Late cardioprotection of exercise preconditioning against exhaustive exercise-induced myocardial injury by up-regulatation of connexin 43 expression in rat hearts
2015-11-30KaiWangBaiChaoXuHaiYunDuanHuaZhangFuSongHu
Kai Wang, Bai-Chao Xu, Hai-Yun Duan, Hua Zhang, Fu-Song Hu
1College of Sports Science, Qufu Normal University, Qufu 273165, China
2Department of Physical Education, Hainan Medical University, Haikou 571100, China
3QiongZhou University Physical Test Room, Sanya 572022, China
1. Introduction
Cardiovascular disease, which is induced by ischemic, is the major cause of death in the industrialized world. Repeated transient ischemia, which is called ischemic preconditioning (IP), have been shown to protect myocardium against a subsequent more sustained ischemic insult[1-3]. Until now, many studies have evidenced that IP shows cardioprotection by enhancing myocardial tolerance to ischemia-reperfusion (I/R)injury and improving myocardial function[4,5]. Myocardium connexin 43 (Cx43)is a predominant protein forming gap junctions and non-junctional hemichannels in ventricular myocardium, through which ions and small molecules diffuse between cells[6]. It plays a key role in IP cardioprotection against I/R injury and IP have been indeed documented to affect the phosphorylation status of Cx43. Jain et al showed that IP attenuates ischemia-induced dephosphorylation of myocardial Cx43 and the resulting electrical uncoupling[7]. The preserved phosphorylation of Cx43 during IP may be related to the enhanced association of Cx43 with PKC and p38 mitogen activated protein kinases[8]. Furthermore, IP cardioprotection disappears in heterozygous Cx43-deficient mice[9,10], indicating that Cx43 is very important in relieving IP cardioprotection against I/R injury.Like IP cardioprotection, exercise preconditioning (EP), which is brief episode regular exercises, is also widely demonstrated to protect heart during an I/R insult[11-15]. At present, many researches about myocardial protection have focused especially on EP, which includes a biphasic protection manner, ie., early cardioprotection of exercise preconditioning (EEP)occurring immediately after the exercise and late cardioprotection of exercise preconditioning(LEP)developing 24 h post exercise. For example, EP can improve myocardial function and enhance myocardial tolerance to I/R injury.Its endogenous myocardial protection mechanisms may be closely related to channels opening, attenuation of apoptosis, and proteins activation closely[12,15-17]. EP was also found to markedly attenuate exhaustive exercise-induced myocardial injury, and PKC is probably involved in EP cardioprotection[18].
Although many studies have demonstrated the protective effect of EP against exhaustive exercise-induced myocardial injury,its mechanisms are still unclear. Considering the same powerful cardioprotective effects of both EP and IP as well as the key role of increased expression of Cx43 in IP cardioprotection, we hypothesized that Cx43 may involve in cardioprotective signaling transduction of EP. It has been observed that endurance exercise training preserves a higher expression level of Cx43 in heart under resting conditions[19]. Moreover, the expression level of Cx43 is down-regulated in trained mice during acute exercise[20]. However,to our knowledge, no research has been done to study the regulatory effects of Cx43 on EP cardioprotection against exhaustive exerciseinduced myocardial injury. Therefore, this study aimed to determine the relationship between Cx43 and LEP cardioprotection against exhaustive exercise-induced myocardial injury
2. Materials and methods
2.1. Rats
Eight-week-old adult male Sprague Dawley rats weighting 220–300 g were purchased from Chinese Academy of Sciences (Shanghai,China). All rats were housed in standard rat cages and maintained at a constant temperature and humidity with a 12 h:12 h light–dark cycle. They were fed and watered ad libitum. All animal care and experimental procedures were conducted in accordance with the Guiding Principles for the Care and Use of Animals in the Field of Physiological Sciences and approved by the Ethics Committee for Science Research of the Qufu Normal University.
2.2. Experimental protocol
All rats were given an adaptive training on a treadmill for 5 d, with an adaptive training velocity of 15 m/min and a time course of 10–20 min/d. After 1-day rest, all rats were randomly divided into four groups, eight in each group: (Ⅰ)sedentary control group (group C),the rats of which were placed on the treadmill without any treadmill exercise; (Ⅱ)exhaustive exercise group (group EE), rats of which run to exhaustion on the 0% grade treadmill at a speed of 35 m/min to induce myocardial injury; (Ⅲ)late exercise preconditioning group(group LEP), rats of which were allowed to run on the treadmill for four periods of 10 min, each at 30 m/min, with intervening periods of rest of 10 min at 0% grade. Exercise began and ended with 5-minute'warm up' and 'cool down' periods at 15 m/min and 0% grade.The rats were sacrificed 24 h after the exercise. (Ⅳ)late exercise preconditioning plus exhaustive exercise group (group LEP+EE),rats of which were treated as those in the group LEP, except that they run to exhaustion 24 h after the exercise and were sacrificed 0.5 h after the exhaustion.
Animals were anesthetized with trichloroacetaldehyde monohydrate(400 mg/kg intraperitoneally)and fixed in the dorsal position on an animal operation table. After abdominal cavity was quickly opened and heart was exposed, blood was drawn via inferior caval vein.Partial heart was rapidly excised, and left ventricle (LV)free wall was isolated at the level of the near apex for real-time fluorescent quantitative PCR and western blot analysis. Once the tissue was divided, the pieces were quick frozen in liquid nitrogen. The rest heart was exposed to perfusion fixation for in situ hybridization and immunohistochemistry.
2.3. Detection of serum cTnⅠ and NT-proBNP
The serum levels of cTnⅠ and NT-proBNP in the inferior caval vein blood samples were quantified. The serum cTnⅠ levels were determined by automated immunochemiluminescence on Access 2 immunoassay system (Beckman Coulter, USA), while the serum NT-proBNP levels were determined by using rat NT-proBNP ELISA kits (R&D Systems, Minneapolis, MN, USA)in accordance with the manufacturer's instructions. The sensitivity thresholds for cTnⅠ and NT-proBNP were 0.01 μg/L and 1 μg /L, respectively.
2.4. Hematoxylin basic fuchsin picric acid (HBFP)staining
Following deparaffinization and rehydration, tissue section was stained in alum hematoxylin for 5 min and differentiated in 1% acid alcohol for 4-6 s. After being stained in 0.1% basic fuchsin for 3 min, the section was rinsed with absolute acetone for 15-22 s. Then,it was differentiated in 0.1% picric acid in absolute acetone for 5-10 min. Finally, it was cleared with xylene and covered with a coverslip.The ischemic/hypoxic myocardial fibers stain a crimson red color under light microscope.
2.5. In situ hybridization
The tissue section was first deparaffinized and rinsed with distilled water and phosphate-buffered saline (PBS). After that, it was soaked in 3% hydrogen peroxide for 30 min, followed by pepsin digestion for 5-7 min. After the second washing with PBS and distilled water and prehybridization at 42 ℃ thermostat, the section was hybridized,blocked with serum, incubated, washed with PBS, colorated,counterstained with hematoxylin, dehydrated in graded series of ethanol and mounted. Serial adjacent sections were hybridized, and probe hybridization solution was replaced by PBS as a negative control. Cx43 in situ hybridization kit and DAB chromogenic reagents were purchased by Wuhan Boster Biological Engineering(Wuhan, China). The sequences of three intermediate mRNA fragment probes for rat Cx43 gene are as follows: 5'-TCT CTC ACG TGC GCT TCT GGG TCC TTC AGA TCA TA-3', 5'-CTC ATC CAG TGG TAC ATC TAT GGG TTC AGC TTG AG-3', and 5'-AAC AAT TCC TCG TGC CGC AAT TAC AAC AAG CAA GC-3'.
2.6. Real-time fluorescent quantitative PCR
Total RNA was isolated from LV myocardium using a commercial kit (RNeasy, Mini Kit, Qiagen)according to the manufacturer's instructions. Strand cDNA was synthesized with random hexanucleotides from 1.5 μg of total RNA using a reverse transcription system kit (SuperScriptTMⅢ Reverse Transcriptase,Invitrogen). Real-time quantitative PCR was done using Biorad's iQ 10 000× SYBR Green Supermix (Invitrogen). The primer sequences are as follows: 5'-AAA GGC GCG TTA AGG ATC GCG TG-3' (the first primer for Cx43 gene), 5'-GTC ATC AGG CCG AGG CCT-3'(the second primer for Cx43 gene), 5'-TGA CGT GGA CAT CCG CAA AG-3' (the first primer for GAPDH gene), and 5'-CTG GAA GGT GGA CAG CGC GAG G-3' (the second primer for GAPDH gene). The quality of the RT-PCR product was routinely checked by the thermal denaturation curve following RT-PCR reactions. The expression of RNA encoding the channel subunits was normalized to glyceraldehyde-3-phosphate dehydrogenase (GAPDH), and relative mRNA levels were quantified by the ΔCT method.
2.7. Western blotting
The LV tissue was homogenized in lysis buffer and centrifuged at 14 000×g for 15 min at 4 ℃. Then the supernatant was collected to get total Cx43. Electrophoresis was performed on 10% SDS-polyacrylamide gels to transfer proteins to 0.2 μm Sequi-Blot PVDF membranes (200 Ma, 4 ℃). After incubation with 5% BSA, the membranes were incubated with rabbit anti-rat Cx43 antibodies(1:5 000 dilution, Boster Biological Engineering, Wuhan, China).Rabbit anti-rat GADPH antibodies(1:10 000 dilution, Kang Biological Engineering, Shanghai, China)overnight at 4 ℃. After being washed, the membranes were incubated with horseradish peroxidase (HRP)-conjugated goat anti-rabbit IgG antibodies(1:5 000 dilution, Boster Biological Engineering, Wuhan, China).Results were visualized by the enhanced chemiluminescence method and evaluated by Image J software (NIH, Bethesda, MD, USA).
2.8. Immunohistochemistry
The tissue section was deparaffinized, rehydrated, washed,immersed in 3% hydrogen peroxide, and then digested with pepsin for antigen retrieval. After the blocking of unspecific binding by serum, the section was incubated with rabbit anti-rat Cx43 antibodies (1:200, Boster Biological Engineering, Wuhan, China)at 4 ℃ overnight. Then, it was detected using the streptavidinbiotin complex kit (1:200, goat anti-rabbit IgG, Boster Biological Engineering, Wuhan, China). Diaminobenzidine/peroxidase substrate was used to produce a brown-colored signal. The section was counterstained, dehydrated, cleared, and coverslipped. PBS was used to replace primary antibody and adjacent sections were used as negative control.
2.9. Statistical analysis
Results are expressed as mean±SD, and values were compared using the one-way analysis of variance (SPSS 10.0; SPSS, Chicago,IL). Upon confirmation of a significant main effect, individual differences were determined with post hoc analysis. A value of P<0.05 was considered significant.
3. Results
3.1. Establishment of animal model
Table 1 shows the serum levels of cTnⅠ and NT-proBNP. The serum levels of both cTnⅠ and NT-proBNP were significantly higher in the group EE than in the group C (P<0.05). However, the serum levels of cTnⅠ and NT-proBNP were significantly lower in the group LEP+EE than in the group EE (P<0.05). No significant difference in the serum cTnⅠ level was found between the group C and group LEP.
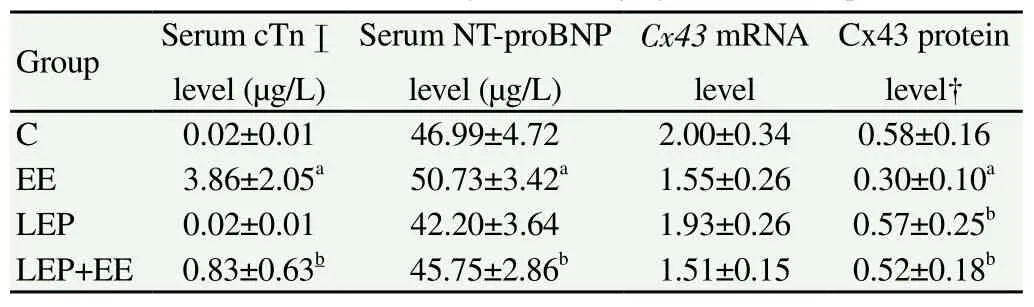
Table 1Effects of LEP on biomarkers of myocardial injury and Cx43 expression.
HBFP-stained cardiomyocytes of the LV free wall are represented in Figure 1. The ischemic cardiomyocytes were stained vivid crimson red, and the non-ischemic tissues were stained light brown.A light brown color was seen in the cardiomyocytes of the group C,while crimson red displayed in the majority of cardiomyocytes of the group EE. There is less crimson red color and the red patchy stain was scattered in the cardiomyocytes of the group LEP+EE, compared with that of the group EE. Thus, detection of serum cardiac biomarkers and an obvious HBFP staining pattern in cardiomyocytes confirmed the successful establishment of rat model of exhaustive exercise-induced myocardial injury and cardiopretection induced by exercise preconditioning.
3.2. Observation of myocardial Cx43 mRNA by in situ hybridization
The Cx43 mRNA in the cardiomyocytes of the LV anterior free wall at the level of the near apex was detected by in situ hybridization(Figure 2). The cardiomyocytes of the group C displayed a light brown color and granular distribution in the cytoplasm, while those of the group EE showed no obvious changes, and No obvious difference in the Cx43 mRNA was found between the group EE and group LEP+EE.
3.3. Changes of Cx43 mRNA and protein levels in myocardium
No miscellaneous peak and abnormal broadening of the main peak was found in the myocardial Cx43 melting curve, indicating that Cx43 was the only product of PCR amplicon. Table 1 represents the real-time fluorescent quantitative PCR results of myocardial Cx43 gene. No significant differences in the Cx43 mRNA level in myocardium were found between the four groups.
As shown by the western blotting (Table 1), the Cx43 protein level in myocardium was significantly lower in the group EE than in the group C (P<0.05), but it was significantly higher in the group LEP and group LEP+EE than in the group EE (P<0.05).
3.4. Immunohistochemical detection of myocardial Cx43
The Cx43 protein in cardiomyocytes was also detected by the immunohistochemical method (Figure 3). The Cx43 protein in the cardiomyocytes of the group C displayed a light brown color and the cardiomyocytes showed a rod-shaped or punctate distribution pattern in end-to-end connection (intercalated disc)and side to side connection. Similar appearance was found in the rest groups.Compared with the group C, the group EE showed only less rodshaped or punctate Cx43 in the cardiomyocytes. In contrast, the group LEP and group LEP+EE had considerable more rod-shaped or punctate Cx43 in the cardiomyocytes, compared with the group EE,and more Cx43 can be found in intercalated disc than in side to side connection of cardiomyocytes.
4. Discussion
In the present study, we evaluated the expression of Cx43 in the cardioprotection of LEP against exhaustive exercise induced myocardial injury, by using the established rat model. Exercise training, a way for progressive adaptation of the organism to physical activity, can make the subject to acquire new abilities and increase resistance for better performances. A lot of studies have also proved that endurance exercise is the practical and sustainable countermeasure which can provide cardioprotection against myocardial injury[21-25]. Moreover, some studies have documented that EP)which is brief episode regular exercise training, can protect the heart against a subsequent ischemic insult[12,14,16,18,26].Especially, EP cardiopretection against exhaustive exercise induced myocardial injury have been recently reported in rats trained in a once EP program. Some researchers have been proved that exhaustive exercise renders heart injury in rats, and EP markedly attenuates exhaustive exercise-induced myocardial injury[18,27,28].
Our study showed that the serum levels of NT-proBNP and cTnⅠ, which are biomarkers of myocardial injury, increased when the rats suffered the exhaustive exercise program, but their levels were reduced in the LEP group. In particular, a decrease in the serum levels of NT-proBNP and cTnⅠ appeared in the hearts of rats in the group LEP before the exhaustive exercise induced myocardial injury.The HBFP staining also demonstrated myocardial ischemia in the rat hearts, with the ischemic fibers being stained a vivid crimson color, and crimson red was found in majority of cardiomyocytes in hearts of rats suffering the exhaustive exercise program. However,less crimson red color and red patchy stain were found scattered in the cardiomyocytes of the group LEP before the exhaustive exercise. These results indicated that the successful establishment of animal model of exhaustive exercise-induced myocardial injury and cardiopretection induced by exercise preconditioning. Our results conform to those of Shen et al[18], and similar results have been documented by other researchers[29,30].
In recent years, many studies related to myocardial protection have focused especially on EP, and a few studies have stated the powerful beneficial effect of EP[15,17,31]. Nevertheless, its effectiveness and mechanisms have not yet been clearly demonstrated. Therefore, it is necessary to explore the protective effects of EP on exhaustive exercise-induced myocardial injury.
Functional gap junctions are necessary for the cell-to-cell electrical coupling which is required for synchronized cardiac contraction.Cx43, the main gap junction protein, is predominantly localized at the sarcolemma in myocardium of rats, and it also appears to be one vital element of the signal transduction cascade of the protection by preconditioning. Cx43 in cardioprotection induced by preconditioning to attenuate myocardial I/R injury have been probed in previous studies[8,32-34]. Schwanke et al pointed out that Cx43 knockout mice presented a disappeared cardioprotection by IP[10],which demonstrated that myocardial Cx43 plays a very important role in cardioprotection.
To our knowledge, the effect of EP on Cx43 expression in the cardioprotection against exhaustive exercise-induced myocardial injury have not been examined yet, and this is the first study to probe into the relationship between Cx43 and cardioprotection which is induced by LEP to alleviate exhaustive exercise-induced myocardial injury.
The RT-PCR results showed that no significant differences in the Cx43 mRNA levels were found between the groups, as supported by the in situ hybridization detection. Obvious decreased expression of Cx43 mRNA was found in trained mice during acute exercise[20,35],while chronic exercise training has no effect on the expression of Cx43 mRNA in intact blood vessels in vivo[36]. Therefore, we speculated that different exercise intensity has different effects on the expression of Cx43 mRNA. Further studies are needed to describe the related explicit mechanisms.
As demonstrated by the western blotting, the group EE had less myocardial Cx43 than the group C, while the group LEP and group LEP+EE had more myocardial Cx43 than the group EE. The results demonstrated that LEP could relieve exhaustive exercise-induced myocardial injury by maintaining the spatial distribution of Cx43-formed gap junction channels, through which possibly protecting electrophysiological properties of myocardial tissues. Similar results have been reported[34,37]. Thus, ridistribution of Cx43 and remolding of Cx43-based gap junction channels play a very important role in myocardium preconditioning. Different from the variation of Cx43 mRNA, the Cx43 protein level markedly decreased in the rats with exhaustive exercise-induced myocardial injury, which is consistent with the results of Tiscornia et al[20]. Cx43 expression is regulated by a variety of factors, and the reasons why Cx43 expression was decreased in the rats with exhaustive exercise-induced myocardial injury are unknown.
Cx43 becomes dephosphorylated with prolongation of ischemia[8,38], and its expression area and fluorescence intensity in hypertrophied myocardium are reduced after I/R, compared with non-I/R hypertrophied myocardium[34]. Therefore,dephosphorylation of Cx43 and reduced Cx43 expression in myocardium ischemia injury suggested that modification of gap junction conductance by dephosphorylation of Cx43 induces electrical uncoupling companied with ischemia.
Like I/R injury, exhaustive exercise may render myocardial ischemia injury, and the ischemia injury can decrease Cx43 expression, which suggested that both I/R injury and exhaustive exercise-induced myocardial injury may have the same mechanism to cause myocardial ischemia injury. In addition, LEP alleviated exhaustive exercise-induced myocardial injury, and a remarkable increased Cx43 expression was found in the LEP group. And a higher Cx43 protein level was found in the group LEP+EE than in the group EE. Bellafiore et al also reported similar results, and they found there is a progressive increase of Cx43 expression levels in response to endurance training that reached a peak in ventricular myocardium of mice exercised for 30 d[19].
IP can reduce Cx43 dephosphorylation compared to nonpreconditioned myocardium[7,8]and almost completely abolish the Cx43 dephosphorylation-related electrical uncoupling[39]in rat hearts[7]. Moreover, IP can alleviate ischemia/reperfusioninduced arrhythmias by up-regulating Cx43 expression[34]. The same results of Cx43 expression from cardioprotection of IP and LEP indicated the possible presence of similar signal transduction pathways. LEP can preserve phosphorylation of Cx43, and the putative mechanism may be related to an enhanced association of Cx43 with protein kinases such as PKC and PKA. Bao et al showed that the phosphorylation of Cx43 at Ser368 by PKC induces a closure of hemichannels, whereas the inhibition of PKC induces their opening[40]. In contrast, Ek-Vitorin et al pointed out that phosphorylation of Cx43 at Ser368 by PKC increases gap junctional permeability[41]indicating that Cx43 is a target protein of several kinases. Furthermore, Cx43 is associated not only with protein kinases but also with a variety of other proteins[42-44]. The finding that Cx43 can interact with several proteins and exert different functions indicated that Cx43 not only acts as a channel-forming protein but also is involved in intracellular signaling. More recently,the activation of protein kinase C has been reported as an important element in myocardial protection with EP[18]. In addition, we also found Cx43 was involved in myocardial protection with LEP. Hence,the relationship between Cx43 and protein kinase C in myocardial protection with LEP needs further exploration in future work.
In summary, the LEP attenuated the exhaustive exercise-induced myocardial injury in the cardioprotection. Cx43-formed channels are involved in protection against exercise-induced myocardial injury and exercise preconditioning. Cx43 was markedly up-regulated in the LEP rat myocardium. The exact underlying mechanism, how in fact Cx43 mediates protection, remains to be established. Cx43 can quickly translocate between several organelles under pathologic conditions such as ischemia and exercise. The changes in membrane connexin or gap junction plaque density and the relationship between Cx43 and protein kinase C in myocardial protection with LEP should be further elucidated by other techniques.
Conflict of interest statement
We declare that we have no conflict of interest.
This work was supported by Chinese Postdoctoral Science Foundation (No. 2014N560538), Hainan Province Colleges and Universities Scientific Research Project (No. Hnky2015-34), and Project of Natural Science Foundation of Hainan Province (314090).
[1]Gross GJ, Peart JN. KATP channels and myocardial preconditioning: An update. Am J Physiol-Heart Circ Physiol 2003; 285: H921-H930.
[2]Wang LG, Cherednichenko G, Hernandez L, Halow J, Camacho SA,Figueredo V, et al. Preconditioning limits mitochondrial Ca2+during ischemia in rat hearts: Role of K-ATP channels. Am J Physiol-Heart Circ Physiol 2001; 280: H2321-H2328.
[3]Stein AB, Bolli R, Guo YR, Wang OL, Tan W, Wu WJ, et al. The late phase of ischemic preconditioning induces a prosurvival genetic program that results in marked attenuation of apoptosis. J Mol Cell Card 2007; 42:1075-1085.
[4]Hefti MA, Harder BA, Eppenberger HM, Schaub MC. Signaling pathways in cardiac myocyte hypertrophy. J Mol Cell Card 1997; 29:2873-2892.
[5]Eisen A, Fisman EZ, Rubenfire M, Freimark D, McKechnie R,Tenenbaum A, et al. Ischemic preconditioning: Nearly two decades of research. A comprehensive review. Atherosclerosis 2004; 172: 201-210.
[6]Sáez JC, Berthoud VM, Brañes MC, Martínez AD, Beyer EC. Plasma membrane channels formed by connexins: their regulation and functions.Physiol Rev 2003; 83: 1359-400.
[7]Jain SK, Schuessler RB, Saffitz JE. Mechanisms of delayed electrical uncoupling induced by ischemic preconditioning. Circ Res 2003; 92:1138-1144.
[8]Schulz R, Gres P, Skyschally A, Belosjorow S, Konietzka I, Heusch G.Ischemic preconditioning preserves connexin 43 phosphorylation during sustained ischemia in pig hearts in vivo. FASEB J 2003; 17: 1355-1357.
[9]Schwanke U, Konietzka I, Duschin A, Li X, Schulz R, Heusch G. No ischemic preconditioning in heterozygous connexin43-deficient mice. Am J Physiol Heart Circ Physiol 2002; 283: H1740-H1742.
[10]Schwanke U, Li X, Schulz R, Heusch G. No ischemic preconditioning in heterozygous connexin 43-deficient mice a further in vivo study. Basic Res Cardio 2003; l98: 181-182.
[11]Kavazis AN. Exercise preconditioning of the myocardium. Sports Med 2009; 39: 923-935.
[12]Domenech R, Macho P, Schwarze H, Sánchez G. Exercise induces early and late myocardial preconditioning in dogs. Cardiovasc Res Aug 2000;55: 561-566.
[13]Parra VM, Macho P, Domenech RJ. Late cardiac preconditioning by exercise in dogs is mediated by mitochondrial potassium channels. J Cardiovasc Pharmacol 2010; 56: 268-274.
[14]Powers SK, Quindry YJ C, Kavazis AN. Exercise induced cardioprotection against myocardial ischemia-reperfusion injury. Free Radic Biol Med 2008; 44: 193-201.
[15]Yamashita N, Hoshida S, Otsu K, Asahi M, Kuzuya T, Hori M. Exercise provides direct biphasic cardioprotection via manganese superoxide dismutase activation. J Exp Med 1999; 189: 1699-1706.
[16]Sánchez G, Escobar M, Pedrozo Z, Macho P, Domenech R, Härtel R,et al. Exercise and tachycardia increase NADPH oxidase and ryanodine receptor-2 activity: possible role in cardioprotection. Cardiovasc Res 2008; 77: 380-386.
[17]Melling CW, Thorp DB, Milne KJ, Noble EG. Myocardial Hsp70 phosphorylation and PKC-mediated cardioprotection following exercise.Cell Stress Chaperones 2009; 14: 141-150.
[18]Shen YJ, Pan SS, Ge J, Hao Z. Exercise preconditioning provides early cardioprotection against exhaustive exercise in rats: potential involvement of protein kinase C delta translocation. Mol Cell Biochem 2012; 368: 89-102.
[19]Bellafiore M, Sivverini G, Palumbo D, Macaluso F, Bianco A, Palma A,et al. Increased cx43 and angiogenesis in exercised mouse hearts. Int J Sports Med 2007;28: 749-755.
[20]Tiscornia GC, Moretta R, Argenziano MA, Amorena CE, Garcia Gras EA. Inhibition of connexin43 in cardiac muscle during intense physical exercise. Scand J Med Sci Sports 2014; 24: 336-344.
[21]Brown DA, Chicco AJ, Jew KN, Johnson MS, Lynch JM, Watson PA,et al. Cardioprotection afforded by chronic exercise is mediated by the sarcolemmal, and not the mitochondrial, isoformof the KATP channel in the rat. J Physiol 2005; 569: 913-924.
[22]Ascensão A, Magalhães J, Soares JM, Ferreira R, Neuparth MJ, Marques F, et al. Endurance training limits the functional alterations of rat heart mitochondria submitted to in vitro anoxia-reoxygenation. Int J Cardiol 2006; 109: 169-178.
[23]Patil HR, O'Keefe JH, Lavie CJ, Magalski A, Vogel RA, McCullough PA. Cardiovascular damage resulting from chronic excessive endurance exercise. Mo Med 2012; 109: 312-321.
[24]Yankelson L, Sadeh B, Gershovitz L, Werthein J, Heller K, Halpern P, et al. Life-threatening events during endurance sports: is heat stroke more prevalent than arrhythmic death? J Am Coll Cardiol 2014; 64: 463-469.
[25]Li Y, Cai M, Cao L, Qin X, Zheng T, Xu X, et al. Endurance exercise accelerates myocardial tissue oxygenation recovery and reduces ischemia reperfusion injury in mice. PLoS One 2014; 9: e114205.
[26]Shen YJ, Pan SS, Zhuang T, Wang FJ. Exercise preconditioning initiates late cardioprotection against isoproterenol-induced myocardial injury in rats independent of protein kinase C. J Physiol Sci 2011; 61: 13-21.
[27]Hao Z, Pan SS, Shen YJ. Exercise preconditioning-induced late phase of cardioprotection against exhaustive exercise: possible role of protein kinase C delta. J Physiol Sci 2014; 64: 333-345.
[28]Hao Z, Pan SS, Shen YJ, Ge J. Exercise preconditioning-induced early and late phase of cardioprotection is associated with protein kinase C epsilon translocation. Circ J 2014; 78: 1636-1645.
[29]King DW, Gollnick PD. Ultrastructure of rat heart and liver after exhaustive exercise. Am J Physiol 1970; 218: 1150-1155.
[30]Li S, Zhong S, Zeng K, Luo Y, Zhang F, Sun X, et al. Blockade of NF-kappaB by pyrrolidine dithiocarbamate attenuates myocardial inflammatory response and ventricular dysfunction following coronary microembolization induced by homologous microthrombi in rats. Basic Res Cardiol 2010; 105: 139-150.
[31]Yellon DM, Downey JM. Preconditioning the myocardium: from cellular physiology to clinical cardiology. Physiol Rev 2003; 83: 1113-1151.
[32]Schulz R, Heusch G. Connexin43 and ischemic preconditioning.Cardiovasc Res 2006; 42: 213-227.
[33]Vetterlein F, Muhlfeld C, Cetegen C, Volkmann R, Schrader C, Hellige G. Redistribution of connexin43 in regional acute ischemic myocardium:influence of ischemic preconditioning. Am J Physiol Heart Circ Physiol 2006; 291: H813-H819.
[34]Chen Z, Luo H, Zhuang M, Cai L, Su C, Lei Y, et al. Effects of ischemic preconditioning on ischemia/reperfusion-induced arrhythmias by upregulatation of connexin43 expression. J Cardiothorac Surg 2011; 6:80.
[35]Chang Y, Yu T, Yang H, Peng Z. Exhaustive exercise-induced cardiac conduction system injury and changes of cTnT and Cx43. Int J Sports Med 2015; 36: 1-8.
[36]Bearden SE, Linn E, Ashley BS, Looft-Wilson RC. Age-related changes in conducted vasodilation: effects of exercise training and role in functional hyperemia. Am J Physiol Regul Inter Comp Phtsiol 2007; 293:R1717-R1721.
[37]Garcia-Dorado D, Ruiz-Meana M, Padilla F, Rodriguez-Sinovas A,Mirabet M. Gap junction-mediated intercellular communication in ischemic preconditioning. Card Res 2002; 55: 456-465.
[38]Miura T, Ohnuma Y, Kuno A, Tanno M, Ichikawa Y, Nakamura Y, et al.Protective role of gap junctions in preconditioning against myocardial infarction. Am J Physiol 2004; 286: 214-221.
[39]Beardslee MA, Lerner DL, Tadros PN, Laing JG, Beyer EC, Yamada KA,et al. Dephosphorylation and intracellular redistributoin of ventricular connexin43 during electrical uncoupling induced by ischemia. Circ Res 2000; 87: 656-662.
[40]Bao X, Reuss L, Altenberg GA. Regulation of purified and reconstituted connexin43 hemichannels by protein kinase C-mediated phosphorylation of Serine 368. J Biol Chem 2004; 279: 20058-20066.
[41]Ek-Vitorin JF, King TJ, Heyman NS, Lampe PD, Burt JM. Selectivity of connexin 43 channels is regulated through protein kinase C-dependent phosphorylation. Circ Res 2006; 98: 1498-1505.
[42]Giepmans BN. Gap junctions and connexin-interacting proteins.Cardiovasc Res 2004; 62: 233-245.
[43]Akiyama M, Ishida N, Ogawa T, Yogo K, Takeya T. Molecular cloning and functional analysis of a novel Cx43 partner protein CIP150. Biochem Biophys Res Commun 2005; 335: 1264-1271.
[44]Matsuda T, Fujio Y, Nariai T, Ito T, Yamane M, Takatani T, et al.N-cadherin signals through Rac1 determine the localization of connexin43 in cardiac myocytes. J Mol Cell Cardiol 2006; 40: 495-502.
杂志排行
Asian Pacific Journal of Tropical Medicine的其它文章
- Antidiabetic and antioxidant activities of Nypa fruticans Wurmb. vinegar sample from Malaysia
- Anti-inflammatory and analgesic activities with gastroprotective effect of semi-purified fractions and isolation of pure compounds from Mediterranean gorgonian Eunicella singularis
- Natural products: Perspectives in the pharmacological treatment of gastrointestinal anisakiasis
- Upregulated hepatic expression of mitochondrial PEPCK triggers initial gluconeogenic reactions in the HCV-3 patients
- Analysis of human B cell response to recombinant Leishmania LPG3
- Rifabutin reduces systemic exposure of an antimalarial drug 97/78 upon co- administration in rats: an in-vivo & in-vitro analysis
