Herbal and dietary supplement induced liver injury: Highlights from the recent literature
2021-10-11StephanieWooWilliamDavisSooryaAggarwalJosephClintonSaraKiparizoskaJamesLewis
Stephanie M Woo, William D Davis, Soorya Aggarwal, Joseph W Clinton, Sara Kiparizoska, James H Lewis
Stephanie M Woo, William D Davis, Joseph W Clinton, Sara Kiparizoska, Department of Internal Medicine, MedStar Georgetown University Hospital, Washington, DC 20007, United States
Soorya Aggarwal, James H Lewis, Department of Gastroenterology, MedStar Georgetown University Hospital, Washington, DC 20007, United States
Abstract Herbal-induced liver injury (HILI) is an important and increasingly concerning cause of liver toxicity, and this study presents recent updates to the literature. An extensive literature review was conducted encompassing September 2019 through March 2021. Studies with clinically significant findings were analyzed and included in this review. We emphasized those studies that provided a causality assessment methodology, such as Roussel Uclaf Causality Assessment Method scores. Our review includes reports of individual herbals, including Garcinia cambogia, green tea extract, kratom as well as classes such as performance enhancing supplements, Traditional Chinese medicine, Ayurvedic medicine and herbal contamination. Newly described herbals include ashwagandha, boldo, skyfruit, and ‘Thermo gun’. Several studies discussing data from national registries, including the United States Drug-Induced Liver Injury (DILI) Network, Spanish DILI Registry, and Latin American DILI Network were incorporated. There has also been a continued interest in hepatoprotection, with promising use of herbals to counter hepatotoxicity from anti-tubercular medications. We also elucidated the current legal conversation surrounding use of herbals by presenting updates from the Federal Drug Administration. The highlights of the literature over the past year indicate interest in HILI that will continue as the supplement industry in the United States grows.
Key Words: Herbal-induced liver injury; Dietary supplement-induced liver injury; Druginduced liver injury; Roussel Uclaf Causality Assessment Method; Hepatotoxicity; Liver toxicity
INTRODUCTION
Reports of herbal-induced liver injury (HILI) and dietary and weight loss supplement liver injury (DSLI) continue to be published at an increasing rate, highlighting the growing interest in the field, as well as enhanced recognition of HILI by clinicians. For example, a routine PubMed search revealed eight systematic reviews and metaanalyses on HILI published in 2020 and four in 2019, compared to none published earlier than 2002[1-8]. In this review, we discuss the highlights chosen from the recent literature regarding HILI and DSLI liver injury since our last review period[9]. New information on the incidence of HILI and DSLI, reports of new herbal hepatotoxins and updates on previously described HILI are included, along with the current regulatory status of kratom and other agents.
METHODOLOGY
A literature review for this paper was performed utilizing PubMed and Google Scholar search engines spanning September 1, 2019 through March 31, 2021. Keywords utilized included “hepatotoxicity,” “hepatic toxicity,” “liver toxicity,” “herbal induced liver injury,” “HILI” and “dietary supplements.” Using both search engines, we came across approximately 1800 publications. In order to narrow down this extensive search we focused on case reports, case series, review articles and original research that were published in journals with an impact factor ≥ 1 based on listings contained in Scholar One[10]. Of note, seven of the 85 discrete journals that we reviewed had an impact factor < 1 or none at all. However, we felt the information within those particular articles was important enough to include in our review. The range of journal impact factors (IF) was 0.28-60 (Zhongguo Zhong Yao Za Zhi and Lancet respectively), mean IF was 5.46, and median was 3.37[10]. Additionally, we focused on recent literature reporting new cases of HILI/DSLI along with particular herbal agents of interest such as green tea extract (GTE) and kratom along with performance enhancing supplements (PES), traditional Chinese medicines (TCM) and Ayurvedic medicines. Many reports described the cytoprotective effects of herbal compounds, and we focused on those utilized in human studies. Legal and regulatory ramifications were also addressed in particular with regard to kratom. As in past years, we emphasized those studies that provided a causality assessment methodology, such as RUCAM scores, believing that this enhanced their validity[11]. Through this selection process we narrowed our review to approximately 150 publications (Figure 1). Given the number of publications reviewed, the omission of any specific article should not be viewed as lacking importance or significance.
Figure 1 Study selection flowchart.
INCIDENCE RATES OF HILI/DSLI
Data on the true frequency of HILI/DSLI are generally lacking, in part due to underdiagnosis and under-reporting[12]. The incidence of HILI in mainland China, which we would expect to be among the highest worldwide, is estimated to be 6.38 per 100000 based on the large retrospective study by Shenet al[13], who described DILI incidence to be 23.8 per 100000 of which 26.8% of single agents were TCM. In the United States, the estimated incidence of HILI was 1.16 per 100000 based on a small prospective study conducted in Delaware[14]. Perhaps a better estimation for a Western country comes from a prospective population-based study from Iceland that found an incidence of 3 per 100000[15]. While these estimates are lower than China’s, HILI cases reported to the United States DILI Network have increased from 7% of all drug-induced liver injury cases in 2005 to 20% in 2014, with herbal and dietary supplements (HDS) representing the second leading class of compounds causing liver injury after antibiotics[16]. The most recent update of the United States DILI Network contained 404 cases of HILI enrolled between 2003 to 2019[17].
Registry-based frequency data demonstrates HDS responsible for 8% and 4% of DILI cases reported by the Latin DILI and Spanish DILI Networks, respectively[18]. Data from a single German hospital dedicated to TCM indicates a HILI frequency of 0.12% over a 20-year period[19]. The increasing number of reports of HILI are likely explained by the combination of more widespread HDS use as well as clinician awareness[13].
An ongoing difficulty with assessing a true incidence of HILI relates to the fact that herbal supplements commonly contain multiple ingredients, and several products are often used concurrently. As a result, it is challenging, if not impossible, to determine which specific HDS component might be responsible for the hepatotoxicity[16]. Frequent mislabeling of supplements, patient non-disclosure, and physician lack of awareness further complicate the diagnosis of HILI[16,20]. Nevertheless, it is crucial that clinicians maintain awareness of HILI, as it may have a greater potential for acute liver failure than DILI[16].
REGULATORY STATUS OF HERBAL AND HDS PRODUCTS
In contrast to the United States, herbal supplements undergo much more regulatory scrutiny in member states of the European Union (EU), where according to Directive 2004/24/EC, herbal medicinal products are required to not only register with the EU, but also comply with specific manufacturing and quality standards[21]. Herbal supplements are widely accessible to Americans both online and in nutrition stores and pharmacies, and their appeal is heightened by marketers portraying them as natural and healthy[22]. In 2019, Americans spent $9.6 billion on herbal supplements alone, (exclusive of vitamins and other complementary and alternative therapies, which represented an 8.6% increase from the previous year[23]. As the use of HDS continues to climb, clinicians and patients alike will be faced with the challenge of recognizing and managing potential hepatic injury. The relative lack of regulatory control over HDS in the US compared to conventional medications, means there are fewer protections available to the consumer, such as quality control.
Despite the general lack of regulation of the herbal and supplement industry, the Food and Drug Administration (FDA) does maintain a role in providing for their safety. Herbal medications and vitamin supplements have long been categorized as food supplements and thus have a lower threshold required to maintain evidence for safety[24]. This was changed when the Dietary Supplement Health and Education Act of 1994 (DSHEA) was passed that named the FDA as responsible for safety concerns and for taking action against dietary supplements if needed[25]. Unfortunately, the provision only takes effect after supplements reach the market and supplement companies are not required to register themselves or their products with the FDA before offering them for sale. Until recently, the FDA mainly monitored product information through a voluntary dietary supplement adverse reporting system and took action retroactively against companies when necessary[25,26].
In recent years however, there has been an outcry regarding the sheer number of herbal supplements that have come to market with little to no consumer protection regarding their claims[27,28]. In 2019, then FDA Commissioner, Dr. Scott Gottlieb, issued a statement announcing plans for major policy changes toward the oversight of the dietary supplement industry. This included improvements in the adverse reporting system and a proposal to require the listing of the ingredients of dietary supplements with the FDA[29]. Since then, the proposal to register ingredients of various supplements has been a primary objective of the FDA with both the 2020 and 2021 budget proposals to Congress including a provision for this. In addition, they have asked for a mandate to allow them to act against products and manufacturers providing misleading information to the FDA[30]. However, both proposals have been met with significant resistance from the industry and have yet to become enacted into law.
CAUSALITY ASSESSMENT OF HILI AND DSLI
Diagnosing HILI remains a challenge and while there are several assessment tools used to determine causality, ultimately it is a diagnosis of exclusion[31]. Currently, Roussel Uclaf Causality Assessment Method (RUCAM), designed in 1993, is the most widely used assessment tool for determining causality[32]. Indeed, Teschkeet al[33] identified 12068 HILI cases reported in the recent literature in which RUCAM was used as the basis of causality.
In another retrospective review, Teschkeet al[34] analyzed 11,160 HILI cases from Asian countries - mainland China, Hong Kong and Taiwan, Korea, Singapore, and Japan - collected from 1964 to 2019. They identified China and Korea as being exemplary in their use of RUCAM to evaluate HILI cases. They suggest that RUCAM will be a particularly valuable tool when assessing causality of liver injury occurring during the COVID-19 pandemic, which may confound findings given the high incidence of liver test abnormalities associated with the infection[34,35]. Anirvanet al[36] described the effects of COVID-19 on the liver, concluding it has both direct viral cytopathic mechanisms and also acts indirectly, through immune-mediated, druginduced, and other pathways. These investigators suggest that acute non-icteric hepatitis may precede pulmonary symptoms in COVID-19 infection[36].
RUCAM, however, is an imperfect tool, and some authors argue that it should be developed further as some of its criteria are not evidence-based[31]. For example, RUCAM does not accommodate evaluation of the several individual hepatotoxins that may comprise a single HDS[4]. Other assessment tools include the Clinical Diagnostic Scale (CDS) and Digestive Disease Week Japan 2004 Scale (DDW-J). Liuet al[37] compared RUCAM, CDS, and DDW-J in a cohort of 458 DILI patients at a hospital in Tianjin, China and found the CDS to be the most accurate in diagnosing DILI. The six variables that CDS employs are comparable to RUCAM’s seven, though the former allocates different point values for timing of drug administration to onset of symptoms in addition to assigning points for extrahepatic manifestations including rash, fevers, eosinophilia, arthralgia, and cytopenia[38]. Of note, the most common causative agents for liver injury in this cohort were TCM, used in 52.41% of patients.
BIOMARKERS AND GENETICS
This past year showed continued interest in innovative tools for diagnosing HILI. Liuet al[37] investigated the potential role of anin vitromonocyte-derived hepatocyte-like (MH) cell test in diagnosing HILI. Investigators identified 47 patients in Munich and Hong Kong who were determined by RUCAM to have had HILI. Among these patients, the MH cell test exhibited sensitivity and specificity of 90.6% and 86.7%, respectively. In a prior study, the MH cell test was shown to have higher specificity than RUCAM[39]. Thus, the MH cell test may be a valuable test in diagnosing HILI in the future.
Studies have investigated potential biomarkers for specific agents. Pyrrolizidine alkaloids (PA) are hepatotoxins commonly found in food items and herbs used in TCM, includingGynura japonica (G. japonica). Pyrrole-hemoglobin adducts and three miRNAs - has-miR-148a-3p, has-miR-362-5p, and hs-miR-194-5p - have been shown to increase diagnostic accuracy of PA-induced liver injury. Similarly,Polygonum multiflorum(P. multiflorum) is an herbal popular in TCM. Metabolomics profiling has shown to successfully differentiate between DILI caused byP. multiflorumand autoimmune hepatitis (AIH) as well as hepatitis B virus[40]. Given the widespread ingestion of PA andP. multiflorum, these pioneering diagnostic tests may help guide clinicians in managing liver injury caused by these herbals.
UPDATES IN HILI REGISTRIES
United States
The United States DILI Network (DILIN) examined the association between GTE and the proinflammatory allele HLA-B*35:01 (see “Green Tea Extract”)[41]. The other update focused on ashwagandha, a popular Ayurvedic medicine using data from the United States DILIN and Iceland (see “Ayurveda”)[42].
Spain
Two updates from the Spanish DILI registry were published in 2020. While mainly focused on DILI, there is also comment on HILI. In a study of liver injury in the elderly, Weersinket al[43] found herbal products accounted for 4% of cases in younger patients, with a decreasing overall incidence with increased age. Similarly, in their comprehensive review of DILI over the span of 20 years up until 2018, Stephenset al[44] identified 843 cases of liver injury, 29 (3.4%) of which were attributable to HDS and an additional 22 (2.6%) were caused by selective androgen receptor modulators (SARMs).
Latin America
The Latin DILI Network (LATINDILI) comprises a group of seven countries that collect DILI cases prospectively, using RUCAM to determine causality. Bessoneet al[18] published an analysis of HDS in Latin America from 2011 to 2019, and found that, similar to the findings from the prospective Spanish DILI and United States DILIN, HILI was more common among young women attempting to lose weight[18,45,46]. Rates of acute liver failure were 17%, 16%, and 6%, respectively for the LATINDILI, DILIN, and Spanish DILI networks. In another study using LATINDILI data, Santoset al[5] reviewed 17 records of HILI and foundCentiella asiatica,Carthamus tinctorius, and the weight loss supplement ‘HerbaLife’ (that previously contained GTE and ephedra), as the most common causes[47]. They also found weight loss to be the most common reason for supplement use, which was also the most common indication reported by Bessoneet al[18] Interestingly, whileGarcinia cambogia(G. cambogia) is the third most frequent cause of liver injury in Latin America, as reported by Bessoneet al[18], it was not present in the Spanish DILI registry. The authors suspected this was due to native cultural influences and surrounding geography, as well as the growing potential of different regions.
Malaysia
In Malaysia, a national centralized database of hepatic adverse drug reactions sponsored by the Ministry of Health was used to collect cases retrospectively and Leeet al[47] presented data from 2000 to 2017. They presented 2090 cases of DILI, 11.24% of which were attributable to HDS. Causality was determined using WHO-Uppsala Monitoring Center criteria employed by physician and pharmacist members of the Malaysian Adverse Drug Reactions Advisory Committee (MADRAC). Of note, only 27.1% of products causing liver injury in this study were registered with the Ministry of Health, meaning the vast majority were unregulated. This highlights the similar regulatory challenges faced by authorities in Asia and in the West.
China
There are no significant updates to the China registry since Shenet al[13] extrapolated data from the National Health and Family Planning Commission to conduct the first nationwide study on HILI in mainland China, published in 2019. However, given the surge in literature investigating the impact of COVID-19 on liver injury, a potential confounder, we expect updates to Chinese HILI and DILI registries will be forthcoming.
LiverTox
Livertox is a database founded and maintained by the National Institute of Health. At present, it lists 1095 drugs, including 66 herbal and dietary supplements, and their potential for hepatotoxicity[48]. Likelihood scores are attributed to each herbal or supplement, ranging A-E, as designed by the United States DILIN to determine causality. In the LiverTox compendium, 24 (36.4%) of listed herbs or supplements have an A, B, or C rating, meaning a drug has “well known or more than 50 cases described”, “known or highly likely or 12-50 cases described”, or “probable or less than 12 cases described” to cause liver injury, respectively, based on published reports. In 2020, entries for 11 (16.6%) herbal and dietary supplements were updated on the website (Table 1).
NEWLY DESCRIBED HEPATOTOXINS
Peumus boldus
Peumus boldus(P. boldus) has been implicated as a cause of hepatotoxicity when consumed orally as an infusion, like a tea, especially in elderly patients[49]. The compound Epigallocatechin gallate (EGCG) has been identified as the underlying cause of hepatotoxicity[49]. Oliveiraet al[49] describe a case report of an 87-year-old male patient who presented with weakness, anorexia, and jaundice. He was found to have a hepatocellular injury pattern. It was later discovered that the patient had been orally ingesting infusions ofP. boldusover the past month as a treatment for dyspepsia. After exclusion of other causes of liver injury, the authors determinedP. bolduswas the probable cause of HILI, although they did not include a causality assessment score[49]. The patient’s liver tests returned to baseline with conservative management.
Skyfruit
Skyfruit, also known as Xiang-tian-guo, is used to treat diabetes and hypertension, and was first reported to be hepatotoxic in 2018[50]. Since then, fewer than five case reports are documented in the literature. A 67-year-old woman with skyfruit exposure for six months and presenting with jaundice received a RUCAM score of 7, indicating ‘probable’ causality, described by Shaoet al[51]. Xiaet al[52] describe another case of a 63-year-old woman with a three day history of skyfruit use, who developed epigastric pain, nausea, and fever, and was given a RUCAM score of 10, indicating ‘highly probable’ causality. As diabetes and hypertension are common afflictions and clinicians become more aware of skyfruit’s hepatotoxic potential, the incidence of skyfruit-induced liver injury may increase.
Ashwagandha
Ashwagandha, from the roots ofWithania somnifera,is an Ayurvedic medication used to treat anxiety, depression, and erectile dysfunction. Björnssonet al[42] published a case series, drawing from an Icelandic registry and the United States DILIN, of five patients with Ashwagandha-induced liver injury. The authors used DILI expert opinion to determine causality in these patients who developed jaundice and pruritus after a latency period ranging from two to twelve weeks. The pattern of liver injury was cholestatic or mixed and liver enzyme abnormalities self-resolved within one tofive months in four of the five patients; the fifth patient was lost to follow up. Prior to this paper, only one case report had been published on the topic.
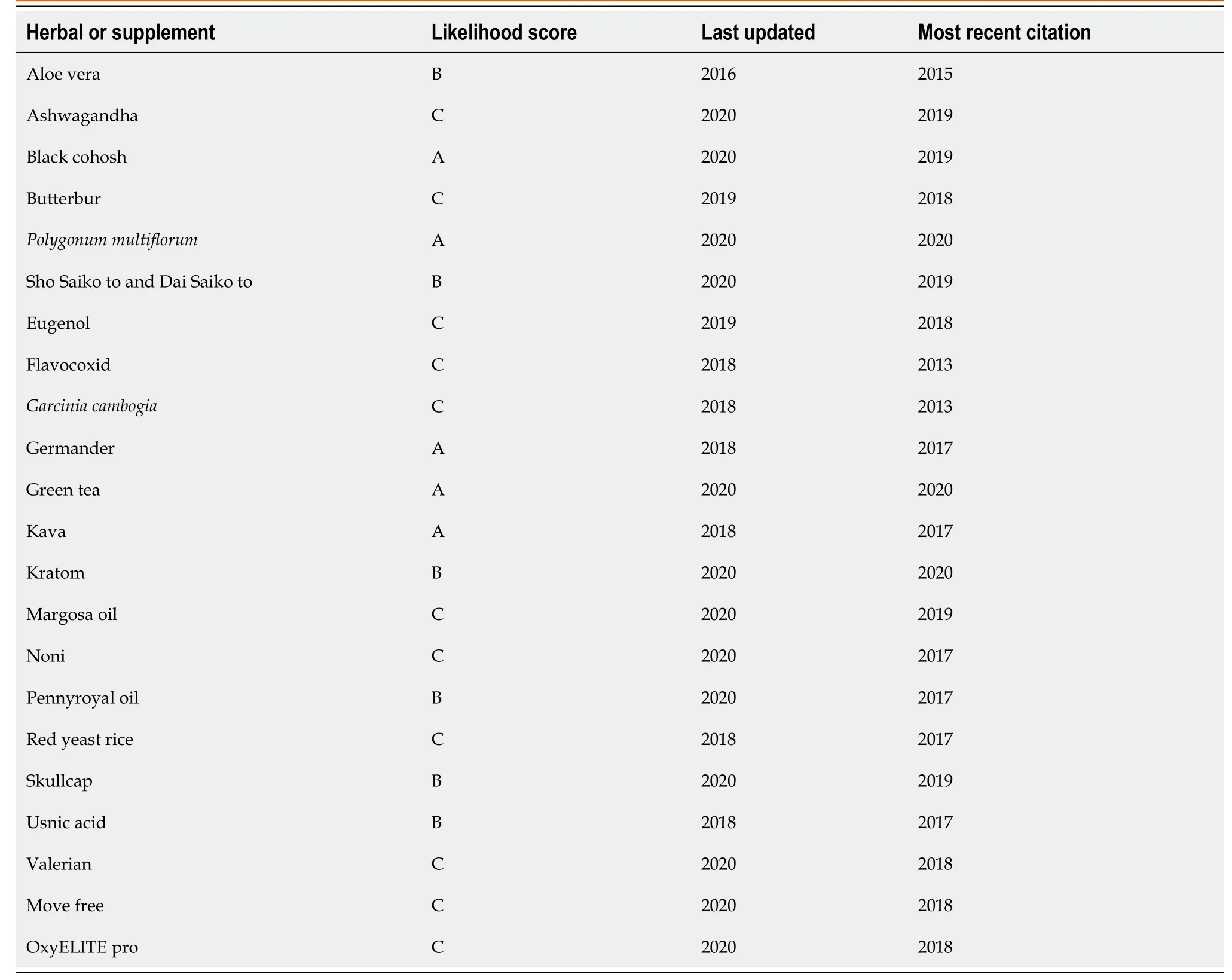
Table 1 Herbs or supplements with an A, B, or C rating as listed in LiverTox
‘Thermo gun’
Ferreiraet al[53] described a case of a 36-year-old male who presented to the hospital with jaundice one week after taking the dietary supplement ‘Thermo gun’. The authors reported no previous reports of a HILI association, but noted that oxilofrine, white willow, and caffeine could all play a possible role. Laboratory exams showed a cholestatic liver injury pattern. The drug was discontinued but the patient's liver function continued to deteriorate and he eventually developed acute liver failure. He successfully underwent liver transplantation and continued to do well at long term follow up. The authors assigned this case a RUCAM score of 7, indicating ‘probable’ causality by ‘Thermo gun’.
UPDATES ON KNOWN HEPATOTOXINS
Kratom
Kratom is a controversial herbal compound derived fromMitragyona speciosaand originating in Southeast Asia. It has dominated headlines in recent years because of its popularity as a stimulant and the associated legal ramifications of its use to reduce opiate withdrawal symptoms[54,55]. The active components are believed to be Mitragyona and 7-Hydroxymitragynine (7-HMG)[56]. Kratom has been used as a stimulant at lower doses or to treat pain and precipitate euphoria at higher doses. At even higher doses, it acts as a sedative. Although it has found use in people who suffer from opioid addiction to prevent withdrawal, at present there are no medical indications for kratom use in the United States, and the FDA has labeled it a ‘drug of concern’. Despite being banned in countries including Thailand and Malaysia, it remains widely available in the United States over the internet - although it is banned in Alabama, Arkansas, Indiana, Rhode Island, Vermont, and Wisconsin.
Despite these admonitions, it is becoming more mainstream with over $207 million in annual sales[57]. Of note, in 2021, Schimmelet al[58] published the first national survey of kratom use in the United States. Using data from the Non-Medical Use of Prescription Drugs (NMURx) Program, these investigators conducted a cross-sectional study of kratom users in the United States from 2018 to 2019, and found kratom users were more likely to be young, male, and have more severe substance abuse profiles, as measured using DAST-10, than cannabis, alcohol, or cigarette users[58]. They also estimated a prevalence of kratom use of 0.8%.
Cultural differences may influence the use of kratom, and subsequently its adverse effects. Ramanathan and McCurdy argue that kratom has been more harmful in the west as compared to Southeast Asia. These authors propose this is because western users are more likely to ingest kratom recreationally[59]. To further delineate the motivations for using kratom in their Malaysian cohort, they found that current opioid users were more likely to use kratom to ameliorate withdrawal symptoms as compared to former opioid users, who used kratom recreationally (OR 1.9,P< 0.035)[60].
Current legal status
In 2016, the Drug Enforcement Agency (DEA) attempted to classify kratom as a Schedule I drug, meaning it has no medical indication and high potential for abuse, alongside heroin, lysergic acid diethylamide, and methylenedioxymethamphetamine (ecstasy). However, this effort was met with pushback from lobbying groups, members of congress, and the public. A bipartisan group of senators, including Bernie Sanders and Orrin Hatch, signed a letter protesting the FDA’s immediate scheduling of kratom, and encouraged a lengthier investigation into the safety of kratom given its long history of use in other countries and growing popularity in the United States[54]. Moreover, some researchers believe that restricting kratom as a Schedule I drug would prevent advancement of research because of increased bureaucratic processes previously illustrated by studies on marijuana and psychedelic-assisted therapies[61]. Thus, kratom remains legal at the federal level, despite its known hepatotoxic potential.
Polypharmacy
Polypharmacy plays a significant role in kratom’s potential for hepatotoxicity. Mitragynine inhibits glucuronidation by UDP-glucuronosyltransferases (UGT), which may explain kratom’s increased toxicity when co-administered with other substances, such as UGT substrates including buprenorphine and ketamine[56]. Polysubstance abuse with kratom furthermore increases rates of death. The CDC collects data on death from substance abuse in the State Unintentional Drug Overdose Reporting System (SUDORS), and has investigated kratom, most recently publishing updated data in 2019[62]. Of the 27338 deaths due to overdose reported to SUDORS from July 2016 to December 2017, kratom was implicated in 152 (0.56%) cases. Among the 152 cases, medical examiners determined kratom to be a cause of death in 91 (59.9%), with kratom identified as the only substance in seven cases. Egglestonet al[63] conducted a retrospective review using kratom exposures reported to the National Poison Data System and New York State’s county medical examiner’s office records, and found 2312 cases of kratom exposure, of which 935 reported kratom as the only substance used.
Product contamination
The potential lethality of kratom is heightened by issues with product contamination, with both heavy metals and organisms that may cause illness. Most recently, in 2018, the FDA/DEA completed an investigation of kratom products contaminated with salmonella resulting in an outbreak affecting 199 individuals across 41 states[64]. Contaminants in kratom products were most recently found in a survey of kratom use in a Chicago suburb, which also revealed the presence of heavy metals, fungi, and bacteria[65,66].
New reports of kratom liver injury
Despite the safety concerns surrounding kratom, its popularity is continuing to rise. According to data from the System to Retrieve Information from Drug Evidence/STARLiMS, the DEA’s registry for seized drugs, reports of kratom increased to 589 in 2018 from one in 2010[67]. A PubMed query for “kratom” revealed 101 articles published in 2020 compared to just 11 in 2010. The United States DILIN reported 11 cases of kratom-induced hepatotoxicity in the United States from 2003 to 2019, and causality was determined by expert consensus opinion[17].
Schimmel and Dart published a review of 85 kratom cases that nicely summarizes its clinical signature with respect to liver injury[68]. Using published case reports and abstracts, cases in the United States DILIN, FDA databases, and online user forum, they found most patients presented with abdominal discomfort, jaundice, pruritus, and dark urine. While liver tests revealed a mixed injury pattern, histology often showed cholestasis. The authors were only able to calculate a RUCAM score for 20 cases, with a median modified RUCAM score of 5 and mean of 4.5 (range 1-8), indicating ‘possible’ causality.
A newly reported form of kratom-induced injury is cholestasis resembling primary biliary cholangitis. A case report by Gandhiet al[69] from India, is only the second in the literature, reported. Causality in this case was determined by clinical judgment using symptoms of nausea, decreased appetite, fatigue, and jaundice with associated elevated bilirubin levels in the setting of kratom use two weeks prior to presentation. Cholestatic liver injury consistent with primary biliary cholangitis, was confirmed by histology revealing centrilobular cholestasis, moderate chronic portal tract inflammation, and brisk lymphocytic-predominant bile duct injury. Symptoms resolved with supportive care and steroids.
GTE
Green tea is one of the most widely consumed drinks worldwide, and is not considered a hepatotoxin[70]. In contrast, GTE has gained significant popularity for its weight loss enhancing potential and can be found in over a 100 herbal preparations in varying concentrations[70]. and have been associated with the potential for hepatotoxicity[71]. A systematic review of GTE performed by the United States Pharmacopeial Convention (USP) in 2008 and revisited in 2019 urged the use of cautionary labels to warn the general public of such causal relationships[71].
The USP reviewed both human case reports and animal studies to establish the role of GTE in hepatotoxicity. EGCG, a highly bioactive phytochemical, is felt to be the main compound implicated in liver injury and is seen in approximately 10% of GTE formulations at varying concentrations[70-72]. Indeed, the concentration of EGCG has been directly correlated to risk of liver injury[71]. The review conducted by the USP of human cases determined the median intake of 720 mg/d of EGCG for at least two weeks was related to liver injury[71]. Notably, the average over-the-counter GTE supplement contains an EGCG concentration from 45-1575 mg/d[71]. In addition, the bioavailability of EGCG increases in a fasting state, increasing serum concentrations at lower consumed dosages[71].
GTE-related hepatotoxicity almost always presents as an acute hepatitis with a hepatocellular injury pattern[70,71]. While the exact pathogenesis of injury is unclear, proposed mechanisms include the interaction of cytochrome P450 and EGCG, direct mitochondrial toxicity from reactive oxygen species produced by EGCG, or possibly, bactericidal effects of EGCG causing endotoxic induced liver injury[72,73]. Additionally, there is believed to be an idiosyncratic, dose-independent cause in genetically susceptible individuals related to individual HLA phenotype[41,72].
Hoofnagleet al[41] performed a retrospective review of 1414 cases of drug induced liver injury, of which 40 were attributed to GTE. 95% of these patients had the typically hepatocellular injury pattern with 3 ultimately requiring liver transplant. Notably, an HLA analysis on these 40 patients found that 72% had HLA-B*35:01[41]. There have been reports of other drugs causing idiosyncratic liver injury related to HLA-B *35:01, including trimethoprim-sulfamethoxazole andP. multiflorum[74,75]. This pharmacogenetic association suggests there may be a possible immunologic susceptibility in GTE-related HILI.
G. cambogia
G. cambogiais derived from the fruit of the Malabar tamarind tree found in South East Asia[76-78]. This herb continues to be an increasingly popular over the counter herbal supplement for its potential for enhancing weight loss[78,79]. Its weight loss potential stems from the active agent withinG. cambogia, hydroxyl citric acid (HCA). HCA is thought to be an appetite suppressant which has demonstrated weight loss in rat models[78,80]. Additionally, HCA prohibits cholesterol and fatty acid synthesis in tissue through inhibition of adenosine triphosphate-dependent citrate lyase enzyme helping in weight reduction[78,81]. Although it is an OTC supplement, caution must be taken as there have been rare, but serious cases of serotonin syndrome, rhabdomyolysis and hepatotoxicity[78].
It has been estimated that approximately 1 in 10000 individuals usingG. cambogiaexperience significant liver-related injury[76,78]. Onset of injury generally occurs over one week to a few months after initiation[77]. The pattern of liver injury is typically hepatocellular. This year, cases ofG. cambogia-induced liver injury with a pattern similar to autoimmune hepatitis appeared[76-79]. Injury and subsequent recovery is frequently managed with abstinence from offending supplements and supportive care[77-79]. However, there have been instances of individuals requiring liver transplant or even death related to such liver injury[78]. Although the pathogenesis of liver injury is unclear, proposed mechanisms through rat models include excessive production of reactive oxygen radicals from lipid peroxidation resulting in increased oxidative stress and cytoplasmic vacuolization signaling hepatocyte injury[77,79]. Nonetheless, there is thought to be two broader mechanisms; a dose-dependent mechanism through HCA consumption and an idiosyncratic, dose-independent etiology[78].
One of the most well-known examples ofG. cambogiaassociated hepatotoxicity was seen in the weight loss supplement, “Hydroxycut™”[81]. This product was recalled in 2009 after the FDA issued a warning of its potential hepatotoxic effects based on numerous case reports reporting severe hepatotoxicity[81,82]. Anduezaet al[82] summarized 21 cases ofG. cambogiarelated liver injury of which seven were attributed to the use of Hydroxycut. RUCAM was utilized in two of seven cases and was deemed ‘highly probable’ in both cases with a score of 9. Hydroxycut™ has been newly formulated in the absence ofG. cambogiaand continues to be marketed. Despite the new formulations however, new cases of Hydroxycut-related liver injury continue to be reported. Yousafet al[77] described a tabulated summary of eight reported cases of non-G. cambogiacontaining Hydroxycut induced hepatotoxicity cases from found in 2010-2018. Of the eight reported cases, RUCAM was used in six, with scores ≥ 6[77]. An additional case associated with the use of “Proclinical Hydroxycut™” over a twelve weeks period presented with tremor, progressive fatigue, chest pain and hepatocellular liver injury on laboratory tests. RUCAM was 9, indicating a ‘highly probable’ causality with this new formulation of Hydroxycut[81].
In addition to the cases of liver injury from Hydroxycut, there have been other notable cases ofG. cambogia-induced liver injury from otherG. cambogiacontaining products this year[77,80,83]. Three recent cases described liver injury related to GCcontaining products occurring four weeks to seven months after ingestion[77,80]. Both were in young patients and presented with hepatocellular injury patterns[77,80,83]. It is important to note, however, that the patient presenting seven months after ingestion was also taking GTE[80]. Two of the three patients ultimately required liver transplant due to failed conservative management[80,83]. RUCAM scoring was used in one of three cases, who did not require liver transplant and recovered with conservative management. The RUCAM score in this case was 9, deeming causality ‘highly probable’[77].
An additional noteworthy case was the first presentation ofG. cambogia-induced liver injury with a pattern of AIH. A 39-year-old female presented with jaundice, hepatomegaly and fatigue five weeks after using “slimming tea” containingG. cambogia[76]. Liver tests demonstrated a hepatocellular injury pattern with positive ANA and anti-smooth muscle antibodies. A liver biopsy was suggestive of DILI with superimposed AIH[76]. Given these findings, the patient was treated with high-dose prednisone but relapsed after a steroid taper, and was eventually transitioned to chronic immunosuppressive agents. No causality score was presented for this patient.
PES
The use of PES has become a billion dollar industry[84]. Usage of multiple different PES is commonplace, confounding the ability to determine causality in many cases of liver injury[84].
SARMs have become increasingly popular outside the fields of bodybuilding and professional athletics[85]. Their selective tissue effects on muscle and bone allow for the benefit of building muscle mass without unwanted side effects[86,87]. SARMs act intracellularly through the binding of androgen receptors that subsequently regulate the production of androgen genes within the cell's nucleus[87]. Due to these effects, SARMs are being actively investigated in the management of sarcopenia, osteoporosis and profound nutritional deficiency. However, they are not approved by the United States FDA for such uses[87].
In fact, the FDA warns users of such supplements due to their hepatotoxic effects[87]. Several recent reports have described both SARM-induced cholestatic as well as hepatocellular injury, all starting within two weeks to four months after ingestion[86-89]. The SARMs described in these cases were Ligandrol (Alpha Elite), RAD-140 (Alpha Bolic) and enobosarm[86-89]. Liver enzymes improved with conservative management in all cases. In the cases described by Floreset al[89], liver injury was related to Ligandrol and RAD-140, presenting five weeks and four months respectively after initial ingestion. Laboratory findings were consistent with hepatocellular and hepatocellular-cholestatic injury respectively. RUCAM scoring deemed both cases as ‘probable’. RUCAM was not used in the other cases, with causality determined simply by ruling out viral, autoimmune and possible other medicationinduced liver injuries[86-88].
Stimulant workout supplements have also been implicated in DSLI[90-92]. These mixtures may vary in concentrations of ingredients or contain undeclared active ingredients that can result in harm[90]. Eiswerthet al[91] described a case of hepatocellular liver injury in a previously healthy 38-year-old male after using a popular preworkout brand “Bucked Up.” It is thought the component “deer antler extract,” which contains insulin-like growth factor, was the culprit for such injury[8]. Liver enzymes were shown to downtrend with supportive care[90]. A RUCAM causality score was deemed ‘probable’ with a score of 7[91].
Two additional cases of pre-workout PES-induced liver injury were reported in previously healthy young adults[90,92]. In one case, the patient was found to have a cholestatic injury pattern[92]. He admitted to taking creatine, whey protein powder and “Mr. Hyde” pre-workout, containing the ingredient theacrine which was thought to be the cause of liver injury[92]. Indeed, rats exposed to theacrine in high concentrations demonstrated centrilobular hepatocellular necrosis[92]. Additionally, the coingestion of caffeine, which is also found in “Mr. Hyde” pre-workout, has been shown to increase the bioavailability of theacrine, potentially raising serum concentrations to hepatotoxic levels[92]. The other case described hepatocellular liver injury after ingesting of “Dust V2” pre-workout consistently for four months [90]. While the patient’s liver enzymes declined with conservative management, his clinical course was further complicated by severe aplastic anemia two months after the initial presentation requiring hematopoietic stem cell transplant[90]. RUCAM was not used to assess causality in either of these two cases.
Additional brief reports of DSLI noted on our literature review included usage of creatine and glutamine powder[84,93].
TCM
TCM aims to establish and maintain balance in patients through acupuncture, massage, tai chi, and herbals, and its influence continues to grow[94]. In 2019, TCM was officially recognized by the World Health Organization[95]. TCM is included in Chapter 26 of the 11th ICD, set to roll out in 2022, which will broaden its reach worldwide[96]. However, controversy exists over this decision, as some clinicians argue it is dangerous to perpetuate practices that are not evidence based[97].
Our search yielded 264 results on TCM published during 2020. The interested reader is referred to a comprehensive review by Panet al[98] emphasizing the complexity of TCM agents and their mechanisms for hepatotoxicity. We will highlight a few examples of TCM liver injury.
P. multiflorum
P. multiflorumis a commonly used and widely researched herbal within TCM, with its major active ingredients being stilbene glucosides and anthroquinones[99]. Although believed to have therapeutic effects on the liver, it is also a known hepatotoxin and is the only TCM listed on LiverTox with a likelihood score of A[49]. Much of the current literature onP. multifloruminduced liver injury is focused on its mechanism of toxicity. Liet al[99] argue thatP. multiflorumliver toxicity is idiosyncratic and immunemediated, rather than direct as previously proposed in the literature. Zhanget al[100] conducted a prospective study using metabolomics to examine serum samples, and identified 25 metabolites that could distinguish between groups susceptible to or tolerant ofP. multifloruminduced liver injury. In another study investigating risk factors forP. multiflorum-induced hepatotoxicity, Yanget al[101] identified HLA-B35:01 as a potential susceptibility factor.
San-Qi and G. japonica
San-Qi is a TCM that is used for hemostasis and to treat trauma and ischemic cardiovascular disease, with the main component beingPanax notoginseng[102]. Two other herbals, both called Tu-San-Qi, one of which contains the pyrrolizydine alkaloid (PA)-producingG. Japonicaand the other isSedum aizoon(S. Aizoon), which does not produce PAs[102].G. JaponicaandS. Aizoonare also known to induce blood flow and detumescence as well as treat pain. The similarity of the names has led to confusion with regard to usage which has led to cases of liver injury, as PAs are known hepatotoxic agents, specifically causing hepatic sinusoidal obstruction syndrome (HSOS). A review by Zhuet al[102] identified 2156 incidences of Tu-San-Qi induced HSOS. While the authors used the ‘Nanjing Criteria’, developed by the Hepatobiliary Diseases Committee of the Chinese Society of Gastroenterology, to evaluate PAinduced HSOS, it is unclear how causality was determined for the patients identified in this study. Furthermore, the authors conceded that in many of the cases, they did not specify which agent was included in the specific formulation of Tu-San-Qi.
Bu Gu Zhi and Psoralea corylifolia
Bu Gu Zhi (BGZ) is a TCM used to treat osteoporosis, and the main ingredient isPsoralea corylifolia(P. corylifolia). In a retrospective review conducted by Wanget al[103], 40 cases of BGZ-induced liver injury were identified at a single hospital in Beijing. Causality was determined using presence of clinical symptoms, namely decreased appetite, dark urine, and fatigue, as well as liver enzyme abnormalities, 92% of which were consistent with hepatocellular injury. Zero patients died or required liver transplantation. This is the first study of this size examining BGZ-induced liver injury.
Rhubarb
Rhubarb, also known as dahuang in TCM, possesses anti-inflammatory properties through its anthraquinones, specifically rhein, emodin, aloe-emodin[104]. Zhuanget al[104] reviewed the literature on the dual protective and toxic properties of rhubarb on the liver, and concluded rhubarb’s hepatotoxicity increases with higher doses and less processing of the product. More studies are required to make definitive conclusions regarding rhubarb’s effect on the liver.
Ayurveda
Ayurveda is another form of ancient medicine, and while not as mainstream in the United States, interest in the field is growing. The practice of Ayurveda comes from India and is based on balancing the five elements to optimize bodily humors[105].
Assessing HILI due to Ayurvedic medication is difficult because labeling of ingredients is often incomplete or incorrect. In one case series, a woman is described to have developed acute liver injury after consumption of a combination powder medication called “puriyas” prescribed by a local healer[106]. When Ayurvedic ingredients are identified, the literature commonly describes ashwagandha, brahmi/gotu kola, turmeric, guggul, bakuchi, Indian senna, aloe vera, Indian mulberry, pyrrolizidine alkaloids[107].
In their case series, Karousatoset al[108] present three patients with HILI from three different Ayurvedic preparations. The medications presented were Giloy kwarth containing the hepatotoxicTinospora cordifolia, followed by a combination of Manjishthadi kwatham and Aragwadhi kwatham, containing 52 and 10 individual plant extracts with 23 and nine known hepatotoxins, respectively, and finally Kanchnar guggulu, comprised of 10 individual plant extracts of which nine are known hepatotoxins. The individual RUCAM scores for each product ranged from 7 to 8, indicating ‘probable’ HILI. The complexity of these preparations highlights the need for clinician awareness with regard to HILI from Ayurveda.
Turmeric
Turmeric has been suggested to have hepatotoxic effects through its active ingredient of curcumin. Lombardiet al[109] published a series of cases of acute liver injury in Tuscany following ingestion of turmeric, using RUCAM to establish a causal relationship that was supported by a positive de-challenge response in six of seven ‘possible’ and ‘probable cases, although the actual RUCAM scores were not provided. A systematic review identified 23 cases of ‘possible’ to ‘probable’ turmeric-induced liver injury, but the majority of patients had a concomitant exposure to another medication. A case reported by Leeet al[110] described a patient who developed AIH following turmeric ingestion, established using a RUCAM of score 9, indicating turmeric was ‘highly probable’. This patient also was using piperine, and the authors propose the combined use of turmeric and piperine increased the absorption of turmeric and increased the risk for liver injury.
Ayurveda and autoimmune hepatitis
Ayurvedic medicine has also been shown to worsen liver injury in patients with existing liver disease. In their single-center case-control study, Philipset al[111] found that in patients diagnosed with AIH who are treated with Ayurvedic and herbal medicines, defined in this study broadly as complementary alternative medicine, had significantly worse biomarkers and changes on pathology, leading to reduced shortterm survival compared to those who were treated with conventional medicine. Specifically, patients treated with polyherbal Ayurvedic compounds, which comprised the majority of complementary and alternative medicine (CAM) therapy employed, displayed significantly higher Child-Pugh, chronic liver failure, and discriminant function scores. When comparing the two groups at the end of one-, three-, and sixmonth follow-up periods, authors found a significantly higher mortality among CAM patients, with sepsis the most common cause of death in both groups. Authors also identified the contamination of the CAM compounds with heavy metals, antibiotics, chemotherapy agents, nonsteroidal anti-inflammatory drugs, alcohols, antidepressants, anxiolytics, and recreational drugs.
OTHER HERBAL AND DIETARY SUPPLEMENTS
Khat
Khat is an herbal stimulant originating in Ethiopia and used in Eastern Africa, Somalia, and Yemen that can be chewed, ingested, or smoked[112]. The main active components are cathine and cathinone. A number of case reports depicting khatinduced liver injury have been published, but Arguetaet al[113] present the first case of hepatotoxicity due to khat in the United States. The patient was a 28-year-old man from Yemen who presented with hepatotoxicity in the setting of regular recreational khat use until one week prior to presentation. The authors identified abnormal liver enzymes consistent with hepatocellular injury. Cessation of Khat resulted in clinical improvement, indicated a positive de-challenge response which was the basis of causality as RUCAM scoring was not mentioned. Of note, American clinicians are likely unfamiliar with the presentation of Khat, as it is illegal in the United States.
Skullcap
Skullcap comes from the root ofScutellaria baicalensisand is commonly used in TCM. There are previously published case reports of skullcap causing liver injury through the active ingredient wogonin. Skullcap has a designated LiverTox likelihood score of B[114]. Puriet al[115] imply that these case reports may have overstated the hepatotoxic potential of skullcap, as patients were all concurrently at least one other HDS with established association with hepatotoxicity, and conducted their own prospective study to test their hypothesis. They found that skullcap ingestion did not result in significant liver enzyme abnormalities or hepatic dysfunction.
Black cohosh and arborvitae
Black cohosh, fromCimicifuga racemose, is a well-established hepatotoxin with greater than 50 cases reported cases[116]. It is native to North America and is used to treat menopausal symptoms[117]. Recent studies have investigated the effect of adding black cohosh to clomiphene to treat infertility[118,119]. Black cohosh’s main active ingredients are glycoside and terpene. Arborvitae or white cedar, fromThuja occidentalis (T. occidentalis),is a tree native to North America and is used to treat respiratory infections, uterine malignancy, amenorrhea[120]. Unlike black cohosh, arborvitae has not been described in the literature as a hepatotoxin. Arborvitae’s main active ingredient is thujone. Caruntuet al[120] present a case of a 40-year-old female from Bangladesh and living in the United States who concomitantly used black cohosh and arborvitae to increase her fertility. The combination of these herbal supplements was given a RUCAM score of 6, indicating ’probable‘ HILI. Both agents were discontinued at the same time, neither were re-challenged, and the patient showed clinical improvement. Thus, it is impossible to determine if the liver injury was caused entirely by black cohosh or if arborvitae also contributed. As such, clinicians should remain aware of the possibility of hepatotoxicity from arborvitae use.
HERBAL HEPATOCYTOPROTECTION
A significant number of review articles were identified in the past year dealing with the potential protective effects of herbals on the liver. The majority of reports found however, were conducted either usingin vitroorin vivorat models. In order to provide the most relevant information to clinical practice we focused only of those herbals utilized in human studies, in particular, silibinin (milk thistle) and N-acetylcysteine (NAC) to prevent anti-tuberculosis medication liver injury and vitamin E to protect against methotrexate DILI.
Mycobacteriumtuberculosis continues to be the leading cause of infection related mortality amongst adults worldwide[1]. The mainstay of treatment consists of quadruple therapy [isoniazid (INH), pyrazinamide (PZA), ethambutol and rifampin] for two months followed by rifampin and isoniazid for the remaining four months [121]. Despite adjustments in duration of treatment, the hepatotoxic effects of PZA, INH and rifampin limit their use, leading to therapy discontinuation in approximately 11% of patients[122,123]. The mechanism of hepatotoxicity of PZA and INH is thought to stem from oxidative injury and production of toxic metabolites[122]. Rifampin upregulates hepatic microsomal enzymes accelerating INH metabolism, increasing toxic metabolites thus increasing risk of liver injury[123].
Silibinin (milk thistle) is a TCM flavonoid derived from the extract of the plantSilybum marianum[123]. Gohet al[122] investigated silibinin’s hepatoprotective role against INH, PZA and combination regimen within vitroassays as a prophylactic agent (prior to anti-TB treatment), rescue agent (given with anti-TB treatment), and as a salvage agent (given after onset of hepatotoxicity). They found that silibinin was most effective as a rescue agent by way of reducing intracellular levels of oxidative stress and oxidative damage to intracellular targets and mitochondria, leading to decreased apoptotic activity[122]. Silibinin was not effective as a prophylactic or salvage agent. Additionally, it was found that silibinin was more protective against INH alone compared to PZA or combination regimens suggesting that silibinin does not protect against PZA-induced hepatotoxicity[122].
Additionally, Singhet al[2] performed a systematic review of randomized control trials of chemoprophylaxis in the setting of four-drug regimen anti-tuberculosis treatment. They identified four trials utilizing silymarin/silibinin and three trials utilizing NAC[2]. Only one of four trials demonstrated clinically significant cytoprotection. The study in question, however, was shown to have insufficient power and was stopped prematurely for safety concerns[2]. These findings are concordant with the study performed by Gohet al[122] in which silibinin showed protection against INH, but not PZA. NAC however, showed clinically significant cytoprotection in all three studies reviewed. Its hepatoprotective effect is thought to stem from the increase in glutathione, protecting the liver against oxidative stress[2].
Sanjayet al[123] studied gallic acid, an Ayurvedic herbal medicine that is present in various fruits and vegetables in the setting of INH and rifampin DILI in Wistar rat models. Gallic acid was co-administered with INH and rifampin and was compared to both negative control and positive control (silymarin treated) models[123]. Gallic acid demonstrated a hepatic protective effect with co-administration and was comparable to the protective effect of the silymarin treated group[123]. Mechanism of action was attributed to gallic acid’s antioxidant properties by increasing expression and activation of Nrf2[123].
Vitamin E and methotrexate
Methotrexate is one of the main treatments used in rheumatoid arthritis[124]. However, long term use has been associated with the development of fatty liver disease, fibrosis and cirrhosis[125]. As a result, it is often discontinued when aminotransferases reach 3× upper limit of normal (ULN) or remain persistently above 2× ULN[124]. Vitamin E has been studied for its beneficial effects in patients with nonalcoholic fatty liver disease (NAFLD), and a systematic review and meta-analysis performed by Amanullahet al[126] looked at five randomized controlled trials of adult patients with NAFLD treated with vitamin E that demonstrated biochemical and histological improvement.
Vaidyaet al[124] performed a prospective open-label case-control study over a six month span evaluating the hepatoprotective effects of vitamin E in the setting of methotrexate use. Prior animal studies have demonstrated vitamin E hepatoprotection against methotrexate[124]. The groups were randomized such that each consisted of their individualized methotrexate regimen, folate 1mg/daily along with dietary and exercise advice to minimize lifestyle induced fatty liver disease. The treatment group received vitamin E 400 mg twice a day while the control group did not. This study also included a crossover design in which the control group individuals that were shown to have ≥ 1-fold but less than 3-fold rise in aminotransferase levels at the three month follow up visit were then treated with vitamin E. The study found that the vitamin E treated group had a statistically significant reduction in AST/ALT levels compared to controls. Additionally, those individuals who were crossed over to receive vitamin E also demonstrated a statistically significant decrease in AST/ALT. The authors concluded that vitamin E attenuates methotrexate-induced liver injury. A limitation of this study is not knowing if these patients had underlying fatty liver disease prior to methotrexate initiation.
Numerous additional studies were identified that investigated the hepatoprotective effects of many other herbal medications for their antioxidant, anti-inflammatory and anti-apoptotic roles. These studies were largely conducted throughin vitroorin vivorat models as mentioned above. The individual studies that may be of interest to the reader include polyphenols and acetaminophen (APAP)[127,128], Gamisou-san and APAP[129], Lycopene and tamoxifen[130], and licorice and cisplatin[131]. Additional herbal agents were identified as cytoprotective after induction of liver injury by carbon tetrachloride or APAP[132-142].
HILI MISCELLANY
Psoralen and APAP-induced toxicity
Psoralen, an organic compound found in the seeds ofP. corylifolia, is known for its photosynthesizing properties used to treat psoriasis and vitiligo. Unfortunately, it has also been implicated in hepatotoxicity and is one of the key ingredients responsible for liver injury in the popular TCM, buguzhi. Britzaet al[143] conducted anin vitrostudy using a line of liver carcinoma cells and showed that psoralen exacerbates APAP hepatotoxicity. Interestingly, when non-toxic doses of psoralen and APAP were concurrently applied to the cell cultures, they synergistically induced liver injury. These findings have yet to be applied toin vivoanimal models.
Selenium
Selenium is a trace element abundant in brazil nuts and fish and believed to protect against oxidative stress and infection[144]. In a cross-sectional study conducted by Aktaryet al[145], a negative association was observed between selenium intake and the presence of NAFLD in a Canadian cohort. Similar findings suggesting selenium’s hepatoprotective effect was seen in multiple rat models[146,147]. However, in a population-based study in China, Wuet al[148] found a significant association between dietary selenium intake and the presence of NAFLD, consistent with a dose-response relationship. Our understanding of selenium’s effect on the liver therefore remains inconclusive.
Usnic acid
Usnic acid derived from lichens is a well-documented agent of liver injury with first reported cases dating to 2000 in relation to the dietary supplement, LipoKinetix[149}. Approximately 21 cases of LipoKinetix-induced liver toxicity were reported leading to one death and one liver transplant[149]. This dietary supplement has since been removed from the market[150]. Usnic acid’s known mechanism of liver injury is a dose- and time-dependent manner through decoupling oxidative phosphorylation along with inducing oxidative stress through glutathione depletion[149].
Contaminants
Herbal products are not subjected to the same quality control measures as prescription drugs and such can lead to contaminations and subsequent liver injury. Quanet al[4] describes contaminates of herbal products as nonphyto-hepatoxins. These contaminates can be divided into heavy metals, biologic factors, pesticide and herbicidal residue[4]. Of the heavy metal arsenic, mercury, cadmium, nickel and lead are most commonly detected[4]. A study performed by Abualhasanet al[151] analyzed 18 green and herbal tea samples. Seven of 18 samples were detected to contain chromium and lead at concentrations above set limits set by WHO. In this study microbial contamination were also detected in six of these seven metal containing samples[151]. These microbial contaminations have been shown to be hepatotoxic through decreasing antioxidation, increasing lipid peroxidation and upregulating apoptotic genes.[4] Additionally, the use of pesticides and herbicides have been shown to cause hepatotoxicity through hepatic mitochondrial toxicity and obstructive cholestasis[4].
CONCLUSION
HILI continues to be a growing concern for clinicians both in the United States and worldwide. While currently considered a subtype of DILI, differences in composition, application, and outcomes of HDS compared to conventional medications indicate that HILI may deserve to be considered independently.
The lack of HDS regulation in the United States limits our understanding of their potential for hepatotoxicity. Even an accurate estimate of the incidence of HILI is difficult to ascertain, and the frequencies that are reported using registries, single center hospitals, and population-based cohorts, make them difficult to compare.
Moreover, the diagnosis of HILI remains a challenge, and while assessment tools are valuable in determining causality, even the widely applied RUCAM scale - designed to evaluate DILI - falls short of adequately evaluating HILI[31]. Complete data are required for proper utilization of RUCAM, thus highlighting the importance of prospective registries. While imperfect, the RUCAM scale is currently the most widely used tool available, and until a better alternative is developed, we encourage its continued use and refinement to help identify verifiable HILI cases[31]. Development of prospective HILI/DSLI registries in Asia would also improve the overall utility of RUCAM and provide a more reliable and standardized causality scoring system. Future studies in HILI should examine (1) causality assessment scores; (2) clinical significance of using multiple herbal ingredients simultaneously; and (3) prospective studies to better understand incidence in Western countries. By improving assessment tools and expanding the data, advocates may be able to make stronger arguments to regulatory boards in support of consumer protection laws with regard to HDS.
The use of pharmacogenetics has identified susceptibility factors to HILI in the case of GTE and HLA-B *35:01. The search for other associations showing a strong correlation to idiosyncratic HILI is ongoing[41].
The use of herbals in hepato-protection continue to show promising outcomes in preventing and/or attenuating DILI from anti-TB liver injury. Further human clinical trials are still required in order to assess the true therapeutic benefit of cytoprotective herbals in other settings.
Kratom and its legal status will undoubtedly remain a hotly debated topic in coming years, as the opioid epidemic continues. At present, kratom is legal at the federal level, but banned in several states and countries. The literature indicates kratom is potentially lethal, not only through overdose but also by contaminated products, and some degree of regulation certainly seems warranted[63-65,152]. However, it has yet to be determined which end of the spectrum, through a ban or legalization, would best serve consumers.
The highlights of the updated literature over the past year indicate interest in HILI that we expect will continue to increase as the multi-billion-dollar supplement industry in the United States grows.
杂志排行
World Journal of Hepatology的其它文章
- Coronavirus disease 2019 and non-alcoholic fatty liver disease
- Epigenetic mechanisms of liver tumor resistance to immunotherapy
- Advances in the management of cholangiocarcinoma
- Challenges in the discontinuation of chronic hepatitis B antiviral agents
- Liver Kidney Crosstalk: Hepatorenal Syndrome
- Hepatitis C virus treatment failure: Clinical utility for testing resistance-associated substitutions
