Successful treatment of relapsed acute promyelocytic leukemia with arsenic trioxide in a hemodialysis-dependent patient: A case report
2020-04-07HeeJeongLeeSangGonPark
Hee Jeong Lee, Sang-Gon Park
Hee Jeong Lee, Department of Hemato-Oncology, Chosun University Hospital, Gwang-ju 501-717, South Korea
Sang-Gon Park, Department of Internal Medicine, Hemato-oncology, Chosun University Hospital, Gwangju 61453, South Korea
Abstract BACKGROUND Arsenic trioxide (ATO) is recommended for patients who do not achieve molecular remission or who have molecular or morphologic relapse. However,there are no guidelines for adjusting ATO dosage in patients with severe renal failure or on dialysis. Herein, we report the successful treatment of relapsed acute promyelocytic leukemia (APL) in a patient on hemodialysis with ATO single agent and review the cases in literature.CASE SUMMARY A 46-year-old woman who has been on hemodialysis to chronic glomerulonephritis for 15 years visited our hospital for pancytopenia. She had been seen for pancytopenia 3 years ago and had been diagnosed with APL. She also received chemotherapy for APL but unfortunately was lost to follow-up after her second consolidation chemotherapy. She was noted to have pancytopenia by her nephrologist during hemodialysis 1 mo ago. Bone marrow biopsy and reverse transcriptase-polymerase chain reaction (RT-PCR) tests revealed a diagnosis of relapsed APL. Treatment for relapsed APL with ATO single agent was started and she achieved molecular remission after administering 24 doses of ATO. Thus far,four consolidation therapies have been performed with the ATO single agent,and, to date, the molecular remission has been maintained as negative promyelocytic leukemia/retinoic acid receptor-α fusion gene as confirmed by RTPCR testing for two years.CONCLUSION This is a rare case of relapsed APL successfully treated with the single agent ATO in a patient on hemodialysis.
Key Words: Arsenic trioxide; Acute promyelocytic leukemia; Pancytopenia; Hemodialysis;Promyelocytic leukemia/retinoic acid receptor-α fusion gene; Case report
INTRODUCTION
Acute promyelocytic leukemia (APL) is a subtype of acute myelogenous leukemia(AML) and accounts for 10%-15% of all AML diagnoses[1]. APL is cytogenetically distinguished by thet(15:17) chromosomal translocation. The translocation of the promyelocytic leukemia (PML) gene on chromosome 15 to the retinoic acid receptor-α(RAR-α) gene on chromosome 17 produces a PML-RARα fusion gene. APL is distinguished from other types of AML by its unique morphology and clinical presentation. APL is more aggressive than other subtypes of AML because of high early death rates due to fatal coagulopathy but is curable[1-4].Approximately 90%-95%of patients who receive induction chemotherapy achieve complete remission, and more than 85%-90% of patients can be cured[5].
Arsenic trioxide (ATO) is recommended for patients who do not achieve molecular remission after consolidation treatment or who subsequently demonstrate molecular or morphologic relapse. As a single agent, ATO produces complete remission rates of 80% to 90% in patients with hematologic relapse and helps achieve molecular remissions in 70% to 80% of those patients[6-9]. Generally, ATO is well tolerated, but patients with severe renal impairment or on hemodialysis have been difficult in use for safety because of elimination to urine. The standard therapy for APL in patients on dialysis or chronic renal failure has not been established. Only a few cases have been reported regarding ATO use in patients with APL on dialysis[10-16].
Herein, we describe a case of relapsed APL successfully treated with ATO as a single agent in a patient on hemodialysis and review the literature on previously reported cases.
CASE PRESENTATION
Chief complaints
A 46 years old woman patient admitted for worsening pancytopenia for 1 mo.
History of present illness
The patient had her regular hemodialysis scheduled at the nephrology department and we were consulted by the nephrology department for pancytopenia. Bone marrow biopsy and reverse transcriptase-polymerase chain reaction (RT-PCR) tests were performed. Bone marrow examination showed hypercellular marrow with 54%promyelocytes. Cytogenetics revealed FISH fort(15:17) in 10 of 20 cells, while RT-PCR revealed the PML/RARα fusion gene. She received induction chemotherapy with alltrans retinoic acid (ATRA) and idarubicin. After induction chemotherapy, follow-up bone marrow biopsy was performed; however, morphologic remission for promyelocyte was observed, and molecular remission was not achieved. Therefore, the first consolidation chemotherapy with idarubicin was provided with sustained ATRA administration,and molecular remission was confirmed a month later.Unfortunately,after the second consolidation chemotherapy, she was lost to follow-up. She maintained only her regular hemodialysis schedule at the nephrology department where pancytopenia was reconfirmed.
History of past illness
The patient has been on regular hemodialysis for 15 years due to chronic glomerulonephritis.
Personal and family history
She has been gotten only erythropoietin treatment for mild anemia.
Physical examination
No signs of bleeding such as mucosal bleeding or petechia, commonly observed in APL patients, were noted. Her vital signs were stable.
Laboratory examinations
The complete blood count results was normal in parentheses, as shown below: White blood cells (WBCs): 1080 × 103/μL (4.0-10.0 103/μL); hemoglobin: 8.9 g/dL (12-16 g/dL); platelets: 69 × 103/μL (150-400 103/μL); absolute neutrophil count (ANC): 420 ×103/μL (1.5-7.5 103/μL). Blood biochemistry results were as follows: Total bilirubin:0.55 mg/dL (0.2-1.1 mg/dL); aspartate aminotransferase: 23 U/L (5-40 U/L); alanine aminotransferase: 17 U/L (5-40 U/L); albumi: 4.54 g/dL (3.5-5.2 g/dL); blood urea nitrogen: 86.6 mg/dL (8-20 mg/dL); creatinine: 11.6 mg/dL (0.5-1.3 mg/dL); Creactive protein (CRP): 0.13 mg/dL (0-0.5 mg/dL). The results of the coagulation profile were as follows: Prothrombin time (PT): 11.3 s (9.4-12.5 s); activated partial thromboplastin time: 31.9 s (28.0-44.0 s); fibrinogen: 189 mg/dL (200-400 mg/dL); Ddimer: 9537 (0-255 ng/mL).
Further diagnostic work-up
Bone marrow examination and RT-PCR tests were performed for the evaluation of pancytopenia. Bone marrow aspiration demonstrated relapsed APL with promyelocytes comprising 40% of differential leukocyte counts, as well as thet(15:17)(q22; q12) translocation and RML/RARα fusion transcript.
FINAL DIAGNOSIS
She was diagnosed with relapsed APL based on bone marrow examination and RTPCR results.
TREATMENT
We treated her with ATO as a single agent because ATRA was not covered by insurance for the treatment of relapse in South Korea. As she was on hemodialysis, she received a decreased dose of ATO of 0.1 mg/kg/d after hemodialysis three times a wk. During the first wk of ATO initiation, complete blood counts, sodium, potassium,chloride, blood urea nitrogen, creatinine, glucose, PT, activated partial thromboplastin time, fibrinogen, calcium, magnesium, and electrocardiogram were monitored daily.Electrolytes were stable in the normal range, and QTc duration was normal. As no side effects were observed, from the second wk of treatment, the dose was increased to the recommended dose of 0.15 mg/kg/d after dialysis, and a total of 24 doses were administrated throughout 8 wk. RT-PCR performed after the administration of 16 doses was positive for the PML/RARα fusion gene but became negative after 19 doses.Four wks later, consolidation chemotherapy was started with ATO 0.15 mg/kg/d after hemodialysis three times a wk as well, and 12 doses were administrated for 4 wk. The consolidation chemotherapy was performed four times at 2 mo intervals. We performed RT-PCR after each consolidation chemotherapy and confirmed that the patient had a negative result for the PML/RARα fusion gene.
OUTCOME AND FOLLOW-UP
After induction chemotherapy with ATO, molecular remission was achieved and has been maintained for 2 years without recurrence.
DISCUSSION
ATO is the basis of APL treatment. In the past, ATO was used mainly for treating relapsed APL, but recent NCCN guidelines recommend that ATO can be used in the frontline setting[17-19]. However, there is a limit to using ATO treatment in patients with severe renal impairment or on hemodialysis because ATO is excreted by the kidneys[20].Indeed, there are no guidelines for the use of ATO in such patients. Thus far, six case reports and one pilot study involving four patients with renal failure have been published (Table 1)[10-16].
In patients with normal renal function, ATO has demonstrable efficacy during the induction therapy when administered at a fixed daily dose (10 mg or 15 mg daily) and a weight-based dose (0.15-0.30 mg/kg/d)[17,18,21]. When arsenic plasma concentrations range from 0.1 to 0.5 µmol/L (7.4-37.6 ng/mL), PML/RARα proteins are degraded and leukemic promyelocyte differentiation is promoted[22]. When arsenic plasma concentrations range from 0.5 to 2 µmol/L (37.6-150 ng/mL), leukemic cells undergo apoptosis[22]. When ATO is administered at 10 mg daily to APL patients with normal renal function, the arsenic plasma concentration reaches a steady-state of approximately 1 to 1.5 µmol/L after a wk and remains stable until the completion of 28 d of treatment[16,23]. On the other hand, hemodialysis reduces plasma arsenic concentrations by approximately 38% to 40%, but plasma arsenic levels gradually increase because of redistribution between the blood and other tissues with continuous administration of ATO[12,13]. Most of the cases reported were treated by checking arsenic levels. However, many hospitals cannot measure arsenic levels. Further, the reason for measuring the arsenic level is to predict the toxicity of ATO. Moreover,there is no correlation between arsenic levels and QTc interval change or AV block degree[13]. Ultimately, direct measurement of arsenic concentration may help to prevent toxicity, but if it cannot be measured, the dosage can be determined by monitoring ATO side effects. We also started to treat our patient with a lower dose than the recommended dose for a single dose. As no special side effects were observed in the first wk of treatment, the dose was increased to the recommended dose from the second wk.
Generally, treatment ofde novoAPL consists of induction chemotherapy,consolidation chemotherapy, and maintenance therapy; however, in relapsed APL treatment, induction chemotherapy and six consolidation chemotherapy are recommended[19].There is no consensus on the duration of ATO treatment for patients with severe renal failure or on dialysis. Even in the patients mentioned above, the ATO dose schedule was different.
Although there is not much evidence, we reduced the initial dose of ATO and started treatment immediately after hemodialysis three times a week. No serious side effects were observed during the weekly administration; hence, we continued to monitor toxicity and increased ATO up to the usual recommended dose. Although data suggest that the duration of ATO administration was insufficient, our patient somewhat tolerated the treatment and was started on consolidation therapy after a rest period of four weeks following induction chemotherapy. Consolidation treatment was performed for 4 wk every 8 wk. Our patient received consolidation therapy four times and has remained relapse free for two years to date. As our treatment strategy was based on data from other case reports, our case of successful treatment without serious side effects can, in turn, be of great help to clinicians.
CONCLUSION
The use of ATO has not been well studied in dialysis patients, and there is a paucity of data in the literature to support its use in such a situation. Hence, this is a valuable report because it is a case of successful treatment in which complete remission is achieved by administering ATO to a patient with relapsed APL on hemodialysis.
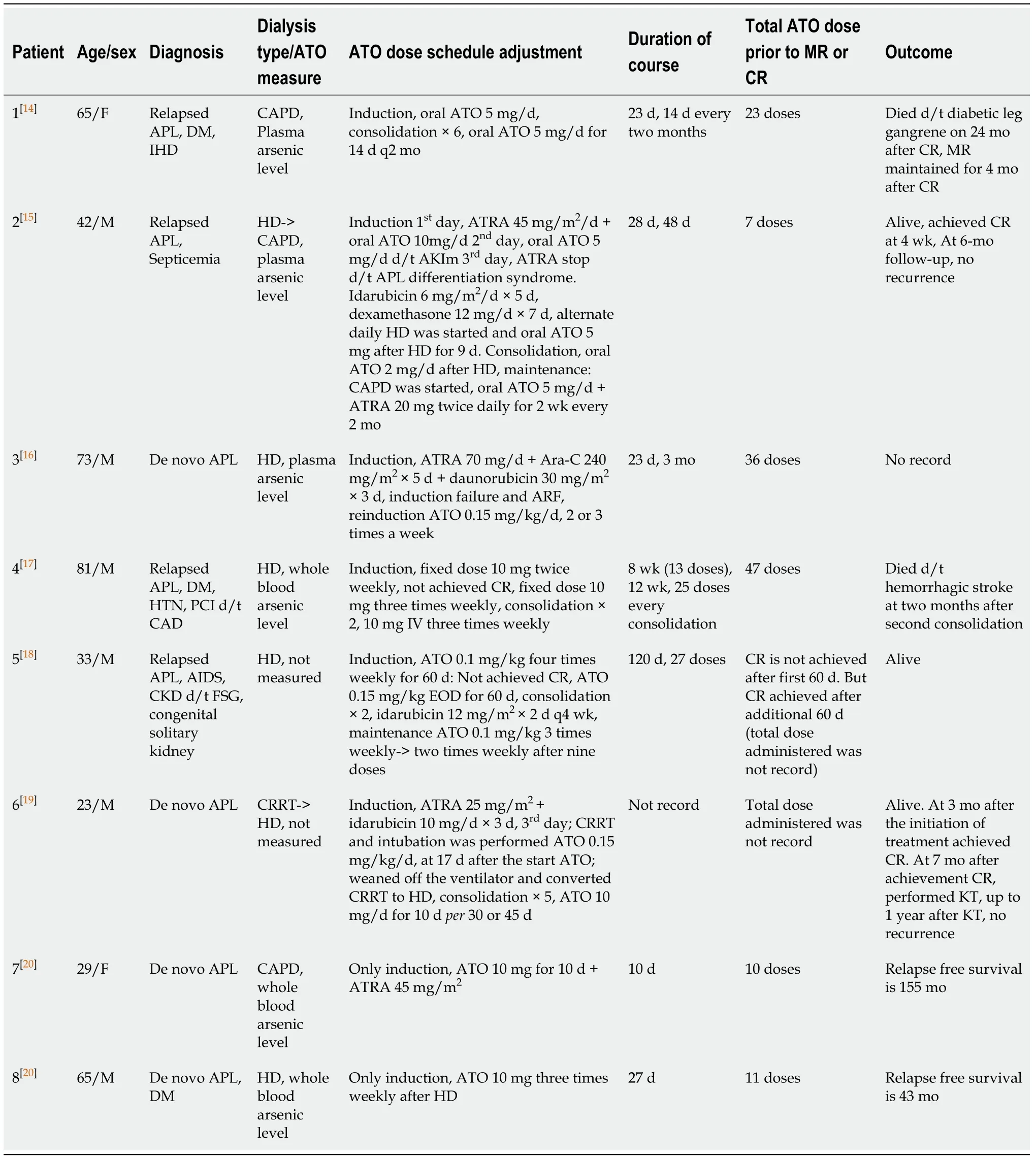
Table 1 Arsenic trioxide treatment in acute promyelocytic leukemia patients with severe renal impairment or on hemodialysis
杂志排行
World Journal of Clinical Cases的其它文章
- Strategies and challenges in the treatment of chronic venous leg ulcers
- Peripheral nerve tumors of the hand: Clinical features, diagnosis,and treatment
- Treatment strategies for gastric cancer during the COVID-19 pandemic
- Oncological impact of different distal ureter managements during radical nephroureterectomy for primary upper urinary tract urothelial carcinoma
- Clinical characteristics and survival of patients with normal-sized ovarian carcinoma syndrome: Retrospective analysis of a single institution 10-year experiment
- Assessment of load-sharing thoracolumbar injury: A modified scoring system
