列与立枯丝核菌AG-2-1的参考序列聚在同一分支,且亲缘关系置信度为100%,另外18条与AG-1-IB参考序列聚在另一分支,而外群菌株则以较远的亲缘关系处在系统发育树的外围(图2)。序列测定和系统发育分析结果表明,共有32株菌(占64%)属于立枯丝核菌AG-2-1,另外18株(36%)为AG-1-IB。

M:DNA分子量标准(5 000 bp)DNA Marker (5 000 bp);1—24:分离菌株经ITS和TEF-1α通用引物扩增后的产物电泳条带 The amplification of R. solani fragment by universal primers of ITS and TEF-1α;25:阴性对照 Negative control
2.2 特异性引物的扩增结果
用F-Common/R-AG-2-1和F-Common/R-AG-1-IB特异性引物分别对代表菌株进行PCR扩增,可分别产生约300和250 bp的扩增片段(图3)。特异性扩增结果表明,该引物对可准确鉴定分离菌株为立枯丝核菌AG-2-1和AG-1-IB。
2.3 分离菌株对白菜品种的致病力测定
致病力试验结果表明,接种后两融合群菌株在20个白菜品种上均出现死棵症状,融合群AG-1-IB致病力稍强于AG-2-1,对叶片及茎基部均有致病力,而AG-2-1对金娃娃及小皇宝两个品种的白菜叶片不具有致病力。从病情指数来看,两融合群在不同白菜品种上存在差异(表3),茎基部病情指数≥50的品种,AG-2-1仅有3个,而AG-1-IB占9个,且均高于AG-2-1。
2.4 rDNA-ITS基因片段的克隆与鉴定
以白菜死棵病组织提取总DNA为模板,运用F-RS/R-RS引物进行常规PCR扩增,其中实验室保存的立枯丝核菌DNA作为阳性对照,对照菌基因组DNA及健康白菜样品DNA作阴性对照,ddH2O作为空白对照。PCR扩增产物经1%的琼脂糖凝胶电泳,结果表明(图4),白菜病株样品出现特异单一条带,位置在389 bp,同阳性对照条带大小一致,而阴性对照和空白对照则无特异性条带出现。将目的条带切胶回收,经连接克隆载体至博迈德基因技术有限公司进行测序,将测序结果在NCBI数据库中进行BLASTn比对,结果证实所获得的序列为ITS基因片段。

表3 WWC18081640和WWC18081618对20个白菜品种的致病力测定(病情指数)

图2 基于rDNA-ITS 和TEF-1α核酸序列的系统发育树

M:DNA分子量标准(5 000 bp) DNA marker (5 000 bp);1—8:F-Common/R-AG-2-1特异性扩增 Amplification by primer F-Common/R-AG-2-1 for AG-2-1;9—17:立枯丝核菌融合群AG-3—AG-11菌株R. solani AG3 to AG11;18:阴性对照negative control;19—23:F-Common/R-AG-1-IB特异性扩增 Amplification by primer F-Common/R-AG-1-IB for AG-1-IB

M:DNA分子量标准(5 000 bp) DNA marker (5 000 bp);1:阳性对照Positive control;2:感染立枯丝核菌的白菜样品R. solani-infected cabbage sample;3—21:立枯丝核菌融合群AG-3—AG-11菌株、水稻纹枯病菌、双核丝核菌AG-A、小麦纹枯病菌、茄镰孢、尖镰孢、胶孢炭疽菌、瓜果腐霉、终极腐霉、芸薹生链格孢菌和致病疫霉R. solani AG-3toAG-11, R. solani AG-1-IA, binulceate Rhizoctonia AG-A, R. cerealis, F. solani, F. oxysporum, C. gloeosporioides, P. aphanidermatum, P. ultimum, A. brassicicola, P. infestans;22:健康白菜样品 Healthy cabbage sample;23:空白对照 Blank control
2.5 标准曲线的建立
以10倍梯度进行稀释的7个质粒样品作为模板,进行real-time PCR,结果表明随着阳性质粒模板浓度逐渐降低,Ct值出现递增趋势,并且在质粒标准品浓度8.41×104—8.41×108copies/μL范围内,Ct值同质粒标准品浓度之间线性关系良好,标准曲线斜率为-3.546,决定系数2=0.9983,扩增效率为91%,直线方程为=-3.546+ 44.241(代表循环阈值即 Ct 值,代表质粒初始浓度)(图5)。该标准曲线将用于立枯丝核菌含量的测定。

图5 Real-time PCR 标准曲线图
2.6 质粒标准品灵敏度检测
Real-time PCR和常规PCR的灵敏度检测比对结果表明,real-time PCR的立枯丝核菌检测方法能够最低检测出8.41×103copies/μL的样品(图6),而常规PCR仅能够检测到8.41×105copies/μL的样品(图7),real-time PCR检测立枯丝核菌的灵敏度是常规PCR的100倍。
2.7 白菜病株及根际土壤样品检测
利用real-time PCR检测技术对人工接种的白菜茎基部和根际土壤进行检测,结果表明人工接种白菜植株和土壤后第5天,立枯丝核菌在白菜植株体内和根际土壤中积累量最大(表4)。对田间采集病株及根际土壤进行real-time PCR检测,除了阴性对照和空白对照,其他样品均可检测到相应的荧光信号值(图8)。此外,对上述样品进行常规PCR检测,发现大多数样品均在相同位置检测到特异性单一条带(图9),但有1个样品(第16孔)未出现明显条带,因此常规PCR的检出率低于real- time PCR。

1—7:8.41×109—8.41×103 copies/μL质粒标准品8.41×109-8.41×103 copies/μL standard plasmids;8:空白对照 Blank control
表4 Real-time PCR检测人工接种植株及根际土壤的带菌量

M:DNA分子量标准(5 000 bp) DNA marker (5 000 bp);1—7:8.41×109—8.41×103 copies/μL质粒标准品8.41×109-8.41×103 copies/μL standard plasmids;8:空白对照 Blank control
3 讨论
白菜在我国种植范围广,市场消费较大。甘肃以干燥、冷凉的气候特点,将白菜作为重要的蔬菜品种,其种植面积逐年增加。近年来由于栽培面积扩大,死棵问题不断出现。本研究发现引起白菜死棵的病原菌是立枯丝核菌。立枯丝核菌是一类在自然界中广泛存在的土壤习居真菌,能引起多种植物病害[28-29],根据菌丝融合、培养形态、致病力以及DNA同源性等,被划分为14个不同的AG以及一些融合亚群[30-32]。传统分类学基于形态特征、菌丝融合、同工酶、可溶性蛋白等生物学和生化的方法对融合群进行划分,耗时长、信息量少[33],目前越来越多学者通过分子学方法进行融合群的分类,其中核糖体DNA(rDNA)内部转录间隔区(ITS)区域已被证明能够准确划分融合群及亚群,但该区域在进行融合群群内各菌株之间遗传差异分析时缺乏多态性[34]。TEF-1是蛋白翻译及延伸中的重要蛋白质,该基因具有丰富的信息量,比ITS的多态性更高[35],其序列在不同物种中高度相似,已被用于多种病原菌的鉴定及系统发育研究,如Geiser等[36]利用此基因分析镰孢菌系统发育关系及Anees等[37]用来研究木霉属真菌。本研究结合PCR反应、基于ITS和TEF-1基因序列的系统发育分析及苗期品种抗病性鉴定,明确了从死棵白菜上分离到的病原菌主要为立枯丝核菌AG-2-1和AG-1-IB,此后还会继续进行病株采集和鉴定。
近年来对立枯丝核菌检测的相关报道也逐渐增多,目前使用较为普遍的方法是real-time PCR,其中基于TaqMan探针的real-time PCR越来越多的应用于AG检测,如Budge等[38]检测到土壤中11个融合群,Woodhall等[39]检测到引起马铃薯黑痣的4个融合群,但其检测成本均较高,而基于荧光染料SYBR Green的检测方法能够很好地弥补上述缺陷,并已广泛应用于多种融合群的检测和研究中。本研究选取ITS的保守区域设计出可同时检测AG-2-1和AG-1-IB的real-time PCR特异性引物F-RS/R-RS,通过常规PCR扩增发现AG-2-1和AG-1-IB样品均在目的大小处条带单一,并且不能扩增出其他AG和对照菌,说明F-RS/R-RS特异性较强,能够满足后续验证试验;real-time PCR检测结果发现F-RS/R-RS扩增曲线良好,有特异性熔解峰,结合本研究摸索的反应体系和条件,能够最大程度保证反应过程中目标基因的有效扩增,增强反应特异性。本研究选用纯化的重组质粒DNA作为标准品,与普通试剂盒提取的DNA相比,具有稳定易保存等特点[40]。以立枯丝核菌重组质粒DNA作为标准品构建的标准曲线,质粒标准品中立枯丝核菌拷贝数的对数值同Ct值之间呈现良好的线性关系,相关系数达0.9983,扩增效率为91%,完全满足样品中立枯丝核菌含量测定的需求。通过比较常规PCR和 real-time PCR的检测灵敏度,发现real-time PCR的检测灵敏度是常规PCR的100倍,因此对田间低丰度样品的准确有效检测成为现实。本研究对人工接种的白菜、根际土壤以及田间采集的死棵病株和土壤进行检测,通过分析扩增曲线、熔解曲线并与阳性、阴性和空白对照进行对比,均检测到了病原菌,证实了该方法的检测适用性。对人工接种的不同天数白菜植株和根际土壤样品进行检测,发现带菌量均在接种后第5天最大,此时植株的发病程度也最大,但土壤带菌量均低于茎基部带菌量,很可能是在土壤DNA提取过程中造成了部分损失[41],需要进一步筛选出更好的提取土壤DNA方法。
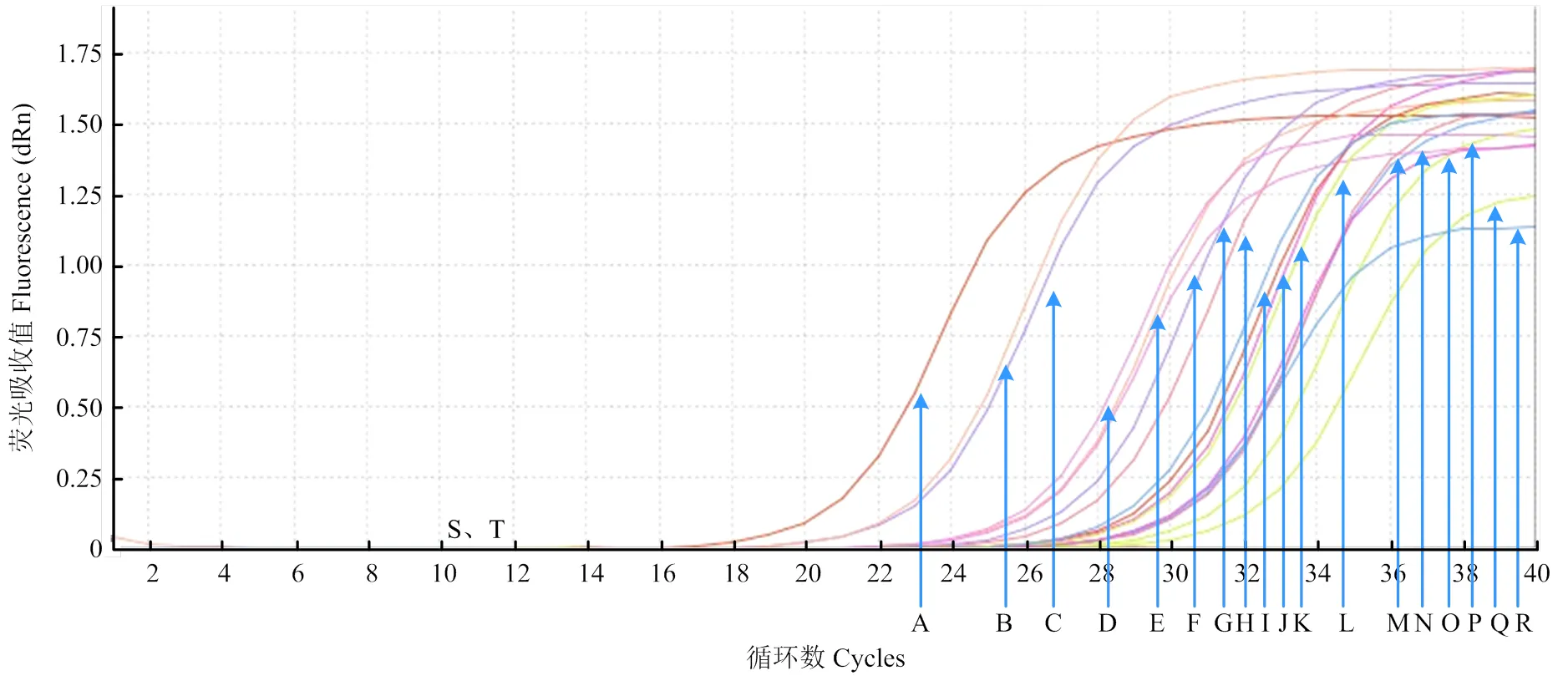
A:阳性对照 Positive control;B—Q:田间样品 Samples in field;S:阴性对照negative control;T:空白对照blank control
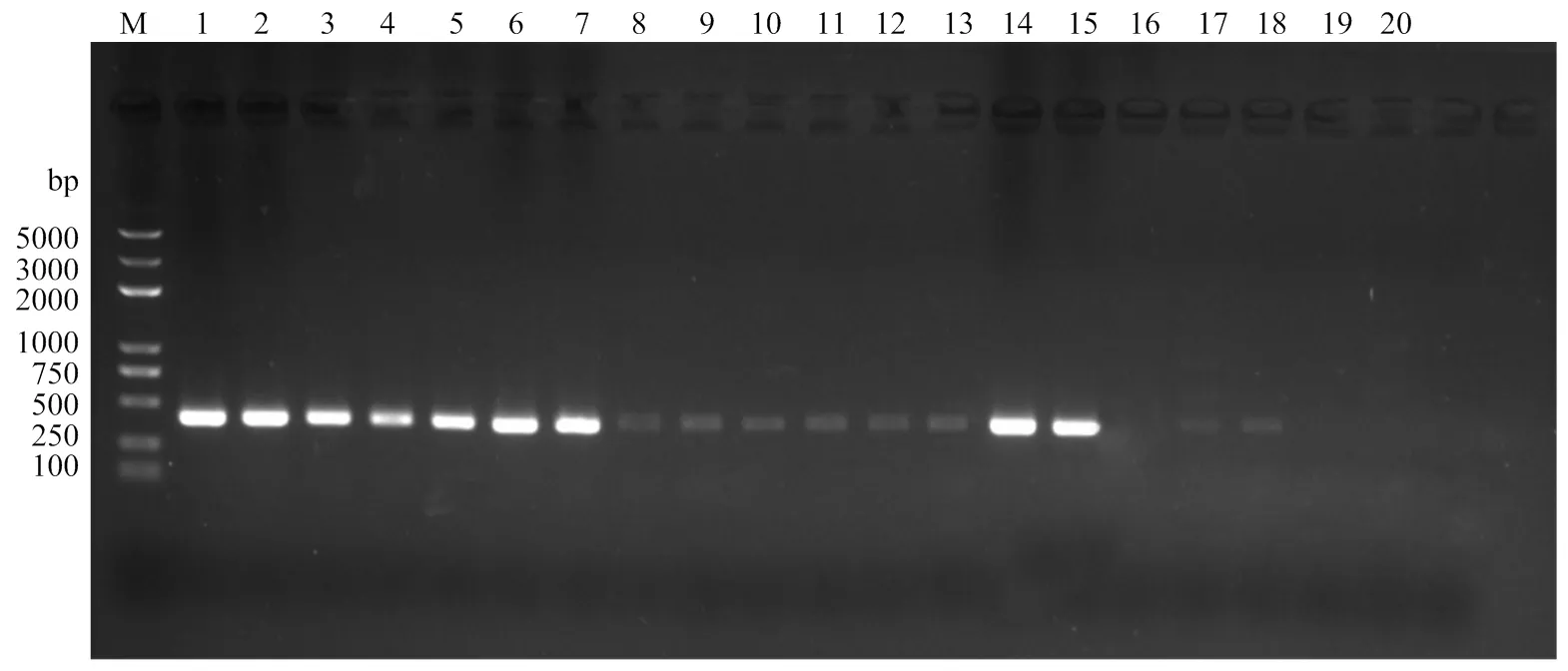
M:DNA分子量标准(5 000 bp) DNA marker(5 000 bp);1:阳性对照Positive control;2—8:田间死棵病株 Died plants in fields;9—18:田间病株根际土壤 Soils on died plants;19:阴性对照Negative control;20:空白对照 Blank control
4 结论
甘肃省白菜主产区死棵病的病原菌是立枯丝核菌AG-2-1和AG-1-IB。基于SYBR Green I建立的real-time PCR 检测方法特异性强、灵敏度高,能够满足针对田间植物及根际土壤中立枯丝核菌的检测和含菌量测定,可为田间白菜死棵病的预测预报提供技术支持。
[1] STRAUSBAUGH C A, EUJAYL I A, PANELLA L W, HANSON L E. Virulence, distribution and diversity offrom sugar beet in Idaho and Oregon., 2011, 33(2): 210-226.
[2] MELZER M S, YU H, LABUN T, DICKSON A, BOLAND G J. Characterization and pathogenicity ofspp. from field crops in Canada., 2016, 38(3): 367-374.
[3] ANDERSON N A. The genetics and pathology of., 1982, 20: 329-347.
[4] OGOSHI A. Ecology and pathogenicity of anastomosis and intraspecific groups ofKühn., 1987, 25: 125-143.
[5] CUBETA M A, Vilgalys R. Population biology of thecomplex., 1997, 87(4): 480-484.
[6] CARLING D E, KUNINAGA S, BRAINARD K A. Hyphal anastomosis reactions, rDNA-internal transcribed spacer sequences, and virulence levels among subsets ofanastomosis group-2 (AG-2) and AG-BI., 2002, 92(1): 43-50.
[7] Cappelli C, CORAZZA L, LUONGO L, STRAVATO V M. Interactions between crucifers andAG 2-1, AG 2-2IIIB, AG 2-2IV, AG 4., 1999, 38(1): 37-39.
[8] ZHOU Q X, HWANG S F, FU H T,STRELKOV S E, GOSSEN B D. Genetic variation ofisolates from canola in Alberta, Canada., 2014, 94(4): 671-681.
[9] MISAWA T, KUROSE D, MORI M, TODA T. Characterization of JapaneseAG-2-1 isolates using rDNA-ITS sequences, culture morphology, and growth temperature., 2018, 84(6): 387-394.
[10] 段春芳, 杨根华, 尼章光, 刘光华, 吴华英. 立枯丝核菌AG-1-IB引起白菜、薄荷、莴苣叶腐病的研究. 云南农业大学学报, 2008, 23(3): 422-425.
DUAN C F, YANG G H, Ni Z G, LIU G H, WU H Y. Occurrence of foliar rot of Chinese cabbage, mint and lettuce caused byAG-1-IB in China., 2008, 23(3): 422-425. (in Chinese)
[11] DE MELO M P, CABRAL C S, REIS A, MATOS K S, MARTINS P P, BESERRA JúNIOR J E A, NECHET K L, HALFELD-VIEIRA B A.AG 1-IB and AG 4 HG-I causing leaf blight and root rot in plants from the Lamiaceae family in Brazil., 2018, 43(2): 152-159.
[12] SHARON M, Kuninaga S, Hyakumachi M, NAITO S, SNEH B. Classification ofspp. using rDNA-ITS sequence analysis supports the genetic basis of the classical anastomosis grouping., 2008, 49(2): 93-114.
[13] LEES A K, CULLEN D W, SULLIVAN L, NICOLSON M J. Development of conventional and quantitative real-time PCR assays for the detection and identification ofAG-3 in potato and soil., 2002, 51(3): 293-302.
[14] 申永铭, 郭成瑾, 王喜刚, 沈瑞清, 陈爱昌, 胡小平. 土壤中立枯丝核菌AG3菌核的荧光定量PCR快速检测. 菌物学报, 2017,36(10): 1383-1391.
SHEN Y M, GUO C J, WANG X G, SHEN R Q, CHEN A C, HU X P. Rapid detection ofAG3 sclerotia in soil by quantitative real-time PCR., 2017, 36(10): 1383-1391. (in Chinese)
[15] SAYLER R J, YANG Y. Detection and quantification ofAG-1 IA, the rice sheath blight pathogen, in rice using real-time PCR., 2007, 91(12): 1663-1668.
[16] REN S F, ZHANG Z Y, DONG W H, BAO W J, YUE R, LIU L, LI C Y, YANG G H. The application of lettuce in the qualitative and quantitative detection ofAG-1 IA toxins., 2017, 45(4): 583-589.
[17] ZHOU Q, CHEN Y, YANG Y, AHMED H U, HWANG S F, STRELKON S E. Effect of inoculum density and quantitative PCR-based detection ofAG-2-1 andon canola., 2014, 59: 71-77.
[18] OKUBARA P A, SCHROEDER K L, PAULITZ T C. Identification and quantification ofandusing real-time polymerase chain reaction., 2008, 98(7): 837-847.
[19] WOODHALL J W, BROWN M J, PERKINS K, VALDEOLMILLOS E S, BOONHAM N, RAY R V. A TaqMan real-time PCR assay forand its use in wheat and soil., 2017, 148(2): 237-245.
[20] 周而勋, 杨媚. 从植物病组织中分离立枯丝核菌的快速、简便技术. 华南农业大学学报, 1998, 19(1): 125-126.
ZHOU E X, YANG M. A rapid and simple technique for the isolation offrom diseased plant tissues., 1998, 19(1): 125-126. (in Chinese)
[21] YANG G H, WANG X Y, CHEN H R, AKIRA O. A method for long-term storage ofspp., 2002, 21(1): 140.
[22] WHITE T J, BRUNS T, LEE S, TAYLOR J. Amplification and direct sequencing of fungal ribosomal RNA genes for phylogenetics//, 1990, 38: 315-322.
[23] 张春艳. 马铃薯黑痣病菌系统发育分析及融合群快速鉴定[D]. 保定: 河北农业大学, 2014.
ZHANG C Y. Phylogenetic analysis offrom potato and the rapid identification of their anastomosis groups[D]. Baoding: Hebei Agricultural University, 2014. (in Chinese)
[24] MATSUMOTO M. Trials of direct detection and identification ofAG1 and AG2 subgroups using specifically primed PCR analysis., 2002, 43(2): 185-189.
[25] YANG G H, CHEN H R, NAITO S, WU J Y, HE X H, DUAN C F. Occurrence of foliar rot of pak choy and Chinese mustard caused byAG1-IB in China., 2005, 71(5): 377-379.
[26] SHIM C K, KIM M J, KIM Y K, JEE H J, HONG S J, PARK J H, HAN E J, YUN J C. Leaf rot and leaf ring spot caused byin Chinese cabbage., 2013, 19(4): 300-307.
[27] 丁天波, 刘晓蓓, 李洁, 魏可可, 褚栋. 番茄褪绿病毒实时荧光定量PCR检测技术的建立. 中国农业科学, 2018, 51(10): 2013-2022.
DING T B, LIU X B, LI J, WEI K K, CHU D. Development of a real-time fluorescent quantitative PCR method for the detection ofand its application., 2018, 51(10): 2013-2022. (in Chinese)
[28] SINGH V, AMARADASA B S, KARJAGI C G, LAKSHMAN D K, HOODA K S, KUMAR A. Morphological and molecular variability among Indian isolates ofcausing banded leaf and sheath blight in maize., 2018, 152(1): 45-60.
[29] Zhao C, Li Y, Wu S, Wang P, Han C, Wu X. Anastomosis group and pathogenicity ofspp. associated with seedling damping-off of sugar beet in China., 2019, 153(3): 869-878.
[30] GODOY-Lutz G, STEADMAN J R, HIGGINS B, POWERS K. Genetic variation among isolates of the web blight pathogen of common bean based on PCR-RFLP of the ITS-rDNA region., 2003, 87(7): 766-771.
[31] HARIKRISHNAN R, YANG X B. Recovery of anastomosis groups offrom different latitudinal positions and influence of temperatures on their growth and survival., 2004, 88(8): 817-823.
[32] TSROR L. Biology, epidemiology and management ofon potato., 2010, 158(10): 649-658.
[33] 黄雯雯, 王玲, 刘连盟, 刘恩勇, 黄世文. 水稻纹枯病立枯丝核菌的分类及遗传多样性研究进展. 中国稻米, 2010, 16(3): 34-38.
HUANG W W, WANG L, LIU L M, LIU E Y, HUANG S W. Research advances of taxonomy and genetic diversity in., 2010, 16(3): 34-38. (in Chinese)
[34] HARVESON R M, WATKINS J E, GIESLER L J, CHAKY J L. EC05-1894 dry bean disease profiles II: Fungal root rot and wilt diseases., 2005(32): 1613.
[35] 吕顺, 曾莉莎, 刘文清, 王芳, 赵志慧, 周建坤, 李洪波, 杜彩娴, 陈石, 韩秀香, 向欣叶. 大蕉枯萎病病原菌鉴定及序列分析. 植物病理学报, 2014, 44(4): 337-348.
LüS, ZENG L S, LIU W Q, WANG F, ZHAO Z H, ZHOU J K, LI H B, DU C X, CHEN S, HAN X X, XIANG X Y. Identification andsequence analysis ofwilt pathogens from Dajiao., 2014, 44(4): 337-348. (in Chinese)
[36] GEISER D M, JIMENEZ-GASCO M, KANG S, MAKALOWSKA I, VEERARAGHAVAN N, WARD T J, Zhang N, Kuldau G A, O’Donnell K. FUSARIUM-ID v. 1.0: A DNA sequence database for identifying., 2004, 110(5/6): 473-479.
[37] ANEES M, TRONSMO A, EDEL-HERMANN V, HJELJORD L G, HERAUD C, STEINBERG C. Characterization of field isolates ofantagonistic against., 2010, 114(9): 691-701.
[38] BUDGE G E, SHAW M W, COLYER A, PIETRAVALLE S, BOONHAM N. Molecular tools to investigatedistribution in soil., 2009, 58(6): 1071-1080.
[39] WOODHALL J W, ADAMS I P, PETERS J C, HARPER G, BOONHAM N. A new quantitative real-time PCR assay forAG3-PT and the detection of AGs ofassociated with potato in soil and tuber samples in Great Britain., 2013, 136(2): 273-280.
[40] 胡加谊, 罗志文, 范鸿雁, 李向宏, 刘志昕, 何凡. 菠萝凋萎相关病毒-2实时荧光定量RT-PCR检测方法的建立. 园艺学报, 2014, 41(6): 1257-1266.
HU J Y, LUO Z W, FAN H Y, LI X H, LIU Z X, HE F. Development of a real-time fluorescent quantitative RT-PCR method for the detection of., 2014, 41(6): 1257-1266. (in Chinese)
[41] 孙炳剑, 陈清清, 袁虹霞, 施艳, 李洪连. SYBR Green I实时荧光定量PCR检测小麦纹枯病菌体系的建立和应用. 中国农业科学, 2015, 48(1): 55-62.
SUN B J, CHEN Q Q, YUAN H X, SHI Y, LI H L. Establishment of SYBR Green I real-time PCR for quantitatively detectingin winter wheat., 2015, 48(1): 55-62. (in Chinese)
Identification of the Pathogen Causing Cabbage Died in Gansu Province and Detection of Anastomosis groups
WANG Duo, XIE XueWen, CHAI ALi, SHI YanXia, LI BaoJu
(Institute of Vegetables and Flowers, Chinese Academy of Agricultural Sciences, Beijing 100081)
【】The objective of this study is to identify the pathogen causing cabbage died in Gansu Province, develop a molecular method for identification, and to provide a reference for prevention and control of the disease.【】Diseased samples collected from Gansu Province were purified and cultured, and isolates were characterized by colony morphology, it was initially identified the pathogen as. For accurate identification, the internal transcribed spacer region of the ribosomal DNA (rDNA-ITS) and translation elongation factor 1-alpha (TEF-1) of isolates were amplified and sequenced. Mega 7.0 was used to draw the phylogenetic tree of strains and other related sequences. Molecular identification of the pathogen was carried out by PCR amplification using a specific primer pair F-RS/R-RS. Pathogenicity on 20 cabbage cultivars was verified by spray inoculation and irrigating roots treatment. Based on the conserved region of rDNA-ITS, a pair of specific primer was designed and the SYBR Green I real-time PCR reaction system was established. The specificity, sensitivity and repeatability of conventional PCR and real-time PCR were also evaluated, andAG3-AG11, binulceate Rhizoctonia AG-A,spp.,spp. were used as control fungi. The established system was used to detectin the cabbage and rhizosphere soils. 【】A total of 86 strains ofwere isolated. The results of conventional PCR specific test showed that most strains belonged to AG-2-1 (68/86) and others were AG-1-IB (18/86). The phylogenetic analyses showed that 50 strains were divided into AG-2-1 and AG-1-IB, and the homology of each strain in two groups was 100%. Pathogenicity test results showed that the two anastomosis groups could both cause lesions on stems, and AG-2-1 was not pathogenic to the leaves of Jinwawa and Xiaohuangbao. The pathogenicity of AG-1-IB was slightly stronger than that of AG-2-1. The primers were of great specificity, the specific PCR fragment was amplified from the DNA ofstrains, but not from the DNA of other fungal strains by conventional PCR.The real-time PCR assays also did not amplify DNA from control fungi. The sensitivity of conventional PCR was 8.41×105copies/μL plasmid, while the sensitivity of real-time PCR was 8.41×103copies/μL, which increased two orders of magnitude. The standard curve established by recombinant plasmid showed a fine linear relationship between threshold cycle and template concentration. The melt curve was specific with the correlation coefficient of 0.9983 and with high amplification efficiency (91%). The method could successfully detect the pathogens in cabbage and rhizosphere soils collected in fields. For the indoor potted experiments, the detection results of real-time PCR of infected cabbage samples inoculated after different days showed that the stems and rhizosphere soils had the largest amount ofon the 5th day after inoculation. 【】Through molecular biological identification and pathogenicity test of pathogen of cabbage died, it was confirmed that the causal agent of cabbage died in Gansu wereAG-2-1 and AG-1-IB. The developed real-time PCR system forhas strong specificity and high sensitivity, it can realize the detection and monitoring ofin plant and soil, and can provide technical support for early warning and management of diseases.
cabbage; anastomosis groups of; real-time fluorescent quantitative PCR; detection
2019-04-04;
现代农业产业技术体系北京叶类蔬菜创新团队(BAIC-2018)、中国农业科学院科技创新工程(CAAS-ASTIP-IVFCAAS)
王朵,E-mail:15713890570@163.com。
石延霞,E-mail:shiyanxia@caas.cn。通信作者李宝聚,E-mail:libaoju@caas.cn
(责任编辑 岳梅)
