The emerging role of autophagic-lysosomal dysfunction in Gaucher disease and Parkinson’s disease
2017-04-07
1 Institute of Healthy Ageing and Department of Genetics, Evolution and Environment, University College London, London, UK
2 Institute of Neurology, University College London, London, UK
3 Department of Basic and Clinical Neuroscience, King’s College London, Maurice Wohl Clinical Neuroscience Institute, London, UK
4 London Central & West Unscheduled Care Collaborative, St. Charles Centre for Health and Wellbeing, Exmoor Street St, Charles Hospital, London, UK
5 Research Division, Joslin Diabetes Center, Boston, MA, USA
6 Department of Genetics and Harvard Stem Cell Institute, Harvard Medical School, Boston, MA, USA
The emerging role of autophagic-lysosomal dysfunction in Gaucher disease and Parkinson’s disease
Kerri J. Kinghorn1,2,3,*, Amir M. Asghari4, Jorge Iván Castillo-Quan1,5,6
1 Institute of Healthy Ageing and Department of Genetics, Evolution and Environment, University College London, London, UK
2 Institute of Neurology, University College London, London, UK
3 Department of Basic and Clinical Neuroscience, King’s College London, Maurice Wohl Clinical Neuroscience Institute, London, UK
4 London Central & West Unscheduled Care Collaborative, St. Charles Centre for Health and Wellbeing, Exmoor Street St, Charles Hospital, London, UK
5 Research Division, Joslin Diabetes Center, Boston, MA, USA
6 Department of Genetics and Harvard Stem Cell Institute, Harvard Medical School, Boston, MA, USA
How to cite this article:Kinghorn KJ, Asghari AM, Castillo-Quan JI (2017) The emerging role of autophagic-lysosomal dysfunction in Gaucher disease and Parkinson’s disease. Neural Regen Res 12(3):380-384.
Open access statement:This is an open access article distributed under the terms of the Creative Commons Attribution-NonCommercial-ShareAlike 3.0 License, which allows others to remix, tweak, and build upon the work non-commercially, as long as the author is credited and the new creations are licensed under the identical terms.
Gaucher disease (GD), the commonest lysosomal storage disorder, results from the lack or functional deficiency of glucocerebrosidase (GCase) secondary to mutations in theGBA1gene. There is an established association betweenGBA1mutations and Parkinson’s disease (PD), and indeedGBA1mutations are now considered to be the greatest genetic risk factor for PD. Impaired lysosomal-autophagic degradation of cellular proteins, including α-synuclein (α-syn), is implicated in the pathogenesis of PD, and there is increasing evidence for this also in GD andGBA1-PD. Indeed we have recently shown in aDrosophilamodel lacking neuronal GCase, that there are clear lysosomal-autophagic defects in association with synaptic loss and neurodegeneration. In addition, we demonstrated alterations in mechanistic target of rapamycin complex 1 (mTORC1) signaling and functional rescue of the lifespan, locomotor defects and hypersensitivity to oxidative stress on treatment of GCase-deficient flies with the mTOR inhibitor rapamycin. Moreover, a number of other recent studies have shown autophagy-lysosomal system (ALS) dysfunction, with specific defects in both chaperone-mediated autophagy (CMA), as well as macroautophagy, in GD andGBA1-PD model systems. Lastly we discuss the possible therapeutic benefits of inhibiting mTOR using drugs such as rapamycin to reverse the autophagy defects in GD and PD.
Gaucher disease; Parkinson’s disease; Drosophila; autophagy; lysosome; glucocerebrosidase; GBA
The Link between Gaucher Disease and Parkinson’s Disease
Homozygous loss-of-function mutations in theGBA1gene cause Gaucher disease (GD), the commonest lysosomal storage disorder. This gene encodes glucocerebrosidase (GCase), a lysosomal enzyme responsible for the hydrolysis of the glycolipid substrate glucosylceramide (GlcCer) to ceramide and glucose. Reductions in enzymatic activity result in the lysosomal accumulation of GlcCer, as well as glucosylsphingosine, within reticulo-endoendothelial cells, leading to the systemic sequelae of GD, including organomegaly, bone disease, anaemia and thrombocytopenia. The clinical spectrum of GD has historically been sub-divided into Type 1 GD, the so-called non-neuronopathic milder form of the disease, and Types II and III GD, which are severe acute and chronic neuronopathic forms respectively (Cox, 2010). In recent years heterozygousGBA1mutations have been identified as key genetic risk factors for Parkinson’s disease (PD), and increase the risk of developing PD by approximately 20-fold (Sidransky et al., 2009; Migdalska-Richards and Schapira, 2016).
The Role of Autophagy in Cellular α-Synuclein Degradation
The neuropathological hallmark of PD is the presence of proteinacious intraneuronal inclusions, known as Lewy bodies, predominantly composed of aggregated α-synuclein (α-syn), in addition to other proteins such as ubiquitin and p62 (Zatloukal et al., 2002; Shults, 2006). α-syn is a presynaptic protein of unknown function, which is thought to play a central role in the pathogenesis of PD. Moreover, there is mounting evidence that soluble α-syn aggregation intermediates (so-called oligomeric or protofibrillar species) represent the most toxic form of the protein (Volles and Lansbury, 2003). α-syn is removedfrom the cell by both macroautophagy and chaperone-mediated autophagy (CMA) (Cuervo et al., 2004; Watanabe et al., 2012), and indeed α-syn aggregates accumulate in response to the pharmacological inhibition of autophagy in mice (Klucken et al., 2012). α-syn is a well-characterised substrate of CMA due to the presence of a CMA-specific pentapeptide sequence motif. It is selectively translocated across the lysosomal membrane in a complex with the heat shock cognate protein 70 (hsc70), a process dependent on lysosomal-associated membrane protein 2A (LAMP-2A). Furthermore pathogenic forms of α-syn inhibit CMA through the blockade of receptor-mediated uptake into the lysosome (Cuervo et al., 2004). Macroautophagy is a less selective degradative pathway, which is responsible for the bulk removal of defective organelles and mis-folded cytoplasmic proteins, including α-syn, from the cell. It involves the sequestration of the cytosolic contents into double-membrane autophagosomes, which are then delivered to the lysosome to form a single-membrane autophagolysosome. The cargo is then degraded by lysosomal hydrolases. Macroautophagy dysregulation is increasingly being recognized as a pathogenic factor in neurodegeneration, including in PD (Ravikumar et al., 2010). Consistent with this, the selective suppression of autophagy, through the neuronal loss of the autophagy genesatg7oratg5, results in a number of phenotypes in mice, including locomotor defects, accumulation of polyubiquitinated proteins and neurodegeneration (Hara et al., 2006; Komatsu et al., 2006).
The Role of GCase Deficiency in PD
Both loss-of-function and toxic gain-of-function hypothesizes have been put forward to explain the link betweenGBA1mutations and PD (Kinghorn, 2011; Migdalska-Richards and Schapira, 2016). There is growing evidence supporting the role of GCase loss-of-function in PD. The majority ofGBA1mutations, including missense, nonsense and frame-shift mutations, insertions or deletions, are associated with reduced lysosomal GCase levels (Montfort et al., 2004; Sidransky et al., 2009). Moreover, milder mutations, associated with slightly diminished GCase levels, confer a much lower risk of PD than more severe mutations resulting in severe enzymatic dysfunction (Swan and Saunders-Pullman, 2013). Moreover, GCase protein levels and enzymatic activity are both decreased in the post-mortem brain tissue from patients with both idiopathic andGBA1-linked PD (Gegg et al., 2012). A number of mechanisms linking GCase loss-of-function with neurodegeneration have been demonstrated (Kinghorn, 2011; Migdalska-Richards and Schapira, 2016). Loss of GCase activity has been shown to lead to GlcCer accumulation, resulting in stabilisation of α-syn oligomers and α-syn accumulation. The subsequent increase in α-syn may inhibit GCase ER-Golgi trafficking and lysosomal GCase levels, thus creating a bidirectional pathogenic loop (Mazzulli et al., 2012).
The Emerging Role of Autophagy-lysosomal Dysfunction in GD and PD
We recently investigated the role of the autophagy-lysosomal (ALS) system in neuronopathic GD usingDrosophila melanogaster(Kinghorn et al., 2016). Employing homologous recombination techniques we developed a fly model lacking GCase in the brain. GCase-deficient flies displayed reduced lifespan, age-dependent locomotor abnormalities, as well clear evidence of synaptic loss and neurodegeneration. In keeping with the hallmark lysosomal dysfunction seen in GD cells, staining with LysoTracker® revealed numerous abnormally enlarged lysosomes in the brains of flies lacking GCase. This abnormal lysosomal pathology was associated with the accumulation of GlcCer, similar to that seen in neuronopathic GD brains (Conradi et al., 1984) (Figure 1).
Normal lysosomal functioning is required for the formation of autolysosomes and thus the autophagic removal of misfolded proteins and defective organelles from the cell (Ravikumar et al., 2010). During macroautophagy LC3-I is converted into its lipidated form, LC3-II, which is recruited to autophagosomal membranes (Ravikumar et al., 2010). We therefore probed macroautophagy function by measuring the levels of the fly homologue of LC3, Atg8, by western blot analysis. This demonstrated that both Atg8(LC3)-I and Atg8(LC3)-II are increased in the GCase-deficient fly brain, in addition to the ratio of Atg-II/Atg-I, suggestive of increased autophagosome number and a block in autophagy flux. This mirrors the increased LC3-II levels seen in the substantia nigra of PD patients (Dehay et al., 2010). In addition, we demonstrated increased accumulation in GCase-deficient fly brains of autophagy substrates, namely polyubiquinated proteins and Ref(2)P, theDrosophilahomologue of p62. Both ubiquitin and p62 are abundant constituents of protein inclusions associated with neurodegenerative diseases, including LBs in PD (Zatloukal et al., 2002; Bartlett et al., 2011). Further interrogation of autophagy at the regulatory level in fly brains revealed a decrease in the phosphorylation of p70S6 kinase, a substrate of the energy sensing mammalian target of rapamycin (mTOR)-complex 1 (mTORC1). mTORC1 plays a central role in macroautophagy by initiating the formation and elongation of the autophagosomal membrane (Ravikumar et al., 2010). Thus the decrease in mTORC1 activity seen in GCase-deficient fly brains likely represents a compensatory response to the autophagy block and is similar to that seen in fibroblasts derived from PD patients harbouringGBA1mutations (Magalhaeset al., 2016). In keeping with the impaired autophagy, abnormal giant mitochondria were observed in the brains of GCase-deficient flies, a reflection of the inability of the cells to clear aged and defective mitochondria by autophagy (the process of mitophagy). Interestingly GCase-deficiency in flies appeared to phenocopyMitf-knockdown.Mitf(microphthalmia transcription factor) is the fly homologue of mammalianTFEB(transcription factor EB), the master regulator of lysosomal biogenesis. It has many conserved functions in the fly including regulation of the lysosomal-autophagy pathway and lipid metabolism (Bouche et al., 2016). GCase deficiency in the fly was also associated withMitfgene up-regulation, likely in response to the lysosomal-autophagy block. Lastly treatment of GCase-deficient flies with the mTOR inhibitor rapamycin resulted in a significant rescue of the lifespan and locomotor phenotypes, as well as resistance to oxidative stress (Figure 1).
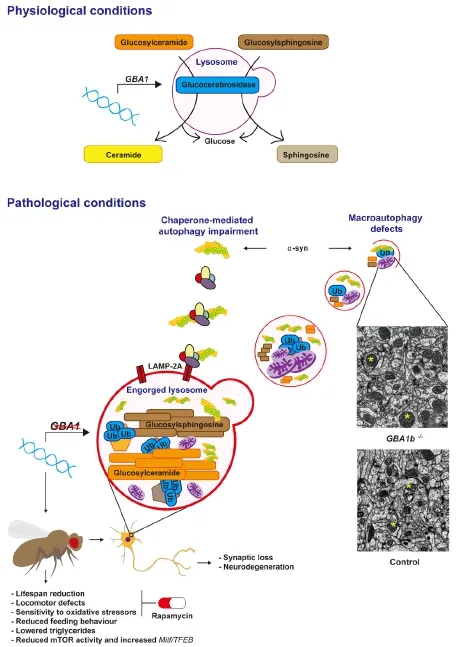
Figure 1 Glucocerebrosidase (GCase) deficiency results in autophagy-lysosomal system (ALS) dysfunction.
ALS dysfunction is increasingly being implicated in the pathogenesis of GD and PD, and has been identified in several GD and GCase-deficient PD models. Autophagy defects, in association with inflammasone activation, were observed in GD macrophages (Aflaki et al., 2014). Pharmacological inhibition of GCase in mice, through the chronic systemic treatment with conduritol-β-epoxide, lead to the accumulation of insoluble α-syn aggregates and neurodegeneration, in addition to an increase in the CMA-associated protein LAMP-2A and the macroautophagy marker LC3-II (Rocha et al., 2015b). GCase siRNA knockdown in cell culture also inhibited macroautophagy flux with a rise in LC3-II and p62, andGBA1knockout in mouse embryonic fibroblasts lead to a compromise in CMA. Furthermore the autophagy defects were associated with loss of autophagic lysosomal reformation and maturation of endosomes. They also observed a decrease in functional acidified lysosomes within GCase-deficient cells (Magalhaes et al., 2016). Enlargement of the lysosomal compartment, as observedin GCase-deficient fly brains, was also seen inGBA1mutant PD induced pluripotent stem cell (iPSC)-derived dopamine neurons, in association with autophagy defects and accumulation of intracellular p62 (Fernandes et al., 2016).
It has been suggested that both GD patients andGBA1mutation carriers display a similar risk of developing PD (Schapira, 2015). Consistent with this,in vitrowork has shown that the degree of autophagy inhibition and α-syn accumulation is comparable in iPSC-derived neurones from GD patients andGBA1mutation carriers with PD (Schöndorf et al., 2014). Moreover, autophagic lysosomal reformation defects in GCase-deficient cellular models were also similar in heterozygote and homozygote mutantGBA1models (Magalhaes et al., 2016). These observations are in keeping, at least in part, by the complex interplay between the loss and toxic gain-of-functions of GCase activity, the ageing cellular degradative machinery, including the ALS, and additional genetic risk factors predisposing to PD. Work in our GCase knockout fly model, as well as in other cellular and mouse models, has demonstrated that loss of GCase activity is sufficient to cause neurodegeneration, even in the absence of α-syn, when GCase levels are sufficiently low (Kinghorn et al 2016). As already mentioned, α-syn can further potentiate GCase deficiency through the generation of a bidirectional feedback loop, leading to lysosomal dysfunction and the generation of toxic oligomeric α-syn species (Mazzulli et al., 2012). In addition to the effects of GCase loss-offunction, the cellular sequelae of aging undoubtedly play a role. Indeed both lysosomal and autophagic function decline with age (Martinez-Vicente et al., 2005; Cuervo, 2008). During ageing GCase activity also declines in the substantia nigra and putamen of healthy controls, and is comparable to the enzymatic activity in PD patients withGBA1mutations (Rocha et al., 2015a). This suggests that an age-dependent reduction in GCase activity may lower the threshold for developing PD, and contribute to the lysosomal dysfunction that occurs in ageing and PD. Terefore moderate reductions in GCase activity inGBA1mutation carriers, in addition to the age-related decline in the ALS, and other cellular perturbations secondary to additional genetic and environmental risk factors, may be sufficient to promote α-syn accumulation and neurodegeneration. Further studies are required to better characterize the relationship between specific pathogenicGBA1mutations, their corresponding GCase activity and ALS function, in addition to their dose effect and clinical PD phenotypes. Such studies will undoubtedly aid our understanding of the variable penetrance ofGBA1mutations, in both the heterozygous and homozygous state, in causing PD.
Targeting Autophagy in GD and PD using the mTOR Inhibitor Rapamycin
The beneficial effects of rapamycin, demonstrated in our GCase knockout fly model, suggest that the inhibition of mTORC1 may represent an additional therapeutic strategy in the treatment of GD and PD, as well as other neurodegenerative disorders, in which there is significant ALS dysfunction. In support of this, rapamycin increased the clearance of exogenously expressed human wild-type and mutant α-syn in an inducible cell model (Webb et al., 2003) as well as endogenous α-syn inGBA1-knockdown primary cortical neurons (Du et al., 2015). Rapamycin has also been shown to mediate neuroprotective effects in other neurodegenerative diseases, including in a TDP-43-expressing amyotrophic lateral sclerosis mouse model (Bové et al., 2011; Wang et al., 2012). However, inhibition of mTOR by rapamycin is not invariably protective in allGBA1mutant models, and indeed rapamycin was found to trigger cell death in GD iPSC-derived neuronal cells harboring aGBA1mutation (Awad et al., 2015). Furthermore chronic rapamycin treatment aggravated the pathology and muscle weakness in a mouse model of VCP (valosin containing protein)-associated myopathy, despite a defect in mTOR signaling (Ching and Weihl, 2013). Tus the effects of inhibiting mTORC1 activity appear to vary depending on disease pathology, the model organism, and also likely the timing of the therapy. Further studies in mammalian models are now required to verify the benefits of rapamycin, and other inhibitors of mTOR, in ameliorating the neuropathology in GD and PD. It is likely that the clinical success of pharmacological mTOR inhibitors will rely on the precise titration of mTOR activity, in order to stimulate autophagy appropriately in the face of ALS dysfunction, minimalizing unwanted side effects.
Author contributions:KJK, AMA and JIC-Q all wrote the manuscript.
Conflicts of interest:None declared.
Aflaki E, Stubblefield BK, Maniwang E, Lopez G, Moaven N, Goldin E, Marugan J, Patnaik S, Dutra A, Southall N, Zheng W, Tayebi N, Sidransky E (2014) Macrophage models of Gaucher disease for evaluating disease pathogenesis and candidate drugs. Sci Transl Med 6:240ra73.
Awad O, Sarkar C, Panicker LM, Miller D, Zeng X, Sgambato JA, Lipinski MM, Feldman RA (2015) Altered TFEB-mediated lysosomal biogenesis in Gaucher disease iPSC-derived neuronal cells. Hum Mol Genet 24:5775-5788.
Bartlett BJ, Isakson P, Lewerenz J, Sanchez H, Kotzebue RW, Cumming RC, Harris GL, Nezis IP, Schubert DR, Simonsen A, Finley KD (2011) p62, Ref(2)P and ubiquitinated proteins are conserved markers of neuronal aging, aggregate formation and progressive autophagic defects. Autophagy 7:572-583.
Bouche V, Espinosa AP, Leone L, Sardiello M, Ballabio A, Botas J (2016) Drosophila Mitf regulates the V-ATPase and the lysosomal-autophagic pathway. Autophagy 12:484-498.
Bové J, Martínez-Vicente M, Vila M (2011) Fighting neurodegeneration with rapamycin: mechanistic insights. Nat Rev Neurosci 12:437-452.
Ching JK, Weihl CC (2013) Rapamycin-induced autophagy aggravates pathology and weakness in a mouse model of VCP-associated myopathy. Autophagy 9:799-800.
Conradi NG, Sourander P, Nilsson O, Svennerholm L, Erikson A (1984) Neuropathology of the Norrbottnian type of Gaucher disease. Morphological and biochemical studies. Acta Neuropathol 65:99-109.
Cox TM (2010) Gaucher disease: clinical profile and therapeutic developments. Biologics 4:299-313.
Cuervo AM (2008) Autophagy and aging: keeping that old broom working. Trends Genet 24:604-612.
Cuervo AM, Stefanis L, Fredenburg R, Lansbury PT, Sulzer D (2004) Impaired degradation of mutant alpha-synuclein by chaperone-mediated autophagy. Science 305:1292-1295.
Dehay B, Bové J, Rodríguez-Muela N, Perier C, Recasens A, Boya P, Vila M (2010) Pathogenic lysosomal depletion in Parkinson’s disease. J Neurosci 30:12535-12544.
Du TT, Wang L, Duan CL, Lu LL, Zhang JL, Gao G, Qiu XB, Wang XM, Yang H (2015) GBA deficiency promotes SNCA/alpha-synuclein accumulation through autophagic inhibition by inactivated PPP2A. Autophagy 11:1803-1820.
Fernandes HJ, Hartfield EM, Christian HC, Emmanoulidou E, Zheng Y, Booth H, Bogetofte H, Lang C, Ryan BJ, Sardi SP, Badger J, Vowles J, Evetts S, Tofaris GK, Vekrellis K, Talbot K, Hu MT, James W, Cowley SA, Wade-Martins R (2016) ER stress and autophagic perturbations lead to elevated extracellular α-synuclein in GBAN370S Parkinson’s iPSC-derived dopamine neurons. Stem Cell Rep 6:342-356.
Gegg ME, Burke D, Heales SJ, Cooper JM, Hardy J, Wood NW, Schapira AH (2012) Glucocerebrosidase deficiency in substantia nigra of parkinson disease brains. Ann Neurol 72:455-463.
Hara T, Nakamura K, Matsui M, Yamamoto A, Nakahara Y, Suzuki-Migishima R, Yokoyama M, Mishima K, Saito I, Okano H, Mizushima N (2006) Suppression of basal autophagy in neural cells causes neurodegenerative disease in mice. Nature 441:885-889.
Kinghorn KJ (2011) Pathological looping in the synucleinopathies: investigating the link between Parkinson’s disease and Gaucher disease. Dis Model Mech 4:713-715.
Kinghorn KJ, Gronke S, Castillo-Quan JI, Woodling NS, Li L, Sirka E, Gegg M, Mills K, Hardy J, Bjedov I, Partridge L (2016) A drosophila model of neuronopathic Gaucher disease demonstrates lysosomal-autophagic defects and altered mtor signalling and is functionally rescued by rapamycin. J Neurosci 36:11654-11670.
Klucken J, Poehler AM, Ebrahimi-Fakhari D, Schneider J, Nuber S, Rockenstein E, Schlötzer-Schrehardt U, Hyman BT, McLean PJ, Masliah E, Winkler J (2012) Alpha-synuclein aggregation involves a bafilomycin A 1-sensitive autophagy pathway. Autophagy 8:754-766.
Komatsu M, Waguri S, Chiba T, Murata S, Iwata J, Tanida I, Ueno T, Koike M, Uchiyama Y, Kominami E, Tanaka K (2006) Loss of autophagy in the central nervous system causes neurodegeneration in mice. Nature 441:880-884.
Magalhaes J, Gegg ME, Migdalska-Richards A, Doherty MK, Whitfield PD, Schapira AH (2016) Autophagic lysosome reformation dysfunction in glucocerebrosidase deficient cells: relevance to Parkinson disease. Hum Mol Genet 25:3432-3445.
Martinez-Vicente M, Sovak G, Cuervo AM (2005) Protein degradation and aging. Exp Gerontol 40:622-633.
Mazzulli JR, Xu YH, Sun Y, Knight AL, McLean PJ, Caldwell GA, Sidransky E, Grabowski GA, Krainc D (2012) Gaucher’s disease glucocerebrosidase and alpha-synuclein form a bidirectional pathogenic loop in synucleinopathies. Cell 146:37-52.
Migdalska-Richards A, Schapira AH (2016) The relationship between glucocerebrosidase mutations and Parkinson disease. J Neurochem Suppl 1:77-90.
Mills K, Eaton S, Ledger V, Young E, Winchester B (2005) The synthesis of internal standards for the quantitative determination of sphingolipids by tandem mass spectrometry. Rapid Commun Mass Spectrom 19:1739-1748.
Montfort M, Chabás A, Vilageliu L, Grinberg D (2004) Functional analysis of 13 GBA mutant alleles identified in Gaucher disease patients: Pathogenic changes and “modifier” polymorphisms. Hum Mutat 23:567-575.
Ravikumar B, Sarkar S, Davies JE, Futter M, Garcia-arencibia M, Green-thompson ZW, Jimenez-sanchez M, Korolchuk VI, Lichtenberg M, Luo S, Massey DCO, Menzies FM, Moreau K, Narayanan U, Renna M, Siddiqi FH, Underwood BR, Winslow AR, Rubinsztein DC (2010) Regulation of mammalian autophagy in physiology and pathophysiology. Physiol Rev 90:1383-1435.
Rocha EM, Smith GA, Park E, Cao H, Brown E, Hallett P, Isacson O (2015a) Progressive decline of glucocerebrosidase in aging and Parkinson’s disease. Ann Clin Transl Neurol 2:433-438.
Rocha EM, Smith GA, Park E, Cao H, Graham A-R, Brown E, McLean JR, Hayes MA, Beagan J, Izen SC, Perez-Torres E, Hallett PJ, Isacson O (2015b) Sustained systemic glucocerebrosidase inhibition induces brain α-synuclein aggregation, microglia and complement C1q activation in mice. Antioxid Redox Signal 23:550-564.
Schapira AH (2015) Glucocerebrosidase and Parkinson disease: Recent advances. Mol Cell Neurosci 66:37-42.
Schöndorf DC, Aureli M, McAllister FE, Hindley CJ, Mayer F, Schmid B, Sardi SP, Valsecchi M, Hoffmann S, Schwarz LK, Hedrich U, Berg D, Shihabuddin LS, Hu J, Pruszak J, Gygi SP, Sonnino S, Gasser T, Deleidi M (2014) iPSC-derived neurons from GBA1-associated Parkinson’s disease patients show autophagic defects and impaired calcium homeostasis. Nat Commun 5:4028.
Shults CW (2006) Lewy bodies. Proc Natl Acad Sci U S A 103:1661-1668.
Sidransky E, Nalls MA, Aasly JO, Aharon-Peretz J, Annesi G, Barbosa ER, Bar-Shira A, Berg D, Bras J, Brice A, Chen CM, Clark LN, Condroyer C, De Marco EV, Dürr A, Eblan MJ, Fahn S, Farrer MJ, Fung HC, Gan-Or Z, et al. (2009) Multi-center analysis of glucocerebrosidase mutations in Parkinson disease. N Engl J Med 361:1651-1661.
Swan M, Saunders-Pullman R (2013) The association between beta-glucocerebrosidase mutations and parkinsonism. Curr Neurol Neurosci Rep 13.
Volles MJ, Lansbury PT Jr (2003) Zeroing in on the pathogenic form of alpha-synuclein and its mechanism of neurotoxicity in Parkinson’s disease. Biochemistry 42:7871-7878.
Wang IF, Guo BS, Liu YC, Wu CC, Yang CH, Tsai KJ, Shen CKJ (2012) Autophagy activators rescue and alleviate pathogenesis of a mouse model with proteinopathies of the TAR DNA-binding protein 43. Proc Natl Acad Sci U S A 109:15024-15029.
Watanabe Y, Tatebe H, Taguchi K, Endo Y, Tokuda T, Mizuno T, Nakagawa M, Tanaka M (2012) p62/SQSTM1-dependent autophagy of Lewy body-like α-synuclein inclusions. PLoS One 7:e52868.
Webb JL, Ravikumar B, Atkins J, Skepper JN, Rubinsztein DC (2003) Alpha-synuclein is degraded by both autophagy and the proteasome. J Biol Chem 278:25009-25013.
Zatloukal K, Stumptner C, Fuchsbichler A, Heid H, Schnoelzer M, Kenner L, Kleinert R, Prinz M, Aguzzi A, Denk H (2002) p62 Is a common component of cytoplasmic inclusions in protein aggregation diseases. Am J Pathol 160:255-263.
*Correspondence to: Kerri J. Kinghorn, M.D., Ph.D., k.kinghorn@ucl.ac.uk.
orcid: 0000-0003-2048-4332 (Kerri J. Kinghorn) 0000-0002-6324-2854 (Jorge Iván Castillo-Quan)
10.4103/1673-5374.202934
Accepted: 2017-03-06
杂志排行
中国神经再生研究(英文版)的其它文章
- Telomerase and mTOR in the brain: the mitochondria connection
- Impacts of the retinal environment and photoreceptor type on functional regeneration
- Tissue-type plasminogen activator is a homeostatic regulator of synaptic function in the central nervous system
- Novel insights into the role of NF-κB p50 in astrocytemediated fate specification of adult neural progenitor cells
- Anesthetic considerations for patients with acute cervical spinal cord injury
- Neuromelanin, one of the most overlooked molecules in modern medicine, is not a spectator
