Racial disparities in nonalcoholic fatty liver disease clinical trial enrollment: A systematic review and meta-analysis
2021-01-14ParitaPatelCharlesMullerSonaliPaul
Parita Patel, Charles Muller, Sonali Paul
Parita Patel, Charles Muller, Sonali Paul, Section of Gastroenterology, Hepatology, and Nutrition, University of Chicago Medical Center, Chicago, IL 60637, United States
Abstract
Key words: Non-alcoholic fatty liver disease; Non-alcoholic steatohepatitis; Hispanic; Racial disparities; Meta-analysis
INTRODUCTION
Non-alcoholic fatty liver disease (NAFLD) is the most common cause of chronic liver disease in the United States, affecting up to 25% of the global adult population[1]. NAFLD ranges from simple steatosis to non-alcoholic steatohepatitis (NASH), with some patients experiencing eventual cirrhosis. Given the rising incidence, NAFLD is poised to become the leading indication for liver transplantation in the coming years[2].
Risk factors for the development of NAFLD include insulin resistance and metabolic syndrome (encompassing elevated fasting glucose levels, hypertension, dyslipidemia, and central obesity). However, not all individuals with these risk factors develop NAFLD. In a recent systematic review and meta-analysis, heterogeneity in NAFLD burden between racial and ethnic groups was noted, with the highest prevalence seen in Hispanic populations (pooled prevalence 22.9%)[3].
Although it remains unclear why Hispanics are at a higher risk of developing NAFLD and NASH, there is likely an interplay of multifactorial causes. Genetic risk factors play a large role in the pathogenesis of NAFLD. Studies have shown the single nucleotide polymorphisms in patatin-like phospholipase domain-containing protein 3 (PNPLA3), transmembrane 6 superfamily member 2 (TM6SF2), and membrane bound O-acyl transferase (MBOAT) play various roles in different races[4]. For example, the isoleucine to methionine substitution at position 148 (I148M) variant inPNPLA3, has been strongly linked to hepatic fat content. This variant occurs more frequently in Hispanics (49%) compared to non-Hispanic whites (23%) or African Americans (17%)[4]. Additionally, other factors such as culture, environment, and socioeconomic status, play an important role.
Although weight loss through lifestyle interventions or bariatric surgery can reverse the effects of NAFLD, there are currently no Food and Drug Administration (FDA) approved therapies for the treatment of NAFLD. Several promising therapies are currently being investigated in clinical trials. Although the burden of NAFLD on Hispanics is significant, it is unknown if this population is represented in these clinical trials. Identifying possible racial disparities is the first step in improving targeted interventions for patient subgroups. The aim of this study was to evaluate the enrollment of Hispanics in NAFLD trials conducted in the United States and Canada. We hypothesized that the expected rate of Hispanics in NAFLD therapy trials should be proportionate to the burden of disease among Hispanics within the NAFLD population.
MATERIALS AND METHODS
Literature search strategy
The literature search was performed using the PubMed (United States National Institutes of Health, Bethesda, MD, United States) database from January 1, 2005 to March 31, 2019. Three index search terms for nonalcoholic fatty liver disease, nonalcoholic steatohepatitis, and fatty liver were combined. Other potential studies were identified from reference lists of previously published review articles. The search was restricted to English-language articles. Conference abstracts were excluded. Three investigators (Patel P, Muller C and Paul S) reviewed articles for study inclusion. Discrepancies were resolved by consensus.
Study selection and data collection
Published studies of patients with NAFLD or NASH receiving any therapeutic intervention were included. NAFLD and NASH were independently defined by each study, usually either by imaging or histology.
Randomized controlled trials (RCTs) or prospective cohort studies conducted in the United States and Canada with human subjects aged 18 years or older were included. Retrospective studies, case-control, case series, case reports, reviews, and studies with non-human subjects or non-English language were excluded. Three investigators (Patel P, Muller C and Paul S) reviewed articles for study inclusion with discrepancies resolved by consensus. All data were extracted by 1 researcher and verified by another independent researcher and included study author, country, publication date, study design, intervention, sex, age, and race and/or ethnicity. Enrollment demographic information regarding race and ethnicity, when available, was recorded as defined in each individual study. For the purposes of analysis, ethnicity referred to designations of “Hispanic” or “non-Hispanic”, reported along with an independent racial designation for each participant.
An assessment of risk of bias was not performed as we had a heterogenous inclusion criteria, and a risk assessment is not applicable to our study design. Additionally, given the framework of our research question, we have demonstrated that these studies are, in fact, biased towards patient selection.
Data synthesis and analysis
NAFLD prevalence data was obtained using a recent systematic review and metaanalysis that examined racial and ethnic disparities in NAFLD prevalence among adult patients in the United States through August 2, 2016[3]. In this study, the prevalence of NAFLD in the Hispanic population was 22.9% compared to 14.4% in white persons and 13.0% in black persons[3]. Additionally, the prevalence of NASH followed similar trends in this analysis with Hispanics disproportionately affected with a prevalence of 45.4%[3].
Descriptive statistics were performed with frequencies and proportions reported. Two-tailedz-test was performed to compare differences in proportions. All metaanalyses were performed using random effects models and results were pooled using the maximum likelihood estimation. The arcsine transformation was used to estimate the absolute proportion of Hispanics participating in each study. Study heterogeneity was assessed using the CochraneI2statistic. All statistical analyses were performed using OpenMeta software. The statistical methods of this study were reviewed by Dr. Sonali Paul.
Prespecified subgroup analyses explored differences in Hispanic trial participation by specifically a diagnosis of NASH, mode of NAFLD diagnosis, and type of therapeutic intervention. Further subgroup analyses examined the effect of study design (RCT versus prospective cohort) on enrollment by ethnicity.
RESULTS
The search strategy yielded 14406 citations using the relevant search terms, with 38 meeting eligibility criteria (Figure 1). Thirty-two studies (84%) were conducted in the United States, 4 studies (11%) performed in Canada, and 2 studies (5%) were multinational (Table 1). Twenty-six (68%) studies were randomized controlled trials and 12 (32%) were prospective cohort or open label studies. When reported, median age of enrolled subjects was 49 years old (range 41.5-58) with 56% female participants. NAFLD was defined through biopsy findings in 79% (n= 30) of the studies. Of the included articles, treatment modalities ranged from medications (n= 28, 74%), lifestyle interventions (n= 5, 13%), bariatric surgery (n= 4, 11%) and phlebotomy (n= 1, 2%).
Reporting of racial data
Of the 38 identified trials, 25 (66%) included racial data with a total of 2531 total enrolled patients. Twenty-one (84%) trials were conducted in the United States, 2 (8%) trials were performed in Canada and 2 (8%) were multinational trials. The median age of enrolled patients was 49.5 years (range 41-58). NAFLD was diagnosed by biopsy in 80% (n= 20) of the trials, with 20% (n= 5) diagnosed by imaging. Interventions included medications (n= 23, 92%) or bariatric surgery (n= 2, 8%) (Table 1).
Enrollment of Hispanic patients
Among the 38 eligible trials, only 17 (44.7%) included information regarding patient ethnicity. Of the 2983 patients enrolled in all eligible trials, a total of only 346 (11.6%) Hispanic participants was reported. Among the 25 studies that included data on race, 14 included data on Hispanic participation. Of note, 3 studies that did not have racial data did provide data on Hispanic participation (Table 1).
Among the 17 trials that reported Hispanic participation, there were 346 Hispanic patients out of 1577 total enrolled patients with a participation rate of 21.9% compared to 74.8% of Caucasian participants among those including data on Caucasian participation. The 21.9% unadjusted pooled prevalence of Hispanic trial participants was similar to the 22.8% unadjusted pooled NAFLD Hispanic prevalence (990/4332 total patients) in the recent systematic review by Richet al[3](P= 0.365).
A meta-analysis was then performed to estimate pooled prevalence while taking heterogeneity of included studies into consideration. The pooled prevalence was found to be 24.3% [95% confidence interval (CI) 16.6-32.0] with significant heterogeneity(I2= 94.6%) (Figure 2).
Further sub-group meta-analyses were performed in patients with biopsy proven NASH and found Hispanic participation to be 24.7% (Table 2), considerably lower than the 45.4% prevalence of NASH in the Hispanic population found in the recent meta-analysis by Richet al[3]. Caucasian and African American participation in studies using NASH as inclusion criteria, was slightly lower than those of NAFLD studies (67.3%vs63.9% and 8.0%vs2.7%, respectively).
To determine if rates of Hispanic enrollment changed over time, studies conducted before and after 2015 were compared. The pooled prevalence of Hispanic patients in studies from 2005-2014 was 15%, compared to 37% for studies from 2015-2019. Trends in Hispanic study participation over time are displayed in Figure 3.
DISCUSSION
The purpose of this systematic review and meta-analysis was to characterize the participation rate of Hispanic patients in clinical trials investigating therapies for NAFLD. Despite the importance of genetics and race in the prevalence of NAFLD, our results show that racial/ethnic demographic data are under-reported, with only 25 of 38 (66%) eligible clinical trials reporting race or ethnicity. Both the FDA and the National Institutes of Health (NIH) have published recommendations on how to report race and ethnicity data in clinical trials, however in practice these guidelines are not strictly followed or enforced[5,6]. Previous studies have demonstrated reporting of race/ethnicity to be similarly suboptimal in clinical trials across several specialties[7,8]. An analysis of clinical trial enrollment for several disease processes (spanning general medicine, oncology, cardiovascular disease, and infectious diseases) from 2009, found that 21% of studies failed to include racial or ethnic demographic data[7].
In this review, of the 38 trials that met eligibility criteria, 25 reported racial information. Among these only 17 (68%) provided data on ethnicity (participation of Hispanic patients). It is well established that the prevalence of NAFLD and NASH ishigher among Hispanic patients than among either non-Hispanic whites or other minority groups, with a prevalence of 25%-40% and 25%, respectively[9-13]. AlthoughHispanic participation among trials that included information about Hispanic enrollment (24.3%) was close to that of the United States Hispanic population (18.3%)[14], it does not provide an accurate reflection of the racial and ethnic makeup of the NAFLD population, which is closer to 30% Hispanic[12]. There was also significant discrepancy between Hispanic participation in NASH trials (24.7%) and the prevalence of NASH (45.5%) reported in a recent meta-analysis[3]. It is not known whether the low rate of Hispanic participation in these trials is due to lack of collection of ethnic demographic data on behalf of the investigators, failure to report ethnicity by subjects, or true under-enrollment.

Table 1 Summary of non-alcoholic fatty liver disease studies included in systematic review

1Race and ethnicity categorized together.2Definition of NAFLD included presence of steatosis on MRI.3Patients were categorized as either white or non-white.4Patients were categorized as either Hispanic or non-Hispanic. NAFLD: Non-alcoholic fatty liver disease; NASH: Non-alcoholic steatohepatitis; Y: Yes; N: No; MRI: Magnetic resonance imaging; NR: Not recorded; RCT: Randomized control trial.

Table 2 Proportion of trial patients with non-alcoholic steatohepatitis by race/ethnicity
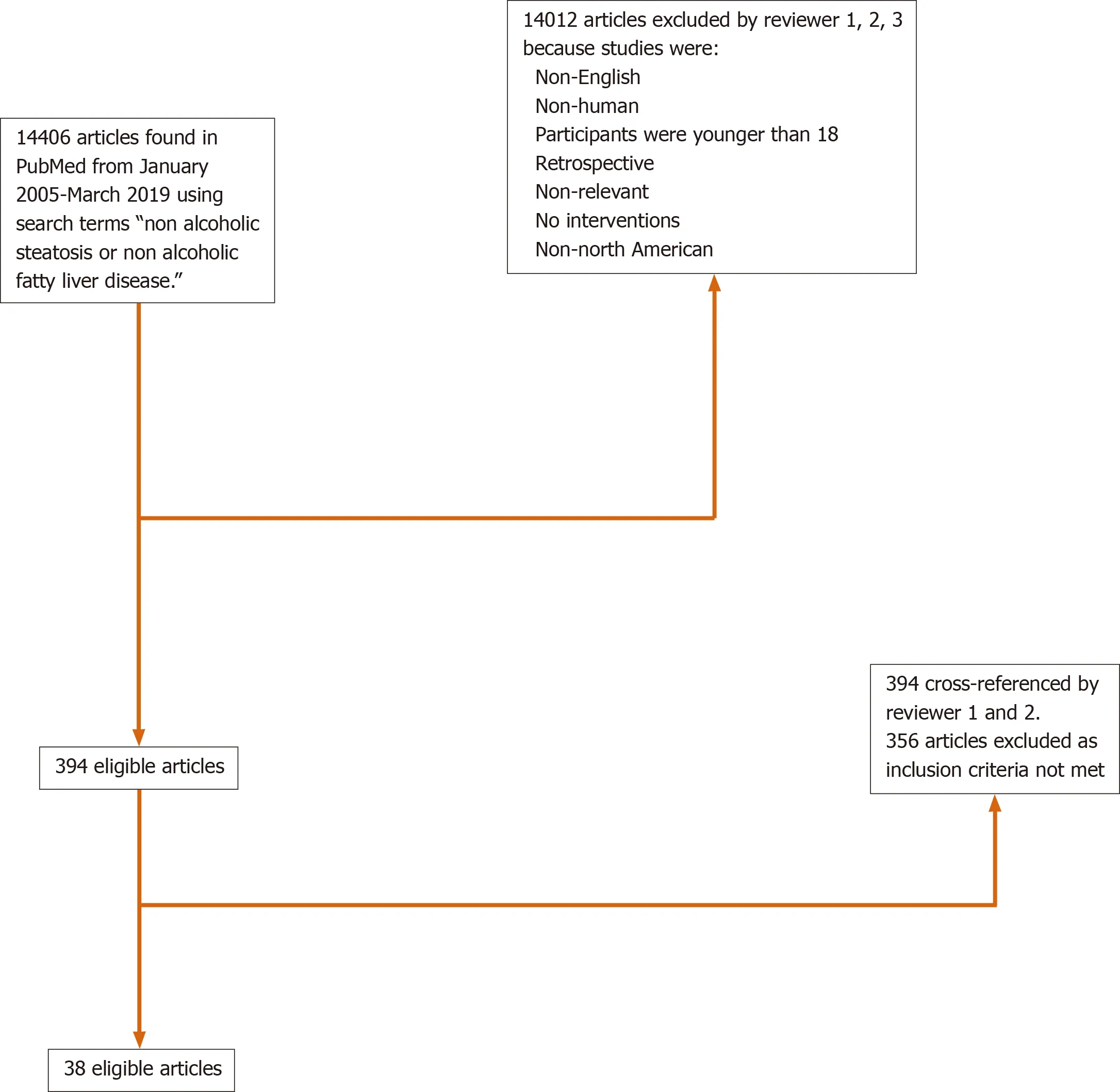
Figure 1 Summary of review process of all PubMed articles using search terms.
Under-reporting of Hispanic trial participants could be in part due to heterogeneity in self-reported ethnicity among Hispanic patients and diversity of their country of origin and race[15]. Health sciences typically follow the United States Census practice of categorizing “Hispanic” as an ethnicity that is distinct from race. This practice can result in discordance between patients’ self-perceived race/ethnicity and their associated categorization in health systems in addition simplification of a diverse, heterogenous group, and inaccuracy in reporting[15,16].

Figure 2 Pooled prevalence of Hispanic patients among studies reporting ethnicity.
Heterogeneity of enrollment practices for the included trials is also likely contributing significantly to Hispanic under-enrollment. Hispanic enrollment (among those reporting any Hispanic participants) ranged from 4%-67%. TheI2statistic of 94% highlights the significant heterogeneity of Hispanic enrollment among studies included in this meta-analysis. The finding that 10 of the 17 trials (59%) including data on Hispanic enrollment were conducted in states that shared a border with Mexico[17-26]highlights the opportunistic, rather than systematic, nature of trial recruitment and enrollment. The generalizability of such clinical trials is significantly compromised when they fail to include information about key demographics.
When comparing racial and ethnic enrollment between studies using NAFLD and NASH as inclusion criteria, we found that Hispanic enrollment in NASH trials increased relative to enrollment of Caucasian and African American participants. These findings are consistent with those of prior studies demonstrating that Hispanic NAFLD patients are more likely to progress to steatohepatitis than Caucasians or African Americans[3,27]. However, given that 45% of Hispanic NAFLD patients experience progression to steatohepatitis, compared to 32% and 20% of Caucasian and African American NAFLD patients, respectively[3], Hispanics are likely even more under-represented in studies of NASH relative to the disease burden in that population. The observed proportion of African American NASH participants in our study was particularly low (2.7%), but is likely a reflection of the low rate of African American enrollment in included studies in general.
Acknowledging the importance of diverse trial participation to generalizability of findings in the development of new therapies, the NIH has stipulated that all sponsored clinical trials include women and minority patients since 1993[7]. Aside from ethical issues related to equity and justice, racial differences in response to pharmacologic therapy identified in diverse trials have been used to guide current clinical practice[28,29]. Recent work suggesting differences in the underlying genetic contributions to NAFLD in patients of different racial/ethnic backgrounds[30,31]highlight the importance of diversity in clinical trial participation. Genetic variants onPNPLA3, TM6SF2, and neurcan (NCAN)can increase the heritability of NAFLD by up to 27% within families. A missense mutation ofPNPLA3has a strong association with hepatic fat accumulation and with a higher susceptibility to develop more severe histologic liver damage, irrespective of the degree of obesity or presence of diabetes[32-34]. This variant inPNPLA3gene has been observed in highest frequency in Hispanics[4].

Figure 3 Percentage of Hispanic enrollment in trials.
Despite the benefits of diversity in trial enrollment, minority patients have historically been underrepresented in clinical trials. Barriers to minority participation in clinical trials include mistrust of providers/research, reduced access to healthcare, financial and time constraints, lack of education about clinical trials, and cultural or language differences impairing communication with trial recruiters or providers[35]. Hispanic patients, in particular, have been underrepresented in clinical trials for multiple conditions. In a 2004 analysis of colorectal, lung and prostate cancer studies, Murthyet al[36]found that Hispanic patients only constituted 3.1% trial participants. Compared to African American patients, Hispanic patients are less likely to be aware of or recruited to participate in clinical research[37-39]. Although lack of access to healthcare resources and lower socioeconomic status are shared among multiple minority groups, language barriers create a burden for Hispanic patients in particular. Interventions such as provision of Spanish-speaking recruitment materials or personnel have been shown to improve enrollment of Hispanic patients in clinical trials[40,41]and serve as potential targets for increasing diversity of study populations for NAFLD. In spite of these historic barriers to Hispanic participation in clinical trials, a trend toward increasing Hispanic enrollment over time was observed in our study, with Hispanic enrollment in studies conducted after 2015 nearly triple that of studies from 2005-2014. While these results are encouraging, future efforts are needed to standardize reporting of race/ethnicity in clinical trials and encourage diverse, representative enrollment.
A major limitation of this study is the low rate of reporting demographic data on Hispanic participation among the trials analyzed. Although many trials did not include any racial/ethnic demographic data, the rate of inclusion of data on Hispanic participation was particularly poor (44% of eligible trials). From the information available, it is not known if these trials did not actually recruit any Hispanic participants or if they simply failed to collect or report data on their inclusion.
In conclusion, North American clinical trials of NAFLD from 2015-2019 did not consistently include data on Hispanic participation. Among trials that did include racial/ethnic demographic data, Hispanic patients may be underrepresented relative to the burden of NAFLD and NASH among this population. Future efforts aimed at improving or standardizing reporting of race in clinical trials and at increasing enrollment of diverse and representative study populations are needed to address this disparity.
ARTICLE HIGHLIGHTS
Research background
Non-alcoholic fatty liver disease (NAFLD) is the most common cause of chronic liver disease in the United States and has a heterogeneous distribution across racial and ethnic groups, with a disproportionate burden among Hispanics. Although it remains unclear why Hispanics are at a higher risk of developing NAFLD and nonalcoholic steatohepatitis (NASH), there is likely an interplay of multifactorial causes including genetics, culture, socioeconomic status and environment. Despite this high burden of disease, there are currently no approved therapies for the treatment of NAFLD.Several promising therapies are currently being investigated in clinical trials but it is unknown if Hispanics are appropriately represented in these clinical trials.
Research motivation
Identifying possible racial disparities is the first step in improving targeted interventions for patient subgroups. The purpose of this systematic review and metaanalysis was to characterize the participation rate of different races and ethnicities in clinical trials investigating therapies for NAFLD.
Research objectives
The aim of this study was to evaluate the enrollment of Hispanics in NAFLD trials conducted in the United States and Canada. We hypothesized that the expected rate of Hispanics in NAFLD therapy trials should be proportionate to the burden of disease among Hispanics within the NAFLD population.
Research methods
The literature search was performed using the PubMed (US National Institutes of Health, Bethesda, MD, United States) database from January 1, 2005 to March 31, 2019 using the following search terms: Nonalcoholic fatty liver disease, nonalcoholic steatohepatitis, and fatty liver. Randomized controlled trials (RCTs) or prospective cohort studies conducted in the United States and Canada with human subjects aged 18 years or older were included. Descriptive statistics were performed with frequencies and proportions reported. Two-tailed z-test was performed to compare differences in proportions. All meta-analyses were performed using random effects models and results were pooled using the maximum likelihood estimation.
Research results
Of the 38 trials that met eligibility criteria, twenty-five reported racial information. Among these only 17 (68%) provided data on ethnicity (participation of Hispanic patients). Among the 2983 patients enrolled in all eligible trials, a total of only 346 (11.6%) Hispanic participants was reported. Among the 17 trials that reported Hispanic participation, there were 346 Hispanic patients out of 1577 total enrolled patients with a participation rate of 21.9% compared to 74.8% of Caucasian participants among those including data on Caucasian participation. A meta-analysis was then performed to estimate pooled prevalence while taking heterogeneity of included studies into consideration. The pooled prevalence was found to be 24.3% (95%CI: 16.6-32.0) with significant heterogeneity(I2= 94.6%). To determine if rates of Hispanic enrollment changed over time, studies conducted before and after 2015 were compared. The pooled prevalence of Hispanic patients in studies from 2005-2014 was 15%, compared to 37% for studies from 2015-2019.
Research conclusions
North American clinical trials of NAFLD from 2015-2019 did not consistently include data on Hispanic participation. Among trials that did include racial/ethnic demographic data, Hispanic patients may be underrepresented relative to the burden of NAFLD and NASH among this population.
Research perspectives
Future efforts aimed at impr oving or standardizing reporting of race in clinical trials and at increasing enrollment of diverse and representative study populations are needed to address this disparity. It is not known whether the low rate of Hispanic participation in these trials is due to lack of collection of ethnic demographic data on behalf of the investigators, failure to report ethnicity by subjects, or true underenrollment. Despite the benefits of diversity in trial enrollment, minority patients have historically been underrepresented in clinical trials. Barriers to minority participation in clinical trials include mistrust of providers/research, reduced access to healthcare, financial and time constraints, lack of education about clinical trials, and cultural or language differences impairing communication with trial recruiters or providers. Interventions such as provision of Spanish-speaking recruitment materials or personnel have been shown to improve enrollment of Hispanic patients in clinical trials and serve as potential targets for increasing diversity of study populations for NAFLD.
杂志排行
World Journal of Hepatology的其它文章
- Mechanisms and consequences of COVID-19 associated liver injury:What can we affirm?
- Review: Pathogenesis of cholestatic liver diseases
- Lipidomics in non-alcoholic fatty liver disease
- Update on diagnosis and management of sepsis in cirrhosis: Current advances
- Cell competition in liver carcinogenesis
- Management of hepatitis C in children and adolescents during COVID-19 pandemic
