Prognostic significance and relationship of SMAD3 phosphoisoforms and VEGFR-1 in gastric cancer: A clinicopathological study
2024-01-26ShiLinLvPeiGuoJunRongZouRenShengChenLingYuLuoDeQiangHuang
Shi-Lin Lv,Pei Guo,Jun-Rong Zou,Ren-Sheng Chen,Ling-Yu Luo,De-Qiang Huang
Abstract BACKGROUND The TGF-β/SMAD3 and VEGFR-1 signaling pathways play important roles in gastric cancer metastasis.SMAD3 phosphorylation is a crucial prognostic marker in gastric cancer.AIM To determine the prognostic value and relationship of SMAD3 phospho-isoforms and VEGFR-1 in gastric cancer.METHODS This was a single-center observational study which enrolled 98 gastric cancer patients and 82 adjacent normal gastric tissues from patients aged 32-84 years (median age 65) between July 2006 and April 2007.Patients were followed up until death or the study ended (median follow-up duration of 28.5 mo).The samples were used to generate tissue microarrays (TMAs) for immunohistochemical (IHC) staining.The expressions of TGF-β1,pSMAD3C(S423/425),pSMAD3L(S204),and VEGFR-1 in gastric cancer (GC) tumor tissue and normal tissue were measured by IHC staining using TMAs obtained from 98 GC patients.Prognosis and survival information of the patients was recorded by Outdo Biotech from May 2007 to July 2015.The relationship between TGF-β1,pSMAD-3C(S423/425),pSMAD3L(S204),and VEGFR-1 protein expression levels was analyzed using Pearson's correlation coefficient.The relationship between protein expression levels and clinicopathological parameters was analyzed using the Chi-squared test.A survival curve was generated using the Kaplan-Meier survival analysis.RESULTS TGFβ-1 and VEGFR-1 expression was significantly upregulated in gastric cancer tissue compared to adjacent noncancerous tissue.The positive expression of phosphorylated isoforms of Smad3 varied depending on the phosphorylation site [pSMAD3C(S423/425): 51.0% and pSMAD3L(S204): 31.6%].High expression of pSMAD-3L(S204) was significantly correlated with larger tumors (P=0.038) and later N stages (P=0.035).Additionally,high expression of VEGFR-1 was closely correlated with tumor size (P=0.015) and pathological grading (P=0.013).High expression of both pSMAD3L(S204) and VEGFR-1 was associated with unfavorable outcomes in terms of overall survival (OS).Multivariate analysis indicated that high expression of pSMAD3L(S204) and VEGFR-1 were independent risk factors for prognosis in GC patients.VEGFR-1 protein expression was correlated with TGFβ1 (r=0.220,P=0.029),pSMAD3C(S423/425) (r=0.302,P=0.002),and pSMAD3L(S204) (r=0.201,P=0.047),respectively.Simultaneous overexpression of pSMAD3L(S204) and VEGFR-1 was associated with poor OS in gastric cancer patients.CONCLUSION Co-upregulation of pSMAD3L(S204) and VEGFR-1 can serve as a predictive marker for poor gastric cancer prognosis,and pSMAD3L(204) may be involved in enhanced gastric cancer metastasis in a VEGFR-1-dependent manner.
Key Words: Gastric cancer;pSMAD3L(S204);pSMAD3C(S423/425);Survival;Transforming growth factor-β1;VEGFR-1
INTRODUCTION
Gastric cancer (GC) is one of the leading causes of cancer-related deaths worldwide[1].Despite improvements in therapeutic strategies,GC,especially advanced GC,has a high mortality rate[2,3].Tumor metastasis,in which angiogenesis plays a key role,is the primary cause of the poor prognosis[4].Therefore,targeting tumor angiogenesis is an essential strategy in the treatment of GC[5].
VEGFR-1 (Vascular endothelial growth factor receptor-1) is a tyrosine kinase receptor for VEGF-A,VEGF-B,and placental growth factor (PlGF)[6].VEGFR-1 is mainly expressed in endothelial cells,smooth muscle cells,and macrophages,where it is involved in regulating cell migration and survival[6].Additionally,increasing evidence has associated VEGFR-1 with enhanced invasiveness of various malignancies such as glioma[7],non-small cell lung cancer[8],colorectal cancer[9],liver cancer[10],and oral squamous cell carcinoma[11].However,the functional role and prognostic significance of VEGFR-1 in GC are still not fully understood.
TGF-β is a pleiotropic cytokine that regulates various biological functions,including angiogenesis,tumorigenesis,proliferation,differentiation,and fibrosis[12].The TGF-β/SMAD axis is a primary signaling pathway that promotes epithelial-mesenchymal transition (EMT)[13].TGF-β can mediate SMAD2 and SMAD3 phosphorylation,allowing them to affect the transcription of genes related to tumor invasiveness[14,15].SMAD3,a key component of this pathway,contains highly conserved MAD homology domains (MH1 and MH2) connected by a less conserved junction region (LR).This linker region contains four proline-directed kinase phosphorylation sites: Thr179,Ser204,Ser208,and Ser213.Phosphorylation of SMAD3 leads to the formation of three phosphorylated forms: Carboxyl-terminal phosphorylated SMAD3 (pSMAD3C),linker-phosphorylated SMAD3 (pSMAD3L),and carboxyl-linker-dual phosphorylated SMAD3 (pSMAD3L/C)[16].
The carboxyl-terminal phosphorylation of SMAD3 is essential for TGF-β-mediated signal transduction.Blocking or attenuating carboxyl-terminal phosphorylation of SMAD3 at Ser423/425 significantly inhibits TGF-β expression levels in cancer cells[17,18].In lung cancer,TGF-β-induced epithelial cell transformation depends on pSMAD3C[19].High levels of pSMAD3L have been considered a primary risk factor in the clinical management of liver cancer,and inhibitors targeting the JNK/pSMAD3L axis have been shown to suppress hepatocellular carcinoma (HCC) progression[20].Mutations in the phosphorylation sites of SMAD3 LR and carboxyl-terminus can impair primary tumor growth and metastasis,respectively,in breast cancer[21].Furthermore,TGF-β promotes cancer cell growth in a SMAD3L/C-dependent manner[22].These observations highlight the importance of SMAD3 phosphorylation in cancer cell behavior,suggesting a need for better understanding of SMAD3 phospho-isoforms in GC.
It has been shown that TGF-β1 elevates VEGF and VEGFR-1 expression through the SMAD pathway in mouse dendritic cells[23].Additionally,PGF1 expressed on the surface of lung cancer cells can bind to VEGFR-1 on macrophages,resulting in increased TGF-β1 production and enhanced lung cancer angiogenesis[24].These findings suggest that the TGF-β1/SMAD3 pathway is involved in VEGFR-1-promoted tumor angiogenesis.Moreover,the phosphorylation status at both the carboxyl-terminus and LR determines the activity of SMAD3[25].For instance,TGF-β can phosphorylate SMAD3 at its LR,and consequently regulate its function[26].However,the association of pSMAD3L with VEGFR-1 is unclear in GC.
To examine the relationship between SMAD3 phospho-isoforms and VEGFR-1,we measured the expression levels of pSMAD3C(S423/425),pSMAD3L(S204),VEGFR-1,and TGF-β1 in GC and investigated how their expression was related to the clinicopathological features and prognosis of patients with GC.Moreover,to investigate the association between TGF-β/SMADs signaling and VEGFR-1 protein expression,we analyzed the correlation between pSMAD3C(S423/425),pSMAD3L(S204),TGF-β1,and VEGFR-1 expression,and explored the significance of pSMAD3L(S204) and VEGFR-1 coexpression in GC.
MATERIALS AND METHODS
Patients and specimens
Ninety-eight GC tissue samples (n=98) and adjacent normal gastric tissues (n=82) were postoperatively collected from patients aged 32-84 years (median age of 65 years) between July 2006 and April 2007 by Outdo Biotech (Shanghai,China).Samples were used to generate the tissue microarrays for immunohistochemical (IHC) staining.Prognosis and survival information were recorded by Outdo Biotech from May 2007 to July 2015.TNM staging was conducted according to the 7thedition criteria of the American Joint Committee on Cancer[27].Histological grading of tumors followed the WHO guidance on digestive system tumor classification[28].The study protocol was approved by the hospital ethics committee.
IHC staining
IHC was performed as previously described[29].Briefly,paraffin-embedded sections (4 µm) were dewaxed and hydrated,followed by heat-induced antigen retrieval in citrate buffer (pH 6.0).Subsequently,endogenous peroxidases were blocked with H2O2,and sections were incubated with primary antibodies at 4°C overnight: anti-TGF-β1 (Abcam,66043,1:150),anti-pSMAD3C(S423/425) (Abcam,52903,1:150),anti-pSMAD3L(S204) (Thermo-Fisher Scientific,2816414,1:150),anti-VEGFR-1 (Abcarta,PA359,1:100).The next day,sections were washed with PBS three times and incubated with antirabbit HRP-conjugated IgG secondary antibody (Zsbio,China) for 1 h at 37°C.Sections stained with PBS were used as the negative control.Subsequently,sections were washed and incubated with peroxidase substrate (Zsbio,China) for 20 min at 37°C.Finally,nuclei were counterstained with hematoxylin,and sections were processed for dehydration and covered with a mounting medium.
Two pathologists blinded to the study scored the degree of staining according to the German semi-quantitative statistical methods[30].The overall staining intensity and percent of positively stained cells were used to reflect protein expression levels.
Overall staining intensity was divided into four categories: No staining (0);light yellow staining (1);yellow staining (2);and dark yellow/brown staining (3).The percent positive staining was defined as the percent of glandular epithelial cells within the tissue that were positively stained on the slide.The corresponding scoring criteria are as follows: <5% (0);5%-25% (1);25%-50% (2);50%-75% (3);>75% (4).The overall staining intensity and percent positive staining scores were combined to describe the level of protein expression: negative (0-2),+(3-5),++(6-8),+++(9-12).Ultimately,we classified all of the tissues into a low expression group (-or +) and high expression group (++or +++)[29].
Statistical analysis
Statistical analyses were performed using SPSS 11.5 (SPSS Inc,Chicago,United States).The relationship between protein expression levels [TGF-β1,pSMAD3C (S423/425),pSMAD3L (S204),and VEGFR-1] and clinicopathological parameters were analyzed using the Chi-squared test.A survival curve was generated using the Kaplan-Meier survival analysis.Univariate and multivariate survival analyses were performed using the Cox proportional hazards regression model.The relationship between TGF-β1,pSMAD3C(S423/425),pSMAD3L(S204),and VEGFR-1 protein expression levels was analyzed using Pearson’s correlation coefficient.The receiver operating characteristic (ROC) curve was generated using the R package “pROC”.Nomograms and calibration plots were constructed using the R package “rms”.APvalue of <0.05 was considered statistically significant.
RESULTS
pSMAD3C(S423/425), TGF-β1, pSMAD3L(S204), and VEGFR-1 expression in GC
The expression and localization of phosphorylated SMAD3 isoforms,TGF-β1,and VEGFR-1 were evaluated in GC and adjacent normal tissues (Figure 1).TGF-β1 exhibited dominant cytoplasmic localization,while the pSMAD3C(S423/425) was predominantly observed in the nucleus.Statistical results showed significantly higher levels of pSMAD3C(S423/425) (P<0.05) and TGF-β1 (P<0.001) in GC tissues compared to adjacent normal tissues (Table 1).Notably,minimal pSMAD3L(S204) was detected in normal gastric tissue,but its immune reactivity was observed exclusively in the nuclei of cancer cells.IHC analysis revealed that 31.6% (31/98) of the GC samples exhibited pSMAD3L(S204) positivity.Moreover,VEGFR-1 displayed immune reactivity in both the cell membrane and cytoplasm of tumor cells,as well as tumor stromal vessels.Semi-quantitative analysis demonstrated significantly higher VEGFR-1 expression in GC compared to adjacent normal tissues (P<0.001) (Table 1).
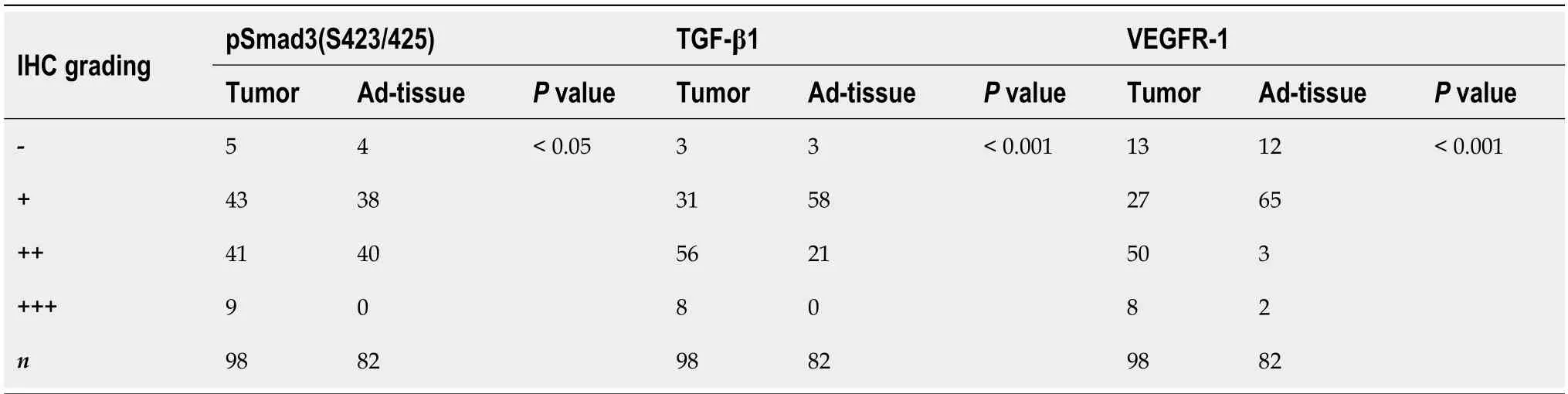
Table 1 Differential expression of pSmad3(S423/425),TGF-β1 and VEGFR-1 protein in gastric cancer tissue

Figure 1 The expression of TGF-β1,pSMAD3C,pSMAD3L,and VEGFR-1 in gastric cancer. Representative immunohistochemical images showing the expression of TGF-β1,pSMAD3C,pSMAD3L,and VEGFR-1 in cancerous (tumor tissue) and normal (Ad-tissue) gastric tissue.The right panel provides an enlarged view of the specific features highlighted in the rectangular image presented in the left panel.(Scale bar,100 μm).
Correlation between the expression of pSMAD3C(S423/425), TGF-β1, pSMAD3L(S204), and VEGFR-1, and clinicopathologic characteristics of GC
We investigated the correlation of pSMAD3C(S423/425),TGF-β1,pSMAD3L(S204),and VEGFR-1 and clinicopathological factors of the 98 GC patients (Table 2).The results revealed significant associations between the expression levels of these proteins and specific clinicopathological features.Elevated expression of pSMAD3C(S423/425) was significantly associated with distant metastasis (M staging) (P=0.042).High expression of TGF-β1 was correlated with advanced T stage (P=0.042) and tumors located in the gastric antrum (P=0.022).Furthermore,higher expression of pSMAD3L(S204) was positively associated with larger tumors (P=0.038) and advanced N stage (P=0.035).In addition,increasedexpression levels of VEGFR-1 were associated with larger tumors (P=0.015) and higher tumor pathological grades (P=0.013).However,no significant correlations were found between the expression levels of these proteins (pSMAD3C(S423/425),TGF-β1,pSMAD3L(S204),and VEGFR-1) and other clinicopathological parameters (P=>0.05).
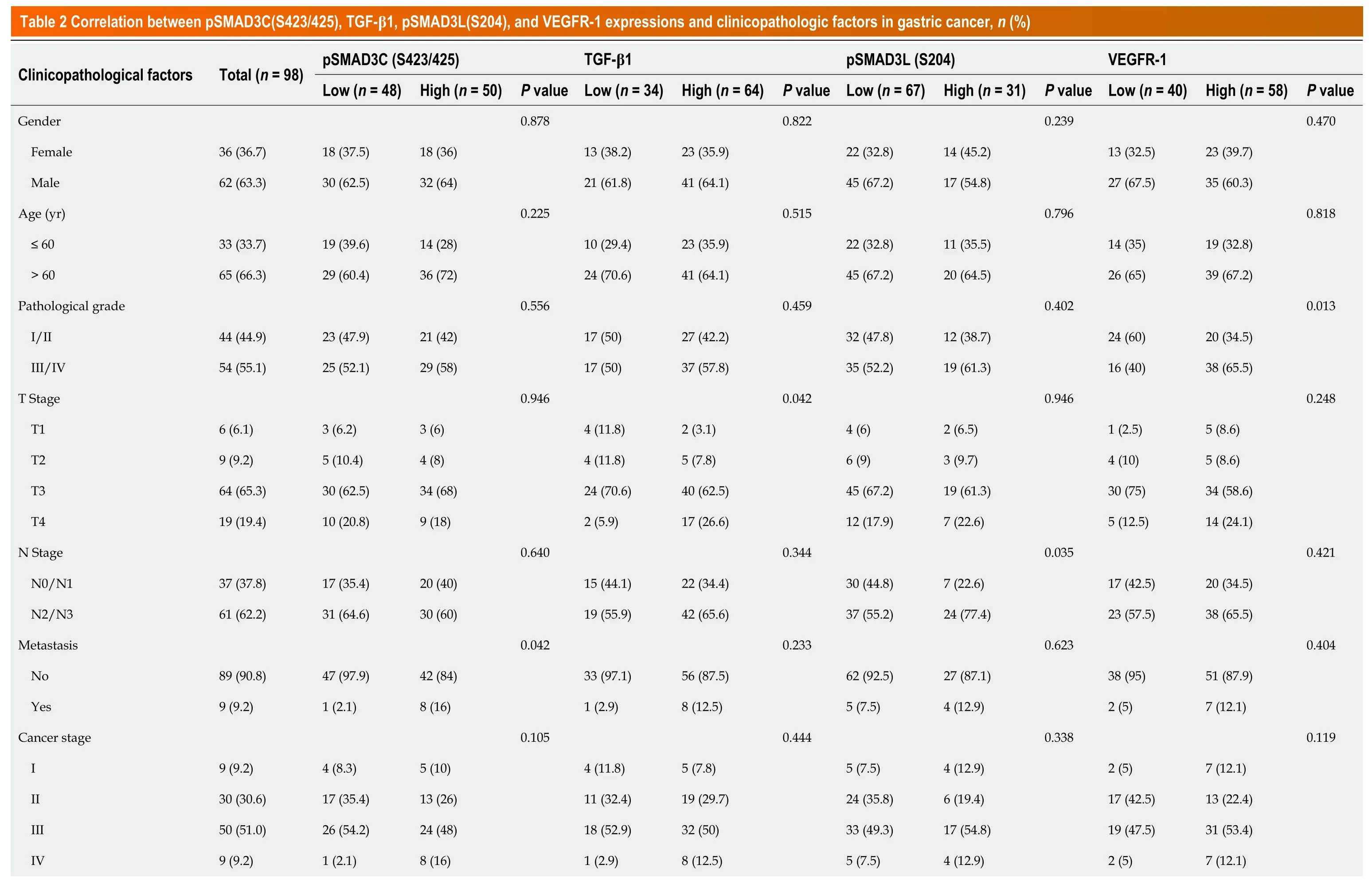
Correlation between pSMAD3C(S423/425), TGF-β1, pSMAD3L(S204), and VEGFR-1 expressions in GC
Quantification of protein levels revealed that expression of TGF-β1,pSMAD3C(S423/425),and pSMAD3L(S204) was positively correlated with VEGFR-1 (r=0.220,P=0.029;r=0.302,P=0.002;r=0.201,P=0.047,respectively).These results indicate that SMAD3 phosphor-isoforms may regulate TGF-β1-mediated tumor migration in a VEGFR-1-dependent manner (Table 3).

Table 3 Correlation between pSMAD3C(S423/425),TGF-β1 pSMAD3-S204 and VEGFR-1 expression in gastric cancer (n=98)
Prognostic value of pSMAD3L(S204) and VEGFR-1 expression
We assessed the overall survival (OS) of 98 GC patients and found that the 3-year and 5-year OS rates were 45.9% and 38.8%,respectively.Kaplan-Meier analysis revealed a significant association between higher expression of pSMAD3L(S204) and reduced OS (P<0.001),and a significant correlation between higher expression of VEGFR-1 and decreased OS (P=0.002).Notably,although patients with higher expression of TGF-β1 exhibited a trend of decreased OS,this trend did not reach statistical significance (P=0.227).Interestingly,the expression of pSMAD3C(S423/425) did not significantly impact patient prognosis (Figure 2).

Figure 2 The prognostic value of TGF-β1,pSMAD3C,pSMAD3L,and VEGFR-1 in gastric cancer. A-D: The correlation between the expression of(A) pSMAD3C (S423/425),(B) pSMAD3L(S204),(C) TGF-β1,and (D) VEGFR-1 and the overall survval of gastric cancer patients was analyzed using Kaplan-Meier analysis.
To further investigate the impact of pSMAD3L(S204) and VEGFR-1 expression on other prognostic factors,subgroup survival analyses were performed.The results showed that elevated pSMAD3L(S204) was significantly associated with OS in different cancer stages (I/II stage,P=0.001;III/IV stage,P=0.028),TNM stages (M0,P<0.001;T1/T2,p=0.028;T3/T4,P<0.001;N1/N2,P=0.006;N3/N4,P=0.022),and pathological grades (Grade I/II,p=0.009;Grade III/IV,P=0.005) (Figure 3A-I).The results also showed that elevated VEGFR-1 was significantly associated with OS of cancer stages (III/IV stage,P=0.008),TNM stages (M0,P=0.01;T3/T4,P=0.002;N3/N4,P=0.008),and pathological grades (Grade III/IV,P<0.001) (Figure 3J-N).

Figure 3 Evaluating the prognostic significance of pSMAD3L(S204) and VEGFR-1 expression in gastric cancer subgroups. A-I: Kaplan-Meier analysis revealed that higher pSMAD3L(S204) expression was associated with shorter overall survival (OS) in several subgroups of gastric cancer (GC) patients,including T1/T2 (A),T3/T4 (B),N1/2 (C),N3/4 (D),M0 (E),Grade I/II (F),Grade III/IV (G),Clinical stage I/II (H),and Clinical stage III/IV (I);J-N: Moreover,higher VEGFR-1 expression was associated with shorter OS in subgroups of GC patients with T3/T4 (J),N3/4 (K),M0 (L),Grade III/IV (M),and Clinical stage III/IV (N).
In addition,we investigated the impact of pSMAD3L(S204) and VEGFR-1 expression on GC prognosis through univariate and multivariate Cox regression analyses.The results showed that tumor size,N stage,M stage,pSMAD3L(S204) expression,VEGFR-1 expression,and TNM stage were significantly associated with poor prognosis (P<0.05) (Table 4).Furthermore,multivariate survival analysis revealed that pSMAD3L(S204) and VEGFR-1 expression,as well as tumor size,were independent prognostic factors in GC patients (P<0.05) (Table 4).
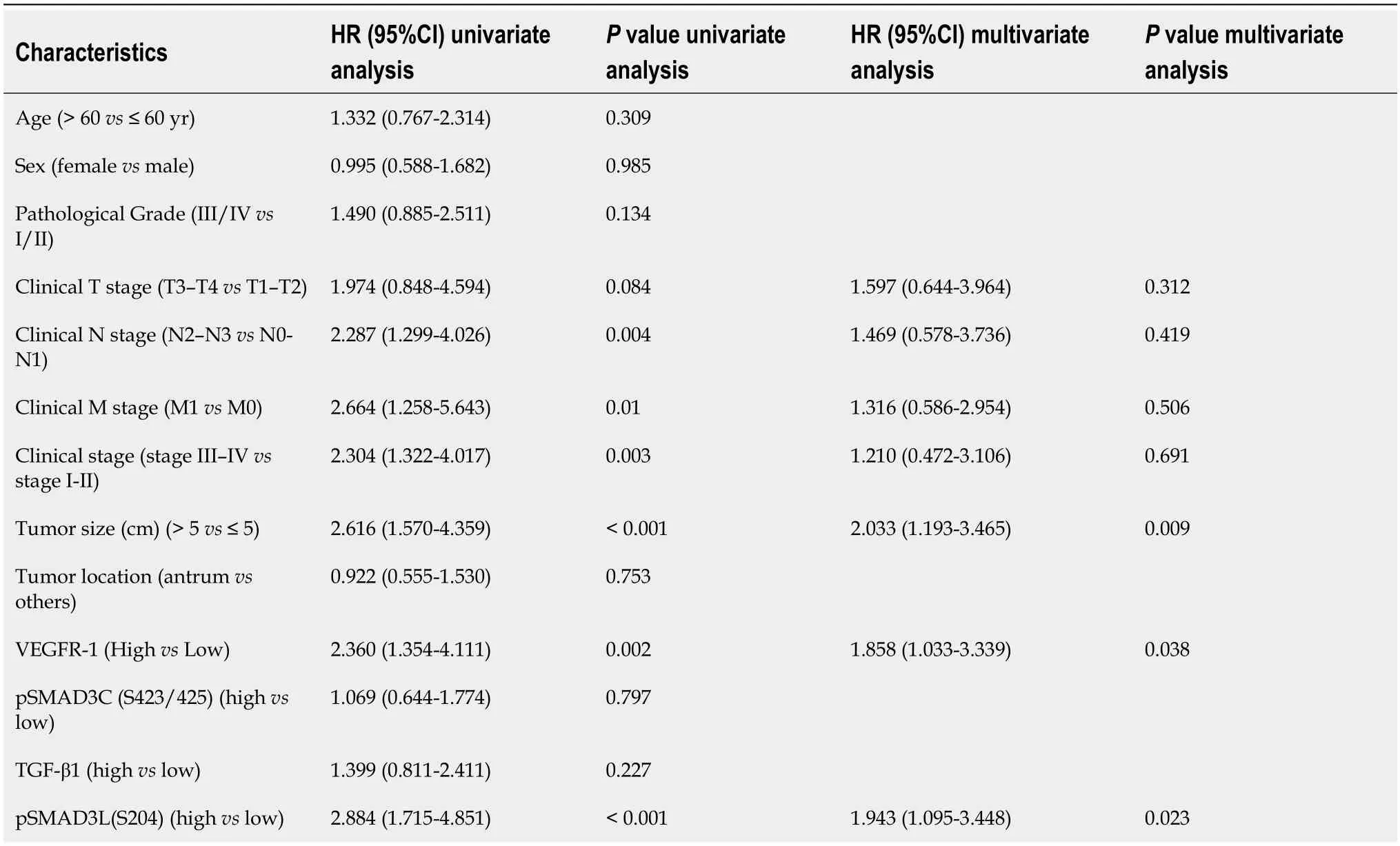
Table 4 Association of various factors with overall survival as determined by Cox regression
To predict OS,a prognostic nomogram was constructed incorporating pSMAD3L(S204) and VEGFR-1 expression levels,as well as tumor size,based on the statistically significant independent prognostic factors identified by the Cox regression analysis.The efficiency of the nomogram was assessed using calibration curves,which demonstrated optimal predictions for 1-,2-,and 3-year clinical outcomes (Figure 4).The C-index was 0.690 (0.656-0.724),indicating its potential as a prognostic tool.
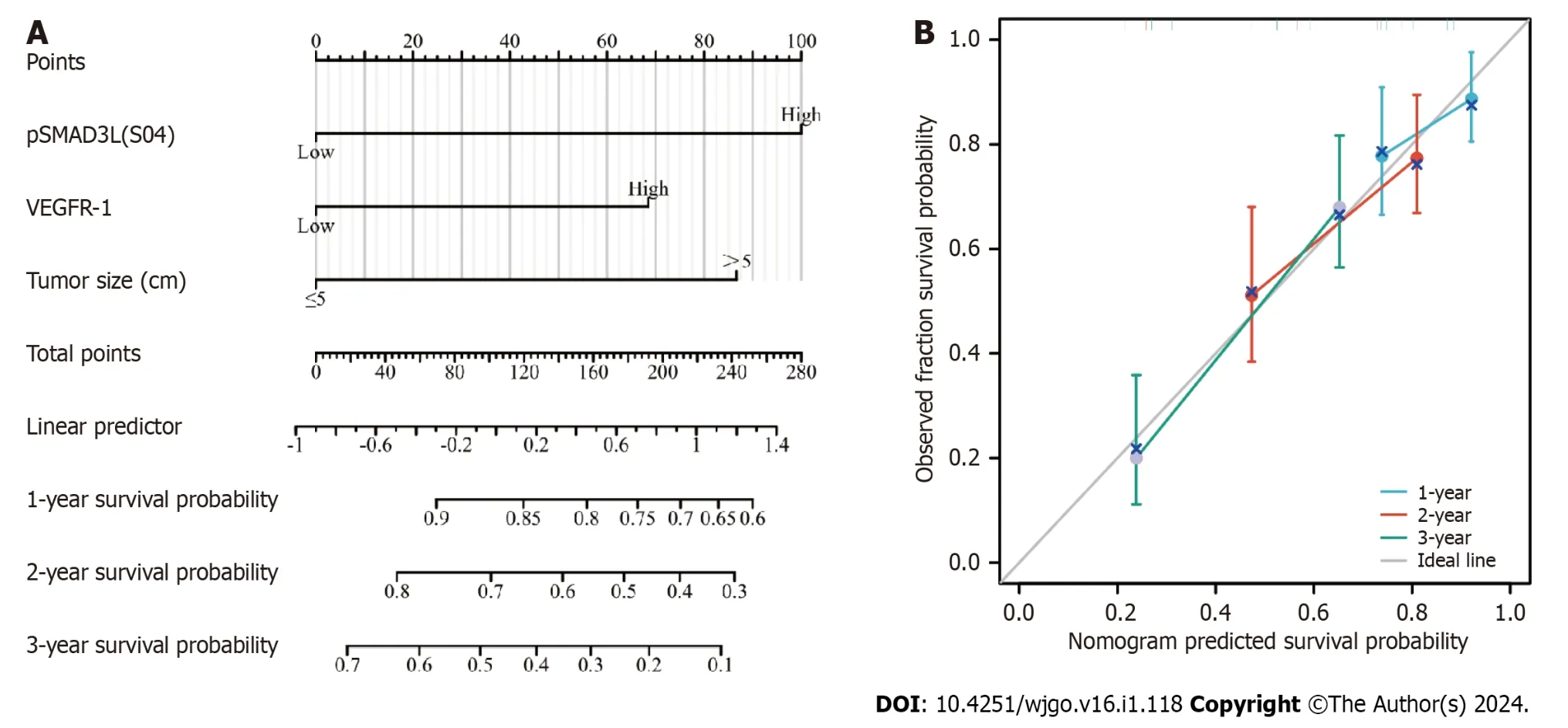
Figure 4 Construction and validation of the nomogram based on pSMAD3L(S204) and VEGFR-1 expression for predicting 1-year,2-year,and 3-year overall survival in gastric cancer patients. A: Nomogram;B: Calibration curve.
Prognostic values of co-expression of pSMAD3L(S204) with VEGFR-1, pSMAD3C(S423/425), or TGF-β1
To investigate the prognostic value of co-expression patterns involving pSMAD3C(S204) and TGF-β1,VEGFR-1,or pSMAD3L(S423/425),we conducted a retrospective evaluation of the OS rate in 98 patients,with a median follow-up duration of 28.5 mo.The patients were categorized into four groups based on their expression levels of pSMAD3L(S204) and VEGFR-1: pSMAD3L low/VEGFR-1 low (n=33),pSMAD3L High/VEGFR-1 low (n=7),pSMAD3L low/VEGFR-1 high (n=34),and pSMAD3L high/VEGFR-1 high (n=24).Our results revealed that patients with high levels of both pSMAD3L(S204) and VEGFR-1 expression had the poorest OS (Figure 5A;P<0.001).Furthermore,when comparing the OS of this group to that of all other biomarker combinations,the difference remained significant (Figure 5B;P<0.001).
Similarly,we stratified the cohort based on the levels of pSMAD3L(S204) and pSMAD3C(S423/425) and assessed the OS of each group.Our findings showed that the OS of patients with pSMAD3C High/pSMAD3L High was significantly lower than that of patients with pSMAD3C High/pSMAD3L Low,pSMAD3C Low/pSMAD3L High,or pSMAD3C Low/pSMAD3L Low (Figure 5C;P<0.001).Additionally,when comparing the OS of this group to that of all other biomarker combinations,the difference remained significant (Figure 5D;P<0.001).
Furthermore,patients with high expression levels of both pSMAD3L and TGF-β1 (pSMAD3L high/TGF-β1 high) exhibited shorter OS compared to patients with pSMAD3L High/TGF-β1 Low,pSMAD3L Low/TGF-β1 High,or pSMAD3L Low/TGF-β1 Low) (Figure 5E and F).
These findings suggest that the co-expression patterns of pSMAD3C(S204) and TGF-β1,VEGFR-1,or pSMAD3L(S423/425) have prognostic i mplications and may provide valuable insights into the OS of GC patients.
Diagnostic value of TGF-β1 expression in gastric cancer
We further interrogated the diagnostic value of TGF-β1 expression in GC by generating a ROC curve.The results show that the area under the curve (AUC) of TGF-β1 is 0.704.We also analyzed the diagnostic power of TGF-β1 expression in different stages and showed that the AUC of TGF-β1 in stages I,II,III,and IV were 0.658,0.683,0.704,and 0.818,respectively (Figure 6).

Figure 6 Diagnostic value of TGF-β1 expression in gastric cancer patients. Receiver operating characteristic (ROC) curves were generated based on TGF-β1 protein expression levels in patient samples.A: ROC curve for TGF-β1 expression in normal and tumor tissues;B-E: Subgroup analysis for stages I,II,III,and IV,respectively.
DISCUSSION
TGF-β signaling can promote the malignant progression of cancer by promoting EMT,which facilitates tumor cell invasion and chemoresistance[31].TGF-β1 is abundantly expressed in various cancers,and high levels of TGF-β1 usually predict adverse clinical outcomes[32,33].Therefore,targeting TGF-β production is an i mportant anticancer strategy.
Our results showed that TGF-β1 expression was significantly higher in GC than in normal tissues and that high TGF-β1 expression was associated with T stages (P=0.042),consistent with the findings of previous studies[32,33].We also found that TGF-β1 expression is discriminative in all stages of GC,but especially in late-stage tumors (stages III and IV),indicating its prognostic value.TGF-β signaling requires phosphorylation at Ser423/425 in SMAD3.Therefore,inhibiting or attenuating SMAD3(Ser423/425) phosphorylation can significantly inhibit TGF-β1 expression levels in cancer cells.Our study found that pSMAD3C(S423/425) was highly expressed in GC,and its high expression was associated with the M stage (P=0.042).
The SMAD3 phospho-isoform,pSMAD3L,has been reported to play a pro-cancer role in different types of cancer[34,35],but its clinical significance in GC remains unclear.Based on the results of the IHC staining,we found that 31.6% (31/98) of GC patients expressed a high level of pSMAD3L(S204) and that the increased levels of pSMAD3L(S204) were associated with larger tumors (P=0.038) and advanced N stage (P=0.035).Moreover,our univariate and multivariate COX regression analysis revealed,for the first time,that the expression of pSMAD3L(S204) is an independent predictor of GC prognosis.This is consistent with previous studies that have shown that SMAD3 phospho-isomers have also been associated with poor prognosis in colorectal cancer,HCC,and esophageal squamous cell carcinoma by promoting tumor growth and metastasis[35-37].Our results also showed that high expression of pSMAD3L(S204) was associated with low OS,suggesting that pSMAD3L(S204) is a molecular biomarker for outcome prediction in GC.
The SMAD3 LR has been reported to mediate TGF-β signaling transduction as TGF-β-mediated SMAD3 carboxylterminus phosphorylation is a precursor for TGF-β-induced SMAD3 LR phosphorylation[26].pSMAD3L/C has been demonstrated to promote tumor progressionviathe facilitation of tumor cell metastasis and proliferation[34].Our study found that the OS of GC patients was not associated with increased TGF-β1 and pSMAD3C(S423/425),but rather with concomitant elevation of pSMAD3L(S204)/pSMAD3C(S423/425),or pSMAD3L(S204)/TGF-β1.These observations are consistent with previous studies[34,38].In addition,strong pSMAD3L/C expression was observed in tumors with EMT features[34] and also led to the activation of alternate oncogenic pathways in colorectal cancer.This suggests that the pSMAD3L/C pathway simultaneously promotes tumor-promoting TGF-β signaling and activates invasive behavior[38].
It has been reported that functional VEGFR-1 in cancer cells promotes invasion and growth and that high expression of VEGFR-1 is associated with poor prognosis in HCC[39].Our study found that VEGFR-1 expression was significantly higher in GC than in normal tissues.We also found that high expression of VEGFR-1 was associated with larger tumors (P=0.015) and higher tumor pathological grades (P=0.013).Univariate and multivariate COX regression analyses highlighted that the expression of VEGFR-1 is an independent predictor of prognosis in patients with GC.These results suggest that VEGFR-1-related signaling may play a tumor-promoting role in GC.Of note,TGF-β1 effectively induces VEGFR-1 expression in vascular endothelial cells,thus preventing oxygen-induced vascular lossin vivo[40].Moreover,upregulated TGF-β was observed in HCC and could induce the secretion of VEGF,the major activator of angiogenesis[41].However,the relationship between the TGF-β/SMAD3 pathway and VEGFR-1 in GC is unclear.Our results showed that TGF-β1,pSMAD3L(S204),and pSMAD3C(S423/425) were positively correlated with VEGFR-1 expression.These results suggest that VEGFR-1 may be associated with TGF-β1/SMAD3-mediated angiogenesis.Our study revealed a significant association between high expression of VEGFR-1 and poor prognosis in patients with GC.Notably,when both pSMAD3L(S204) and VEGFR-1 are upregulated,OS rates are significantly reduced,providing evidence for a potential interplay between these proteins in cancer progression.Evaluating the expression pattern of these two proteins together may help predict prognosis.
CONCLUSION
In conclusion,this study is the first to characterize the relationship between pSMAD3L(S204) and VEGFR-1 expression in GC.Our current results suggest that these proteins may lead to GC metastasis by promoting angiogenesis.We also found that the simultaneous upregulation of pSMAD3L(S204) and VEGFR-1 was an unfavorable prognostic factor for GC and that targeting VEGFR-1 and pSMAD3L(S204) could represent a promising treatment strategy for GC.Our study provides novel insights into the tumorigenesis of GC and has identified potential therapeutic targets for this malignancy.
ARTICLE HIGHLIGHTS
Research background
Gastric cancer is a significant health concern and understanding the molecular mechanisms underlying its progression and metastasis is crucial.The TGF-β/SMAD3 and VEGFR-1 signaling pathways have been identified as important players in gastric cancer metastasis,with SMAD3 phospho-isoforms emerging as a critical prognostic marker.
Research motivation
Given the clinical significance of gastric cancer,there is a pressing need to elucidate the prognostic value and interrelationship of SMAD3 phospho-isoforms and VEGFR-1 in gastric cancer.This study aims to address this gap in knowledge.
Research objectives
The primary objective of this study was to determine the prognostic significance and relationship between SMAD3 phospho-isoforms (pSMAD3C(S423/425) and pSMAD3L(S204)) and VEGFR-1 in gastric cancer.
Research methods
This observational single-center study enrolled 98 gastric cancer patients and 82 adjacent normal gastric tissues.Immunohistochemical staining and tissue microarrays were utilized to measure the expression levels of TGF-β1,pSMAD3C(S423/425),pSMAD3L(S204),and VEGFR-1.Prognosis and survival information of the patients were recorded,and statistical analyses including Pearson's correlation coefficient,Chi-squared test,and Kaplan-Meier survival analysis were employed.
Research results
The study revealed that TGF-β1 and VEGFR-1 expression were significantly upregulated in gastric cancer tissue compared to adjacent non-cancerous tissue.High expression of pSMAD3L(S204) and VEGFR-1 was associated with larger tumors,later N stages,tumor size,and pathological grading,and was correlated with unfavorable overall survival outcomes.Multivariate analysis identified high expression of pSMAD3L(S204) and VEGFR-1 as independent risk factors for the prognosis of GC patients.
Research conclusions
The co-upregulation of pSMAD3L(S204) and VEGFR-1 may serve as a predictive marker for poor gastric cancer prognosis,suggesting a potential role for pSMAD3L(204) in enhanced gastric cancer metastasis in a VEGFR-1-dependent manner.
Research perspectives
Future research could focus on elucidating the underlying molecular mechanisms linking SMAD3 phospho-isoforms and VEGFR-1 in gastric cancer metastasis,potentially paving the way for targeted therapeutic interventions.
FOOTNOTES
Author contributions:Huang DQ and Lv SL conceived and designed the study,analyzed and interpreted the data,and wrote the manuscript;Guo P,Zou JR,Luo LY and Chen RS collected clinical samples and completed the related experiments;all authors approved the final manuscript.
Supported byNational Nature Science Foundation of China,No.82060450,No.82360517,No.81460374,and No.31 460304;and Nature Science Foundation of Jiangxi Province of China,No.20232BAB206086,No.20192BAB205072,No.20203BBGL73206,No.2017BCB23086,No.2017BAB205062,and No.20181BAB205050.
Institutional review board statement:This study involving patient participants was reviewed and approved by the Ethics Committee of Shanghai Outdo Biotech Company (Shanghai,China) (NO: SHYJS-CP-1801009).
Informed consent statement:Written informed consent was obtained from all participants.
Conflict-of-interest statement:The authors declare that they have no competing interests.
Data sharing statement:The datasets used and/or analyzed during the current study are available from the corresponding author on reasonable request.
STROBE statement:The authors have read the STROBE Statement—checklist of items,and the manuscript was prepared and revised according to the STROBE Statement—checklist of items.
Open-Access:This article is an open-access article that was selected by an in-house editor and fully peer-reviewed by external reviewers.It is distributed in accordance with the Creative Commons Attribution NonCommercial (CC BY-NC 4.0) license,which permits others to distribute,remix,adapt,build upon this work non-commercially,and license their derivative works on different terms,provided the original work is properly cited and the use is non-commercial.See: https://creativecommons.org/Licenses/by-nc/4.0/
Country/Territory of origin:China
ORCID number:Ling-Yu Luo 0000-0002-1090-0198;De-Qiang Huang 0000-0003-3467-5359.
S-Editor:Lin C
L-Editor:A
P-Editor:Zhao S
杂志排行
World Journal of Gastrointestinal Oncology的其它文章
- Comprehensive analysis of the role of ubiquitin-specific peptidases in colorectal cancer: A systematic review
- Colonoscopy plays an important role in detecting colorectal neoplasms in patients with gastric neoplasms
- Analysis of the potential biological value of pyruvate dehydrogenase E1 subunit β in human cancer
- Colorectal cancer’s burden attributable to a diet high in processed meat in the Belt and Road Initiative countries
- Emerging role of liquid biopsy in rat sarcoma virus mutated metastatic colorectal cancer: A case report
- Comprehensive evaluation of rare case: From diagnosis to treatment of a sigmoid Schwannoma: A case report
