Nitric oxide detection methods in vitro and in vivo
2020-01-07EktaGoshiGaoxinZhouQianjunHe
Ekta Goshi,Gaoxin Zhou,Qianjun He,
1 Guangdong Provincial Key Laboratory of Biomedical Measurements and Ultrasound Imaging,National-Regional Key Technology Engineering Laboratory for Medical Ultrasound,School of Biomedical Engineering,Health Science Center,Shenzhen University,Shenzhen,Guangdong,China
2 Center of Hydrogen Science,Shanghai Jiao Tong University,Shanghai,China
Abstract
Key words:nitric oxide;detection method;nitric oxide synthase;nitric oxide therapy;colorimetric;chemiluminescence;fluorescence;electrochemical;gas chromatography;magnetic resonance imaging
I NTRODUCTION
Since its discovery by Joseph Priestly in 1722 as a kind of colorless gas,nitric oxide (NO) was majorly considered as an environmental pollutant.Until 1987,NO was recognized as an important molecule that can regulate endothelial functions in the body.Ignarro et al.1proved that endothelial derived relaxing factor was NO,thus giving a boost for in depth research on NO.This discovery paved way for later recognization of NO synthesis pathways and its various biological functions.In 1988,L-arginine was found to be the precursor for generation of NO which could be converted to L-citrulline by NO synthases (NOSs)in vivo.Three isoforms of NOSs were named according to their functions and the type of tissues in which they were firstly found.The neuronal isoform was named as neuronal NOS (nNOS,NOS1) found in neuronal cells,inducible NOS (iNOS,NOS2) was found in cells responsible for inflammation,such as macrophages and microglias,while endothelial NOS (eNOS,NOS3) was found in endothelial cells (Figure1).2The importance of NO was established over years of research,where NO was involved in the variety of pathological and physiological functions such as endothelial vasorelaxation,cardiovascular functions,antimicrobial action,wound healing,tissue repair,neurotransmission,immune functions,blood pressure regulation,cytotoxicity,relaxation of the human penile corpus cavernosum,etc.1,3,4

Figure1:General functions of three isoforms of nitric oxide synthases.
NO is an unstable,highly lipophilic,free radical molecule made up of an unpaired electron.3It has poor solubility(1.9 mM) in water but relatively higher solubility in lipid membranes and in nonpolar solvents.4-6The diffusion rate of NO is found to be 50 μm/s in a single direction in biological systems.7The half-life of NO in biological tissues is observed to be only 3-5 seconds,as opposed to 500 seconds in pure aqueous solutions.5Due to the radical nature of NO,it is readily oxidized into nitrite (NO2-) or even nitrate (NO3-).5It is dif ficult to directly measure NOin vivo,and it is frequent to indirectly measure its oxidative products such as NO2-and NO3
Decrease and defective release of NO can lead to atherosclerosis generation,coronary vasopasm and restenosis after angiopathy.On the other hand,the increase in NO synthesis can cause hypotension in patients suffering from liver cirrhosis and failure,and also cause hemorrhagic and anaphylactic shock.Endogenous release of NO at basal levels maintains the low resting stage of pulmonary vasculature.3The use of NO prodrugs and nanomaterials has proved useful to develop promising NO-based therapy.NO gas at a higher concentration range (> nM) is known to exhibit anti-Warburg effect inhibiting tumor occurrence and development.On the other hand,concentration greater than 1 mM can result in NO poisoning,whereas concentration in the range of pM to nM in the tumor cells can promote tumor cell growth.9,10The use of nanomaterials have helped to overcome the limitations of NO having short half-life,instability during storage and possibility of toxicity.Many types of nanomaterials such as polymers,dendrimers,hydrogels,liposomes,gold and silica nanoparticles,certain quantum dots with NO donors have been exploited in the past under various conditions to release NO.Controlled release of NO from intelligent nanoparticles makes it an attractive tool for cancer therapy due to its tumoricidal effects.11The vital roles performed by NO in regulating a variety of functions make it necessary for its rapid and accurate determinationin vitroandin vivo.Over the past few years,various detection methods including colorimetry,chemiluminescence,fluorescence,electrochemical sensing,gas chromatography,electron spin resonance (ESR)spectroscopy and magnetic resonance imaging (MRI) have been developed to help to understand the pathology and to treat various diseases.The strategy involves searching each of these methods with the proper keywords and listing the various ways employed by researchers to detect and measure NOin vitroandin vivo.This review summarizes these advanced methods and discusses their advantages and shortcomings as following.
COLORIMETRIC METHOD
Colorimetric method for NO detection utilizes the quantification of color change caused by reaction between indicator and NO.NO concentrationin vitrocan be measured directly by the nitrosation of hemoglobin and myoglobin (Figure2).12This method depends on the change of multiple absorption bands and does not need standard curve,exhibiting high sensitivity and accuracy.Besides,Griess assay is one of most general colorimetric methods forin vitroNO detection.Due to its relative simplicity,7Griess assay has been extensively used since its discovery in 1879 for analysis of biological samples like saliva,urine,serum,cerebrospinal fluid and culture media.13Griess assay provides an indirect approach for NO detection by measuring nitrite,nitrate and nitrosating agents as an index for NO.7The two-step diazotization reaction is illustrated in Figure3.First,dinitrogen trioxide (N2O3) which is obtained from acidified nitrite (or from NO autoxidation) reacts with sulfanilamide to form diazonium derivative diazobenzenesulfonic acid.The derivative then interacts with N-(1-napthyl)ethylenediamine to produce purple colored diazo product azo-α-aminonaphthalene-parabenzene-sulfonic acid which exhibits a strong absorbance at 540 nm.5,7,13

Figure2:Hemoglobin/myoglobin methods for detection of nitric oxide (NO) concentration.

Figure3:Chemical reaction process involved in Griess assay.
A few variants of Griess assay have been extensively employed for NO measurement.For example,in conjunction with nitrate reductase,Griess assay has proved to be an accurate,sensitive and inexpensive tool to measure nitrite and nitrate in biological fluids,tissue and serum samples.7,14In another variant,Griess assay is coupled with microtitre plate assay to determine the kinetics of NOS enzymatic reaction in crude or purified enzyme solutions.The oxidation of ferroheme by NO causes the increase in absorbance and hence can be used to measure the generation of NO.15Flow injection method was also used to determine NOin vivowhen NOS activity affected the acute renal failure in rats by detecting NOx as the final metabolite of NO with a high sensitivity of above 0.5 µM.16Griess assay can be employed to detect NO2-and NO3-in the extracellular fluid as an index for NO by using Aspergillus nitrate reductase (NADPH,nitrate oxidoreductase),as it can reduce very small amounts of NO3-.17It can also be used in human plasma cells to detect nitrite and nitrate in the plasma of healthy volunteers with an automated analyzer.18-20Macrophage in Drosophila melanogaster can express NOS and generate NO as an important signaling molecule during its immune response,where Griess assay was used by Ajjuri et al.21to measure nitrites as an index of NO in the brain tissue of the organism in response to neuroinflammation.Lipopolysaccharide (LPS)-stimulated murine RAW264.7 macrophages produced NO which once readily converted to nitrite could be detected by Griess assay.22The production of NO as an antiinflammatory response from LPS-induced macrophages could be measured inMagnolia sieboldiiextract,23Garcinia xanthochymusextracts,24Sambucus australis,25Ovis canadensisandOvis aries,26peat moss extracts27viaGreiss assay.In addition,Griess assay can also be used to determine NO concentration in plants like cucumber and tomato.28
Colorimetric method provides measurement of NO with good sensitivity and high accuracy with inexpensive tools and is economically feasible.7However,NO measurement in whole blood with colorimetric method seems to be not feasible because of the disturbance of nitrogen oxide species.29,30
CHEMILUMINESCENCE METHOD
The chemiluminescence method for detecting NO is considered to be a useful technique.The principle of chemiluminescence detection is based on NO-triggered chemiluminescence reactions.Chemiluminescence can be realized by the oxidization of NO with ozone (O3) into nitrogen dioxide in the excited state (NO2*),which emits a photon spontaneously when it decays back to its basal lower energy state (Figure4A).2,3,7,31-33Another route to chemiluminescence is the use of chemiluminescence agent such as luminol to be excited by highly oxidative peroxynitrite (ONOO-) which is generated by the reaction of NO with H2O2.34The emission of photon or luminescence is measured by a photomultiplier.Photomultiplier converts this luminescence into electrical signal which is proportional to NO concentration (Figure4A).35The high specificity of NO detection is attributed to its unique properties,which includes its ability to exist as a gas and its quick reaction rate with ozone.32
It is extremely difficult to directly detect the NO level in blood.Lopez et al.36used acidic vanadium III to reduce the intracellular metabolic products of NO including nitrite and nitrate in the collected serum into NO at 98°C,which was then quantified by the chemiluminescence method.Chemiluminescence assay has also proved to be efficient in measuring NO generated by NOS in endothelial cells.7In the treatment of persistent pulmonary hypertension in newborns and respiratory distress syndrome in adults,exhaled NO levels of asthma patients as an important indicator were also sequentially measured successfully.37Kikuchi et al.38measured the continuous release of NO from the isolated perfused rat kidney organ along with changes of perfusion pressure by the luminol-based chemiluminescence method,achieving a high determination limit of approximately 100 fM.
In addition,Woldman et al.35developed a chemiluminescence method for detection of NO generation in cell cultures by a luciferin-luciferase system (Figure4B).The activation product (pyrophosphate) of guanylyl cyclase by NO reaction was converted to adenosine triphosphate by adenosine triphosphate sulfurylase which further excited the luciferin-luciferase system to generate chemiluminescence.The luciferin-based chemiluminescence method was proved to be two orders of magnitude more sensitive than fluorescent method using 4-amino-5-methylamino-2′,7′-difluorofluorescein (DAF-FM)(mentioned in details in the subsequent section) when tested on cell cultures of bovine aortic endothelial cells and activated murine macrophages.

Figure4:General mode of operation of the chemiluminescence method for detection of NO.
In order to increase the operability and applicable range of the chemiluminescence method,the microdialysis technique based on gas permeable porous membrane is frequently integrated.Yao et al.39realizedin vivoNO measurement with high sensitivity by combining microdialysis technique with chemiluminescence method.Special probe designed allowed the detection of NO in blood and brain tissue of rat and rabbit in spite of the influence of various physiological conditions(Figure5A).The luminol-based chemiluminescence method was used by Robinson et al.40to measure NO in exhaled breath.They used a kind of gas permeable porous hollow polypropylene fiber membranes to make gaseous NO react with the solution of luminol and H2O2in the interface,successfully detecting NO with a limit of 0.3 ppb and a response time of 2 seconds (Figure5B).Zhou and Arnold41developed a kind of gas permeable silicone membrane to detect NO concentration in solution by the luminol-based chemiluminescence method,achieving a detection limit of 1.3 µM,a response time of 8-17 seconds,and a dynamic range from 5 to 40 μM (Figure5C).Chemiluminescence method provides highly sensitive,real time monitoring of NO with rapid and reliable results.7However,this method is not feasible to monitor NOin vivo.Moreover,the detection can be interfered by ozone gas stream,since the ozone generators are difficult to provide a stable,repeatable gas stream.7Liquid phase detection was made feasible by the luminol/H2O2assay,but has low specificity and luminol can react with free radicals causing interference.2Measurement of exhaled breath can be achieved which can aid diagnosis of respiratory diseases,monitor efficacy of therapies for respiratory diseases.40On the other hand,NO measurement by this method is limited by its bulky and expensive instruments,time consuming factor,low specificity and sensitivity.2,3,42
FLUORESCENCE METHOD
Fluorescence method has emerged as a very useful technique for detection of NO.A number of fluorometric probes and dyes have been devised to exploit the full ability of fluorescence in measurement of NO.The most commonly used fluorescence agent 2,3-diaminonaphthalene (DAN),is exploited for its ability to produce fluorescent N-nitrisating agent 2,3-naphthotriazole from NO (Figure6A).7,34,42Another kind of fluorescent agent diaminofluoresceins (DAF) has also proved to be equally useful for detection of NO (Figure6B).Spatial and temporal aspects of NO production have been monitored using fluorescent probes which mainly involve DAF,DAN and some other fluorescent agents discussed below.34
DAN has been employed as an indicator for NO detection in many studies.In 2002,Wada et al.43developed a high performance liquid chromatography method to determine the concentration of NO in cultivated cells of plant Agavepacifica using fluorescence detection with DAN.This method successfully detected NO with the limit of 3.4 pmol/g cells.43In 2003,Gharavi et al.44employed similar fluorometric high performance liquid chromatography detection for one of the stable oxidation products of NO,i.e.,nitrite in murine hepatoma cells.Fluorescence monitoring provided limit of detection of nitrite to be 10 pM.Similarly,DAN assay was used to determine the endothelial NO release from cultured porcine pulmonary artery endothelial cells by measuring the fluorescence from 1-(H)-naphthotriazole which was formed by the reaction between DAN and nitrite.However,DAN assay failed to detect NO in agonist stimulated cultured cells.When the stimulated pulmonary artery endothelial cells were treated with nitrate reductase,DAN assay was able to detect minor levels of NO generated by these cells.Hence,these results suggested that nitrate reduction was essential for the function of DAN.45
Other agents like 2,7-dichlorfluorescin,cobalt complex,Fe(DTCS)2-based sensor did find their application in fluorescent based NO-sensing,however were limited due to poor detection sensitivity,low specificity for NO,fluorescence from interference,etc.(Figure6G-J).2In order to measure NO under physiological conditions,Kim et al.46prepared a novel set of fluorescent indicators,which are popularly known as DAFs.Since its discovery,DAF has been used in various studies for detection of NO.DAFs interact with NO in the presence of dioxygen to yield the triazole derivatives which are highly fluorescent.DAFs generate less fluorescence than the triazole derivatives by 180-fold.The probe most commonly employed uses DAF-2 form which exhibits high fluorescence due to formation of triazofluorescin upon reaction with oxidation product of NO.Given this property of fluorescence,DAF-2 has been employed to measure NO in various settings (Figure7C).The membrane permeable form of DAF-2,DAF-diacetate was utilized to measure NO in endothelial cells by Leikert et al.47Langendorff perfused rabbit heart was used by Patel et al.48to monitor the change ofNO concentration using DAF-2.Similarly,Strijdom et al.49demonstrated the use of DAF-2-DA to detect intracellular NO in fresh adult rat cardiomycetes by flow cytometry and compared the results with cellular nitrate or nitrite level.Use of DAF-2 as an NO sensitive probe revealed the release of NO around the blood vessel wall and sporadically in the extracellular space of B16 melanomas using multiphoton laser-scanning microscopy.However,in the pres-ence of inhibitors,the fluorescence appeared abolished.Along with the immunohistochemical analysis,eNOS was found to be predominant source of NO in vascular endothelial cells,whereas iNOS was the sporadic source of NO in the stromal cells of B16 melanomas (Figure6C).50

Figure5:Different ways adopted for monitoring NO levels using chemiluminescence method.
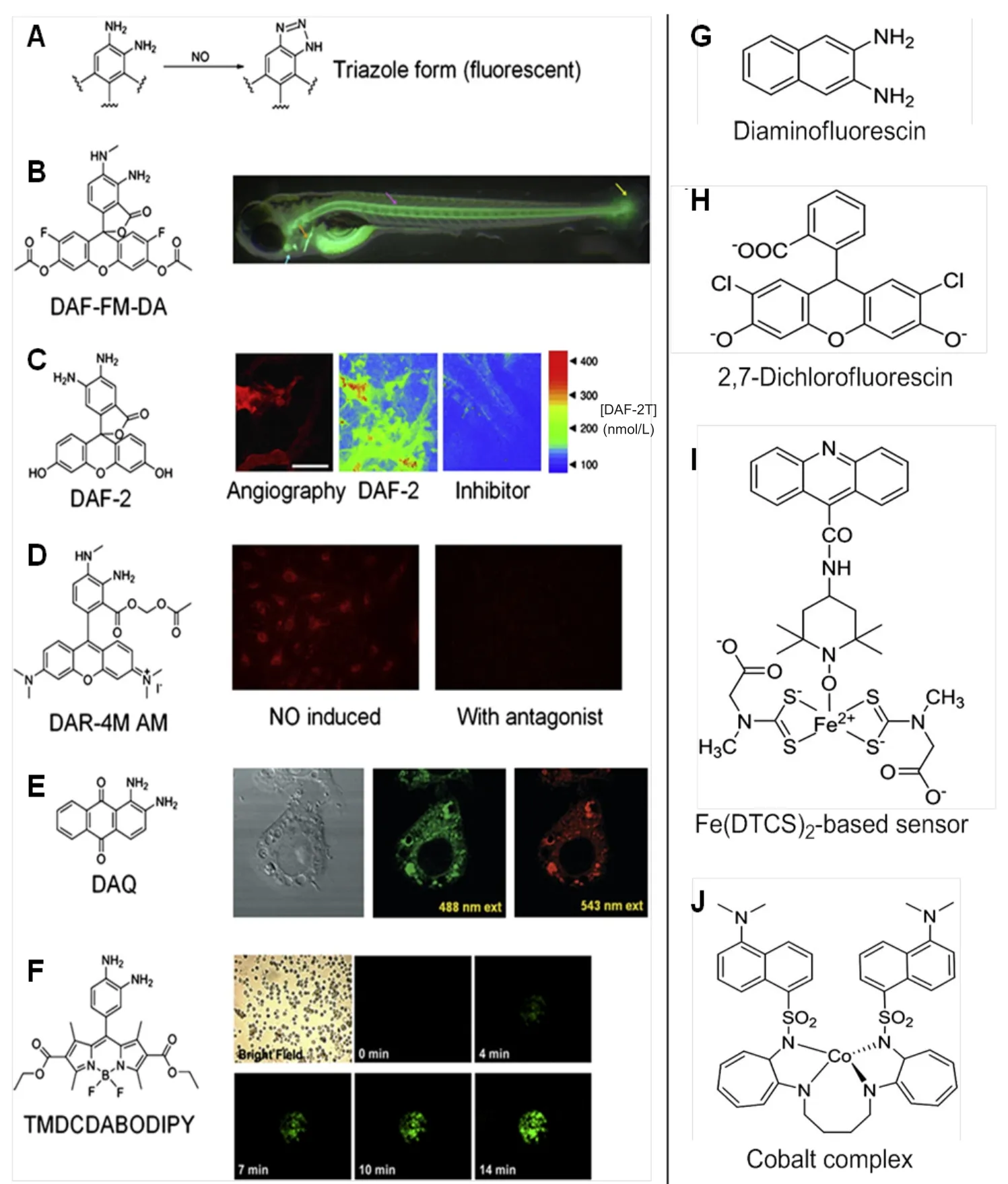
Figure6:Various fluorescent compounds used for imaging of NO.
Another form of DAF,4-amino-5-methylamino-2′,7′-difluorofluorescein diacetate (DAF-FM-DA) (Figure7A)was used by Metto et al.51to detect NO produced in single T1 lymphocytes (Jurkat cells) with the help of a microfluidic device.Standard set of cells was obtained by labeling them with 6-carboxyfluorescein diacetate.Immune cells expressed iNOS upon stimulation by LPS when compared with control set of cells,which exhibited two-fold increase in NO production.51Recently,Agrawal et al.52targeted overexpressing NOS isoform of HEK 293 T cell line imaging to screen the capacity of NOS inhibitors using DAF-FM-DA.Lepiller et al.53employed DAF-FM-DA to detect NO generation sites in a living zebrafish Danio rerio model.The authors observed that NO production changed along the development in the notochord caudal fin.However,no changes were seen in the bulbus arteriosus.This method was also employed by the group to measure local changes in NO production in response to any stress.From their findings,the authors monitored changes in NO production in live zebrafish under physiological and pathophysiological conditions (Figure6B).53
Zhou et al.54previously showed measurements of NO in intact venules by platelet activating factor induced endothelial cells.However,the leakage of the DAF-2 dye after the washout steps compromised the accurate measurement of NO.Hence in 2011,to overcome the dye retention problem and improve the sensitivity for NO measurements,the same group used DAF-2-DA in rat venules.Continuous perfusion of DAF-2-DA in the rat venules was used to measure NO by fluorescence imaging under basal and stimulated conditions.Once DAF-2 achieved a stable state in the endothelial cells,basal and stimulated NO was quantified.This study also showed that,the measurement of fluorescence was mainly due to the hydrolyzed DAF-2 in the cells.With the help of this,NO can be assessed by subtracting non-NO-dependent intracellular DAF-2 in living tissues.
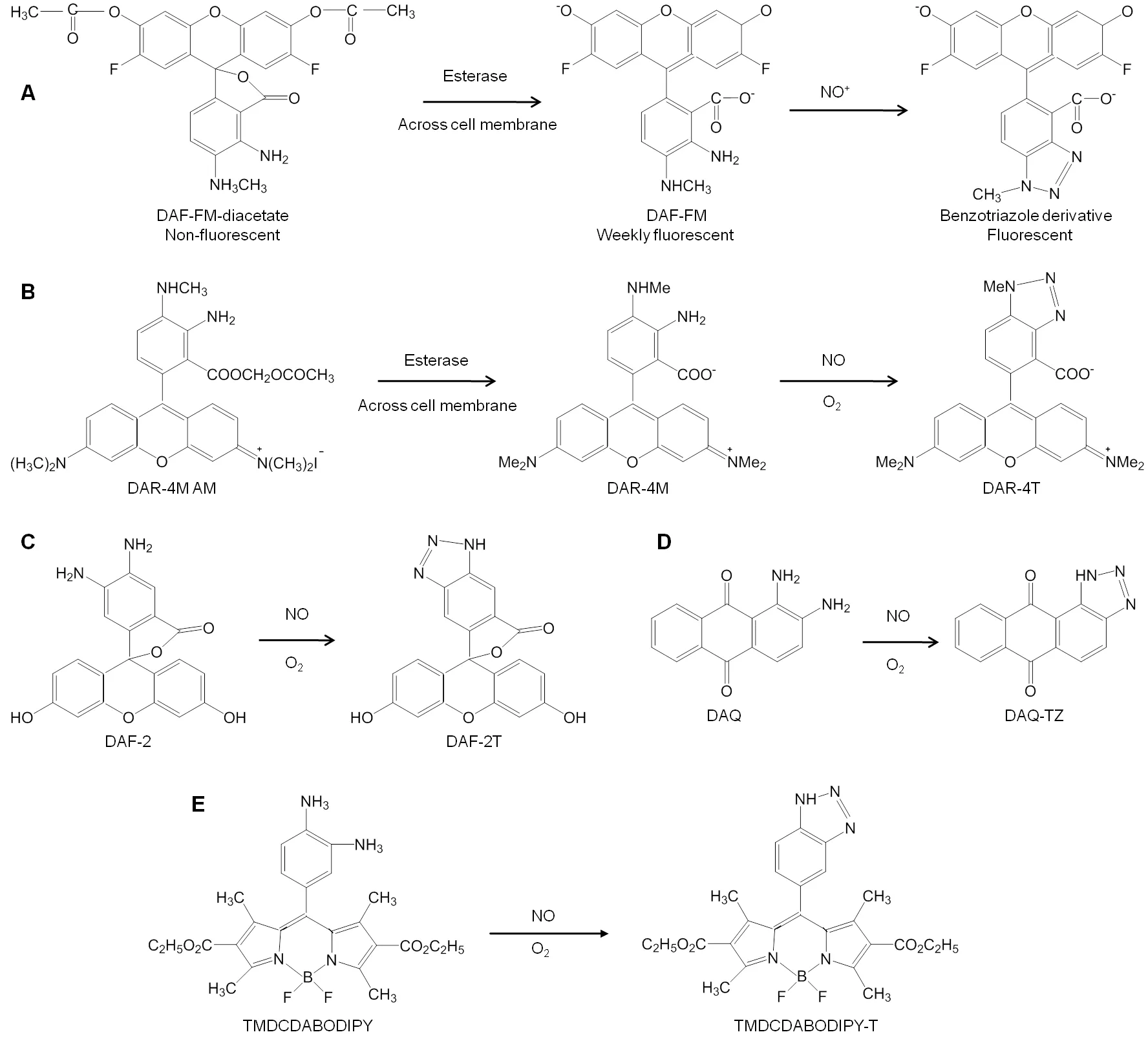
Figure7:Reaction mechanisms of different fluorescent probes for nitric oxide (NO) detection.
Furthermore,in order to overcome the limitations of using DAF-2 in biological applications,in 2001,Kojima et al.55developed a fluorescent indicator on rhodamine chromophore DAR-4M AM (Figure7B) for detection of NO.The membrane permeable property of DAR-4M AM was successfully employed by the authors to carry out bioimaging in bovine aortic endothelial cells for detection of NO with a limit of 7 nM without any pH dependency above pH 7.Human umbilical vein endothelial cells when treated with platelet-activating factor (PAF) could help to determine its effect on NO production.The produced NO was measured using DAR-4M AM and visualized using fluorescence microscopy and also revealed that in the presence of NOS inhibitor and PAF-receptor antagonist,NO production was not achieved,hence confirming the role of PAF in intracellular NO generation by activation of PAF-receptors in the human umbilical vein endothelial cells (Figure6D).56Recently,another rhodaminedeoxylactam based probe was developed and used to detect endogenous and exogenous NO in living HepG2 and RAW 264.7 cells.Once the probe came in contact with NO,it emitted a strong fluorescence which aided the easy detection of NO with high specificity.57
Externally stimulated and non-stimulated RAW 264.7 macrophage cells were treated with 1,2-diaminoanthraquinone(DAA or DAQ) (Figure7D),a non-toxic probe known for its ability to visualize NO in living cells.Its reaction with intracellular NO in the presence of oxygen generated the triazole form of DAA,which could be spectrally visualized using confocal microscopy and fluorescence spectroscopy (Figure6E).58-59The NO reductase property of cytochrome P450 55B1 from Chlamydomonas reinhardtii was exploited by Li et al.60in 2016 to develop a new fluorescence biosensor for detecting NO.The constructed biosensor was employed to detect NO release from L-arginine stimulated rat liver homogenate.The authors concluded that this biosensor can be successfully used for detection of NO in biological samples.Lim et al.61synthesized a fluorescent probe with a copper Cu(II) complex and metal chelating ligand in 2006.The irreversible reduction of Cu(II) to Cu(I) along with the release of nitrosated ligand was determined to be the reaction causing NO induced fluorescence by spectroscopic and mass spectroscopic methods.The Cu(II) based complex was employed by the authors to detect NO generated in macrophages and live neurons by inducible and constitutive NOSs (iNOS and cNOS).MNIP-Cu {Copper derivative of [4-methoxy-2-(1H-napthol[2,3-d]imidazol-2-yl)phenol]} was developed by Jain et al.62to localize NO generation sites in various parts of sunflower (Helianthus annuusL.)almost without lag time which was found to be more specific than DAF probes.
Liu et al.63designed ADNO (2-(α-(3,4-diaminophenoxy)acetyl)-6-(dimethylamino)naphthalene) a new two photon fluorescent probe in 2014 (Figure8A).This probe detected NO on the basis of photoinduced electron transfer mechanism.The NO sensitive fluorescence modulator function of the probe is carried out by o-phenylenediamine moiety and 2-acetyl-6-(dimethylamino) naphthalene moiety functions as the two photon fluorophore.The electron-rich diamine moiety can be altered to quench the fluorescence of 2-acetyl-6-(dimethylamino) naphthalene by transferring the electron to excited fluorophore which can help to detect NO as anticipated by the authors.The experiment carried out in aqueous solution and NIH 3T3 cells presented that ADNO has rapid response rate to NO by exhibiting significant turn-on fluorescence.This probe was found to be less pH dependent and showed high selectivity and can be very useful tool in future biological applications.
Recently,various modifications are done to develop better fluorescent probes.For example,Zhang et al.64developed a dual turn on type BODIPY probe with no intrinsic fluorescence due to photoinduced electron transfer effect.This probe could detect NO and nitrite ion in HepG2 cells with fluorescence confocal microscope under neutral and acidic conditions respectively with DEA-NONOate as the NO donor;(which switches off the photoinduced electron transfer) in assistance with intracellular cysteine and glutathione.Similarly,Chen et al.65developed a BODIPY probe which could simultaneously detect endogenous and exogenous NO and glutathione in macrophage cells to uncover their inter-relation for maintaining the biological system’s redox balance (Figure6F).Another modification in the BODIPY dye was done by Gao et al.66by adding dihydropyridine and triphenylphosphonium moieties to it which could specifically target mitochondria(Mito-DHP probe),tracking the exogenously produced NO in HepG2 cells and endogenously produced NO in stimulated RAW264.7 murine macrophage cells under anaerobic conditions (Figure8B).1,3,5,7-tetramethyl-2,6-dicarbethoxy-8-(3׳,4׳ -diaminophenyl)-difluoroboradiaza-s-indacence(TMDCDABODIPY) (Figure7E) was developed by Huang et al.67which allowed the real-time NO imaging using inverted fluorescence microscope in human umbilical vein endothelial cells,Sf9 cells and PC12 cells when treated with L-arginine.Thus,this combination of the probe with inverted fluorescence microscope could provide selective and sensitive detection of NO released from cells.A hepatocyte targeting fluorescent sensor (Gal-RhB) was developed by Zhang et al.68in 2018(Figure8C).The cellular andin vivoimaging using HepG2 and Zebrafish using this sensor allowed detecting NO about 1.26 nm with good selectivity and sensitivity making it an obvious choice for liver diseases caused due to NO deficiency or surplus amount.

Figure8:Various fluorescent probes employed with different strategies to detect NO.
ELECTROCHEMICAL METHOD
Use of various electrodes for electrochemical detection of NOin vitroandin vivois widely employed.The choice of electrode is of great importance for detection of NO since the electrochemical process takes place at its surface and its quality influences the charge transfer process between target analyst and electrode material.Carbon and noble metal electrodes are most commonly used for NO detection.Other types of materials used for making the electrodes are described in Table1 and Figure9.72Efforts are being made continuously by researchers to modify electrode for better measurement of NO in various settings.This section will focus on such electrodes and their application for NO detection.
The basic of NO detection using amperometric method is the current generated on the surface of electrode due to oxidation of NO.A typical system for NO detection includes use of working (coated with platinum or Teflon) and reference electrodes (Ag/AgCl) immersed in solution containing NO.The application of positive potential (800-900 mV) causes NO to be oxidized at the surface of working electrode by generating a redox current.The oxidation occursviaelectrochemical reaction followed by chemical reaction.One electron transfer from NO molecule to electrode constitutes the electrochemical reaction which generates nitrosonium ion (NO+).This cation reacts with OH-to form nitrite (NO2-) which further gets oxidized to nitrate.NO meter measures the amount of NO oxidized which is proportional to the current flow between working and reference electrodes.An internal electrolyte is enclosed in the NO gas selective permeable membrane of the working electrode.Current due to oxidation gets generated at the surface of working electrode when NO gas passes through the membrane and the electrolyte.However,this method generates redox potential as low as 1-10 pA in biological system.This justifies the need to identify better electrodes to overcome this limitation.ISO-NO Mark II system allows the detection of redox current as low as 0.1 pA.This instrument could measure NO without the need of special electrical screening,ex.Faraday Cage.The development of optically isolated Apollo-4000 has paved the way for detection of NO and other free radicals.73
The use of NO specific electrodes has seen a successful graph in the recent past for monitoring NOin vitroandin vivo.In 1990,NO release stimulated by activation of specific neural elements in brain tissue was measured by inserting an electrochemical microprobe into the molecular level of rat cerebellar slice with a range of 8-58 nM by Shibuki.76This was followed by detecting NO release in real time in the hemoglobin free effluent of guinea pig isolated hearts by Fujita et al.77in 1998.The amount of released NO was measured by comparing the effects of several vasodilator drugs on altering the coronary flow.The same group used Clark type electrode with a selective permeable membrane to measure effluent NO release and bradykinin induced coronary flow in anesthetics treated crystalloid perfused guinea pig hearts.Development of microcoaxial electrode by Kitamura et al.78permitted continuous and direct detection of NO in bovine aortic cultured endothelial cells stimulated by acetylcholine and inhibited NG-nitro-L-arginine methyl esterin the range of 0.1-1.0 µM.They suggested that future application of this electrode could allow measurement of intracellular NOin vitroandin vivobecause of the coaxial arrangements of the working and reference close together.
A few of researchers applied the NO microelectrode to determine the changes in NO concentration in rat superior mesenteric artery by using certain stimulators and attenuators,acetylcholine and NG-nitro-L-arginine.The effect ofthe stimulator and attenuator allowed the authors to conclude that contractility of rat superior mesenteric artery is majorly regulated by endothelium derived NO.79In a similar manner,variety of studies are carried out under various stimulators and attenuators to determine the changes in level of NO in rat superior mesenteric artery,80renal hypertensive rat superior mesenteric artery with impaired endothelium dependent vasorelaxation.81Platinum based NO sensor was used by Griveau et al.82in 2006 to detect NO in tumor bearing mouse.In 2008,real-time detection of NO in tobacco cell suspension treated by cryptogein was studied by Besson-Bard et al.83using NO sensor.The increase in NO concentration due to cryptogein was evaluated in extracellular environment given its role in intracellular environment.

Table1:Types of basic electrodes

Figure9:Types of basic electrodes.
Brown et al.84studied the reaction of NO with metallophthalocyanine (MPc) surface coated electrodes which lead to initial electron transfer from NO to MPc,further resulting in electron to relay to electrode charge sink,hence producing current response.The transient NO+is assumed to be stabilized by MPc before its oxidation to nitrite.Hence,NO signal was amplified and lowered potentials for voltammetric features.Reduced potential for NO oxidation supposedly caused amplification of sensitivity.Selectivity was improved when applied potential was lowered only with high potential interferences such as nitrites.Signal from low potential intereferents such as amino acid was found to be not affected.However,they did hamper continuous detection of NO with good selectivity.As a result,the only feasible way for MPc complexes to improve NO selectivity wasviathe amplification of signal sensitivity.However,by comparison to the permselective membrane,the use of MPc complexes for improved selectivity is achievable but currently limited.
Catheter type NO sensor was employed for real-time NO measurement in the coronary circulations in humans diagnosed with dilated cardiomyopathy.85The NO sensor was inserted in the great cardiac vein (Figure10),and measurement of plasma NO level along with average peak velocity were carried out under the in fluence of acetylcholine and NG-monomethyl-L-arginine.This study demonstrated the clinical application of NO sensor for evaluating endothelial functions in patients with heart condition.They claimed that this sensor can be further employed for diagnostics and therapeutic applications in the future.
Scanning electrochemical microscopy was coupled with amperometric NO nanosensor by Jo et al.,86allowing the realtime imaging of NO releasing sites in nNOS immunoreactive cells which comprise the NO releasing sites of the living mouse brain.The authors successfully providedin vivo2-dimensional images along with 3-dimensional NO distribution data in the living brain with high sensitivity and spatial resolution.Apart from its long data acquisition time,this technique also enlightened the relationship between NO measurements,the size or location of nNOS immunoreactive cells and immunoreactive intensity.A hemin-functionalized graphene field effect transistor sensor constructed by Jiang et al.87was used on macrophage and endothelial cells which provided rapid response time,high specificity and selectivity of generating NO signal with high spatiotemporal resolution.NO selective electrode allowed robust and reproducible detection of subtle changes in NO concentration produced by cultured pulmonary myofibroblasts of rats under wide variety of conditions.88Recently,amperometric NO release was viewed in real time at the stimulated acupoints in rats inserted with an acupuncture microsensor needle made up of gold film and doped with ironporphyrin functionalized graphene composite.89Further,the authors claimed the application of the acupuncture needle to detect other vital signaling molecules (Figure11).
(3)深二含细砂、中细砂夹黏土压缩层(BA7):处于该层组中部至下部,为该层组的构成主体,由数层细砂及中细砂夹数层黏性土组成,底板埋深100~140 m,厚度5~10 m,饱水,中密-密实状态。
These electrochemical sensors are known to have a response time of few seconds with good sensitivity;real-time monitoring and can measure the slight change on NO concentration.71Electrode modification and/or the applied potential allow the enhancing of selectivity and sensitivity.34In general,electrochemical electrodes allow detection of NO in biological samples due to their small size and at low cost.42However,the measurement is affected by gas interference and may provide inaccurate results.3,34
GAS CHROMATOGRAPHY METHOD

Figure10:X-ray pictures with the position of Doppler wire,nitric oxide sensor and infusion catheter.
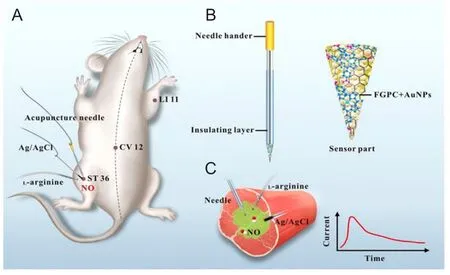
Figure11:Schematic representation of real-time monitoring of NO at acupoints of rats using acupuncture microsensor needle.
The use of gas chromatography for NO detectionin vitroandin vivois currently limited.There is very less data determining NO levels using gas chromatography due to its limited life span.Hence,gas chromatography is employed to measure NO in its different converted forms.The use of nitrite and nitrate as a surrogate to measure NO is commonly employed.Gas chromatography was used to detect oxides of nitrogen in gaseous mixtures like Diesel exhaust gas using argon ionization detection cell.Samples were prepared to measure NO and argon mixture and compared their chromatogram with NO and air mixtures.With the sample size of 0.1 to 1 µL,the increase in retention time is observed with the decrease of NO concentration.Hence,retention time being the function of NO added,it was demonstrated by authors that it might be possible to use this measurement for analytical purposes.90
GC-MS method was freqxuently used to measure nitrite and nitrate for detection of NO.In vivostudies revealed that administration of paracetamol caused the temporary increase in the plasma nitrite concentration which may indicate that eNOS activity and/or expression of eNOS increased after few hours of paracetamol administration.On the other hand,proliferatingin vitroadult rat hepatocytes exhibited weak inhibitory effect in response to paracetamol.91Implications of recent research revealed that NO is responsible for cavernous arterial and trabecular smooth muscle relaxation.Hence,GCMS was employed by Becker et al.92to measure nitrite and nitrate as a determinant of NO metabolites in healthy men and patients with erectile dysfunction under different functional conditions of penis.
ELECTRON PARAMAGNETIC RESONANCE/ELECTRON SPIN RESONANCE SPECTROSCOPY METHOD
Electron paramagnetic resonance (EPR) or ESR has been widely employed for characterizing free radicals with unpaired electrons without need of any interference.4,7Due to unpaired electrons,the free radicals become very reactive and unstable at room temperature.The reactive nature of free radicals and short-life make them difficult being detected.Hence,spin trapping method of EPR was developed.The basic principle of spin trap method involves the use of specific compound to react with short lived radicals to convert them to stable radicals in order to be characterized by EPR.These special compounds are termed as spin trap reagents.A variety of such reagents are currently being used to react with NO in biological systems since it also contains unpaired electron thereby converting it to a stable form to be further characterized using EPR spectroscopy.
The most commonly employed spin trap reagents for NO detection are variants of iron (Fe) complex with dithiocarbamate derivates (Figure12A) since NO has high affinity for them and the resulting stable complexes allow endogenous NO measurementsin vivo.The different types of these complexes are named as diethyldithiocarbamate (DETC) (water insoluble),N-dithiocarcoxy sarcosine (DTCS) (ligand,water soluble),N-methyl-D-glucamine dithiocarbamate (MGD)(water soluble),di(N-(dithiocarboxy)-N-methyl-L-serine(MSD),etc.93,94
Hirayama et al.97carried out NO measurement in live LPS treated mice kidney by using iron and N-dithiocarboxy sarcosine complex (Fe-DTCS)2.NO was spin trapped as NO-Fe(DTCS)2and detected using EPR spectroscopy which resulted in characteristic 3-line EPR spectrum (Figure12B).EPR spectroscopy was also performed on organ homogenates confirming NO distribution in upper abdominal area,renal and hepatic areas (Figure12C).Similar spin trap was employed to detect NO in melanoma tumor in mice and live mice liver.98,99NO was detected by EPR spectroscopy using Fe-(DETC) in upper abdomen of living septic shock mice,100brain of septic shock mice,101kidney of ischemia-reperfusion rats,102forebrain of ischemia rats,103in rabbit vascular tissue.104NO in the blood circulation of conscious mice was detected by EPR spectroscopy using Fe-MGD complex which reacted with NO to form[(MGD)2-Fe2+-NO] complex.The S-band EPR spectrometer resulted in the characteristic 3-line spectrum in the blood circulation of mouse tail administered with sodium nitroprusside as NO donor.105Furthermore,the same complex was also used to detect the NO distribution in micein vivo.106Four kinds of dithiocarbamate iron complexes were synthesized by modifying the functional group of ligand and used as a spin trap to measure NO in LPS-treated mouse liver and blood.The newly synthesized complexes proved to be good traps for NOin vivo.Low frequency ESR spectroscopy data was obtained using Fe(MSD)2,while the other two complexes [Fe(DTCP)2,Fe(DTCTP)2] along with Fe(MSD)2were measured in blood inex vivoexperiments.A unique property was exhibited by the fourth complex Fe(DTCMP)2by giving NO adduct only in blood,reflecting the amount of NO in the blood.107
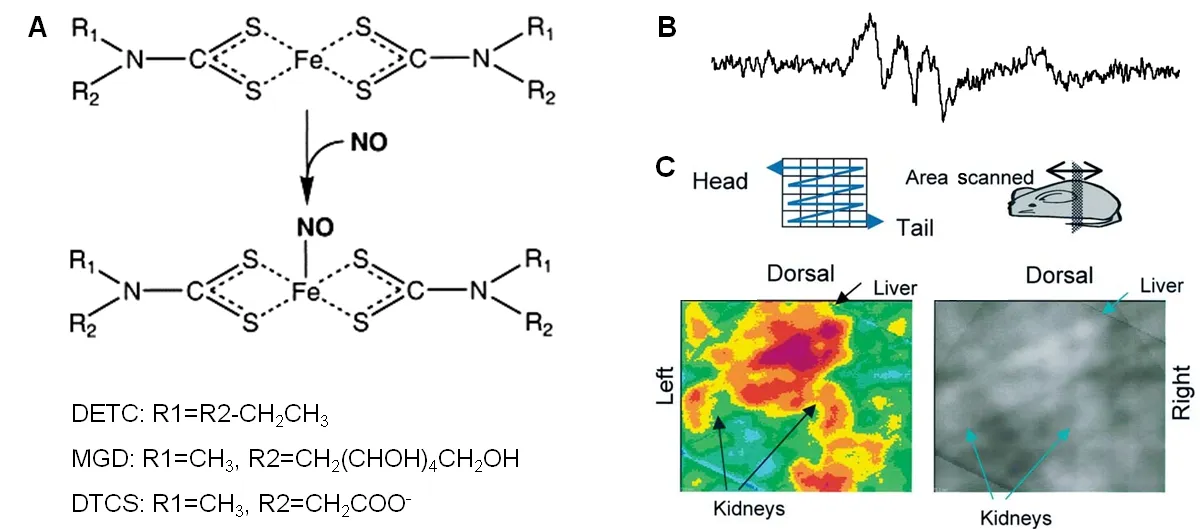
Figure12:Use of various spin traps with different strategies to detect NO.
NO performs numerous functions in plants,including pollen tube growth,root development closure of stomata,defense genes activation,etc.Being a diffusible gas,its presence in the intracellular and extracellular spaces is inevitably allowing it to react with the surrounding environment.Nodule infected tissues contain leghemoglobin which is rich in Fe2+.The Lb2+NO nitrosyl complex formed when NO binds to the iron radical can thus be detected using EPR spectroscopy.This was performed in the nodules of soybean as a direct method to detect NO.108Mice infused with nitrite was induced with cardiopulmonary arrest and subjected to EPR resonator.The EPR spectra achieved was confirmed to be due to the labeled infused nitrite rather than the endogenous tissue nitrite or from enzymatic reactions.The results were compared with control mice,infused with just saline.The NO generation was found to be increased in mice with cardiopulmonary arrest infused with nitrite;NO mainly found in heart,liver and lungs.This study further proved the NO generation due to tissue ischemia by direct reduction of tissue nitrite during the ischemic acidic and reduction conditions.109
However,there are certain limitations using these spin traps.MGD and DTCS need to be prepared just before being administered,since they are unstable,especially in acidic solutions.110Also high concentration of iron complexes might be toxic,and oxidant formation of the complexes is possible which might affect the system being studied.Toxicity of the traps might be system specific depending on their concentration.Furthermore,less water soluble DETC species are unsuitable for NO trappingin vivoeven though it can pass the cell membrane and blood brain barrier.110Nonetheless,EPR technique qualifies as a valuable method to identify free radicals in complex systems along with providing important details regarding the structure,mobility,interactions and kinetics of the radical,111but is limited due to high cost of instrumentation and time consumption to prepare sample.2
MAGNETIC RESONANCE IMAGING METHOD
There are very limited literatures on using MRI modality to detect NO.However,efforts are made to employ EPR spin trap agents which detect NO to be further imaged using MRI(Figure13A and B).Considering this as a powerful tool,MRI enables imaging,visualizing and localizing NO generation sites with the use of specific contrast agents.
Fujii et al.112used this concept and combined NO spin traps with MRI to measure its distribution in rats with septic shock.Adult male Wistar rates were administered with(MGD)2-Fe(II)-NO spin trap and LPS was used to induce NO.EPR measurements were carried outin vivoandin vitrofollowed by MRI.NO signal from excised organs was highest in liver (Figure13C) followed by heart and kidney and very little quantity in brain.The NO spin trap complex was determined to be stable in complex tissues and organs.The paramagnetic properties of the spin traps causes strong proton relaxation enhancement.The spin-spin and spin-trap relaxation of the water protons is stimulated by the magnetic moment of unpaired electrons,which causes the decreases in the T1 and T2 relaxation times,thus enabling the enhancement of signal intensity in T1 and T2 weighted MR images.The relaxivity was found to increase distinctly once NO complexes with (MGD)2-Fe(II) which can enable to view the regions of NO entrapmentin vivo.The authors named this technique as nuclear magnetic resonance-spin traps and mention that this method can be extended for imaging other free radicalsin vivowith suitable spin trap agents.Reaction of NO with oxygenated and deoxygenated hemoglobin generates paramagnetic methemoglobin (Figure13A) and nitrosylhemoglobin (Figure13B) species which are known to significantly cause concentration dependent changes in signal intensity in erythrocytes when imaged using MRI.Healthy male volunteers were administered with regional brain activator ascorbic acid specifically reduced methemoglobin(which measured by EPR) resulting in a significant decrease of functional MRI signal changes thereby suggesting the role of reductant on blood flow independent effect.These signal intensity changes can be attributed to NO generation during brain activation (Figure13D).113
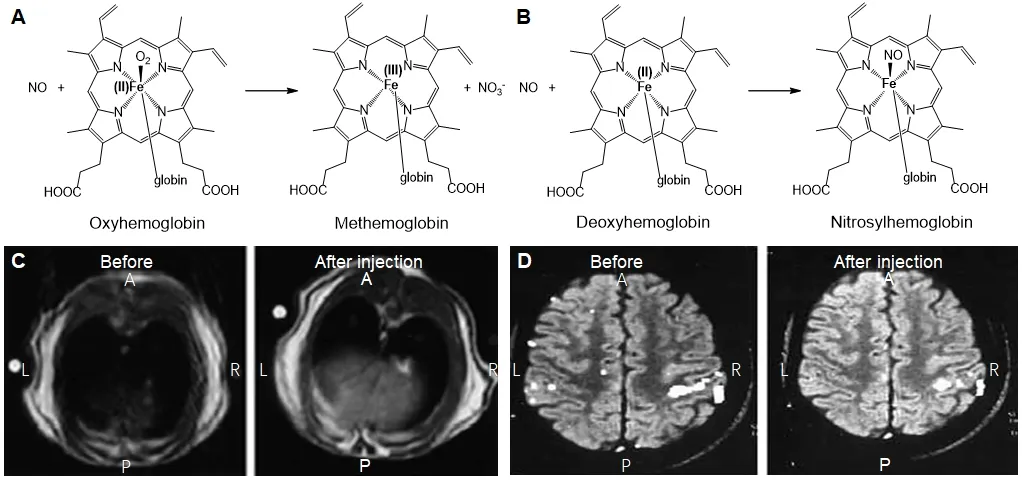
Figure13:General reaction of hemoglobin and deoxyhemoglobin with NO and MR imaging of NO in liver and brain.
The paramagnetic chemical exchange saturation transfer(PARACEST) effect is irreversible disappearance from amide and appearance for amine when observed under MRI caused by PARACEST MRI contrast agent which converts amide to amine in the enzyme catalysis reaction.On this basis,Liu et al.114designed a new PARACEST MRI contrast agent(Yb(III)-(1,4,7,10-tetraazacyclododecane-1,4,7-triacetic acid)-orthoaminoanilide which could measure extracellular pH withinin vivoanimal models to detect NO.The same group developed a similar smart PARACEST MRI agent which after reaction with NO undergoes chemical modifications resulting in the formation of hydrazine,which does not exhibit the PARACEST effect thereby enabling detection of NO.115Chelation of lanthanides (III) such as Yb,Er and Tm with 1,4,7,10-tetraazacyclododecane-1,4,7,10-tetraacetic acid o-aminoanilide is used to synthesize the agent which produces protons with high shift exchange from the functional groups on amide and amine.The absence of PARACEST effect due to reaction of agent with NO is assessed by nuclear magnetic resonance in the presence of NONOate (NO donor).pH and temperature conditions were applied to assess the PARACEST effect before and after the reaction of agent and NO.The absence of the PARACEST effect is attributed to the detection of NO,as PARACEST effect at different pH was achieved from amide and amine.
Trapping of NO in biologic tissues using paramagnetic mono and dinitrosyl-iron complexes (MNIC and DNIC,respectively) which act as magnetic resonance contrast agents and utilizing their ability to enter into dynamic nuclear polarization with water protons with the help of unpaired electrons of these complexes can be imaged using protonelectron double resonance imaging.Aqueous solutions of MNIC and DNIC resulted in alterations of proton-electron double resonance imaging signal intensity.Furthermore,sodium nitroprussside treated rat liver induced a characteristic MNIC and DNIC ESR spectrum.The researchers thus concluded that exploitation of NIC for detection of NO using proton-electron double resonance imaging and MRI (could be employed forin vivoNO imaging).116,117
MRI has proved to possess advantages owing to its availability with good enough field of view.It also provides high resolution imaging along with good contrast for soft tissues and anatomical and functional details.2However,due to its low sensitivity,it can detect NO only at high concentrations which requires high concentration of the contrast agents,which may cause toxic effects.The need for long data acquisition time for generating the MRI image increases the overall time for the detection,which may lead to question the amount of NO detected,owing to its short lifetime.2
SUMMARY AND OUTLOOK
NO plays a vital role in the body carrying out numerous pathological and physiological functions.However,future research focusing on these functions will require identifying better quantification methods for NO.To overcome the disadvantages of a single approach,multimodal approach or hybrid technology can be used to provide synergistic advantages for NO detection.The future application of these approaches in clinical research is worth exploring.NO is a difficult molecule to trap and hence causes difficulty in quantifying it.The measure of NOx as a surrogate for NO detection in various methods provides an indirect approach.The above mentioned approaches come with their own pros and cons,as listed in Table2.However,continuous efforts are being made by researchers for real-timein vitroandin vivomonitoring of NO with reliable data by developing better methods.NO concentration,sample size and interference are some of the factors that direct the choice of method to be used.The choice also depends on the limit of detection,the merits and demerits of the method,accuracy,sensitivity,specificity,direct or indirect measurement,etc.
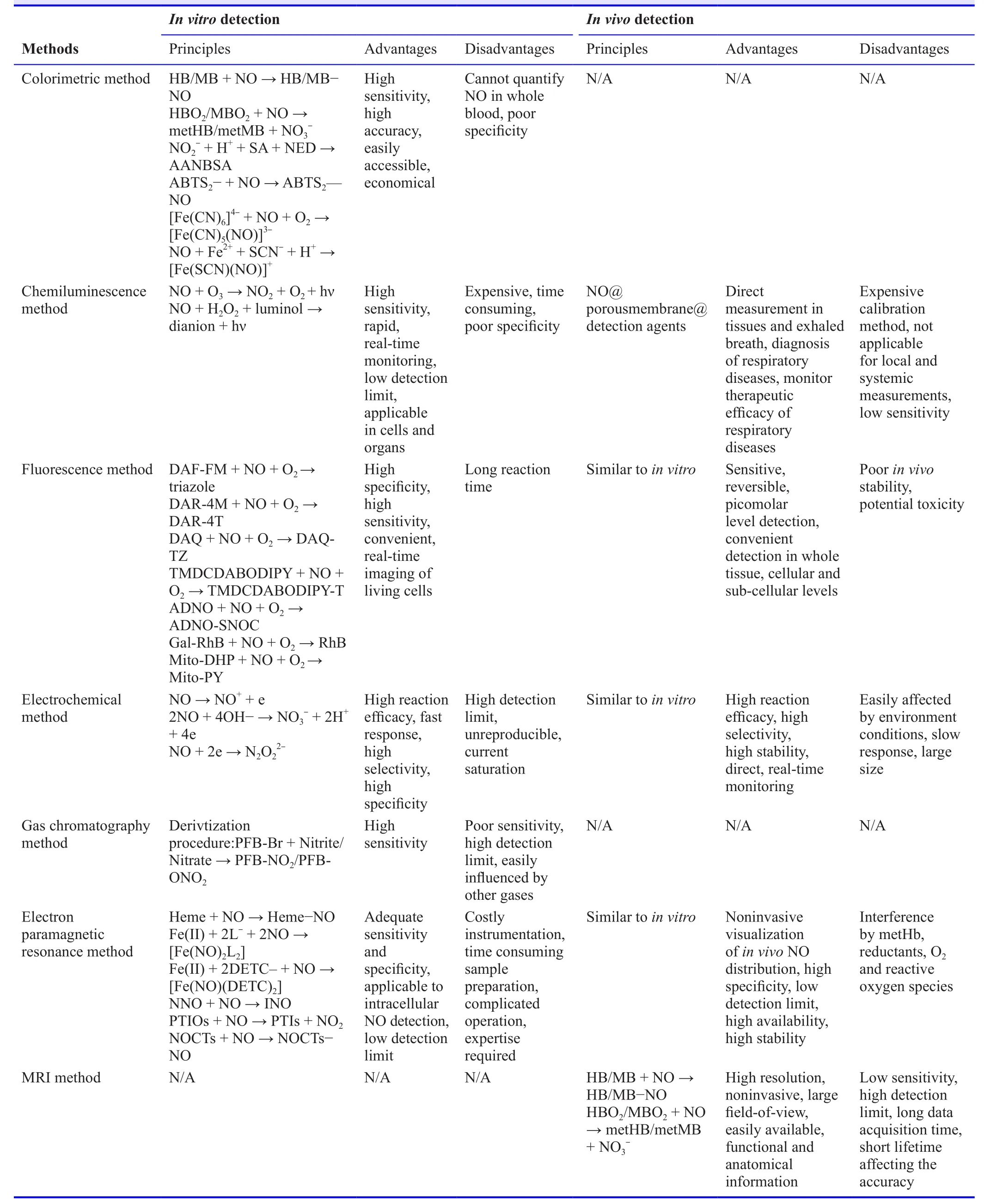
Table2:Comparison of various methods for NO detection in vitro and in vivo.
Author contributions
Review writing:EG;review initial revision:GZ;manuscript revising and supervision:QH.All authors reviewed and approved the final version of this manuscript.
Conflicts of interest
The authors have no conflicts of interests to declare.
Financial support
The work was supported by the National Natural Science Foundation of China,No.51872188,Special Funds for the Development of Strategic Emerging Industries in Shenzhen,China,No.20180309154519685,and Center of Hydrogen Science,Shanghai Jiao Tong University,China.
Copyright license agreement
The Copyright License Agreement has been signed by all authors before publication.
Plagiarism check
Checked twice by iThenticate.
Peer review
Externally peer reviewed.
Open access statement
This is an open access journal,and articles are distributed under the terms of the Creative Commons Attribution-NonCommercial-ShareAlike 4.0 License,which allows others to remix,tweak,and build upon the work non-commercially,as long as appropriate credit is given and the new creations are licensed under the identical terms.
猜你喜欢
杂志排行
Medical Gas Research的其它文章
- Revisiting the expanded use of hyperbaric oxygen therapy for treatment of resistant migraines
- The role of oxygen in cluster headache
- The role of medical gas in stroke:an updated review
- Numerical analysis of mechanical ventilation using high concentration medical gas mixtures in newborns
- Respiratory effects of occupational exposure to low concentration of hydrochloric acid among exposed workers:a case study in steel industry
- Recent developments in nitric oxide-releasing biomaterials for biomedical applications
