The evaluation of biological productivity by triple isotope composition of oxygen trapped in ice-core bubbles and dissolved in ocean:a review
2022-06-23ZHOUYaqianPANGHongxiHUHuantingYANGGuangHOUShugui
ZHOU Yaqian, PANG Hongxi*, HU Huanting, YANG Guang & HOU Shugui
The evaluation of biological productivity by triple isotope composition of oxygen trapped in ice-core bubbles and dissolved in ocean:a review
ZHOU Yaqian1, PANG Hongxi1*, HU Huanting2, YANG Guang1& HOU Shugui2
1Key Laboratory of Coast and Island Development of Ministry of Education, School of Geography and Ocean Science, Nanjing University, Nanjing 210023, China;2Institute of Oceanography, Shanghai Jiao Tong University, Shanghai 200240, China
The17O anomaly of oxygen (Δ17O, calculated from δ17O and δ18O) trapped in ice-core bubbles and dissolved in ocean has been respectively used to evaluate the past biosphere productivity at a global scale and gross oxygen production (GOP) in the mixed layer (ML) of ocean. Compared to traditional methods in GOP estimation, triple oxygen isotope (TOI) method provides estimates that ignore incubation bottle effects and calculates GOP on larger spatial and temporal scales. Calculated from TOI of O2trapped in ice-core bubbles, the averaged global biological productivities in past glacial periods were about 0.83–0.94 of the present, and the longest time record reached 400 ka BP (thousand years before the present). TOI-derived GOP estimation has also been widely applied in open oceans and coastal oceans, with emphasis on the ML. Although the TOI method has been widely used in aquatic ecosystems, TOI-based GOP is assumed to be constant at a steady state, and the influence of physical transports below the ML is neglected. The TOI method applied to evaluate past total biospheric productivity is limited by rare samples as well as uncertainties related to O2consumption mechanisms and terrestrial biosphere’s hydrological processes. Future studies should take into account the physical transports below the ML and apply the TOI method in deep ocean. In addition, study on the complex land biosphere mechanisms by triple isotope composition of O2trapped in ice-core bubbles needs to be strengthened.
triple oxygen isotope, ice-core bubbles, dissolved oxygen in ocean, biological productivity
1 Introduction
Biospheric productivity plays a vital role in controlling the concentration of carbon dioxide in the atmosphere, which influences the global carbon cycle, and subsequently causes global climate change. Both terrestrial and oceanic biological production are primary mechanisms for global oxygen production and carbon uptake. Because the oceans sink around 30% of anthropogenic carbon dioxide emission (IPCC, 2014), evaluating marine photosynthetic productivity is key to understand the global carbon cycle at present and predict the response of carbon cycle to climate forcing in the future.
There are several traditional methods used to assess the biosphere productivity. For instance, H218O spike incubation (Grande et al., 1989) used labeled H218O to estimate GOP based on the evolution of18O2. Because of the large enrichments in18O, this method is easier than triple oxygen isotope (TOI) method in tracing natural variations and is less affected by photosynthetic fractionation (Bender et al., 1987; Stanley and Howard, 2013). However, it is susceptible to sampling, manipulation and containment effects, and it alone cannot characterize respiratory mechanisms in aquatic O2uptake (Luz et al., 2002; Manning et al., 2007a; Tobias et al., 2007; Staehr et al., 2012; Hotchkiss and Hall, 2014). Light or dark incubation (Harris et al., 1989) experiment is an O2-based in vitro approach that is convenient for sampling and is widely applied to assess oceanic production, but its assumption of equal dark and light O2uptake rates will bring about large errors (Manning et al., 2007a). The14C in vitro incubation (Nielsen, 1952) is one of the oldest and most widely used method to quantify primary productivity (Bender et al., 1987; Manning et al., 2017b). However, uncertainties of the14C method remain regarding to whether it reflects the natural environment (“bottle effect”) (Peterson, 1980; Harrison and Harris, 1986) and to its near-instantaneous measurements (6–24 h) (Marra, 2007), as well as to the ambiguity between net community production and gross primary production (Bender et al., 1999). Remote sensing data and satellite algorithms (Behrenfeld and Falkowski, 1997) can estimate marine photosynthetic O2production globally with high spatial and temporal resolution (Juranek and Quay, 2013), but it is lacking in field data for validation, and is confined to measurement of primary production in the surface layer. Fast Repetition Rate Fluorometry (FRRF) (Suggett et al., 2001; Moore et al., 2003) enables high-frequency real-time calculation of primary production, but the assumptions of the FRRF still contain large uncertainties (Fujiki et al., 2008). Having understood the strengths and shortcomings of traditional methods, here we review the newly-developed TOI approach.
Over the past two decades, the triple isotope composition of oxygen (16O,17O and18O) trapped in ice-core bubbles and dissolved oxygen in ocean have been respectively used to evaluate the past global biosphere productivity (including both terrestrial and oceanic biosphere) (Luz et al., 1999; Blunier et al., 2002, 2012) and gross oxygen production (GOP) in the mixed layer (ML) of ocean (Luz and Barkan, 2000; Sarma et al., 2005; Juranek and Quay, 2010; Prokopenko et al., 2011; Bender et al., 2016). Paleo-atmospheric O2and dissolved O2bear the isotopic fractionation from both stratosphere and biosphere, in which oxygen isotopes are fractionated in a mass-independent way and a mass-dependent way, respectively. By measuring Δ17O (defined in section 2.1, equation (2)) in marine dissolved oxygen and paleo O2occluded in ice-core bubbles, accompanied with air-sea gas transfer rates and atmospheric CO2concentration, one can determine the marine photosynthetically produced oxygen and the global biological productivity in the past. TOI method has several advantages in assessing biospheric productivity. First, at a steady state, it reflects community productivity over the residence time (1–3 weeks) of dissolved O2(Reuer et al., 2007; Li et al., 2019), improving the temporal scale of primary productivity assessment. Second, the TOI method can be used in oceanic zones over a wide range of longitudes and latitudes, improving the spatial scale of marine productivity observations (Juranek and Quay, 2013). Also, TOI method can be applied in fresh water ecosystems (Jurikova et al. 2016; Howard et al. 2020). Finally, the calculation of Δ17O is independent on respiration (Quay et al., 1993; Hendricks et al., 2005), which diminishes errors resulted from complex respiratory mechanisms. Although TOI method also has shortcomings, such as uncertainties from analytical errors, gas exchange coefficients (), and gas separation procedures, it is still prospective and worthy of further study in terms of its wide application in assessing marine and global biological productivity. With this in mind, we hope to provide a review of how this approach is applied to evaluation of biological productivity and issues that can be paid attention to in the future.
2 Principles
2.1 Fractionation mechanisms and calculation of Δ17O
Oxygen isotopes of16O,17O,18O have abundances of 99.758%, 0.038% and 0.204%, respectively (Blunier et al., 2002). δ17O and δ18O are used to denote the deviation of measured oxygen isotopes from the standard. Atmospheric O2is usually chosen as the standard for TOI method (Barkan and Luz, 2003). The δ is defined as:

Whererefers to the ratio of heavy isotope (17O or18O) to the light isotope (16O).Subscripts sam and stdrefer to sample and standard, respectively. δ17O and δ18O are expressed in per mil (‰).
Δ17O is denoted as the degree of oxygen isotopic anomaly off mass-dependent fractionation. It is a very small value, usually multiplied by 106, and represented in per meg (1 per meg = 0.001‰). Δ17O of standard air O2is nil. Although represented in several ways (Kaiser, 2011), Δ17O can be defined most commonly as equation 2 (Miller, 2002; Luz and Barkan, 2005).

λ is the mass-dependent fractionation slope, which varies slightly from 0.506 to 0.521 in different biological and isotopic fractionation processes (Luz and Barkan, 2005) (as is shown in Table 1). Most processes in nature discriminates against17O by about 0.52 times as much as it discriminates against18O in a mass-dependent way (Thiemens and Meagher, 1984; Miller, 2002). However, photochemical processes (as follows) occurring in the stratosphere among O2, CO2and O3result in low δ17O and δ18O of O2in a mass-independent way (Thiemens and Heidenreich, 1983; Thiemens et al., 1991; Yung et al., 1991), and the λanomalously turns to 1.7 (Lämmerzahl et al., 2002; Boering et al., 2004).

Table 1 λ for different mass-dependent processes associated with biological productivity
Notes:aAngert et al. (2003);bHelman et al. (2005);cSarma et al. (2005);dLandais et al. (2006).
O2+UV→ 2O,
O + O2→ O3,
O3+ UV → O2+ O (1D),
CO2+ O (1D) → CO3,
CO3→ CO2+ O (3P),
O + O → O2.
Where the UV is ultraviolet, O (1D) is an excited oxygen atom, and O (3P) is a transient oxygen atom.
High-precision determination of λ can help tracing small Δ17O variations (Assonov and Brenninkmeijer, 2005). Studies that analyzed isotopic composition of atmospheric oxygen in ice-core bubbles preferred to use λ = 0.516, while in marine dissolved oxygen preferentially used λ = 0.518 (Luz and Barkan, 2005; Barkan and Luz, 2011).
2.2 Assessing past global biospheric production through TOIs in ice-core bubbles
Gas phase photochemical reactions in stratosphere imparts mass-independently fractionated O2to the atmosphere, while respiration removes O2and photosynthesis replaces the ambient anomalous atmospheric O2with mass dependent fractionated O2. Relative rates of biological O2and stratospheric O2can determine the relationship between δ17O and δ18O of atmospheric O2(Luz et al., 1999; Blunier et al., 2002). Δ17O calculated from paleo atmospheric O2accompanied with data on the atmospheric CO2concentration can be used to infer variations in past biosphere productivity at the global scale. At a steady state, the oxygen isotopic mass balance is represented as equation 3 (Landais et al. 2007a, 2007b):

Where Δ17Obioand Δ17Ostratare Δ17O of biological O2flux (Fbio) and stratospheric O2flux (strat), respectively. Δ17Oatmis measured from air samples. The ratio of biospheric O2production in the past to the present is calculated from equation 4 (Landais et al., 2007a, 2007b):

Here, subscripts bio, strat, atm, past, prst represent biosphere, stratosphere, atmosphere, the past and the present, respectively. Δ17Oatm,prstis defined as 0, and Δ17Oatm,pastis measurable. The former part in the right side of equation 4 can be represented as known CO2concentrations ratio of the past to the present (Luz et al., 1999). Equation 4 is now simplified as an equation relevant to Δ17Obio,prstand Δ17Obio,past, which are determined from both terrestrial (Δ17Oterr)and oceanic (Δ17Oocean)biosphere (equation 5; Landais et al., 2007a).
, (5)
Whereoceanandterrrepresent the fluxes of oceanic and terrestrial oxygen production, respectively.ocean/terrratio in ocean and terrestrial biosphere varied from 0.45 to 0.59 (Bender et al., 1994; Blunier et al., 2002; Hoffmann et al., 2004). The modern Δ17Ooceanis around 249 meg−1(Luz and Barkan, 2000). More details on calculating Δ17Oterrand Δ17Ooceanin the past and Δ17Oterrat present can be found in Landais et al. (2007a).
2.3 Calculating gross oxygen production through the TOIs of dissolved oxygen in ocean
TOI in the oceanic ML is affected by photosynthesis, respiration, air-sea gas exchange and physical transports among water masses. Respiration effect is eliminated because of bearing the same λ (0.518) as Δ17O calculation. Physical transports in the ML are assumed to be neglected. Therefore, the magnitude of the Δ17O in dissolved oxygen is determined by the relative rates of photosynthetic O2to air-sea exchange O2. Dissolved O2produced only from photosynthetic splitting of water bears the same isotopic composition as the seawater (Guy et al., 1989; Yakir et al., 1994), and results in a high Δ17O(Δ17Op)to 249±15 meg−1(Luz and Barkan, 2000). Dissolved O2in equilibrium with atmospheric O2bears a low Δ17O (Δ17Oeq) due to air-sea gas exchange. Luz and Barkan (2009) concluded that Δ17Oeqis positively relative to water temperature (Δ17Oeq=0.6×+ 1.8). Measured Δ17O of dissolved oxygen in ML (Δ17Odiss) falls between the two end-members (van der Meer, 2015; Nicholson et al., 2014). One can thus calculate GOP from Δ17Odiss, along with the parameterizedestimated from wind speed (Watson et al., 1991; Behrenfeld and Falkowski, 1997; Nightingale et al., 2000; Sweeney et al., 2007; Ho et al., 2011).

Here, [O2]eqis the equilibrium concentration of O2(Garcia and Gordon, 1992; Hamme and Emerson, 2004). First proposed by Luz and Barkan (2000), equation (6) is sensitive to errors in Δ17Odiss, Δ17Oeq, and especially in. Thus, Prokopenko et al. (2011) proposed the equation (7), which decreases errors for GOP calculation.

Where the subscripts p, diss and eq refer to O2produced by photosynthesis, dissolved in mixed layer and equilibrated with air, respectively. GOP values calculated by equation (7) are marked with * in Table 3.
3 Methods
Air samples from ice-core bubbles and sea water are collected in pre-evacuated 125 cm3and 500 mL glass flasks sealed with the same Louwers Hapert® O-ring stopcocks. Seawater samples are pre-poisoned with 100 μL of HgCl2saturated solution to avoid biological activity. After 24 hours’ equilibration at room temperature, the water is sucked out leaving only headspace gases inside (Emerson et al., 1995; Sarma et al., 2003; Ash et al., 2020). O2and Ar are separated from air samples through the separation line in Figure 1.
More details of automatic gas separation can be found in Barkan and Luz (2003). Briefly, H2O, N2O and CO2are cryogenically removed by T1 and T2 (Dewar flasks with liquid nitrogen) in 5 min, leaving N2, O2and Ar trapped by molecular sieves at −196°C in T3. Carried by pure helium gas, N2is separated through the chromatographic column (GC) in T4, leaving O2and Ar mixture trapped in T5. By heating the molecular sieves in T5, the gas mixture is then eluted and transferred to the collection finger immersed in T6.
The mixed O2and Ar are analyzed in the dual-inlet mass spectrometry by simultaneously measuring/32, 33, and 34 (Abe and Yoshida, 2003). The average standard analytical errors ofd17O,d18O and Δ17O are about 0.006‰, 0.003‰ and 7 meg−1(standard deviation), respectively (Luz et al., 1999; Sarma et al., 2005; Juranek and Quay, 2010; Munro et al., 2013; Keedakkadan and Abe, 2015).
4 Progress on research of triple isotopes of O2 trapped in ice-core bubbles and dissolved in ocean
4.1 Assessing past global biological productivity through triple isotopes of O2 trapped in ice-core bubbles
The triple isotopic composition of O2from paleo air occluded in ice-core bubbles can be used to estimate the rates of total biological productivity in the past to the present, although uncertainties still remain for terrestrial productivity measurement (Luz et al., 1999; Blunier et al., 2002; Angert et al., 2003; Landais et al., 2007a, 2007b). Here we select records from the Vostok and Greenland Ice Sheet Project 2 (GISP2) as representatives of ice cores in Antarctic and Arctic. The ratios of GOP in the past to the present and Δ17O values of paleo-atmospheric O2are shown in Table 2, and temporal variations of Δ17O values in past glacial and interglacial times are shown in Figure 2.
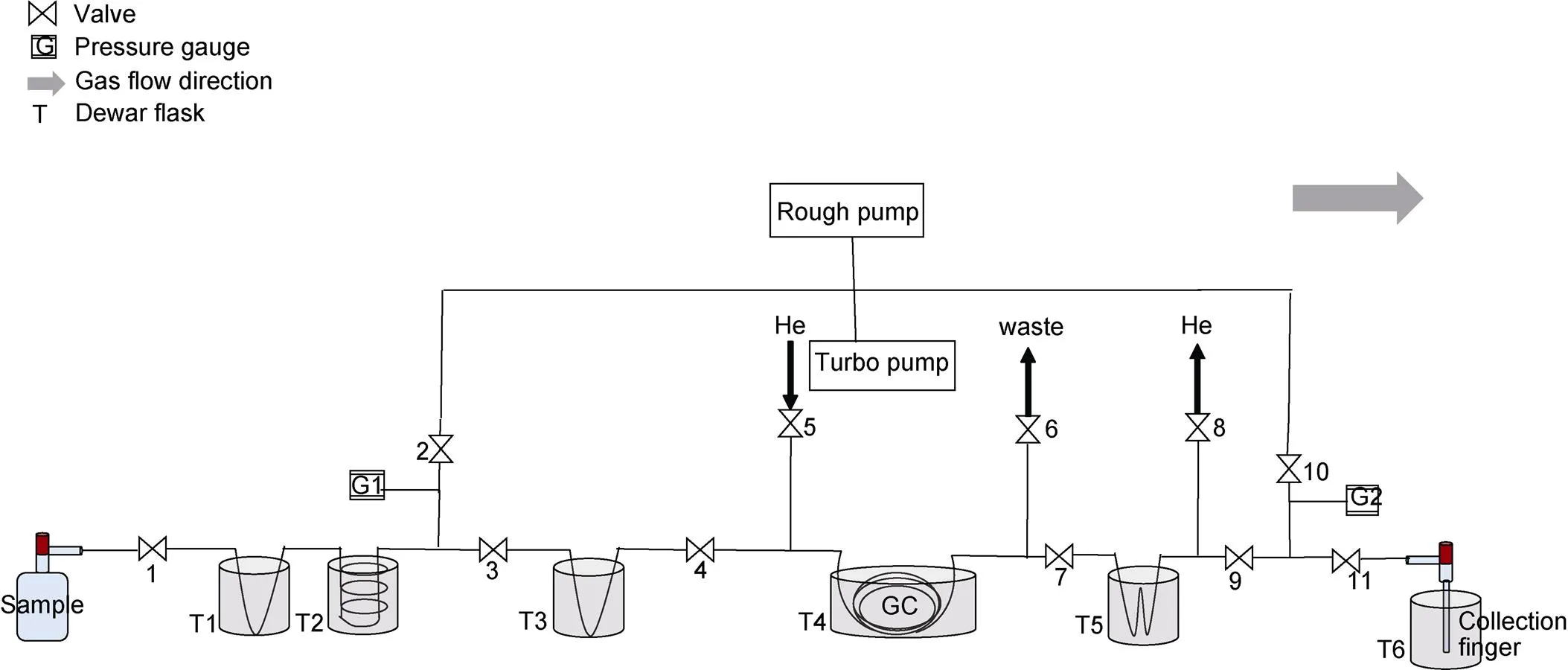
Figure 1 Plot of the separation line.

Table 2 Ratios of GOP in the past to the present and Δ17O values of O2 from ice-core bubbles
Note: * Data referenced from Luz et al. (1999) and Blunier et al. (2002).

Figure 2 Variations of Δ17O in O2trapped in ice-core bubbles from GISP2 (crosses) (a) and Vostok (diamonds) (b) at glacial-interglacial timescale (Luz et al., 1999; Blunier et al., 2012). In addition, the ice δ18O of GISP2 (grey curve; Grootes et al., 1993) and ice δD of Vostok (dark blue curve; Petit et al., 1999) are also shown to indicate the glacial-interglacial cycles.
Luz et al. (1999) first applied the TOI method to estimate the past global biological production through ice cores from GISP2. They concluded that the average estimate of the global GOP ratio in the past 82 ka is 87%–97% of the modern productivity, and proposed that the marine biosphere was almost the same as or just a little higher than the present, while the terrestrial biosphere was slightly lower. Blunier et al. (2002) also calculated the Δ17O of paleo-atmospheric O2trapped in ice-core bubbles from GISP2 over the last 60 ka, and concluded that the GOP rates are only~76%–83% of the modern for the LGM and are slightly lower than today for the glacial-interglacial transition and the early Holocene. Landais et al. (2007a) calculated the biological productivity ratio for the LGM as 60%–75%, and proposed that estimates of the LGM oxygen biosphere productivity obtained by Blunier et al. (2002) may not be adequate (Hoffmann et al., 2004) compared with that in Luz et al. (1999), because of various climatic conditions and plant coverage (Hoffmann et al., 2004). Landais et al. (2007b) emphasized on the relationship between δ17O and δ18O during leaf transpiration, and concluded that the GOP rate during the LGM is 69%±6% of the present, and yielded that the Δ17O is constant around 40 meg−1in glacial times, while changed a lot in the LGM.
Blunier et al. (2012) calculated biospheric productivity from 400 ka BP Δ17O records in Vostok, and concluded that the mean GOP ratios during the past 400 ka are 94%–83% of the modern rates. They also found that oceanic oxygen productivity was elevated by ~20% relative to the present during glacial maximum and the transition of glacial-interglacial, but the increase did not compensate for land productivity decrease.
The variations of biogenic productivity over paleo times in the ocean biosphere are different from that in the land biosphere, especially in glacial times. Blunier et al. (2012) concluded that there were still uncertainties remained to assess both land and ocean productivities from Δ17O data of O2. Distinguished from the global mean value, the marine biological production in glacial times were 88%–140% of the present (Luz et al., 1999; Blunier et al., 2002), while terrestrial biosphere remained uncertain because of complex plant physiology. Angert et al. (2003) showed that at least 15% of the changes in the triple oxygen isotopic composition in paleo-air should be related to the variations among different respiration mechanisms.
4.2 Assessing marine photosynthetically produced oxygen through triple isotopes of O2 dissolved in ocean
The TOI method has been widely applied to measure marine primary productivity in the ML of global open ocean or coastal ocean. Specifically, spatial distribution of TOI-GOP study sites in global ocean is illustrated in Figure 3, and the mean TOI-GOP values are presented in Table 3. According to Juranek and Quay (2010), gross primary productivity (GPP) data were all converted to GOP data based on the photosynthetic quotient (PQ = 1.2) (Burris, 1981; Laws, 1991) and a 15% correction (Bender et al., 1999; Laws et al., 2000) (GPP = GOP × (0.85/PQ)). TOI-GOP data were acquired in the Southern Ocean, the Pacific, the Atlantic and the Arctic oceans, of which the Pacific Ocean and coastal ocean gained wide attention, while the Indian Ocean was blank in TOI-GOP values. So far, TOI-based GOP assessment in the subtropical Atlantic and the subtropical Pacific has mainly collected data from the Bermuda Atlantic Time-series Station (BATS) and the Hawaii Ocean Time-series Station ALOHA, respectively. In the Southern Ocean, TOI-based GOP values focused on theAntarctic Peninsula and the sector close to Australia.

Figure 3 Distribution of TOI-derived GOP values in different oceanic zones.

Table 3 TOI-GOP calculations in different ocean zones

Continued
Notes: * GOP calculated from equation (7);+GOP allowing for production below the ML.
Although widely applied in global ocean regions, TOI method’s assumptions of a steady state (no change of Δ17O with time) in the ML and no physical transports resulted in potential biases in different regions and seasons. It is apparent from Table 3 that the Southern Ocean bears the largest biases for mean GOP values, followed by the coastal ocean. Luz and Barkan (2009) demonstrated that ocean dynamics will affect the Δ17Odissin the ML of subtropical ocean on a seasonal scale. Vertical entrainment or mixing of seawater was the largest sources of the bias, and seasonal variability of Δ17Odisswas another significant source. Nicholson et al. (2014) further demonstrated that biases resulted from physical dynamics tended to incur larger overestimation in midlatitudes and in summer and fall, and biases due to seasonal variability were highest in the fall.
Considering the main contribution of physical transports and seasonal variability to GOP biases in the ML, non-steady state GOP terms were proposed to better estimate primary productivity in the euphotic layer or even the deep ocean (Nicholson et al., 2012; Wurgaft et al., 2013). Luz and Barkan (2000) first calculated the TOI-GOP values covering the euphotic zone with the steady state assumption. Kaiser (2011) and Prokopenko et al. (2011) accounted for disequilibrium terms that affected GOP in the ML, and concluded that exact expressions including both non-steady and steady states performed well in high-productivity aquatic ecosystems. Haskell et al. (2016) then applied the exact expression proposed by Prokopenko et al. (2011) in oligotrophic ocean, and found that the disequilibrium terms would slightly affect GOP values in the ML.
Given to the wide range of time and spatial scales that various productivity methods covered, comparisons were needed to explore their applicability in oceans of different space and timescales (Hamme et al., 2012). TOI-based GOP values more or less exceeded values estimated from traditional methods, which uncovered the underestimation of incubation methods on primary productivity. Quay et al. (2010) concluded that the Δ17O-derived GOP exceeded the labeled18O-GOP by 25%–60%. Reuer et al. (2007) found the TOI-based GOP higher than the prediction in Behrenfeld and Falkowski (1997), and about 2.7 times higher than14C-based gross production. Globally, the underestimation of primary productivity using incubation methods became the greatest in the tropics (Juranek and Quay, 2010). Sarma et al. (2005) attributed the underestimation to unavailability of O2uptake rates and the assumption of equal light and dark respiration mechanisms.
Apart from the application of TOI for GOP calculation, O2/Ar ratios can be concomitantly measured to calculate net oxygen productivity (NOP) (Reuer et al., 2007; Castro-Morales et al., 2013; Munro et al., 2013). More studies have also used recently the NOP/GOP ratios to suggest the potential export efficiency of community ecosystems (Juranek and Quay, 2005; Reuer et al., 2007; Luz and Barkan, 2009), especially in high productivity ecosystems (Prokopenko et al., 2011). Detailed calculations for the weighted gas transfer velocity is key to high-precision NOP calculation (Reuer et al., 2007), while NOP/GOP ratios calculation were free fromerrors, and they were in the range of15N incubation ratios (Sambrotto and Mace, 2000).
5 Problems and prospects
TOI method has been applied widely in assessing past global primary production and modern marine productivity, but problems still remain on possible biases and inadequate understandings, which awaits improvements in future studies.
(1) The assumption of a steady state and neglected physical transports may cause large biases in productivity calculation, thus future studies await further exploration of complex advective mixing or vertical entrainment of seawater, and combined the steady and non-steady state equation to calculate biospheric production in euphotic layer or even deep ocean.
(2) CalculatingD17O-based GOP requires accurate gas separation processes and high-precision measurements of δ18O and δ17O. However, there are only a few technologies in the world that can control the uncertainties and precision in analytically available range. Another big error comes from parameterization of air-sea oxygen exchange rate (Reuer et al., 2007). Thus, more studies need improvements on accurate gas separation and high-precision mass spectrometry analyzing technologies in future works, and a uniform and widely acceptedparameterization standard awaits further exploration.
(3) Inadequate understandings still remain in conversion between GOP and GPP. Therefore, relationships between GOP and GPP need to be better understood by tracking O2and carbon flows in natural communities over long ranges of timescales (Juranek and Quay, 2013).
(4) Complex physiological mechanisms of terrestrial plants hindered the wide application of TOI method in estimating past global biological production, like the inadequate knowledge of leaf water transpiration (Landais et al. 2007b). Studies in the future can move forward on assessment of past global productivity through Δ17O signals in ice-core bubbles earlier than 400 ka BP.
Acknowledgements We appreciate the Institute of Oceanography, Shanghai Jiao Tong University, which provides experiment platforms for us to learn about disciplines of gas separation line and high-precision measurements of TOIs. We are grateful to Zhaojun Zhan, Yanyan Cai and Jiajia Wang, for their help on plots. Gratitude also goes to the technicians who gave suggestions on our pre-processed system. This work was supported by the National Natural Science Foundation of China (Grant nos. 41771031 and 41673125) and the Priority Academic Program Development of Jiangsu Higher Education Institutions (PAPD). We would like to thank two anonymous reviewers, and Associate Editor Dr. Cinzia Verde, for their valuable suggestions and comments that improved this article.
Abe O, Yoshida N. 2003. Partial pressure dependency of17O/16O and18O/16O of molecular oxygen in the mass spectrometer. Rapid Commun Mass Spectrom, 17(5): 395-400, doi:10.1002/rcm.923.
Angert A, Rachmilevitch S, Barkan E, et al. 2003. Effects of photorespiration, the cytochrome pathway, and the alternative pathway on the triple isotopic composition of atmospheric O2. Glob Biogeochem Cycles, 17(1): 1030, doi:10.1029/2002GB001933.
Ash J L, Hu H T, Yeung L Y. 2020. What fractionates oxygen isotopes during respiration? Insights from multiple isotopologue measurements and theory. ACS Earth Space Chem, 4(1): 50-66, doi:10.1021/ acsearthspacechem.9b00230.
Assonov S S, Brenninkmeijer C A M. 2005. Reporting small Δ17O values: existing definitions and concepts. Rapid Commun Mass Spectrom, 19(5): 627-636, doi:10.1002/rcm.1833.
Barkan E, Luz B. 2003. High-precision measurements of17O/16O and18O/16O of O2and O2/Ar ratio in air. Rapid Commun Mass Spectrom, 17(24): 2809-2814, doi:10.1002/rcm.1267.
Barkan E, Luz B. 2011. The relationships among the three stable isotopes of oxygen in air, seawater and marine photosynthesis. Rapid Commun Mass Spectrom, 25(16): 2367-2369, doi:10.1002/rcm.5125.
Behrenfeld M J, Falkowski P G. 1997. Photosynthetic rates derived from satellite-based chlorophyll concentration. Limnol Oceanogr, 42(1): 1-20, doi:10.4319/lo.1997.42.1.0001.
Bender M, Grande K, Johnson K, et al. 1987. A comparison of four methods for determining planktonic community production. Limnol Oceanogr, 32(5): 1085-1098, doi:10.4319/lo.1987.32.5.1085.
Bender M, Orchardo J, Dickson M L, et al. 1999.O2fluxes compared with14C production and other rate terms during the JGOFS Equatorial Pacific experiment. Deep Sea Res Part I: Oceanogr Res Pap, 46(4): 637-654, doi:10.1016/s0967-0637(98)00080-6.
Bender M, Sowers T, Labeyrie L. 1994. The Dole Effect and its variations during the last 130, 000 years as measured in the Vostok Ice Core. Glob Biogeochem Cycles, 8(3): 363-376, doi:10.1029/94GB00724.
Bender M L, Tilbrook B, Cassar N, et al. 2016. Ocean productivity south of Australia during spring and summer. Deep Sea Res Part I: Oceanogr Res Pap, 112: 68-78, doi:10.1016/j.dsr.2016.02.018.
Blunier T, Barnett B, Bender M L, et al. 2002. Biological oxygen productivity during the last 60, 000 years from triple oxygen isotope measurements. Glob Biogeochem Cycles, 16(3): 3-1-3-13, doi:10.1029/ 2001GB001460.
Blunier T, Bender M L, Barnett B, et al. 2012. Planetary fertility during the past 400 ka based on the triple isotope composition of O2in trapped gases from the Vostok ice core. Clim Past, 8(5): 1509-1526, doi:10.5194/cp-8-1509-2012.
Boering K A, Jackson T, Hoag K J, et al. 2004. Observations of the anomalous oxygen isotopic composition of carbon dioxide in the lower stratosphere and the flux of the anomaly to the troposphere. Geophys Res Lett, 31(3): L03109, doi:10.1029/2003GL018451.
Burris J E. 1981. Effects of oxygen and inorganic carbon concentrations on the photosynthetic quotients of marine algae. Mar Biol, 65(3): 215-219, doi:10.1007/BF00397114.
Castro-Morales K, Cassar N, Shoosmith D R, et al. 2013. Biological production in the Bellingshausen Sea from oxygen-to-argon ratios and oxygen triple isotopes. Biogeosciences, 10(4): 2273-2291, doi:10. 5194/bg-10-2273-2013.
Emerson S, Quay P D, Stump C, et al. 1995. Chemical tracers of productivity and respiration in the subtropical Pacific Ocean. J Geophys Res: Oceans, 100(C8): 15873-15887, doi:10.1029/95JC 01333.
Fujiki T, Hosaka T, Kimoto H, et al. 2008.observation of phytoplankton productivity by an underwater profiling buoy system: use of fast repetition rate fluorometry. Mar Ecol Prog Ser, 353: 81-88, doi:10.3354/meps07151.
Garcia H E, Gordon L I. 1992. Oxygen solubility in seawater: better fitting equations. Limnol Oceanogr, 37(6): 1307-1312, doi:10.4319/lo.1992. 37.6.1307.
Grande K D, Williams P J L, Marra J, et al. 1989. Primary production in the North Pacific Gyre: a comparison of rates determined by the14C, O2concentration and18O methods. Deep Sea Res A Oceanogr Res Pap, 36(11): 1621-1634, doi:10.1016/0198-0149(89)90063-0.
Grootes P M, Stuiver M, White J W C, et al. 1993. Comparison of oxygen isotope records from the GISP2 and GRIP Greenland ice cores. Nature, 366(6455): 552-554, doi:10.1038/366552a0.
Guy R D, Berry J A, Fogel M L, et al. 1989. Differential fractionation of oxygen isotopes by cyanide-resistant and cyanide-sensitive respiration in plants. Planta, 177(4): 483-491, doi:10.1007/BF00392616.
Hamme R C, Cassar N, Lance V P, et al. 2012. Dissolved O2/Ar and other methods reveal rapid changes in productivity during a Lagrangian experiment in the Southern Ocean. J Geophys Res: Oceans, 117(C4): C00F12, doi:10.1029/2011JC007046.
Hamme R C, Emerson S R. 2004. The solubility of neon, nitrogen and argon in distilled water and seawater. Deep Sea Res Part I: Oceanogr Res Pap, 51(11): 1517-1528, doi:10.1016/j.dsr.2004.06.009.
Harris G P, Griffiths F B, Thomas D P. 1989. Light and dark uptake and loss of14C: methodological problems with productivity measurements in oceanic waters. Hydrobiologia, 173(2): 95-105, doi:10.1007/BF000 15519.
Harrison W G, Harris L R. 1986. Isotope-dilution and its effects on measurements of nitrogen and phosphorus uptake by oceanic microplankton. Mar Ecol Prog Ser, 27: 253-261, doi:10.3354/meps 027253.
Haskell II W Z, Prokopenko M G, Stanley R H R, et al. 2016. Estimates of vertical turbulent mixing used to determine a vertical gradient in net and gross oxygen production in the oligotrophic South Pacific Gyre. Geophys Res Lett, 43(14): 7590-7599, doi:10.1002/2016GL069523.
Helman Y, Barkan E, Eisenstadt D, et al. 2005. Fractionation of the three stable oxygen isotopes by oxygen-producing and oxygen-consuming reactions in photosynthetic organisms. Plant Physiol, 138(4): 2292-2298, doi:10.1104/pp.105.063768.
Hendricks M B, Bender M L, Barnett B A. 2004. Net and gross O2production in the southern ocean from measurements of biological O2saturation and its triple isotope composition. Deep Sea Res Part I: Oceanogr Res Pap, 51(11): 1541-1561, doi:10.1016/j.dsr.2004.06.006.
Hendricks M B, Bender M L, Barnett B A, et al. 2005. Triple oxygen isotope composition of dissolved O2in the equatorial Pacific: a tracer of mixing, production, and respiration. J Geophys Res: Oceans, 110(C12): C12021, doi:10.1029/2004JC002735.
Ho D T, Wanninkhof R, Schlosser P, et al. 2011. Toward a universal relationship between wind speed and gas exchange: Gas transfer velocities measured with3He/SF6during the Southern Ocean Gas Exchange Experiment. J Geophys Res: Oceans, 116(C4): C00F04, doi:10.1029/2010JC006854.
Hoffmann G, Cuntz M, Weber C, et al. 2004. A model of the Earth’s Dole effect. Global Biogeochem Cycles, 18(1): GB1008, doi:10.1029/ 2003gb002059.
Hotchkiss E R, Hall R O. 2014. High rates of daytime respiration in three streams: Use of δ18OO2and O2to model diel ecosystem metabolism. Limnol Oceanogr, 59(3): 798-810, doi:10.4319/lo.2014.59.3.0798.
Howard E M, Spivak A C, Karolewski J S, et al. 2020. Oxygen and triple oxygen isotope measurements provide different insights into gross oxygen production in a shallow salt marsh pond. Estuaries Coasts, 43(8): 1908-1922, doi:10.1007/s12237-020-00757-6.
Huang K, Ducklow H, Vernet M, et al. 2012. Export production and its regulating factors in the West Antarctica Peninsula region of the Southern Ocean. Glob Biogeochem Cycles, 26(2): GB2005, doi:10.1029/2010GB004028.
IPCC. 2014. Climate Change 2014: Synthesis report. IPCC, Geneva, Switzerland.
Ji B Y, Sandwith Z O, Williams W J, et al. 2019. Variations in rates of biological production in the Beaufort Gyre as the Arctic changes: rates from 2011 to 2016. J Geophys Res: Oceans, 124(6): 3628-3644, doi:10.1029/2018JC014805.
Juranek L W, Quay P D. 2005.andgross primary and net community production in the North Pacific Subtropical Gyre using labeled and natural abundance isotopes of dissolved O2. Glob Biogeochem Cycles, 19(3): GB3009, doi:10.1029/2004GB002384.
Juranek L W, Quay P D. 2010. Basin-wide photosynthetic production rates in the subtropical and tropical Pacific Ocean determined from dissolved oxygen isotope ratio measurements. Glob Biogeochem Cycles, 24(2): GB2006, doi:10.1029/2009GB003492.
Juranek L W, Quay P D. 2013. Using triple isotopes of dissolved oxygen to evaluate global marine productivity. Ann Rev Mar Sci, 5: 503-524, doi:10.1146/annurev-marine-121211-172430.
Juranek L W, Quay P D, Feely R A, et al. 2012. Biological production in the NE Pacific and its influence on air-sea CO2flux: Evidence from dissolved oxygen isotopes and O2/Ar. J Geophys Res: Oceans, 117(C5): C05022, doi:10.1029/2011JC007450.
Jurikova H, Guha T, Abe O, et al. 2016. Variations in triple isotope composition of dissolved oxygen and primary production in a subtropical reservoir. Biogeosciences, 13(24): 6683-6698, doi:10. 5194/bg-13-6683-2016.
Kaiser J. 2011. Technical note: Consistent calculation of aquatic gross production from oxygen triple isotope measurements. Biogeosciences, 8(7): 1793-1811, doi:10.5194/bg-8-1793-2011.
Keedakkadan H R, Abe O. 2015. Cryogenic separation of an oxygen-argon mixture in natural air samples for the determination of isotope and molecular ratios. Rapid Commun Mass Spectrom, 29(8): 775-781, doi:10.1002/rcm.7161.
Lämmerzahl P, Röckmann T, Brenninkmeijer C A M, et al. 2002. Oxygen isotope composition of stratospheric carbon dioxide. Geophys Res Lett, 29(12): 1582, doi:10.1029/2001GL014343.
Landais A, Barkan E, Yakir D, et al. 2006. The triple isotopic composition of oxygen in leaf water. Geochimica et Cosmochimica Acta, 70(16): 4105-4115, doi:10.1016/j.gca.2006.06.1545.
Landais A, Lathiere J, Barkan E, et al. 2007a. Reconsidering the change in global biosphere productivity between the Last Glacial Maximum and present day from the triple oxygen isotopic composition of air trapped in ice cores. Glob Biogeochem Cycles, 21(1): GB1025, doi:10.1029/ 2006GB002739.
Landais A, Yakir D, Barkan E, et al. 2007b. The triple isotopic composition of oxygen in leaf water and its implications for quantifying biosphere productivity. Terr Ecol, 1: 111-125, doi:10. 1016/s1936-7961(07)01008-1.
Laws E A. 1991. Photosynthetic quotients, new production and net community production in the open ocean. Deep Sea Res A: Oceanogr Res Pap, 38(1): 143-167, doi:10.1016/0198-0149(91)90059-O.
Laws E A, Landry M R, Barber R T, et al. 2000. Carbon cycling in primary production bottle incubations: inferences from grazing experiments and photosynthetic studies using14C and18O in the Arabian Sea. Deep Sea Res Part II: Top Stud Oceanogr, 47(7-8): 1339-1352, doi:10.1016/S0967-0645(99)00146-0.
Li B D, Yeung L Y, Hu H T, et al. 2019. Kinetic and equilibrium fractionation of O2isotopologues during air-water gas transfer and implications for tracing oxygen cycling in the ocean. Mar Chem, 210: 61-71, doi:10.1016/j.marchem.2019.02.006.
Luz B, Barkan E. 2000. Assessment of oceanic productivity with the triple-isotope composition of dissolved oxygen. Science, 288(5473): 2028-2031, doi:10.1126/science.288.5473.2028.
Luz B, Barkan E. 2005. The isotopic ratios17O/16O and18O/16O in molecular oxygen and their significance in biogeochemistry. Geochimica et Cosmochimica Acta, 69(5): 1099-1110, doi:10.1016/j. gca.2004.09.001.
Luz B, Barkan E. 2009. Net and gross oxygen production from O2/Ar,17O/16O and18O/16O ratios. Aquat Microb Ecol, 56: 133-145, doi:10.3354/ame01296.
Luz B, Barkan E, Bender M L, et al. 1999. Triple-isotope composition of atmospheric oxygen as a tracer of biosphere productivity. Nature, 400(6744): 547-550, doi:10.1038/22987.
Luz B, Barkan E, Sagi Y, et al. 2002. Evaluation of community respiratory mechanisms with oxygen isotopes: a case study in Lake Kinneret. Limnol Oceanogr, 47(1): 33-42, doi:10.4319/lo.2002.47.1.0033.
Manning C C, Howard E M, Nicholson D P, et al. 2017a. Revising estimates of aquatic gross oxygen production by the triple oxygen isotope method to incorporate the local isotopic composition of water. Geophys Res Lett, 44(20): 10511-10519, doi:10.1002/2017GL074375.
Manning C C, Stanley R H R, Nicholson D P, et al. 2017b. Impact of recently upwelled water on productivity investigated usingand incubation-based methods in Monterey Bay. J Geophys Res: Oceans, 122(3): 1901-1926, doi:10.1002/2016JC012306.
Manning C C, Stanley R H R, Nicholson D P, et al. 2019. Changes in gross oxygen production, net oxygen production, and air-water gas exchange during seasonal ice melt in Whycocomagh Bay, a Canadian estuary in the Bras d’Or Lake system. Biogeosciences, 16(17): 3351-3376, doi:10.5194/bg-16-3351-2019.
Marra J. 2007. Approaches to the measurement of plankton production// Williams P J l B, Thomas D N, Reynolds C S. Phytoplankton productivity. Oxford, UK: Blackwell Science Ltd, 78-108, doi:10. 1002/9780470995204.ch4.
Miller M F. 2002. Isotopic fractionation and the quantification of17O anomalies in the oxygen three-isotope system: an appraisal and geochemical significance. Geochimica et Cosmochimica Acta, 66(11): 1881-1889, doi:10.1016/S0016-7037(02)00832-3.
Reuer M K, Barnett B A, Bender M L, et al. 2007. New estimates of Southern Ocean biological production rates from O2/Ar ratios and the triple isotope composition of O2. Deep Sea Res Part I: Oceanogr Res Pap, 54(6): 951-974, doi:10.1016/j.dsr.2007.02.007.
Munro D R, Quay P D, Juranek L W, et al. 2013. Biological production rates off the Southern California Coast estimated from triple O2isotopes and O2: Ar gas ratios. Limnol Oceanogr, 58(4): 1312-1328, doi:10.4319/lo.2013.58.4.1312.
Moore C M, Suggett D, Holligan P M, et al. 2003. Physical controls on phytoplankton physiology and production at a shelf sea front: a fast repetition-rate fluorometer based field study. Mar Ecol Prog Ser, 259: 29-45, doi:10.3354/meps259029.
Nicholson D P, Stanley R H R, Barkan E, et al. 2012. Evaluating triple oxygen isotope estimates of gross primary production at the Hawaii Ocean Time-series and Bermuda Atlantic Time-series Study sites. J Geophys Res: Oceans, 117(C5): C05012, doi:10.1029/2010JC006856.
Nicholson D, Stanley R H R, Doney S C. 2014. The triple oxygen isotope tracer of primary productivity in a dynamic ocean model. Glob Biogeochem Cycles, 28(5): 538-552, doi:10.1002/2013GB004704.
Nielsen E S. 1952. The use of radio-active carbon (14C) for measuring organic production in the sea. ICES Journal of Marine Science, 18(2): 117-140, doi:10.1093/icesjms/18.2.117.
Nightingale P D, Malin G, Law C S, et al. 2000.evaluation of air-sea gas exchange parameterizations using novel conservative and volatile tracers. Glob Biogeochem Cycles, 14(1): 373-387, doi:10. 1029/1999GB900091.
Palevsky H I, Quay P D, Lockwood D E, et al. 2016. The annual cycle of gross primary production, net community production, and export efficiency across the North Pacific Ocean. Glob Biogeochem Cycles, 30(2): 361-380, doi:10.1002/2015GB005318.
Peterson B J. 1980. Aquatic primary productivity and the14C-CO2method: a history of the productivity problem. Annu Rev Ecol Syst, 11(1): 359-385, doi:10.1146/annurev.es.11.110180.002043.
Petit J R, Jouzel J, Raynaud D, et al. 1999. Climate and atmospheric history of the past 420, 000 years from the Vostok ice core, Antarctica. Nature, 399(6735): 429-436, doi:10.1038/20859.
Prokopenko M G, Pauluis O M, Granger J, et al. 2011. Exact evaluation of gross photosynthetic production from the oxygen triple-isotope composition of O2: Implications for the net-to-gross primary production ratios. Geophys Res Lett, 38(14): L14603, doi:10.1029/ 2011GL047652.
Quay P D, Emerson S, Wilbur D O, et al. 1993. The δ18O of dissolved O2in the surface waters of the subarctic Pacific: a tracer of biological productivity. J Geophys Res, 98(C5): 8447-8458, doi:10.1029/92jc 03017.
Quay P D, Peacock C, Björkman K, et al. 2010. Measuring primary production rates in the ocean: Enigmatic results between incubation and non-incubation methods at Station ALOHA. Glob Biogeochem Cycles, 24(3): GB3014, doi:10.1029/2009GB003665.
Quay P, Stutsman J, Steinhoff T. 2012. Primary production and carbon export rates across the subpolar N. Atlantic Ocean basin based on triple oxygen isotope and dissolved O2and Ar gas measurements. Glob Biogeochem Cycles, 26(2): GB2003, doi:10.1029/2010GB 004003.
Sambrotto R N, Mace B J. 2000. Coupling of biological and physical regimes across the Antarctic Polar Front as reflected by nitrogen production and recycling. Deep Sea Res Part II: Top Stud Oceanogr, 47(15-16): 3339-3367, doi:10.1016/S0967-0645(00)00071-0.
Sarma V V S S, Abe O, Hashimoto S, et al. 2005. Seasonal variations in triple oxygen isotopes and gross oxygen production in the Sagami Bay, central Japan. Limnol Oceanogr, 50(2): 544-552, doi:10.4319/lo.2005. 50.2.0544.
Sarma V V S S, Abe O, Hinuma A, et al. 2006. Short-term variation of triple oxygen isotopes and gross oxygen production in the Sagami Bay, central Japan. Limnol Oceanogr, 51(3): 1432-1442, doi:10.4319/lo. 2006.51.3.1432.
Sarma V V S S, Abe O, Saino T. 2003. Chromatographic separation of nitrogen, argon, and oxygen in dissolved air for determination of triple oxygen isotopes by dual-inlet mass spectrometry. Anal Chem, 75(18): 4913-4917, doi:10.1021/ac034314r.
Sarma V V S S, Abe O, Saino T. 2008. Spatial variations in time-integrated plankton metabolic rates in Sagami Bay using triple oxygen isotopes and O2: Ar ratios. Limnol Oceanogr, 53(5): 1776-1783, doi:10.4319/lo.2008.53.5.1776.
Seguro I, Marca A D, Painting S J, et al. 2019. High-resolution net and gross biological production during a Celtic Sea spring bloom. Prog Oceanogr, 177: 101885, doi:10.1016/j.pocean.2017.12.003.
Staehr P A, Testa J M, Kemp W M, et al. 2012. The metabolism of aquatic ecosystems: history, applications, and future challenges. Aquat Sci, 74(1): 15-29, doi:10.1007/s00027-011-0199-2.
Stanley R H R, Howard E M. 2013. Quantifying photosynthetic rates of microphytobenthos using the triple isotope composition of dissolved oxygen. Limnol Oceanogr: Methods, 11(7): 360-373, doi:10.4319/lom. 2013.11.360.
Stanley R H R, Kirkpatrick J B, Cassar N, et al. 2010. Net community production and gross primary production rates in the western equatorial Pacific. Glob Biogeochem Cycles, 24(4): GB4001, doi:10.1029/2009GB003651.
Stanley R H R, Sandwith Z O, Williams W J. 2015. Rates of summertime biological productivity in the Beaufort Gyre: a comparison between the low and record-low ice conditions of August 2011 and 2012. J Mar Syst, 147: 29-44, doi:10.1016/j.jmarsys.2014.04.006.
Suggett D, Kraay G, Holligan P, et al. 2001. Assessment of photosynthesis in a spring cyanobacterial bloom by use of a fast repetition rate fluorometer. Limnol Oceanogr, 46(4): 802-810, doi:10.4319/lo.2001. 46.4.0802.
Sweeney C, Gloor E, Jacobson A R, et al. 2007. Constraining global air-sea gas exchange for CO2with recent bomb14C measurements. Glob Biogeochem Cycles, 21(2): GB2015, doi:10.1029/2006GB 002784.
Thiemens M H, Heidenreich J E. 1983. The mass-independent fractionation of oxygen: a novel isotope effect and its possible cosmochemical implications. Science, 219(4588): 1073-1075, doi:10.1126/science.219.4588.1073.
Thiemens M H, Jackson T L, Brenninkmeijer C A M. 1995. Observation of a mass independent oxygen isotopic composition in terrestrial stratospheric CO2, the link to ozone chemistry, and the possible occurrence in the Martian atmosphere. Geophys Res Lett, 22(3): 255-257, doi:10.1029/94GL02996.
Thiemens M H, Jackson T, Mauersberger K, et al. 1991. Oxygen isotope fractionation in stratospheric CO2. Geophys Res Lett, 18(4): 669-672, doi:10.1029/91GL00121.
Thiemens M H, Jackson T, Zipf E C, et al. 1995. Carbon dioxide and oxygen isotope anomalies in the mesosphere and stratosphere. Science, 270(5238): 969-972, doi:10.1126/science.270.5238.969.
Thiemens M H, Meagher D. 1984. Cryogenic separation of nitrogen and oxygen in air for determination of isotopic ratios by mass spectrometry. Anal Chem, 56(2): 201-203, doi:10.1021/ac00266a 018.
Tobias C R, Böhlke J K, Harvey J W. 2007. The oxygen-18 isotope approach for measuring aquatic metabolism in high productivity waters. Limnol Oceanogr, 52(4): 1439-1453, doi:10.4319/lo.2007.52.4. 1439.
van der Meer A. 2015. Constraining air-sea equilibrium and biological end-members for marine gross productivity estimates using oxygen triple isotopes. Masters thesis, University of East Anglia.
Watson A J, Upstill-Goddard R C, Liss P S. 1991. Air-sea gas exchange in rough and stormy seas measured by a dual-tracer technique. Nature, 349(6305): 145-147, doi:10.1038/349145a0.
Wurgaft E, Shamir O, Barkan E, et al. 2013. Mixing processes in the deep water of the Gulf of Elat (Aqaba): evidence from measurements and modeling of the triple isotopic composition of dissolved oxygen. Limnol Oceanogr, 58(4): 1373-1386, doi:10. 4319/lo.2013.58.4.1373.
Yakir D, Berry J A, Giles L, et al. 1994. Isotopic heterogeneity of water in transpiring leaves: identification of the component that controls the δ18O of atmospheric O2and CO2. Plant Cell Environ, 17(1): 73-80, doi:10.1111/j.1365-3040.1994.tb00267.x.
Yung Y L, DeMore W B, Pinto J P. 1991. Isotopic exchange between carbon dioxide and ozone via O(1D) in the stratosphere. Geophys Res Lett, 18(1): 13-16, doi:10.1029/90GL02478.
10.13679/j.advps.2021.0038
Zhou Y Q, Pang H X, Hu H T, et al. The evaluation of biological productivity by triple isotope composition of oxygen trapped in ice-core bubbles and dissolved in ocean: a review. Adv Polar Sci, 2022, 33(2): 123-134,doi:10.13679/j.advps.2021.0038
21 July 2021;
21 October 2021;
21 November 2021
Corresponding author, ORCID: 0000-0003-3990-6566, E-mail: hxpang@nju.edu.cn
杂志排行
Advances in Polar Science的其它文章
- “Opinion Editorial” category attracts more attention
- One special issue will be published in September 2023
- The older ice,the better science
- Development of the International Polar Years and their benefits for China
- Inventory of unintentional POPs emission from anthropogenic sources in Antarctica
- Community structure of mesopelagic fauna and the length-weight relationships of three common fishes inthe Cosmonaut Sea,Southern Ocean
