不可逆电穿孔消融治疗胰腺癌的现状与展望
2017-08-11周瑜吴志远丁晓毅
周瑜 吴志远 丁晓毅
·综述·
不可逆电穿孔消融治疗胰腺癌的现状与展望
周瑜 吴志远 丁晓毅
不可逆电穿孔消融(纳米刀)作为一项新兴的介入消融技术,不同于传统的物理消融技术如射频消融、微波消融及冷冻消融等。不可逆电穿孔技术利用高电压脉冲使细胞膜出现纳米级微孔,细胞内环境失衡,最终导致细胞凋亡,但却对细胞外基质影响较小,所以此技术可以应用于邻近重要血管或神经的肿瘤消融。胰腺癌是消化道恶性程度最高的肿瘤之一,预后差且较难早期诊断,往往发现时已经是Ⅲ期或者Ⅳ期。近几年来,不可逆电穿孔消融凭借并发症少、安全性高等优点,逐步应用于不可切除胰腺癌的治疗。本文就不可逆电穿孔消融的基础研究、治疗胰腺癌的临床研究等问题作一综述。
不可逆电穿孔;胰腺癌;消融;钠米刀
胰腺癌是恶性程度最高的消化道肿瘤之一,预后较差,其1年和5年生存率分别为27%和6%,而且患病率正以每年1.3%的速度增加[1]。胰腺癌的早期诊断率较低,只有10%的患者被诊断时为Ⅰ期或者Ⅱ期胰腺癌,手术切除后5年生存率仅为24%。50%的患者有转移病灶出现(Ⅳ期),剩余40%的患者(Ⅲ期)都有不同程度的浸润周围重要组织如门静脉、腹主动脉、肠系膜上动脉等,一般认为Ⅲ期、Ⅳ期胰腺癌无法行外科手术切除[2]。不可切除胰腺癌的治疗手段主要有放疗、化疗和局部治疗[3-4],局部治疗主要有动脉灌注化疗、区域放疗和射频消融、微波消融及冷冻消融等常规的消融方法。上述消融方法利用温度变化使目标区域的细胞发生坏死,但肿瘤周围血液流动(热沉效应[5])使靶区周围的温度改变不足以杀死肿瘤细胞、影响消融范围,从而导致肿瘤局部消融不全、复发率增高;与此同时,常规的消融方法也会对胰腺周围的重要结构如胆管、胰管、血管等造成不可逆损伤,因此并发症也较高。
不同于传统的物理消融技术,不可逆电穿孔消融利用高压、高频脉冲电流产生的电场,使细胞膜出现不可逆的纳米孔道,从而导致细胞内稳态失衡,诱发凋亡。基于这一特性,临床已经逐步开展不可逆电穿孔消融治疗胰腺癌,尤其是局部进展期胰腺癌。本文将对不可逆电穿孔消融的临床前研究、治疗胰腺癌的临床研究及其远期展望作一综述。
一、不可逆电穿孔的临床前研究
电穿孔技术最早应用于生物医学工程、药物及生物大分子递送、基因转导等领域[6],其应用形式为低电压、短时间引起的可逆性穿孔,细胞膜上的穿膜孔道会于20 min内关闭[7]。但将细胞置于高压、高频的脉冲电场中时,细胞膜结构破坏,细胞色素C释放,钙离子水平升高,细胞发生凋亡[8]。Lee等[9]以不可逆电穿孔消融兔和猪的正常肝脏组织,扫描电镜证实消融区域的细胞膜上出现大小不一的不可逆孔道,最大可达490 nm。Zhang等[10]建立胰腺癌移植瘤小鼠模型,行不可逆电穿孔消融肿瘤,30 min后透射电镜下观察到肿瘤细胞染色质浓集、凋亡小体形成并且细胞膜和核膜均有纳米级的缺陷形成,免疫组织化学结果显示TUNEL染色和Caspase3染色均为阳性。Petrishia等[11]用分子模型模拟细胞膜结构,动态演示了在高频、高压脉冲电场条件下细胞膜穿膜孔道的形成。以细胞凋亡为理论基础的不可逆电穿孔消融,不受热沉效应的影响[12],对靶区肿瘤的消融较完全,同时对肿瘤周围血管、神经、胰管、胆管等重要组织的影响较小[13]。Bower等[14]研究显示,不可逆电穿孔消融术后胰脂肪酶、胰淀粉酶暂时性升高,并于3 d内恢复正常,病理结果显示消融区域与正常组织分界清晰,血管结构完整。José等[15]建立原位胰腺癌小鼠模型,行不可逆电穿孔消融效果明确,荷瘤小鼠生存时间明显延长,术后病理结果显示肿瘤细胞凋亡,肿瘤微血管结构破坏,并有大量淋巴细胞浸润。Fritz等[16]对猪正常胰腺组织行不可逆电穿孔消融,术后动物生存状况良好,无严重并发症,术后60 min增强CT示门脉期低密度影,提示CT可以作为评价消融是否成功的监测手段。以上临床前研究表明,不可逆电穿孔以诱导细胞凋亡的方式消融肿瘤,同时对肿瘤周围血管、神经等结构损伤较小,比较适合胰腺癌的治疗。
二、不可逆电穿孔的临床研究
由美国Angio Dynamics公司生产的不可逆电穿孔消融设备Nanoknife(纳米刀)已于2012年4月由FDA批准在美国应用于临床,2015年6月我国食品药品监督管理局批准纳米刀消融设备用于肝脏和胰腺肿瘤消融治疗。该设备由高压电流发生器、消融电极针和心电同步监测仪构成。根据瘤体大小和形状,可选择2~6根消融电极,合适的消融参数(电压1 500~3 000 V,脉冲90~100个,脉宽70~100 μs)[17]对肿瘤进行消融。
1.术前评估:准确完整的术前评估是消融成功的基础,包括肿瘤标志物检测、连续的影像学评估、肿瘤分期以及患者身体状态的综合评估等[18]。Martin等[17]认为,行胰腺癌纳米刀消融前,应连续行高质量的三维CT或MRI检查,以确定肿瘤无转移或对术前新辅助化疗反应良好(肿瘤最长径减小>30%),肿瘤最长径≤4 cm,是行胰腺癌纳米刀消融的最佳适应证。手术患者的排除标准应包括不能耐受全麻者、严重心肺功能障碍或心率失常者、有癫痫病史者以及安装有心脏起搏器者[19-22]。
2.纳米刀消融的麻醉管理: 行纳米刀肿瘤消融术需要全身麻醉[23],丙泊酚诱导,空气/氧气/七氟烷混合气体麻醉维持,芬太尼或瑞芬太尼术中镇痛,术中应同时行血压、心电图、血氧饱和度监测。但需要注意的是,高压脉冲电场会引起肌肉收缩,中度以上的后腹膜或横膈膜刺激会导致靶器官的移位,从而增加穿刺电极对靶器官的创伤或影响布针的准确性,因此需要术中联合应用非去极化型神经肌肉阻滞剂(维库溴铵、罗库溴铵等)[23]。此外,行纳米刀消融时,心率、血压有明显增快、增高[23-24],这也提示麻醉医师注意降压药的应用。虽然纳米刀设备配备心电同步监测仪,但仍有研究中出现心律失常的病例[24],说明有房颤病史是否应该为相对禁忌证仍值得商榷。
3.临床研究:自从纳米刀消融设备被批准应用于临床以来,已有多项关于纳米刀消融治疗中晚期胰腺癌的报道,证明纳米刀消融治疗胰腺癌是安全、有效的(表1)。
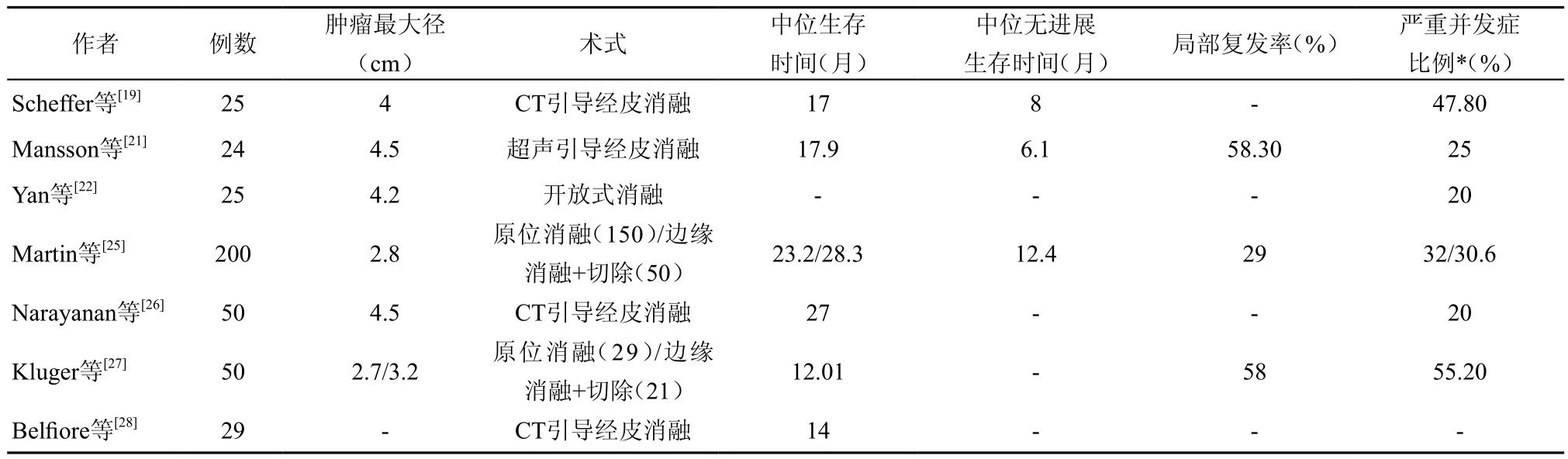
表1 纳米刀消融治疗胰腺癌的安全性和有效性研究
Martin等[29]首次将纳米刀应用于胰腺癌的消融治疗,27例患者中有19例患者行胰腺癌原位消融,8例行手术切除联合纳米刀消融处理可疑阳性切缘,术后随访90 d显示肿瘤消融成功率为100%。胰脂肪酶和胰淀粉酶在术后48 h升至最高,但72 h内降至正常水平,随访90 d有一例患者死亡,其余患者无胰腺炎症状或瘘管形成,说明纳米刀消融用于治疗胰腺癌是可行的。
纳米刀消融治疗胰腺癌被批准应用于临床以后,多项研究不仅证实了其有效性,而且较传统放化疗更能提高患者的中位生存时间,改善患者生活质量。Martin等[25]主持的一项多中心、前瞻性研究中,150例患者接受纳米刀胰腺癌原位消融治疗,50例行手术切除联合纳米刀边缘强化治疗,中位随访时间为29个月。结果显示,术后3个月,有6例患者复发,随访结束时共有58例患者(29%)复发,中位无进展生存时间为12.4个月,远处无进展生存时间为16.8个月,所有患者中位生存时间为24.9个月(12.4~85个月);纳米刀原位消融的患者为23.2个月(4.9~76.1个月),手术切除联合纳米刀边缘强化的患者28.3个月(9.2~85个月)。在Martin等[30]开展的另一项研究中,54例Ⅲ期胰腺癌患者成功实施了纳米刀消融治疗,对照组85例患者则行单纯化疗或放化疗治疗,结果显示纳米刀消融组的患者总体生存时间比对照组明显延长(20 vs.13个月),局部无进展生存时间和远处无转移生存时间也都明显延长(14 vs.6个月,15 vs.9个月)。Lambert等[31]研究则显示,纳米刀消融不仅延长了患者生存期,同时也提高了患者的生活质量—— Karnofsky评分下降缓慢,81%的患者评分≥70分,但在患者死亡前大约8周左右, Karnofsky评分会突然下降。
安全性方面,Scheffer等[19]进行的一项经皮纳米刀消融治疗胰腺癌的研究结果显示,25例患者消融成功率为100%,无一例患者在90 d随访期内死亡,术后总共 23例发生了并发症,其中2例出现Ⅳ级并发症(1例水肿型胰腺炎合并胆漏,1例十二指肠溃疡出血);Ⅲ级并发症有9例,其中胆管梗阻有3例(行ERCP支架置入);Ⅰ/Ⅱ级并发症有12例,对症治疗效果均较理想。此外,该研究中患者中位生存时间为17个月(95%CI:10~24个月),中位无进展生存时间为15个月(95%CI:10个20个月),说明纳米刀治疗胰腺癌是安全、可行的。
经皮纳米刀消融术中消融电极布针的准确性是肿瘤消融成功的关键,许多学者也对此进行了相关研究。Mansson等[32]术前利用CT或增强超声(CEUS)进行肿瘤体表定位,超声引导穿刺布针,5例患者术后增强超声均示无肿瘤残留、无穿刺相关并发症,术后30 d内无严重并发症出现(Clavien-Dindo分级3~5级)。但在实际的临床应用中,大多数报导中还是利用CT定位穿刺布针[26,33]。Bond等[34]则前瞻性的将3D布针导航系统应用于纳米刀经皮消融,相比于未用3D导航系统,平均增加了6.5 min的手术时间,但单纯布针的时间明显减少,同时增强了医生对于布针及手术的自信。
纳米刀消融的术后评估及随访应包括连续2~6个月的影像学资料、实验室检查以及临床症状,以上应作为监测肿瘤复发的优先指标[35]。Vroomen等[36]回顾性分析了25例行经皮纳米刀消融胰腺癌患者的影像资料,包括术前及术后6个月的增强CT和增强MRI,结果显示术后6周内由于水肿和充血,消融区域体积增大,但6周后会下降。25例患者的MR资料显示DWI-b800信号强度术后均明显下降,5例肿瘤复发患者术后6周病灶中均有DWI-b800高信号点,提示增强MRI和DWI-b800检测或许可以预测肿瘤复发。
4.术后并发症:疼痛为纳米刀消融术后最常见的并发症[19,24],常为轻到中度疼痛,给予非甾体抗炎药或阿片类药物即可,但对于有些患者疼痛症状未见减轻,6个月后仍有疼痛,则镇痛药难以治疗[19]。其次常见并发症为消化道症状,包括恶心、呕吐、厌食、脱水等临床表现[25],偶见上消化道出血、十二指肠溃疡或十二指肠瘘等并发症[27]。胰腺作为消融的靶器官,可出现急性胰腺炎、胰瘘等[22]并发症,附近的胆管有可能出现胆管炎、胆道阻塞等[19]并发症。消融区域周围血管相关并发症同样不容忽视,包括门静脉血栓形成、肠系膜上动脉栓塞或出血等[21,37]。此外,还有可能出现尿潴留、呼吸系统并发症[25]等。Mansson等[20]报导一例带金属胆道支架行胰腺癌消融的病例,术后出现十二指肠及横结肠穿孔、肠系膜上动脉出血等严重并发症。
三、展望
1.肿瘤完全消融降低复发率:虽然不可逆电穿孔消融胰腺癌的安全性及有效性已得到临床验证,但同时也存在着肿瘤复发的风险。Philips等[38]研究表明,异位胰腺癌小鼠模型中因消融不完全导致肿瘤复发后,肿瘤细胞的生长速度更快,侵袭性更高,高表达上皮细胞黏附分子(EpCAM),并且有耐药的可能。
肿瘤完全消融是降低复发率的首要条件。(1)不同的消融参数设置(包括电压、脉冲、针距、裸针长度等)会导致不同的病理结果[39],而且由于水肿的因素,CT显示的消融区域往往大于实际的消融区域,所以精确的消融参数是保证肿瘤完全消融的前提[17]。(2)标准化的操作步骤是提高手术成功率的重要一环,比如注意胰头、胰体尾部肿瘤消融[18、40-41]的差异。(3)肿瘤消融指示参数或指示剂。电阻值的改变及电阻曲线的斜率可以预测消融成功与否,但还需长期随访研究[42]。此外,根据胰腺癌肿瘤细胞特点及消融死亡方式——凋亡,并结合材料学研发新的消融指示剂或许可以提高消融的成功率。
2.不可逆电穿孔消融联合其他治疗:Bhutiani等[43]利用不可逆电穿孔造成的细胞膜孔道,促进吉西他滨进入肿瘤细胞,降低了荷瘤小鼠的肿瘤局部复发率。因此,利用细胞膜的孔道联合纳米医学高效递药[44],可以弥补消融区域边缘可逆性穿孔[14,38]的缺点,提高消融成功率。既往研究显示,不可逆电穿孔消融可以激活机体免疫系统[45]、抑制促癌炎症信号通路[46]。Lin等[47]采用不可逆电穿孔消融联合同种异体自然杀伤细胞治疗转移性胰腺癌,安全且短期效果明显,提示不可逆电穿孔消融联合免疫治疗或许可以降低复发率,提升治疗效果。
3.其他:加强围术期营养支持,可以减少术后并发症和患者住院天数,升高术后白蛋白水平,增强治疗效果[48]。精准的数字导航系统引导穿刺布针则可以减少穿刺次数,减少脏器损伤,缩短手术时间,或许可以成为不可逆电穿孔消融的重要辅助手段之一。
1 Siegel RL, Miller KD, Jemal A. Cancer Statistics, 2017[J]. CA Cancer J Clin, 2017,67(1):7-30.
2 Gluth A, Werner J, Hartwig W. Surgical resection strategies for locally advanced pancreatic cancer[J]. Langenbecks Arch Surg, 2015, 400(7):757-765.
3 中国癌症研究基金会介入医学委员会.晚期胰腺癌介入治疗临床操作指南(试行)[J].临床肝胆病杂志,2017,33(2):221-232.
4 Spadi R, Brusa F, Ponzetti A, et al. Current therapeutic strategies for advanced pancreatic cancer: A review for clinicians[J]. World J ClinOncol,2016,7(1):27-43.
5 Lu DS, Raman SS, Vodopich DJ, et al. Effect of vesselsize on creation of hepatic radiofrequency lesions in pigs: assessment of the "heat sink" effect[J]. AJR Am J Roentgenol, 2002, 178(1):47-51.
6 Gehl J. Electroporation: theory and methods, perspectives for drug delivery,gene therapy and research[J]. Acta Physiol Scand, 2003, 177(4):437-47.
7 Chang DC, Reese TS. Changes in membrane structure induced by electroporationas revealed by rapid-freezing electron microscopy[J]. Biophys J, 1990,58(1):1-12.
8 Joshi RP, Schoenbach KH. Bioelectric effects of intense ultrashort pulses[J]. Crit Rev Biomed Eng,2010,38(3):255-304.
9 Lee EW, Wong D, Prikhodko SV, et al. Electronmicroscopic demonstration and evaluation of irreversible electroporation-induced nanopores on hepatocyte membranes[J]. J Vasc Interv Radiol, 2012, 23(1):107-113.
10 Zhang Z, Li W, Procissi D, et al. Rapid dramaticalterations to the tumor microstructure in pancreatic cancer following irreversible electroporation ablation[J]. Nanomedicine (Lond), 2014, 9(8):1181-1192.
11 Petrishia A, Sasikala M. Molecular simulation of cell membrane deformation by picosecond intense electric pulse[J]. J Membr Biol, 2015,248(6):1015-1020.
12 Paiella S, Salvia R, Ramera M, et al. Local ablative strategies for ductal pancreatic cancer (radiofrequency ablation, irreversible electroporation): A review[J]. Gastroenterol Res Pract, 2016, 2016:4508376.
13 Vogel JA, van Veldhuisen E, Agnass P, et al. Time-dependent impact of irreversible electroporation on pancreas, liver, blood vessels and nerves: a systematic review of experimental studies[J]. PLoS One, 2016,11(11):e0166987.
14 Bower M, Sherwood L, Li Y, et al. Irreversible electroporation of thepancreas: definitive local therapy without systemic effects[J]. J Surg Oncol, 2011,104(1):22-28.
15 José A, Sobrevals L, Ivorra A, et al. Irreversible electroporation shows efficacy against pancreatic carcinoma without systemic toxicity in mouse models[J]. Cancer Lett, 2012, 317(1):16-23.
16 Fritz S, Sommer CM, Vollherbst D, et al. Irreversible electroporation of the pancreas isfeasible and safe in a porcine survival model[J]. Pancreas, 2015 ,44(5):791-798.
17 Martin RC 2nd, Durham AN, Besselink MG, et al. Irreversible electroporation in locally advanced pancreatic cancer: A call for standardization of energy delivery[J]. J Surg Oncol, 2016,114(7):865-871.
18 Martin RC. Irreversible electroporation of locally advanced pancreatic headadenocarcinoma[J]. J Gastrointest Surg,2013,17(10):1850-1856. 19 Scheffer HJ, Vroomen LG, de Jong MC, et al. Ablation of locally advanced pancreatic cancer with percutaneous irreversible electroporation: results of the Phase I/II PANFIRE study[J]. Radiology, 2017, 282(2):585-597.
20 Månsson C, Nilsson A, Karlson BM. Severe complications with irreversible electroporation of the pancreas in the presence of a metallic stent: a warning of a procedure that never should be performed[J]. Acta Radiol Short Rep, 2014,3(11):2047981614556409.
21 Månsson C, Brahmstaedt R, Nilsson A, et al. Percutaneous irreversible electroporation for treatment of locally advanced pancreatic cancer following chemotherapy or radiochemotherapy[J]. Eur J SurgOncol, 2016,42(9):1401-1406.
22 Yan L, Chen YL, Su M, et al. A single-institution experience with open irreversible electroporation for locally advanced pancreatic carcinoma[J]. Chin Med J (Engl), 2016 ,129(24):2920-2925.
23 Martin RC, Schwartz E, Adams J, et al. Intra–operative anesthesia management in patients undergoing surgical irreversible electroporation of the pancreas, liver, kidney, and retroperitoneal tumors[J].Anesth Pain Med, 2015, 5(3):e22786.
24 Nielsen K, Scheffer HJ, Vieveen JM, et al. Anaesthetic management during open andpercutaneous irreversible electroporation[J]. Br J Anaesth, 2014, 113(6):985-992.
25 Martin RC 2nd, Kwon D, Chalikonda S, et al. Treatment of 200 locally advanced (stage III) pancreaticadenocarcinoma patients with irreversible electroporation: safety and efficacy[J].Ann Surg, 2015, 262(3):486-494.
26 Narayanan G, Hosein PJ, Beulaygue IC, et al. Percutaneous imageguided irreversible electroporation for the treatment of unresectable, locally advanced pancreatic adenocarcinoma[J]. J VascInterv Radiol, 2017, 28(3):342-348.
27 Kluger MD, Epelboym I, Schrope BA, et al. Single-Institution experience with irreversible electroporation for T4 pancreatic cancer: first 50 patients[J]. Ann Surg Oncol, 2016, 23(5):1736-1743.
28 Belfiore G, Belfiore MP, Reginelli A, et al. Concurrent chemotherapy alone versus irreversible electroporation followed by chemotherapy on survival in patients with locally advanced pancreatic cancer[J]. Med Oncol, 2017, 34(3):38.
29 Martin RC 2nd, McFarland K, Ellis S, et al. Irreversible electroporation therapy in the management of locally advanced pancreatic adenocarcinoma[J]. J Am Coll Surg, 2012, 215(3):361-369.
30 Martin RC 2nd, McFarland K, Ellis S, et al. Irreversible electroporation in locally advanced pancreatic cancer: potential improved overall survival[J]. Ann Surg Oncol, 2013, 20(Suppl 3):S443-449.
31 Lambert L, Horejs J, Krska Z, et al. Treatment of locally advanced pancreatic cancer by percutaneous andintraoperative irreversible electroporation: general hospital cancer center experience[J]. Neoplasma, 2016,63(2):269-273.
32 Månsson C, Bergenfeldt M, Brahmstaedt R, et al. Safety and preliminary efficacy of ultrasound-guided percutaneous irreversible electroporation for treatment of localized pancreatic cancer[J]. Anticancer Res,2014,34(1):289-293.
33 Belfiore MP, Ronza FM, Romano F, et al. Percutaneous CT-guided irreversible electroporation followed by chemotherapy as a novel neoadjuvant protocol in locally advancedpancreatic cancer: Our preliminary experience[J]. Int J Surg, 2015, 21(Suppl 1):S34-39.
34 Bond L, Schulz B, VanMeter T, et al. Intra-operative navigation of a 3-dimensional needle localization system for precision of irreversible electroporation needles in locally advanced pancreatic cancer[J]. Eur J Surg Oncol, 2017 ,43(2):337-343.
35 Akinwande O, Ahmad SS, Van Meter T, et al. CT findings of patients treated with irreversible electroporation for locally advanced pancreatic cancer[J]. J Oncol, 2015,2015:680319.
36 Vroomen LGPH, Scheffer HJ, Melenhorst MCAM, et al. MR
and CT imaging characteristicsand ablation zone volumetry of locally advanced pancreatic cancer treated with irreversible electroporation[J]. Eur Radiol, 2017 ,27(6):2521-2531.
37 Ekici Y, Tezcaner T, Aydın HO, et al. Arterial complication ofirreversible electroporation procedure for locally advanced pancreatic cancer[J].World J Gastrointest Oncol, 2016,8(10):751-756.
38 Philips P, Li Y, Li S, et al. Efficacy of irreversible electroporation in human pancreatic adenocarcinoma: advanced murine model[J]. Mol Ther Methods Clin Dev, 2015 ,2:15001.
39 Wimmer T, Srimathveeravalli G, Gutta N, et al. Comparison of simulation-based treatment planning with imaging and pathology outcomes for percutaneous CT-guided irreversible electroporation of the porcine pancreas: a pilot study[J]. J Vasc Interv Radiol, 2013,24(11):1709-1718.
40 Martin RC 2nd. Irreversible electroporation of locally advanced pancreatic neck/body adenocarcinoma[J]. J Gastrointest Oncol, 2015 , 6(3):329-335.
41 Scheffer HJ, Melenhorst MC, Vogel JA, et al. Percutaneous irreversible electroporation of locally advanced pancreatic carcinoma using the dorsal approach: a case report[J]. Cardiovasc Intervent Radiol , 2015,38(3):760-765.
42 Dunki-Jacobs EM, Philips P, Martin RC 2nd. Evaluation of resistance as ameasure of successful tumor ablation during irreversible electroporation of thepancreas[J]. J Am Coll Surg, 2014, 218(2):179-187.
43 Bhutiani N, Agle S, Li Y, et al. Irreversible electroporation enhances delivery of gemcitabine to pancreatic adenocarcinoma[J]. J Surg Oncol, 2016,114(2):181-186.
44 Li J, Liu F, Gupta S, et al. Interventional nanotheranostics of pancreatic ductal adenocarcinoma[J]. Theranostics, 2016 ,6(9):1393-1402.
45 Li X, Xu K, Li W, et al. Immunologic response to tumorablation with irreversible electroporation[J]. PLoS One, 2012,7(11):e48749.
46 Goswami I, Coutermarsh-Ott S, Morrison RG, et al. Irreversible electroporation inhibits pro-cancer inflammatory signaling in triple negative breast cancer cells[J]. Bioelectrochemistry, 2017,113:42-50.
47 Lin M, Liang S, Wang X, et al. Short-termclinical efficacy of percutaneous irreversible electroporation combined with allogeneic natural killer cell for treating metastatic pancreatic cancer[J]. Immunol Lett, 2017,186:20-27.
48 Martin RC 2nd, Agle S, Schlegel M, et al. Efficacy of preoperative immunonutrition in locally advanced pancreatic cancer undergoing irreversible electroporation (IRE) [J]. Eur J Surg Oncol, 2017,43(4):772-779.
Irreversible electroporation ablation for the treatment of pancreatic cancer:present and prospects
Zhou Yu, Wu Zhiyuan,Ding Xiaoyi. Department of Interventional Radiology, Ruijin Hospital, Shanghai Jiao Tong University School of Medicine, Shanghai 200025, China
Ding Xiaoyi, Email: dxy10456@rjh.com.cn
As an emerging interventional ablation modality which is different from conventional physical ablation such as radiofrequency ablation, microwave ablation and cryoablation, irreversible electroporation(IRE) ablationutilizes high voltage pulses to create permanent nanopores in the cell membrane, which in turn induces apoptosis of targeted cells. The promising technology can be used to ablate tumors adjacent to vital structures due to its less impact on extracellular matrix. Pancreatic cancer, despite extensive research, remains the most aggressive gastrointestinal tumors, having a poor prognosis and ambiguous early symptom usually diagnosed at stage Ⅲ or Ⅳ. Recently, IRE ablation has being progressively applied in the management of unresectable pancreatic cancer by virtue of its high efficiency and safety. This article will review on the basic research of IRE, clinical application in pancreatic cancer and some indefinite questions.
Irreversible electroporation; Pancreatic Cancer; Ablation; Nanoknife
2017-07-01)
(本文编辑:闫娟)
10.3877/cma.j.issn.2095-5782.2017.03.017
200025上海交通大学医学院附属瑞金医院放射介入科
丁晓毅,Email:dxy10456@rjh.com.cn
周瑜,吴志远,丁晓毅. 不可逆电穿孔消融治疗胰腺癌的现状与展望[J/CD].中华介入放射学电子杂志,2017,5(3):194-198.
