Advances, opportunities, and challenge for full-color emissive carbon dots
2022-06-18XinYngXueLiBoyngWngLinAiGuopingLiBiYngSiyuLu
Xin Yng, Xue Li, Boyng Wng, Lin Ai,*, Guoping Li,*, Bi Yng, Siyu Lu,b,*
a Green Catalysis Center, and College of Chemistry, Zhengzhou University, Zhengzhou 450001, China
b State Key Laboratory of Bio-Fibers and Eco-Textiles, Qingdao University, Qingdao 132101, China
c State Key Laboratory of Superhard Materials, College of Physics, Jilin University, Changchun 130012, China
d State Key Lab of Supramolecular Structure and Materials, College of Chemistry, Jilin University, Changchun 130012, China
ABSTRACT Carbon dots (CDs), novel luminescent zero-dimensional carbon nanomaterials, have been widely applied due to their low toxicity, optimal optical properties, and easy modification.However, the current controllable equipment and mechanism explanation of CDs are relatively vague and require urgent resolution.Full-color emission CDs, an essential CDs category, have attracted people’s attention given their light and color-tunable properties.In addition to a wider range of biological and optoelectronic device applications, full-color emission CDs have similar structures and significantly affected the fluorescence mechanism of CDs.At present, few studies have reported on the summary research of CDs emitted by its full color, which greatly limits the development of CDs mechanisms and applications.As such, the present review detailed the full-color CDs development status, to which a suitable method for preparing full-color CDs was presented and the existing fluorescence emission mechanism of full-color CDs was summarized.Herein, we comprehensively introduced full-color CDs applications in biology and optoelectronics.Finally,we made an outlook on the development and potential applications of full-color CDs.The present review aims to contribute novel insights and methods for understanding full-color CDs.
Keywords:Carbon dots Full-color emission Preparation Fluorescence mechanism Application
1.Introduction
In 2004, Xuet al.accidentally discovered fluorescent carbon nanoparticles in the process of separating and purifying singlewalled carbon nanotubes [1]; These fluorescent carbon nanoparticles emit green-blue, yellow, and orange fluorescence.Sunet al.then termed these carbon nanoparticles as carbon dots (CDs) given their similarities to the structures of carbon and quantum dots[2].This study confirmed the excitation dependence.Most previously reported CDs emit blue fluorescence and have a low quantum yields (QYs) [3-7]; As the research continues to deepen, the emission wavelength of CDs continues to red-shift and the QYs gradually increase [7-11].In 2015, Huet al.prepared full-color CDs for the first time [12], thus leading the wave of research in the field of full-color CDs [13-15].The preparation of full-color CDs is closely related to the types of precursors [16].For example, Sunet al.synthesized CDs with different emission colors with high QYs by adjusting the ratio of CA to urea and the reaction temperature[17,18].Wanget al.usedo-phenylenediamine (oPD) as a precursor to obtain CDs with remarkable tunability and stable blue to red as well as white light fluorescence emissions through acid reagent engineering strategy [19].The relative photoluminescence (PL) QYs of these CDs reached 72%.In order to strengthen the further application of CDs [20-22], reasonably obtained CDs with high QYs and long emission wavelength must be generated to replace the toxic quantum dots of heavy metals [8,23,24].
The composition and properties of CDs depend on the influence of many variables in the reaction process [5,22,25-27].As such, the fluorescence mechanism of CDs must be thoroughly examined to understand its structure and create a controllable CDs preparation method [28-30].Although the luminescence mechanism of CDs has been studied on the basis of the sp2/sp3effect, surface state effect,or graphite N, a unified theoretical explanation is yet to be presented [31-33].As the most important category of CDs, full-color CDs have garnered recognition given their properties such as their adjustable light colors [34-36].In addition to its wide application in various fields [37-39], the similar structure between full-color CDs is crucial in explaining the fluorescence mechanism of CDs[40,41].Although several CDs with panchromatic emissions have been synthesized, their very complicated synthesis still requires different precursors and solvents [15,42,43], which greatly limits the development of CDs mechanism and applications [44,45].At present, limited studies have reported on full-color CDs, which limits its understanding.
This review presents a comprehensive summary on the current full-color CDs development statuses and research.In addition, proposed appropriate methods for preparing full-color CDs (solvent adjustment, similar precursors, acid engineering, separation, and purification) were discussed.Some fluorescence emission mechanisms and the application of full-color CDs in the biological and optoelectronic fields were also introduced in detail [46].Finally, an outlook on the development and potential applications of full-color CDs was presented [47].
2.Synthesis methods
In most cases, periodic changes in the carbon source and reaction conditions may result in different CDs optical properties [48-50].In this part, reports on CDs achieving multicolor emission using the same carbon source under different conditions and various carbon sources under the same conditions were summarized and discussed.
2.1.Reaction conditions adjustment
2.1.1.Solvent-engineering
The majority of full-color CDs were previously synthesized by their dissolution in different solvents to achieve full-color emission due to excitation [51].In general, following dispersion in different solvents, the maximum emission wavelength of CDs changed from 431 nm to 641 nm as the dielectric constant (ε) increased.An increase in the solvent polarity reduced the electron-electron power, thereby resulting in PL red-shift [52,53].Similarly, Wanget al.found that CDs emit different PL colors when in different solvents.The solvent-dependent PL characteristics are attributed to the strong dipole interaction between the oxygen and nitrogen functional group structures on the surface of CDs and the solvent[54].
Various methods were developed for the synthesis of full-color CDs [55].Among these methods, reports on the influence of solvents on the synthesis of CDs indicate that this factor is critical to achieving full-color effects.Xionget al.detailed the synthesis of panchromatic CDs by adjusting the ratio of reaction molecules and different solvents (Fig.1A) [56].Solvent conditions can control the dehydration and carbonization processes to produce strong emission CDs with PL emission in the visible light region.
With the development of the solvent effect, the co-regulation of mixed solvents was gradually developed from single-solvent regulation.For example, Wuet al.reported a novel solvent engineering controlled synthesis of full-color CDs [57].Solvent engineering followed two main paths: (1) Using the single solvent (DMF or EtOH)to prepare yellow/red emitting CDs.At present, regulating the carbonization process of CDs using different solvents is the most commonly used method for the preparation of full-color CDs.
2.1.2.Acidity regulation
During the initial formation of CDs, the pH significantly affects the CDs PL fluorescence characteristics.Nowadays, more studies have increasingly discovered regulatory methods for CDs using acidic and alkali conditions.Wuet al.reported an acid reagent strategy that can obtain high-luminescence CDs by adjusting the mass ratio of raw materials and specific acids [58].Fig.1B shows the full-color CDs achieved by different ideal acid reagents.The acidic reagents introduced in the synthesis process play an important role in regulating the PL phenomenon of full-color CDs.
Many studies have indicated that the acid amounts significantly influenced the generated fluorescence phenomena [59].For example, Kailasaet al.reported a simple and green synthesis strategy, using manilkara zapota as the raw materials, and H2SO4and H2O as the solvents to synthesize blue-emitting CDs.CDs emitting yellow and green followed a one-step synthesis with different amounts of H3PO4[60].More interestingly, Yanget al.proposed a unique strategy to achieve acid-mediated full-color fluorescence emission by changing the pH value of the hydrothermal precursor.The obtained CDs showed adjustable fluorescence from blue(B-CDs) to yellow-green (YG-CDs) in aqueous solution.The gradual deepening of research has introduced acidic reagents as an efficient alternative for regulating full-color CDs [61].
2.1.3.Temperature strategy
More importantly, different experimental conditions may seriously affect the PL characteristics, such as the reaction temperature[62], which significantly affects the controllable synthesis of fullcolor CDs.In recent years, more and more full-color CDs have been reported based on the adjustment of the reaction temperature.For example, Yang’s group chose different small organic molecules as single-carbon precursors and nitrogen sources [63].According to Fig.1C, full-color CDs from blue to orange-red were prepared following a temperature increase from 120 °C to 180 °C.Interestingly,the CDs exhibited an increase in the QYs with the pyrolysis temperature.This indicates that the evolution of the surface states is significantly dependent on the temperature.In other words, increased temperature can enhance the surface functionalization and passivation, thus resulting in an emission wavelength red-shift and increased QYs.
Similarly, Zhouet al.detailed the direct hydrothermal carbonization of citric acid (CA) and urea to synthesize nitrogen-based CDs in a DMF solution [64].Interestingly, a series of solutions of different colors (cyan, yellow, and red) were obtained at different temperature.At the same time, as the reaction time increased, the PL peak exhibited a red shift, indicating that the reaction temperature and reaction time were critical to the reaction.Temperature and time are the most direct factors in the synthesis of CDs.It is the easiest way to achieve full-color CDs preparation through temperature and time-control.
2.2.Precursor adjustment
Full-color CDs preparation through the control of similar precursors is a commonly applied control strategy [65].For example,Linet al.selected three isomers of aniline to prepare a full-color CDs.As shown in Fig.1D, the prepared CDs emits bright and stable three primary colors, showing the maximum at 435, 535, and 604 nm, respectively [66].

Fig.1.(A) The synthesis route of CDs with different solvents and appropriate ratios.Copied with permission [56].Copyright 2018, Wiley-VCH.(B) The synthetic route for preparing full-color CDs using oPD and specific acid reagents.Copied with permission [58].Copyright 2020, AAAS.(C) The synthesis of CA-based full-color CDs controlled by temperature.Copied with permission [63].Copyright 2019, Royal Society of Chemistry.(D) CDs preparation with analogs.Copied with permission [66].Copyright 2015,Wiley-VCH.
As compared to the above-mentioned research, some studies conducted under the same carbon source conditions achieved fullcolor emission CDs by adjusting similar nitrogen sources.Yanget al.used CA and different nitrogen sources as the raw materials to synthesize CDs with four colors [67].At the same time, the preparation of full-color CDs was achieved by adjusting the precursor materials to change the degree of graphitization and the surface state.
2.3.Synergy adjustment
An increased number of publications have reported on full-color CDs and significant factors, such as temperature, time, pH, and precursors, that can be adjusted to regulate panchromatic fluorescent CDs.Multiple adjustments can affect their size, graphite N content,and surface groups to better achieve the full-color regulation.
In comparison, full-color CDs can also be obtained by simultaneously adjusting similar precursor molecules and solvents.Zhanget al.reported the adjustment of two isomers, in ethanol or acetone by the hydrothermal method to obtain full-color CDs [68].In the multi-regulation process, the addition of acid not only affects the dehydration and carbonization process of the precursor, but also affects the fluorescence properties of the CDs.As shown in Fig.2A, Yanget al.used 3,5-diaminobenzoic acid and 3,4-diaminobenzoic acid as the carbon sources to successfully synthesize CDs that emit blue (B-CDs) and red (R-CDs) under different heating time conditions (8 h and 12 h) [69].Subsequently, based on the synthesis method of B-CDs, CDs were generated from the addition of phosphoric acid.RGB-CDs have stable optical properties and significant photoluminescence properties that are independent of excitation.As a result, these three CDs exhibited excellent and stable PL properties in both solution and in solid substrates.
Similarly, Fanet al.controlled the mixing of CA and 2,3-diaminonaphthalene or 1,5-diaminonaphthalene (DNA) in an ethanol solution, to which the mixture was solvated in a heat treatment at 200 °C at various times, thereby generating a range of full-color CDs (Fig.2B) [70].Increased CDs conjugatedπ-domains presented a gradual decrease in the band gap, resulting in blue to red fluorescence emission changes.Under the synergistic effect of two or three factors, it is easier to realize the controllable preparation of full-color CDs through multiple adjustments.
2.4.Other methods
In general, many mixtures are produced during the one-pot hydrothermal synthesis of CDs.Purification is a necessary means to obtain single fluorescent CDs.In the field of CDs, purification methods such as silica gel column chromatography, dialysis, and centrifugation are favored.
2.4.1.Purification
Column chromatography is a widely applied and efficient separation method for the purification of individual chemical compounds from compound mixtures [42,71,72], wherein different CDs species are separated from their as-prepared solutions based on their polarity.Liet al.pioneered the use of silica gel column chromatography for CDs separation based on sizes using multicolor emissions [73].Wanget al.recently generated multiple CDs by solvating m-aminophenol under acidic conditions.The crude residue used CH2Cl2/CH3OH (10:1, v/v) as the eluent, which was further separated and purifiedviasilica gel flash column chromatography to obtain seven CDs samples with different fluorescence properties.
Interestingly, Xionget al.used urea andp-phenylenediamine as carbon sources to prepare a mixed CDs solution using a one-pot hydrothermal method.Ethyl acetate and ethanol were added to the eluent of the CDs mixture to adjusted the different polarities,thereby generating CDs emissions ranging from blue to red, which were eluted in batches through silica gel column chromatography(Fig.2C) [74], Further experiments verified the dependence of the CDs red-shift on the carboxyl group content and degree of oxidation.This further proves that the separation of CDs is due to differences in polarity [44].Full-color CDs preparation by column chromatography not only allows for further CDs purification, but also facilitates in the exploration of the PL mechanism of CDs.
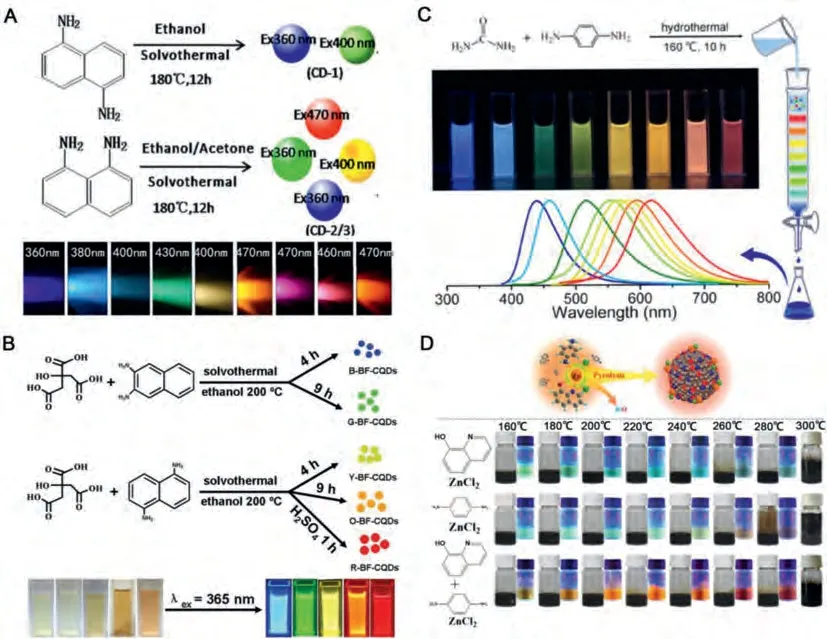
Fig.2.(A) The preparation of full-color single particle luminescence CDs.Copied with permission [69].Copyright 2019, Springer.(B) The preparation of CDs from blue to red by the solvothermal treatment of CA and DAN.Photographs of M-CDs under daylight (left) and fluorescence images (right) under UV light (excited at 365 nm).Copied with permission [70].Copyright 2016, Wiley-VCH.(C) The synthesis and purification of full-color CDs via urea and p-phenylenediamine used as raw materials.Copied with permission [74].Copyright 2015, American Chemical Society.(D) The synthetic route of CDs with high crystallinity and multiple emission was prepared in organic solvent by the ion thermal method.Copied with permission [79].Copyright 2020, Elsevier.
2.4.2.Reflux, electrochemistry, ionic thermal method
The preparation of full-color CDs has mainly adopted solvothermal method and microwave route [75].However, the use of the acid oxidation etching, electrochemical, reflow, and ionothermal methods have gradually increased.For example, Panget al.oxidized carbon fiber with concentrated nitric acid at 120 °C, to which the mixture was allowed to reflux for 6–48 h to generate several CDs emitting blue, green, or red emission [76].Increased reflow research have indicated that the influence of reflow time on the optical properties cannot be ignored.Zhanget al.obtained CDs with different fluorescence properties by refluxing CHCl3and DEA at various times [77].CHCl3and ethylenediamine were refluxed for 1 h to prepare blue-emitting CDs (B-CDs), after which additional reflux for 60 h was applied to prepare full-color CDs (F-CDs).This method was high reproducible and generated stable CDs that were compatible with multiple solvents.For the first time, Yanget al.used a simple electrochemical method to prepare full-color emission CDs (F-CDs) using L-tryptophan as the raw material [78].The prepared F-CDs showed obvious concentration-dependent FL behavior, including full-color emission under the excitation of ultraviolet, blue, and green lights.The CDs exhibited significant biocompatibility and good light stability, which provides a new research mode for multicolor imaging and expands CDs applications.
Recently, the ionthermal synthesis method has been increasingly employed for CDs preparation.Wuet al.thermally synthesized CDs with highly crystalline and emissive CDs in an organic solvent by ion thermal method.ZnCl2was used as a pyrolysis accelerator [79].As shown in the Fig.2D, under the mild reaction conditions of 210 °C, CDs with tunable wavelengths were prepared by direct carbonization from different O and N precursors.However, these methods exhibit certain shortcomings and are not most commonly used for full-color CDs preparation.
3.Fluorescence mechanism of full-color emission CDs
CDs can be prepared using a variety of precursors though different methods, which hardly explains their fluorescence properties using a uniform mechanism [80].As for full-color CDs, several mechanisms have been put forward to explain the fluorescence properties such as quantum size effect, surface modification, and heteroatom doping.Understand the factors that affect the fluorescence characteristics of full-color CDs and the luminescence mechanism is of great significance.
3.1.Effect of graphitic N doping
Apart from those methods, elements doping can be applied to improve fluorescence properties [81-83].The emission wavelength can be adjusted by changing the elements and doping capacities[84-87].Nitrogen is widely used due to its special position in the periodic table.The use of N can also form different structures and possibly produce CDs emission wavelengths [5,39,88].The application of a complex N-doping process may produce multiple nitrogen species on the CDs structure, to which the N-doped CDs properties are largely dependent upon the N atomic configurations [89-91].Four types of nitrogen are formed in CDs: amino, pyrrole, pyridine, and graphite types [92].The mechanism of graphitic N has been extensively studied through experiments and theoretical calculations, wherein results have indicated that the introduction of graphite N results in the red-shift of luminescence.
Zborilet al.synthesized a full-color CDs mixture from CA and urea.Then, according to the difference of the CDs charges, column chromatography was applied to separate the fluorescence components, to which blue, green, yellow, and red luminescent CDs were obtained [93].As shown in Fig.3A, the time-dependent density functional theory (TD-DFT) calculations show that red fluorescence originated from a narrowed band gap due to doped graphitic N.Increased graphitic N on the CDs structure resulted in the emission of red fluorescence by the red-CDs, observations which were confirmed by both experimental and theoretical data.
Different from the above-mentioned research on the same precursor, some studies have reported on the influence of graphite N doping by changing the different nitrogen source.Chen developed a simple strategy to synthesize graphitic N (GN) with controllable content of CDs using precursors at various chemical reducibilities during their solvothermal reactions [94].Simply put, the absence of an additional nitrogen source or weakly reducing additional nitrogen source during the synthesis process resulted in a lack of GN in the sample, such that only a single FL emission was observed in the CDs.GN was only observed in the CDs sample when an additional nitrogen source with a strong enough reducibility was applied, which produced an additional red-shifted FL emission at a longer excitation wavelength.Subsequently, the effect of nitrogen doping on the optical performance of CD was further verified by TD-DFT (Fig.3B).Lowered HOMO-LUMO (H-L) gaps for models GN1 and GN2 indicated that the presence of GN generated a longer FL emission wavelength.In addition, the longer FL emission wavelength in GN2 exhibited a larger relative intensity as compared to GN1, which suggests that the higher doped GNS provided more significant FL emissions at longer wavelengths.The observed trend of the calculated FL results was well in agreement with the experimental results, indicating that both the experimental and theoretical conditions during the introduction of graphite N significantly influence the production of the fluorescence red-shift.

Fig.3.(A) The calculated UV-vis absorption spectra of N-doped model and N-free system P0 of the same size: carbon (green), hydrogen (white), oxygen (red) and nitrogen(blue).Copied with permission [93].Copyright 2017, American Chemical Society.(B) The calculated UV-vis absorption spectra and energy level alignment of the N-doped model (N1, GN1 and GN2) and the non-doped model N0.Copied with permission [94].Copyright 2020, Elsevier.(C) The absorption and emission spectra for a: pristine GQDs and five-edge N-doped, b: pyrazole, c: pyridazine, g: pyridinic, h: pyrrolic, j: amino GQDs in THF; The Absorption spectra of four center N-doped d: graphitic, e: pyridinic, f:pyrrolic, i: amino GQDs in THF, respectively.Copied with permission [95].Copyright 2016, Royal Society of Chemistry.
According to in-depth studies on the mechanism of graphitic N,the fluorescence properties of graphitic N are significantly dependent upon the surface N doping.Wanget al.adjusted the energy level of the excited state through edge N doping, which increased the probability of radiation transition and thus enhanced the fluorescence intensity [95].This work used TD-DFT calculations to theoretically investigate the absorption and emission optical properties of CDs (Fig.3C).The results show that the type and location of N doping play a key role.The CDs produced by edge N doping exhibits higher QYs, and the red shift of absorption and emission spectra is largely determined by graphite N doping.In addition,graphitic N doping can introduce a new intermediate gap state between the highest occupied molecular orbital and the lowest unoccupied molecular orbital, which reduces the band gap, leading to a red-shift.All in all, the doping of graphitic N decreases the band gap in the CDs, thereby the emission of full-color CDs can be achieved by doping different amounts of graphite N.
3.2.Effect of sp2/sp3 hybridization
CDs mainly contain sp2and sp3carbons, and the different content of sp2/sp3carbons in the carbon skeleton will affect the electronic transition, which in turn affects the luminescence properties of CDs.In addition, the emission wavelength of the CDs can be adjusted by controlling the size of conjugated sp2-domains, which were validated by theoretical calculations and experimental results[96].
Our group used the precursor CA, which contains the sp3hybrid structure, and oPD, which contains the sp2hybrid structure,to add acid and adjust the reaction temperature to hydrothermally synthesize full-color CDs [19].A series of theoretical calculations showed that increasing the number of strong sp2hybrid domains in the CDs cores will emit longer wavelengths in the visible spectrum.By designing several sp2hybrid benzene ring structures, the influence of the size of the CDs on fluorescence emission was evaluated.As shown in Fig.4A, the increasing size gradually redshifted the excitation wavelength.The red shift was attributed to the narrowing of the band gap, which was caused by the delocalization ofπelectrons.For CDs of similar sizes, increasing the degree of internal sp2hybridization resulted in red-shifted emission,indicating that the size and sp2/sp3hybridization domain significantly affected the fluorescence emission of the CDs.
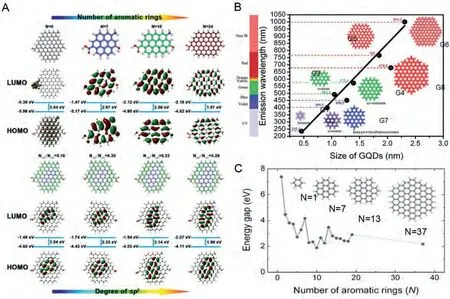
Fig.4.(A) The HOMO and LUMO states of the model were established by the increasing aromatic ring and ratio of sp2/sp3 hybridization domains.Copied with permission[19].Copyright 2020, Wiley-VCH.(B) The function relationship between the emission wavelength and the CDs diameter calculated by the density functional theory (TDDFT)method, where the solid line is a linear fit with different sizes of the sawtooth CDs.Copied with permission [98].Copyright 2014, Royal Society of Chemistry.(C) The relationship between the energy gap of transition and the number of fused aromatic rings calculated by DFT.Copied with permission [99].Copyright 2010, Wiley-VCH.
As compared to the above research, the use of small organic molecules to adjust the solvent can also be similar to the conclusion.For example, Quet al.used two common precursors, namely CA and urea, with three different solvents to prepare full-color CDs [97].Different solvents generally affect the dehydration and carbonization progress during the high-temperature reaction, thus forming sp2domains of the different sizes.The size of the CDs gradually increased in water, glycerol, and DMF Increasing CDs sizes indicates increasing conjugated sp2-domain sizes, which reduces the absorption bands of CDs.The study also suggested that an aprotic solvent such as DMF provided a higher degree of dehydration and carbonization of the CDs precursors than a protic solvent such as water.Based on these observations, conjugated sp2-domains of different sizes can have obtained by turning the volume ratios of the aprotic solvents and protic solvents during solvothermal synthesis.
The CDs band gap openings and its respective PL emissions are highly dependent on the size.That is, larger diameters generate longer emission wavelengths.However, these expected size-related emissions have not been clearly demonstrated by previous studies.This work calculated the emission wavelength of the original sawtooth-edge CDs with different diameters [98].These (G1-G6)have ultraviolet absorption from deep UV to near-infrared (NIR),to which their sizes ranged from 0.46 nm to 2.31 nm (Fig.4B).The obvious size dependence indicates that the diameter of CDs increases from 0.89 nm to 1.80 nm, thus realizing full-color emission.The red shift of the emission wavelength caused by the increase in size is attributed to the reduction of the band gap caused by the delocalization ofπelectrons.
The carbon core is a two-dimensional network composed of sp2and sp3bonded atoms, which contrasts with an ideal graphene sheet composed of 100% sp2hybridized carbon atoms.This unique CDs atom and electronic structure consists of variable sp2/sp3components, which opens up possibilities for new functions.Recently,the PL of chemically derived CDs was investigated.CDs luminescence was observed in the visible light and NIR wavelength ranges.Chowallaet al.observed medium reduction, which consisted of several monolayers of CDs films [99].The CDs films peeled off under different conditions exhibited different fluorescent properties,which were further explained through theoretical calculations.DFT was used to calculate the energy gap of theπ-π*transition as a function of the number of molten aromatic rings (Fig.4C).The energy gap mainly depends on the size or conjugation length of sp2clusters.Therefore, blue emission was assigned to the recombination smaller sp2clusters with fewer aromatic rings.
3.3.Effect of the surface state
Several studies have reported that the surface functional groups of CDs are critical to their properties [100-103].Different precursors and reaction conditions generate different surface groups [39].Usually, the surface contains polar groups, such as carboxyl, hydrophilic hydroxyl or amino groups, and these polar groups have a great influence on their fluorescence properties [51,76].In addition, many studies have shown that the PL center is located in the surface state, which is a hybrid structure formed by the edge or surface oxygen/nitrogen functional groups, which causes the structure to transform into an aromatic nucleus.Therefore, further understanding of the role of functional groups is essential to control the surface functional groups, thus synthesizing CDs with different fluorescent properties.
Quet al.examined the enhancement of NIR fluorescence using surface engineering, wherein CDs were modified with sulfoxide/carbonyl-rich molecules or polymers [104].The X-ray diffraction (XRD), energy dispersive X-ray (EDX), X-ray photoelectron spectroscopy (XPS) and Fourier transform-infrared spectroscopy (FT-IR) results indicate that electron acceptor groups, reacted with the N and O functional groups on the surface of CDs, ultimately interacting with the NH2group.The layered CDs structure exhibited an outer layer and boundary that may exhibit bonding sites (Fig.5A).The S=O/C=O groups interacted with the CDs surface bonding sites.An increase in surface oxidation resulted in a lower outer layer LUMO level as compared to the inner layer, such that the CDs exhibited a NIR absorption band and NIR emission.
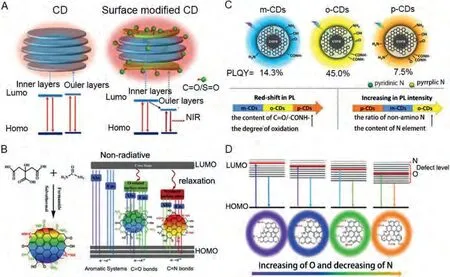
Fig.5.(A) Schematic diagram of the structure and energy level alignment of untreated CD (left) and molecularly modified CDs (right).Copied with permission [104].Copyright 2018, Wiley-VCH.(B) Structure model and fluorescence emission mechanism of CDs with tunable panchromatic luminescence.Copied with permission [105].Copyright 2020, Elsevier.(C) Schematic diagram for fluorescence mechanisms among CDs.Copied with permission [106].Copyright 2018, Royal Society of Chemistry.(D)Model for long-wavelength emission and high quantum yield of CDs with different oxygen and nitrogen contents.Copied with permission [107].Copyright 2019, Royal Society of Chemistry.
In general, solvent adjustments can be performed to change the surface functional groups.In comparison, Wanget al.successfully achieved tunable CDs with panchromatic luminescence on the full visible spectrum through a simple oxidation/reduction method [105].As shown in Fig.5B, both oxidation and reduction treatments can effectively adjust the number of surface functional groups on CDs.The addition of the strong oxidant can convert the reducing functional groups on the surface of CDs into an oxidized state.For CDs, strong reducing agents reduce the oxidizing functional groups on the surface of the original CDs to lower valence functional groups.O-related or N-related defect states were observed following oxygen and N doping in the HOMO and LUMO gap.In addition, the lower energy states exhibited N-related defect states.Therefore, oxidation (o-CDs) and reduction (r-CDs) on the CDs surfaces enhanced PL band transformation into blue/green and red light regions, respectively.
Similarly, Chenet al.prepared three kinds of CDs using phenylenediamine isomers as raw materials.An increase in the C=O/-CONH- content and C=O/C-O ratio produced a gradual change in the PL red-shift from m-CDs and o-CDs to p-CDs (Fig.5C)[106].The pyridine N and pyrrole N contents in o-CDs were significantly higher than those of the m-CDs and p-CDs.Based on these facts, the C=O/-CONH- group can bring about a local delocalization state, thus increasing strain and deformation on the carbon reference plane and ultimately reducing the energy gap and producing a PL red-shift.In short, the surface states significantly affected the fluorescence color and CDs intensities, which were mainly dependent on the nitrogen groups and oxidation degrees.
Xieet al.prepared four bright full-color fluorescent CDs by ethanol solvothermal stripping method.The absorption and emission wavelengths of the four CDs were significantly red-shifted[107].The AFM and TEM studies indicated that the main CDs structures were mainly composed of graphene layers.Oxygen and N doping produced surface defects in the CDs structures due to their electron effect reducing LUMO (Fig.5D).As such, a lower energy gap and red-shifted absorption and emission wavelengths were observed.Interestingly, increased carboxyl group contents resulted in increased QYs values for the four kinds of CDs (v-CDs to o-CDs) as well as the -COOH contents, possibly due to the COOH locations on the CDs surfaces, which tended to enhance the formation of hydrogen bonds.The internal graphene layer hydrogen bonds enhanced the photoluminescence by increasing the structure’s rigidity.
Recently, the amino contents on the surface was strongly related to the fluorescence properties of the CDs.For example, Rogachet al.conducted a comprehensive study of amino functionalized CDs [100].Through model prediction, the CDs functionalized with amino groups were presumably used as amorphous carbon particles, which then carried many N-containing optical centers on their surfaces.As the degree of CDs surface functionalization increased, HOMO-LUMO charge transfer also significantly increased.Previous reports have indicated that CDs composed of benzene rings had a red-shift of similar magnitude with different numbers of -NH2groups.In other words, the PL peak shifted to longer wavelengths as the number of amino groups increased.The red shift of the peak was attributed to the increased contribution of charge transfer to the HOMO-LUMO transition under the high degree of functionalization.
3.4.Synergistic effect
Researchers have proposed a number of fluorescence mechanisms for CDs given the increasing number of synthesized CDs.However, with the in-depth understanding of the fluorescence mechanism, the fluorescence mechanism of CDs cannot be explaining by one mechanism and oftentimes requires a combination of several mechanisms.In the mixed mechanism, graphitic N dominates and the quantum size effect and surface chemistry play synergistic roles.
For example, Liuet al.prepared three kinds of phenylenediamine isomers (m-PD o-PD and p-PD) by oxidative polymerization [108].The results show that the red shift of the emission wavelength depends on the graphite N doping and the size effect.The DFT calculations indicated that graphitic N in the carbon lattice produced a narrower HOMO-LUMO gap, thus producing redshift fluorescence (Fig.6A).It is expected that the red-emitting CDs show the highest graphite N content.This is inconsistent with the XPS data.Therefore, it is assumed that other factors will affect the band gap change of red CDs.Due to the larger particle size, it is easier to realize the quantum effect, thereby reducing the band gap.The above theoretical calculation results show that graphite N doping and increased particle size will produce red-shifted CDs.
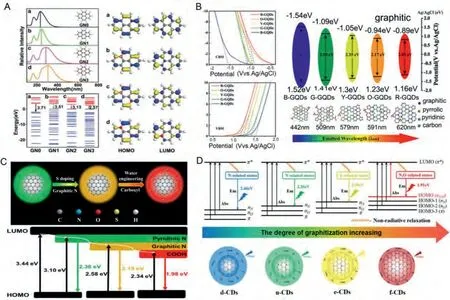
Fig.6.(A) Calculated UV-vis absorption spectra of the N doping model (GN1, GN2 and GN3) and GN0 (N-free system) and the calculated values of HOMO and LUMO and the HOMO-LUMO gap of the N doping system.Copied with permission [108].Copyright 2020, Royal Society of Chemistry.(B) Energy level diagram of PL-tunable GQDs.Copied with permission [109].Copyright 2020, Elsevier.(C) Schematic diagram of the structure model and energy levels of full-color CDs.Copied with permission [13].Copyright 2020, Elsevier.(D) Schematic diagram of the structure, energy levels and electronic transitions among the four kinds of CDs.Copied with permission [67].Copyright 2020,Wiley-VCH.
As compared to the above research, chemical structure engineering can be used to adjust the different N doping by combining different carbon surface sources to change the CDs fluorescence properties.In addition, Wanget al.prepared CDs with blue,green, yellow, orange, and red photoluminescence (PL) by extending theπ-conjugation in the carbon skeleton and introducing different contents of graphitic N according to chemical structural engineering [109].According to Fig.6B, the polyaromatic ring conjugation of the full-color CDs expanded theπ-conjugated sp2-carbon network, thereby reducing the LUMO (π*) level and reducing the band gap, which was largely dependent upon ring size.At the same time, higher graphitic N percentages resulted in decreased band gaps due to the addition of an additional electron in the unoccupiedπ*orbital.The graphitic N exhibited a strong N doping effect due to the presence ofπ*electrons, which will enhance the formation of deep band gap energy levels and reduced the transition energy.Therefore, proper modification of the full-color CDs with different N polyaromatic molecules was able to continuously adjust the HOMO/LUMO energy levels, which resulted in a PL emission shift.Therefore, by expanding theπ-conjugation in the carbon skeleton and introducing different contents of graphitic N according to chemical structure engineering, the regulation of fullcolor CDs can be achieved efficiently.
With the continuous in-depth study of CDs fluorescence emission mechanism, more and more cooperative mechanisms have been discovered.For example, graphite N is dominant and the surface state plays a synergistic effect.Fanet al.using oPD as raw material, obtained fluorescent tunable CDs (G-CDs, Y-CDs and RCDs) from green to red by adding thiourea and water [13].TEM and HR-TEM confirmed that these three CDs have very similar particle sizes, so the red shift of PL has nothing to do with the particle size.It can be seen from the XPS data that the proportion of graphitic N in G-CDs is significantly lower than that of Y-CDs and R-CDs.Combining the spectral data, it is not difficult to find that the content of graphitic N is related to the fluorescence emission wavelength.According to the above results and conclusions,by establishing possible structural models and energy levels, the fluorescence redshift mechanism can be well explained.The fluorescence emission of C-CDs and Y-CDs comes from pyridine N and graphitic N, respectively (Fig.6C).Further, the hydrothermal process increases the hydrophilic functional groups (such as carboxyl groups) on the surface of CDs, resulting in a reduction in the band gap and further red shifting of the emission wavelength.
Yanget al.designed a series of experiments to realize the fullcolor FL emission of CDs [67].By establishing the structure and electronic transition model of CDs, we can further understand the influence of different types of N in the precursor on the fluorescence emission of CDs (Fig.6D).In addition, with the increase of N content, C=N gradually formed on the surface of CDs, and sp2conjugated domains and these surface functional groups together produced more abundant configurations.In short, the emission of various wavelengths of CDs is attributed to the combined effect of eigenstate emission and surface defect state emission.Therefore, by designing a large amount of N doping to achieve a highly graphitized N-rich surface modification structure, it is expected to synthesize high QYs long-wavelength emission CDs.
Since the CDs can be synthesized from different precursors and reaction conditions, the fluorescence mechanism of CDs is different from each other.The purpose of this article is to summarize several fluorescence mechanisms of CDs and hope we can have a good understand of the fluorescence mechanism.We point out that understanding the PL mechanism of the CDs may pave the way to design new strategies for preparing CDs according to our requirement.In this way, it can help us to design CDs according to our meets.As the research deepens, we can explain the fluorescence mechanism of CDs with one unified mechanism in the future.
4.Applications
CDs, as one of the most significant carbon-based fluorescent nanomaterials, are regarded as superior luminescent alternatives with low toxicity and being environment friendly [110-112].The pristine CDs can usually be synthesized from thousands of biological materials and synthetic molecules through a variety of synthetic methods [111-113].They have different surface groups on the surface according to precursors and reaction conditions, which result in superior optical properties [114].Due to their superior optical properties (high photostability, good biocompatibility and facile preparation), CDs have demonstrated many potential applications for modern bioimaging and CDs-based light-emitting diodes (LEDs)[115-117].
4.1.LEDs
Due to their low cost, low toxicity and environmental friendliness compared with rare earth elements, CDs have attracted widespread attention in optoelectronic devices.Up to now, tremendous efforts have been made to improve the properties of utilizing CDs as light-emissive components, for overcoming the aggregationinduced quenching.CDs have excellent performance in LEDs [118-122].Devices can be classified as photoluminescence or electroluminescence according to the cause of light emission.
4.1.1.Photoluminescent LEDs
In recent years, many people have studied the application of multicolor CDs to LEDs.For example, Yanget al.used small aromatic molecules as the raw materials for a one-pot solvothermal reaction to separate from the mixture by silica gel column chromatography [36].As shown in Fig.7A, red (R-), green (G-) and blue(B-) CDs film was obtained by uniformly mixing CDs with three functional groups, phosphor, a dispersant, and a curing agent.The volume ratios of the R-, G-, B-CDs solutions were then adjusted,thus producing CDs phosphor-based white LEDs (WLEDs) with adjustable corresponding color temperature (CCT) and high color rendering index (CRI), which meet outdoor and indoor lighting requirements.Increasing full-color CDs application in LEDs research has resulted in the increased familiarity of full-color luminescent film preparation.
For example, Quet al.used water, glycerol, and DMF individually during solvothermal synthesis to produce full-color CDs from two common precursors [97].A 395 nm-emitting GaN chip was used as the excitation source to prepare full-color LEDs.Fig.7B shows a photo of monochromatic CDs-based LEDs.The device was prepared by continuously covering red and green light-emitting GaN chips with inorganic functional CDs phosphors on a 440 nmemitting GaN chip.This structure helped prevent the red phosphor from absorbing green light and improved the luminous efficiency of the LEDs.The presented results further prove that the microwave-assisted method allowed CDs incorporation into the silica network, which prevented the fluorescence quenching caused by CDs aggregation.
In carbon-based LEDs, full-color LEDs not only emit more colors than full-color CDs, but also allow for high-purity WLED preparation [123].For example, by controlling the molar ratio of citric acid and urea and adjusting the pyrolysis temperature, Sunet al.transferred the maximum emission light of the prepared CDs from blue to red, which covers the entire spectrum [17].First, the CDs dissolved in the epoxy curing agent formed a transparent solution,after which the mixture was cured at room temperature to obtain a CDs/epoxy composite.The emission peak position on the composite material was the same as that of the CDs solution, further confirming that the CDs were well-dispersed in the epoxy matrix.By changing the mass ratio of the three CDs, high-purity WLEDs were prepared.As shown in Fig.7C, the WLEDs exhibited CIE coordinates of (0.33, 0.34) and the CCT and CRI are 5606 K, 92, respectively, which are the highest CRI values reported thus far.Furthermore, the single-component WLEDs exhibited optimal color chromatic stability as the applied current was increased from 20 mA to 90 mA.
4.1.2.Electroluminescent LEDs
Increased CDs application in LEDs has gradually increased the interest in electroluminescence CDs applications.Liet al.chose the triple symmetry of phloroglucinol (PG) as the triangle reagent for the six-member cyclization reaction to form high-purity T-CDs(NBE-TCDs) [124].Using the traditional LEDs structure, NBE-T-CD LEDs were prepared.According to Fig.7D, the EL spectra of NBE-TCDs LEDs are very consistent with the fluorescence spectra of CDs,indicating that NBE-T-CDs has good dispersibility in the PVK host materials.More importantly, the B-, G-, Y- and R-LEDs exhibited half-height widths of 30, 32, 38 and 39 nm, respectively, which was comparable to the developed high color of pure inorganic CDsbased LEDs.The voltage-independent emission color indicated high LEDs color stability, which is of great significance for display technology.
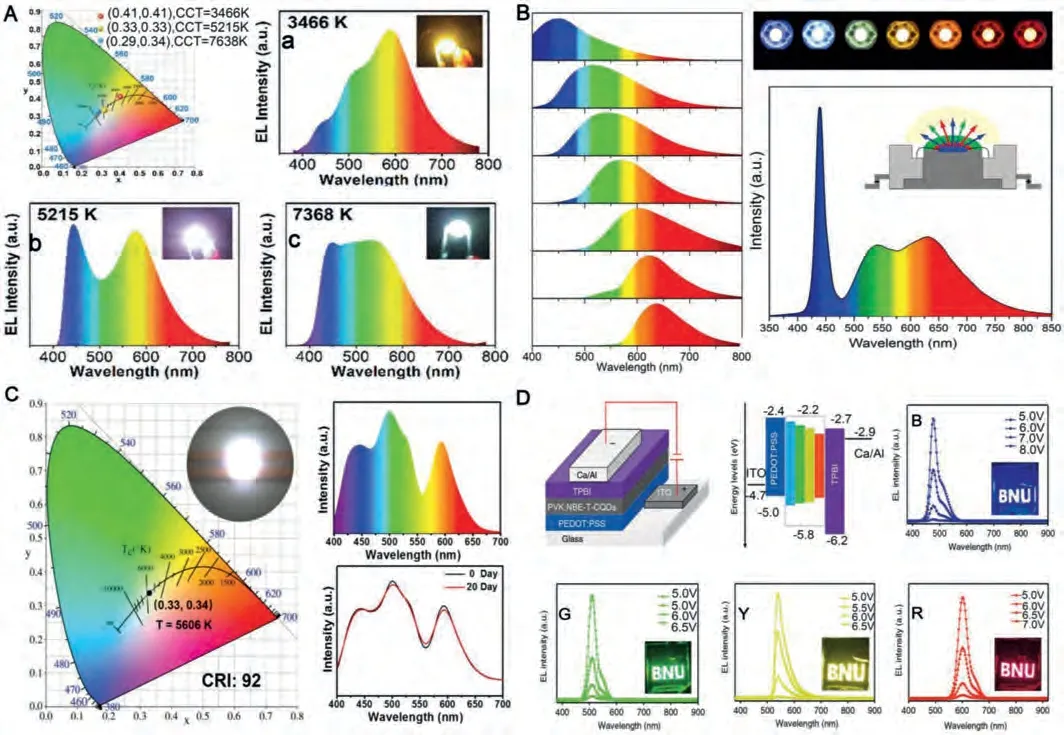
Fig.7.(A) The CIE color coordinates and EL spectra of WLEDs.Reproduced with permission [36].Copyright 2018, Royal Society of Chemistry.(B) The image of a UVpumped WLEDs using the W-CDs as phosphors; and the EL spectrum of the WLEDs lamp.Copied with permission [97].Copyright 2019, the Royal Society of Chemistry.(C)The characteristics of WLEDs and CIE color coordinates (optical photos of WLEDs), the emission spectrum and stability spectrum of WLEDs.Copied with permission [17].Copyright 2017, Wiley-VCH.(D) The device structure and energy level diagram of the NBE-T-CQDs-based LEDs.EL spectra of the B-, G-, Y-, and R-LEDs at different bias voltage, respectively.Copied with permission [123].Copyright 2018, Nature.
With the continuous deepening of LEDs research, Biet al.found new applications for LEDs [125].Since the prepared CDs absorb in the ultraviolet region and the fluorescence emission spectrum covers the entire visible light range, they can fully meet the requirements of plant lighting.CDs can potentially be applied to blue and white LEDs for plant lighting.
Although full-color CDs applications in LEDs have progressed,further improvements in the current photophysical properties are required, particularly for large-scale applications, wherein LEDs luminous efficiency must be addressed.All in all, full-color CDs are highly applicable for LEDs fabrication, though additional investigation is required to generate a cost-effective, high-performance product.
4.2.Bioimaging
Although fluorescent semiconductor quantum dots have made significant progress in biomarking and imaging, the existence of heavy metals and the toxicity problems associated with heavy metals severely limit the application of nanocrystals [126,127].Compared with traditional semiconductor quantum dots, CDs have advanced marking functions, excellent brightness and low toxicity,and are expected to become new medical diagnostic tools [45,128].The biological imaging of fluorescent CDs is mainly divided into cell imaging, tissue imaging, andin vivoimaging [129-132].
4.2.1.In vitro imaging
For full-color CDs, bioimaging generally does not require fullcolor bioimaging, and mainly selects several colors, namely yellow, green, and red [133-135].Because CDs have excellent optical properties and good biocompatibility, the prepared CDs were expected to work as a multiplexed bioimaging reagent [136,137].Linet al.prepared three different emission CDs, using three adjacentp-phenylenediamines as the raw materials for one-pot hydrothermal treatment [66].A preliminary study on the cell imaging capabilities of the CDs was first performed to investigate multiplexed bioimaging applicability.As shown in Fig.8A, the confocal micrographs exhibited different color emissions in the MCF-7 cells.The image displays discharge that was mainly located in the cytoplasmic area, thus indicating that all three CDs entered the cell through the cell membrane.
Similarly, Chenet al.prepared three different CDs photoluminescence (PL), namely blue, yellow, or orange, using the microwave method and the same precursor small molecule [107].As observed in Fig.8B, these CDs passed through the cell membrane, migrated to the intracellular area, and emitted blue, yellow, and orange fluorescence.It is worth noting that this fluorescence tended to be concentrated in a certain location rather than uniformly distributed.This kind of imaging effect with selective labeling of cell nuclei is a very rare phenomenon, which is very useful for biomedical research and disease diagnosis.
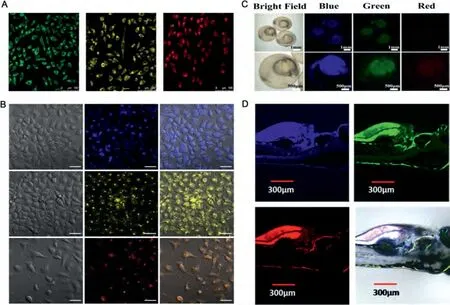
Fig.8.(A) Confocal fluorescence images of m-CDs, o-CDs, and p-CDs at a single 405 nm laser excitation of MCF-7 cells.Copied with permission [66].Copyright 2015, Wiley-VCH.(B) Confocal fluorescence images of m-CDs, o-CDs, and p-CDs incubated MCF-7 cells.Copied with permission [107].Copyright 2018, Springer.(C) Fluorescent images of 2 hpf embryos incubated in CDs solution (400 μg/mL).Copied with permission [138].Copyright 2019, American Chemical Society.(D) Confocal fluorescence images of the ultrabright full color CDs in living zebrafish larvae microinjection.Copied with permission [139].Copyright 2019, American Chemical Society.
4.2.2.In vivo imaging
In the above two studies, full-color CDs positively affected cell imaging, such that Many cells have shown successful effects.In addition to cells, full-color CDs are often used as reagents for tissue imaging (embryos) andin vivoimaging (zebrafish, mice).However, most methods using these imaging techniques were strongly dependent on CDs excitation as compared to full-color imaging,which was achieved under a single excitation light for cell imaging.Full-color CDs were applied to zebrafish embryos to observe their biological imagingin vivo[138].As shown in the Fig.8C, in the control group without CDs, the zebrafish embryos did not show significant fluorescence.On the contrary, the embryos showed strong fluorescence after 22 h of incubation with CDs, indicating that CDs entered the embryo through the chorion.
More and more bioimaging studies have shown that CDs have low toxicity and good biocompatibility, andin vivoimaging has been further studied.Embryos cultured for 5 days were microinjected with CDs solution (50 μg/mL, 50 μL) [139].The samples were then incubated at room temperature for one day.Larval images were then taken with a laser scanning confocal microscope(Fig.8D).The zebrafish images presented strong red, green, and blue fluorescence, including the bone structures, vertebrae, and xiphoid process.As far as we know, this is the first report on the actual tissue application of full-color CDs.
In addition to use in bioimaging, CDs have also exhibited great applicability in selective imaging and drug delivery to tumors.Fanet al.recently synthesized large amino acid mimicking CDs(LAAM TC-CDs) by mixing 1,4,5,8-tetraminoanthraquinone with CA in an aqueous solutionviaa hydrothermal treatment, which can be used in tumor-specific imaging and drug delivery [140].The mice were injected with LAAM TC-CDs, after which fluorescencephotoacoustic imaging was performed.The fluorescence in the tumor region gradually increased with time, such that the highest fluorescence intensity was observed during the 8 days, after which the intensity slowed down.More importantly, fluorescence was not observed in other regions, indicating good selectivity to tumor cells.LAAM TC-CDs exhibited excellent selective imaging properties, suggesting the applicability of the CDs as potential multicolor biomarkers.
5.Conclusions and outlooks
This review presents a comprehensive list of all current synthesis methods, PL mechanisms, and full-color CDs applications.The excellent properties of CDs not only rely on the raw materials, but also on the synthesis procedures, including the reaction solvent,temperature, and acidity.At present, most studies have successfully prepared full-color CDs through slight adjustments in the reaction conditions.
Interestingly, due to the complex and uncontrollable reaction process of CDs, the product is generally a mixture of multiple substances, which can be further purified to obtain full-color CDs.As an environment-friendly material, it is very important to design a synthetic method that could control the production of CDs.As such, thorough understanding of the CDs and PL reaction mechanisms is essential to produce controllable CDs preparation methods.Currently, most studied PL mechanisms focus on graphitic N doping, quantum size, and surface state.In many systems, the fluorescence mechanism of CDs is regulated by multiple actions.Although important achievements have been made in the excitation and emission regulation of polychromatic and full-color CDs, the exploration of required CDs is still in its infancy, such that many challenges still persist in the development of mechanical and practical research.Below, we will share our views and prospects on the field of full-color CDs.
(1) Full-color CDs were generally observed with blue, green, and yellow fluorescence; the controlled generation of full-color CDs is still a big challenge.
(2) The CDs fluorescence mechanism is yet to be fully understood.Although several proposed mechanisms have clarified the PL process and acted as guidance for regulating the PL properties,most are controversial or contradictory.Therefore, there is an urgent need for a comprehensive mechanism to guide effective synthetic routes and innovative applications.
(3) Photophysical characterization is essential for the full disclosure of a more accurate PL mechanism.So far, femtosecond transient absorption,in situPL under high pressure, and absorption measurement have been widely used to assist mechanism analysis.However, advanced test data such asin situdata, multi-photon, and high voltage are insufficient, and further exploration is still needed.
(4) The CDs emissions have been successfully tuned from blue to red and even covered the entire visible precursor area.However, most CDs have exhibited relatively low fluorescence intensity,particularly in the long wavelength range.Manufacturing CDs with efficient and stable emissions at all wavelengths is an urgent problem that must be solved.
(5) CDs have great potential in the application prospects and can be used in sensing, lighting/display equipment, medical, or biological equipment applications.Although many efforts have focused on improving the optical properties of CDs, some limitations still hinder the use of CDs in certain research fields.
(6) For biosensing, improvements in the sensitivity while maintaining selectivity is a daunting challenge.This means that suitable precursors and synthetic methods to determine the chemical properties of CDs need to be carefully selected.Biological imaging requires CDs with long-wavelength emission and high quantum yield to produce high-quality results.
Further research on the mechanism, synthesis optimization,and application development are required.Although these arduous tasks may persist for a long time, huge progress has been made in the field of CDs.Additional research is needed to explore the mysteries of CDs.The future of CDs represents a good vision for basic research and practical applications.
Declaration of competing interest
The authors declare that they have no known competing financial interests or personal relationships that could have appeared to influence the work reported in this paper.
Acknowledgments
This work was financially supported by the National Natural Science Foundation of China (Nos.21905253, 51973200), the China Postdoctoral Science Foundation (Nos.2018M640681,2019T120632), Natural Science Foundation of Henan (No.202300410372), and State Key Laboratory of Bio-Fibers and Eco-Textiles (Qingdao University) (No.KF2020101).
杂志排行
Chinese Chemical Letters的其它文章
- New electron-donating segment to develop thermally activated delayed fluorescence emitters for efficient solution-processed non-doped organic light-emitting diodes
- Microwave-assisted hydrothermal synthesis of NiMoO4 nanorods for high-performance urea electrooxidation
- Self-assemblies of TTF derivatives with fluorinated phenyls and pyridine group
- Plasma treated carbon paper electrode greatly improves the performance of iron-hydrogen battery for low-cost energy storage
- Li-ion charge storage performance of wood-derived carbon fibers@MnO as a battery anode
- Multilayer core-shell nanostructures for enhanced 808 nm responsive upconversion
