Bradykinin Upregulated The Expression of Cyclooxygenase-2 in The Submucosal Plexus of Enteric Nervous System of Guinea Pig*
2022-02-10MAShengYaoWANGWeiYuWANGHuaiJieZHANGDaJinZHANGMeiJiaGAOZhiQinYANGXiaoYunYANGYangQUMeiHua1
MA Sheng‑Yao,WANG Wei‑Yu,WANG Huai‑Jie,ZHANG Da‑Jin,ZHANG Mei‑Jia,GAO Zhi‑Qin,YANG Xiao‑Yun***,YANG Yang,QU Mei‑Hua1,2,***
(1)Translational Medical Center,Weifang Second People’s Hospital,The Second Affiliated Hospital of Weifang Medical University,Weifang 261041,China;2)School of Pharmacy,Weifang Medical University,Weifang 261053,China;3)School of Life Science and Technology,Weifang Medical University,Weifang 261053,China;4)School of Public Health,Qingdao University,Qingdao 266071,China)
Abstract Objective Bradykinin and bradykinin B2 receptors (B2R) play important roles in the enteric nervous system.Bradykinin is usually involved in inflammation and neuroprotection,dependent on the bradykinin‑induced formation of prostaglandins(PGs).Cyclooxygenase‑1(COX1)and cyclooxygenase‑2(COX2)catalyze the conversion of arachidonic acid to PGs.This study aimed to investigate the effect and the signaling mechanism of bradykinin stimulation on the release of prostaglandin E2(pGE2) and the expression of COX2 in the enteric nervous system of guinea pigs.Methods Immunofluorescence was used to detecting the colocalization of COX2 with neural markers Anti‑Hu and chAT in the primary cultured ileal submucosal plexus of guinea pigs.PCR and Western blot were used to detecting the effect of bradykinin evoking COX2 expression.Bradykinin B1 receptor (B1R) antagonist Leu‑8 and B2R antagonist HOE‑140 were preincubated before bradykinin stimulation.COX2 antagonist NS398 and COX1 antagonist FR12207 were used to observing the effect of bradykinin‑induced pEG2 release.Results The results showed that COX2 was co‑localized with neural markers Anti‑Hu and chAT on ileal submucosal plexuses.Bradykinin induced COX2 expression was blocked by the B2R antagonist.The release of pGE2 by bradykinin stimulation in ileal submucosal plexuses was significantly decreased when incubating with the COX2 antagonist.Conclusion COX2 expression evoked by B2R signaling as an excitatory neurotransmitter in bradykinin stimulated pGE2 secretion,which provides a reasonable explanation for the role of bradykinin in intestinal inflammatory diseases.
Key words bradykinin,COX2,pGE2,B2R,enteric nervous system
Bradykinin and kallidin are kinins,a family of endogenous peptides discovered in several pathophysiological events.Kinins are a group of structurally related 9‑11 receptor agonists (Hyp3) ‑bradykinin showed effects comparable to bradykininamino acid peptides that are produced by kallikrein‑mediated enzymatic cleavage of kininogen at the site of tissue injury and inflammation[1‑2].Kinins are formed in plasma and tissuesviathe kallikrein‑kinin system in response to infection,tissue trauma,or inflammatory alterations,such as an increase in vascular permeability,edema formation,and pain[3‑4].Among kinins,bradykinin is widely distributed not only in the periphery but also in the nervous system including the peripheral nervous system,brain,and enteric nervous system (ENS)[5].Bradykinin receptors are cell surface,G‑protein‑coupled receptors of the seven‑transmembrane family.Based on their pharmacological properties,the bradykinin receptor type 1(B1R)and type 2(B2R)are identified[6‑7].
The previous studies showed bradykinin B2R was expressed in a majority of the ganglion cells in the submucosal plexuses in the guinea pig small intestine,suggesting that bradykinin might act in the ENS as a paracrine mediator to alter neural control of secretory and motility functions at the organ level[8‑9].Exposing neurons in the guinea pig small intestinal submucosal plexus to bradykininin vitroevoked slowly activating depolarization of the membrane potential and enhanced excitability characterized by increased firing frequency during intraneuronal injection of depolarizing current pulses in both AH‑and S‑type neurons and the appearance of anodal break excitation at the offset of hyperpolarizing current pulses in AH neurons[10].The results from electrophysiological recording with intracellular microelectrodes suggested that bradykinin acts on bradykinin B2R on submucosal neurons to stimulate the formation of prostaglandins[11].Once formed and released,the prostaglandins (pGs) act to elevate the excitability of the same ganglion cells from which they are released and to diffuse and excite neighboring ganglion cells[12‑13].
Cyclooxygenase (COX) is the rate‑limiting enzyme that catalyzes the formation of prostanoids from arachidonic acid released from membrane phospholipids by phospholipase A2.There are two COX isoforms:COX1 and COX2[14].COX1 is constitutively expressed for basal level as well as for immediate prostaglandin synthesis upon stimulation,particularly at high arachidonic acid concentrations[15].COX2 is induced by cytokines or growth factors and thus contributes to the inflammatory states[16].Bradykinin has previously been reported to induce COX2 expression in various cell types[17‑18].The pro‑inflammatory mediator bradykinin stimulated COX2 expression and subsequently prostaglandin E2 (pGE2)synthesis in dermal fibroblasts[13,19‑20].
The present work aimed to investigate how the signals of bradykinin as an excitatory neurotransmitter on secretomotor neurons at the cellular neurophysiological level are transduced to the physiology of intestinal secretion at the level of the integrated system.As an inflammatory signaling molecule,pGE2 is released by bradykinin through B2R signaling activation and COX2 expression.Therefore,this study provides a theoretical reference for the clinical treatment of neurological diseases.
1 Materials and methods
1.1 Animal and tissue preparation
Adult male guinea pigs (Albino‑Hartley,300-600 g) were sacrificed by stunning followed immediately exsanguination from the cervical vessels according to procedures reviewed and approved by the Weifang Medical University Laboratory Animal Care and Use Committee and Weifang Second People’s Hospital Laboratory Animal Care and Use Committee.A 10‑cm piece of ileum was pinned flat with the mucosal side up to Sylgard 184 encapsulating resin (Dow Corning,Midland,MI,USA) in a dissection dish containing ice‑cold Krebs solution.Fine forceps were used to remove the mucosa and expose the submucosal plexus.Submucosal plexuses preparations were prepared by carefully peeling away the mucosal layer with microdissection.
1.2 Immunohistochemistry
Whole‑mounts of the submucosal plexus were incubated in 10% normal horse serum in PBS for 1 h at room temperature (RT) before exposure to the primary antisera diluted in hyper‑tonic PBS containing 10% normal horse serum,0.3% Triton X‑100,and 0.1%sodium azide.The preparations were placed in humidified chambers and processed for indirect double immunofluorescence staining by incubation for 18 h at RT.The primary antibodies used were rabbit anti‑COX2 (1∶500) (Abcam),mouse anti‑HuC/HuD neuronal protein (1∶200)(Invitrogen),goat anti‑choline acetyltransferase(ChAT) (1∶100) (Chemicon International),sheep anti‑vasoactive intestinal peptide(VIP)(1∶100)(Peninsula Laboratories),sheep anti‑B2R (B2R) (1∶100)(Chemicon International).The preparations were incubated for 24 h in the primary antibody at RT followed by washes with PBS and then incubated in FITC‑labeled donkey anti‑rabbit secondary IgG at RT for 1 h.Then they were incubated with Cy3‑labeled donkey anti‑mouse secondary IgG,donkey anti‑sheep secondary IgG,respectively,for another 1 h at RT.All secondary antibodies were purchased from JacksonImmunoResearch Laboratories (West Grove,PA,USA).The preparations were cover‑slipped in Vectorshield (Vector Laboratories,Burlingame,CA,USA).Fluorescence labeling was examined under a Nikon Eclipse 90i fluorescence microscope (Fryer Company,Cincinnati,OH).The specificity of the COX2 antibodies was tested by pre‑absorbing the antibodies with corresponding blocking peptides provided by the manufacturers,negative controls consisted of omission of either the primary or the secondary antibodies.Double labeling of COX2 with other specific neurochemical markers was used to identify the cell types that express COX2.
1.3 Tissue culture
For these studies,all solutions were treated with 0.1% DEPC water and autoclaved.The guinea pig ileum submucosal plexuses preparations were prepared by carefully peeling away the mucosal layer with microdissection.The submucosal plexuses preparations were incubated in 2 ml Dulbecco’s modified Eagle’s medium (DMEM) with antibiotic antimycotic solution (final concentration contain 100 units/ml penicillin G,100 mg/L streptomycin sulfate,and 0.25 mg/L amphotericin B),glucose(12 mmol/L)in a humidified 37°C,5%CO2incubator before treated by bradykinin or other agents.In the time‑dependent examination,2 pieces of submucosal plexuses were incubated with bradykinin(100µmol/L)for 0,2,4,6,8,24 h before detecting the COX2 expression.To detect whether different concentrations of bradykinin affect the COX2 expression,2 pieces of plexuses were incubated in bradykinin at the concentration of 0,1 nmol/L,10 nmol/L,100 nmol/L,and 1µmol/L respectively for 4 h.
To investigate the signal transduction of COX2 expression,the preparations of the submucosal plexuses of the guinea pig small intestine induced with bradykinin,B1R agonist bradykinin 1‑8(1 mmol/L),or the B2R agonist Hyp3‑bradykinin(1 mmol/L) for 4 h.Other preparations were pre‑incubated with the B1R antagonist Leu‑8 (1 mmol/L)or B2R antagonist HOE‑140 (1 mmol/L) 30 min followed by incubation with bradykinin (100 nmol/L)for 4 h.The tissues were harvested and stored at-80oC for Western blot and RT‑PCR.
1.4 Real-time PCR
Submucosal plexuses preparations,within its matrix of connective tissue,were dissected free and homogenized in Trizol (Invitrogen.Carlsbad,CA 92008,USA) with 100 mg of tissue per 1 ml Trizol.Total RNA was extracted according to the manufacturer’s protocol and dissolved in diethylpyrocarbonate (DEPC)‑treated water.The extracted RNA was treated with DNase and stored at-70oC until use.RT‑PCR was carried out with the SuperScript IIIReverse Transcriptase(Invitrogen)in a 20µl tube containing 2µg of total RNA,2µl(10 mmol/L) dNTP mixture,5 units RNase inhibitor(Invitrogen),600µmol/L random primer (Roche).Reverse transcription was performed according to the manufacturer’s protocol.For PCR,1 µg of the reverse‑transcribed cDNA was placed in a tube that contained 2µl of 2 mmol/L dNTP,1XPCR buffer,1µl dithiothreitol (0.2 mmol/L),200µmol/L sense and antisense primers (Integrated DNA Technologies)mix in a final volume of 50 µl.The final products were run on 1% agarose gels.The primers used in the experiments included COX2,5'‑TGGTGGACTG‑GAATCTTGAA,3'‑CTCTCAGTTGCTCCTGGTCA,and β‑actin,5'‑AGTGTGACGTT‑GACATCCGT,3'‑TGATCTTCATTGTGCTGGGT.
1.5 Western blot
Proteins (100 µg/well) of guinea pig submucosal plexuses preparations treated by bradykinin for different periods or different concentrations of bradykinin (Sigma) were prepared and separated by SDS‑PAGE.The proteins were transferred to nitrocellulose membranes and the membranes were blocked with 5% non‑fat dry milk.After blocking,the membranes were incubated with a polyclonal anti‑COX2 antibody (1∶2 000,Cayman Chemicals,Ann Arbor,MI).The membranes were washed and then incubated with HRP‑conjugated Goat anti‑RabbitIgG (1∶2 000),and then washed again.Pierce ECL Western blot substrate was prepared according to the manufacturers’ instructions and added to the membranes for 1 min.The membranes were exposed to film for the 5 min.
1.6 Chemicals
Bradykinin acetate was purchased from Tocris(Ellisville,MO).Stock solutions were prepared in Krebs solution or deionized H2O.Pharmacological agents were applied by either adding to the Krebs’bathing solution.
1.7 Statistical analyses
2 Results
2.1 COX2 protein localization in the guinea pig enteric nervous system
Under basal conditions,we observed COX2 immunostaining in the submucosal plexus of guinea pig(Figure 1a,b).COX2‑IR was found in(69.7±2.0)% of anti‑Hu‑IR;(90.2 ± 1.3)% of chAT neurons expressed COX2‑IR (Table 1).The results suggested that COX2 is expressed abundantly in guinea pig submucosal plexuses in the enteric nervous system.The results also showed the colocalization of COX2 and B2R in the submucosal plexuses in ENS of the guinea pig ileum(Figure 1c).

Table 1 Distribution of COX2 to chemical codes in the guinea pig submucosal plexus

Fig.1 Immunohistochemical demonstration of COX2 in the ENS
2.2 COX2 mRNA and protein expression in submucosal plexuses induced by bradykinin treatment
A concentration‑response curve for bradykinin was performed with concentrations from 1 nmol/L to 1 μmol/L bradykinin examined by semi‑quantitative RT‑PCR and Western blot (Figure 2).Incubation of submucosal plexus with bradykinin (1 nmol/L-1 µmol/L) for 4 h increased COX2 mRNA expression in a concentration‑dependent manner,reaching the highest at 10-7mol/L (Figure 2a).The expression level was (3.56±0.82) fold increased when incubated with bradykinin at 100 nmol/L compared to the control,while (1.68±0.45) folds,(2.45±0.25) folds,and(2.16±0.15)folds increased at 1 nmol/L,10 nmol/L,and 1 µmol/L bradykinin concentration compared to the control,respectively.Figure 2b showed a similar concentration‑dependent change of COX2 mRNA expression induced by bradykinin detected by the qRT‑PCR.The change of COX2 protein level induced by bradykinin exhibited similar results as the mRNA expression(Figure 2c,d).

Fig.2 Concentration dependence of bradykinin induced COX2 mRNA expression and protein expression in the submucosal plexuses incubated with various concentrations of bradykinin for 4 h
2.3 The submucosal plexuses incubated with bradykinin stimulated the expression of COX2 in a time-dependent manner
Based on the above study,we used 100 nmol/L of bradykinin treatment for the following experiments.The time course of bradykinin induced COX2 mRNA expression and protein level in the submucosal plexuses of the guinea pig small intestine was detected by RT‑PCR,and Western blot,respectively.COX2 mRNA expression by bradykinin(100 nmol/L) treatment in the submucosal plexus was detected at 0,2,4,6,8,and 24 h.The highest COX2 mRNA expression was detected at 4 h of incubation with bradykinin with (5.64±1.91) folds of increase compared to the control sample using β‑actin as an internal reference.COX2 mRNA expression was increased to (1.40±0.33) folds,(4.76±0.51) folds,(4.90±1.24)folds,and(2.82±1.54)folds at 2,6,8,and 24 h,respectively(Figure 3a,b).
Western blot analysis showed that bradykinin induced COX2 protein expression patterns were similar to the mRNA expression(Figure 3c,d).
2.4 Bradykinin induced COX2 expression is mediated through B2R but not B1R in the enteric nervous system
COX2 mRNA expression and protein level were determined with semi‑quantitative RT‑PCR(Figure 4a,b)and Western blot(Figure 4c,d).Bradykinin induced COX2 mRNA(Figure 4a,b) and protein (Figure 4c,d)expression in the submucosal plexus was unaffected by the B1 receptor antagonist Leu‑8 but was suppressed by the B2 receptor antagonist HOE‑140.B1 receptor agonist bradykinin 1‑8 did not increase COX2 expression,but B2 receptor agonist Hyp3‑bradykinin stimulated COX2 expression.The results indicated that bradykinin induced COX2 expression is mediated through B2R but not B1R in the enteric nervous system.

Fig.3 Time course of bradykinin induced COX2 mRNA expression in the submucosal plexuses of the guinea pig small intestine

Fig.4 Bradykinin-induced COX2 expression is mediated through B2 but not B1 bradykinin receptors in the enteric nervous system
2.5 Bradykinin induced pGE2 production was mediated by COX2
COX catalyzes the release of arachidonic acid from membrane phospholipids by phospholipase A2 to form pGE2[21].Bradykinin induced pGE2 releasing was blocked by COX2 inhibitor NS398,but not COX1 inhibitor FR12207 in the submucosal plexuses of the guinea pig small intestine.Preparations of the submucosal and submucosal plexuses were pre‑incubated for 30 min with the COX1 inhibitor,FR12207 (1 mmol/L) or the COX2 inhibitor,NS398(1 mmol/L),followed by incubation with bradykinin(100 nmol/L) for 4 h.pGE2 was analyzed using ELISA kits.The results showed that COX2 inhibitor NS398 significantly decreased the bradykinin evoked releasing of pGE2 (Figure 5a) in the submucosal plexuses.Diagram described a signaling pathway in this study(Figure 5b).
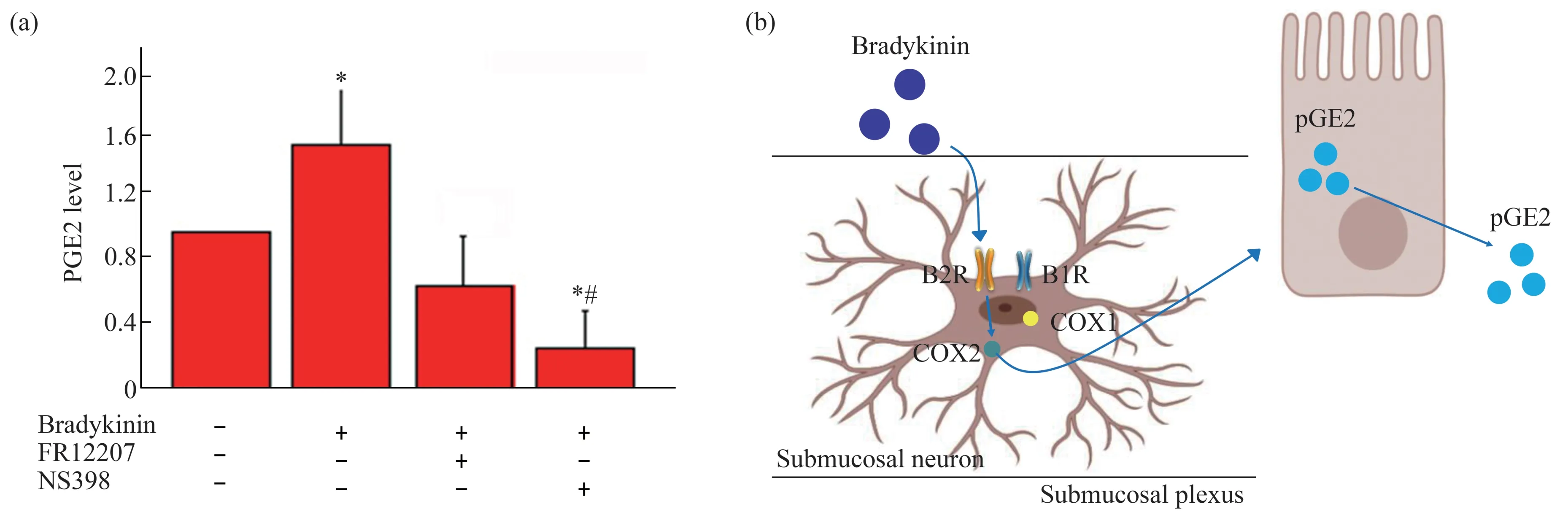
Fig.5 Inhibition of bradykinin induced pGE2 production by COX1 or COX2 inhibitors in the submucosal plexuses
3 Discussion
The enteric immune/inflammatory system communicates with the ENS through the release of chemical mediators,including bradykinin,histamine,prostaglandins,and cytokines[22].Bradykinin belongs to the Kallikrein‑kinin system(KKS),which is widely distributed in the ENS and plays an important regulatory role in various pathological processes and physiological functions.Huet al.[23]showed that bradykinin stimulates enteric muscle neurons of guinea pigs.The cellular effects of kallidin and bradykinin are mediated by two receptors B1R,B2R.B2R is constitutively expressed in a variety of tissues and shows a higher affinity to kinins[24].Our previous results showed that bradykinin stimulates neurogenic chloride secretion in the guinea pig ileum by activating B2R and increasing pGE2 production[25].COX2,known as PGs‑endoperoxide synthase,can be induced by various stimuli in a variety of tissues to promote the biosynthesis of PGs,pGE2 especially,during the inflammatory response of multiple cell types[26‑27].Bradykinin regulates the activity and expression of COX2 through different mechanisms in a variety of cell types including astrocytes[28‑29].This study aimed to reveal the signaling pathway of bradykinin evoking pEG2 releasing in the submucosal plexuses of guinea pig ileum.The current study showed the colocalization of COX2 with B2R and neuron marker Anti‑Hu,ChAT in submucosal plexuses of guinea pigs.Incubated with bradykinin,the submucosal plexuses showed an increased COX2 expression evoked by bradykinin in a time‑ and concentration‑dependent manner.
Amannet al.[30]showed that pGE2 released rapidly after bradykinin stimulating and kept high for at least 3 h from sections of guinea pig ileum tissuein vitro.As the key enzyme in the synthesis of prostaglandins from arachidonic acid,COX was regarded as an inflammation marker and drug target for inflammatory diseases.The two COX isoforms,COX1 and COX2,are reported expressing in neuronal and glial cells responsible for the production of the pGE2[31].In this study,we validated the bradykinin/pGE2 signaling pathway in the submucosal plexuses in the guinea pig ileum.As shown in Figure 5b,bradykinin promoted the expression of COX2 through B2R in the submucosal plexuses in the ileum of guinea pig.This was further supported by the expression of COX2 stimulated by bradykinin was blocked by B2R antagonist HOE‑140,but not B1R antagonist Leu‑8.The secretion of pGE2 in the intestine by bradykinin evoking was blocked by COX2 antagonists other than COX1 antagonists,which implied COX2 was the key factor in pGEs releasing.
4 Conclusion
In summary,we showed that bradykinin induced COX2 protein expression in the submucosal plexuses,which activated COX2/PGE2 signal pathway through B2R.This study revealed the downstream of B2R signaling in the submucosal plexuses and the relationship of pGE2 releasing the COX2 expression,providing targets in the anti‑inflammation drug research in the future.
杂志排行
生物化学与生物物理进展的其它文章
- Protein Aggregation and Phase Separation in TDP-43 Associated Neurodegenerative Diseases*
- 细菌的信号转导系统及其在耐药中的作用*
- CRBGP Inhibited The Activity of Glioma U251 Cells Through Suppressing FAK-AKT Pathway and The Secretion of Interleukin-6*
- 阿尔茨海默病体外诊断纳米技术*
- α-Synuclein as a Diagnostic Marker and Therapeutic Target for Parkinson Disease*
- 甘丙肽对抑郁症状的调控作用及其机制的研究进展*
