运动对肠道屏障和黏膜免疫稳态影响的研究进展
2018-07-03陈佩杰罗贝贝
吴 嵽,陈佩杰,罗贝贝
运动对肠道屏障和黏膜免疫稳态影响的研究进展
吴 嵽,陈佩杰,罗贝贝
上海体育学院 运动科学学院, 上海 200438
肠道屏障和黏膜免疫稳态是机体抵御外来病原微生物入侵的重要防线。运动过程中血液重新分配,消化系统血液灌流量减少,肠道处于低氧环境,诱导转录因子低氧诱导因子(hypoxia-inducible factor, HIF)-1α持续表达,参与调节肠黏膜免疫稳态。相关研究证明,中小强度运动有助于肠黏膜免疫稳态的维持,而大强度运动损伤肠道屏障,破坏肠黏膜免疫稳态。通过回顾肠道屏障结构基础及黏膜免疫机能相关研究报道,综述不同强度运动对肠道屏障功能和黏膜免疫稳态的影响,进一步讨论HIF-1α在运动对肠道屏障功能和黏膜免疫稳态的调节中所起的关键作用,为运动与肠道屏障功能和黏膜免疫相关研究提供新思路。
肠道屏障;黏膜免疫;运动;低氧诱导因子-1α
运动过程中机体的血液重新分配,内脏血流量减少,容易导致肠黏膜缺血,肠通透性增加、细菌易位及内毒素产生[13],可能是运动员出现胃肠道症状(gastrointestinal symptoms)[26]的主要原因[100],主要表现为腹痛、腹泻甚至胃肠道出血。
不同运动强度和持续时间对肠道屏障和黏膜免疫机能的影响各异。定期的适度运动可能会降低机体感染发生率,有研究报道,适度运动能减轻肠道炎症反应,且不会影响正常的肠黏膜屏障组织结构与形态[53]。然而,长时间的剧烈运动可导致机体免疫系统暂时性紊乱,并在运动后持续24 h左右[63,66],主要表现为嗜中性粒细胞的呼吸爆发、淋巴细胞的增殖、自然杀伤细胞活性降低等,并引起肠道屏障功能受损、黏膜免疫稳态变化。运动过程中肠道屏障缺氧程度加剧,导致低氧诱导因子(hypoxia-inducible factor, HIF)-1α稳定表达,可能对肠黏膜免疫稳态的改变起到了一定作用。尽管通常情况下优秀运动员不存在免疫缺陷,但是,免疫指标的小幅变化也将对训练和比赛成绩造成较大影响。因此,了解运动如何影响肠道屏障功能和黏膜免疫稳态,以及在该过程中HIF如何发挥作用,有助于运动员合理安排训练,预防减轻胃肠道症状。
目前,肠黏膜屏障已成为生物医学界的研究热点,Hooper L V[36]和Duerkop B A[19]等对肠黏膜屏障相关研究进行了详细的综述,也有很多报道深入研究了HIF的功能与转录调控机制[20,43,77,78,80],这些研究为进一步了解运动对肠道屏障功能和黏膜免疫稳态的影响提供了理论与实验基础。本文将简要介绍肠黏膜屏障的结构基础及免疫机能,阐述不同强度运动对肠黏膜屏障功能的影响,以及HIF在这些影响中可能的作用机制,为运动与肠道屏障功能和黏膜免疫相关研究提供新思路。
1 肠黏膜屏障的结构基础及免疫机能
肠黏膜屏障是机体屏障系统的重要部分,按功能可分为化学、机械、生物和免疫屏障[2]。化学屏障分泌各种活性物质,保护肠道黏膜。机械屏障通过细胞连接,阻挡病原微生物入侵。大量共生菌群定植在肠黏膜表面,形成生物屏障。正常情况下,肠道共生菌群保持相对稳定;病理情况下,细菌及代谢产物结构与数量失衡,病原微生物可穿透生物屏障。免疫屏障通过黏膜相关淋巴组织及各种免疫活性成分维持肠道正常的免疫机能(图1)。
1.1 肠黏膜屏障的结构基础
黏液素、抗菌肽和溶菌酶共同构成了肠黏膜化学屏障。肠上皮表面杯状细胞(goblet cell)分泌大量黏液素(mucins,MUC)形成黏液层。黏液层由15~30 μm的黏液内层和100~400 μm的黏液外层构成[58]。在黏液外层中,黏液素与菌群相互作用,抵御病原微生物入侵。生理情况下,在人和小鼠肠道中表达的主要是分泌型黏液素MUC2及膜型黏液素MUC13和MUC17[40,46]。Van der Sluis M等发现,MUC2缺失导致小鼠肠黏膜形态异常,表现为肠上皮细胞扁平化和局部溃疡,肠黏膜屏障功能受损,引发炎症反应[91]。在黏液内层中的抗菌肽和溶菌酶主要溶解细菌胞壁、破坏细菌胞膜完整性[81]。抗菌肽主要有α-防御素(α-defensin)和再生胰岛衍生蛋白(regenerating islet-derived protein,Reg)3γ[53]。α-defensin由潘氏细胞(Paneth cell)分泌至隐窝腔[16]。在啮齿类动物肠道中,α-defensin也被称为隐窝素(cryptidins)。Reg3γ由肠上皮细胞分泌,是一种C型凝集素,起到限制细菌与肠上皮接触的作用[90]。肠蠕动时,黏液素、抗菌肽和溶菌酶协同发挥抑菌作用,及时清除入侵病原微生物,并协助修复损伤的肠上皮细胞。

图1 肠黏膜屏障的结构
Figure1. Structure of Intestinal Mucosal Barrier
肠上皮细胞间的紧密连接(tight junction)构成了肠黏膜机械屏障(图2)。紧密连接主要由连接黏附分子的闭合蛋白(occludin)和紧密连接蛋白(claudins)以及细胞内的闭合小带蛋白(zonula occludens)ZO-1、ZO-2、ZO-3构成[8],能将肠上皮细胞连接在一起,阻止大分子和细菌透过。物质通过紧密连接主要经由膜孔途径(the pore pathway)和泄漏途径(the leak pathway)[23]。膜孔途径依赖于钙黏蛋白(E-Cadherins)和紧密连接蛋白[5],是不带电荷的溶质和小分子(半径<4 A)透过紧密连接的主要途径。泄漏途径依赖于ZO-1和occludin[51,89,92,93],只允许少量大分子物质(>10 kDa)包括脂多糖(lipopolysaccharide,LPS)在内通过紧密连接[98]。当肠上皮细胞间紧密连接受损时,膜孔途径与泄漏途径异常开放,肠黏膜屏障通透性病理性升高,大量病原微生物与外来抗原物质透过肠黏膜屏障,从而启动肠黏膜屏障的免疫防御机能[57]。
1.2 肠黏膜屏障的免疫机能
肠黏膜免疫系统(mucosal immune system)是由肠上皮细胞及其分泌物、黏膜相关淋巴组织和栖息菌群构成,是机体抵御肠腔内病原微生物入侵的第一道防线。
肠黏膜免疫系统通过滤泡相关上皮(follicular-associated epithelium,FAE)和树突状细胞(dendritic cell,DC)对抗原不断地进行识别,防止病原微生物入侵[54]。正常情况下,肠固有层(lamina propria)中存在的大量效应性和记忆性淋巴细胞对无害抗原保持低应答或者无应答状态。病理情况下,病原微生物入侵肠固有层,活化DC等抗原提呈细胞,刺激免疫细胞和感染组织分泌多种细胞因子,发挥非特异性免疫效应,诱导急性期反应。
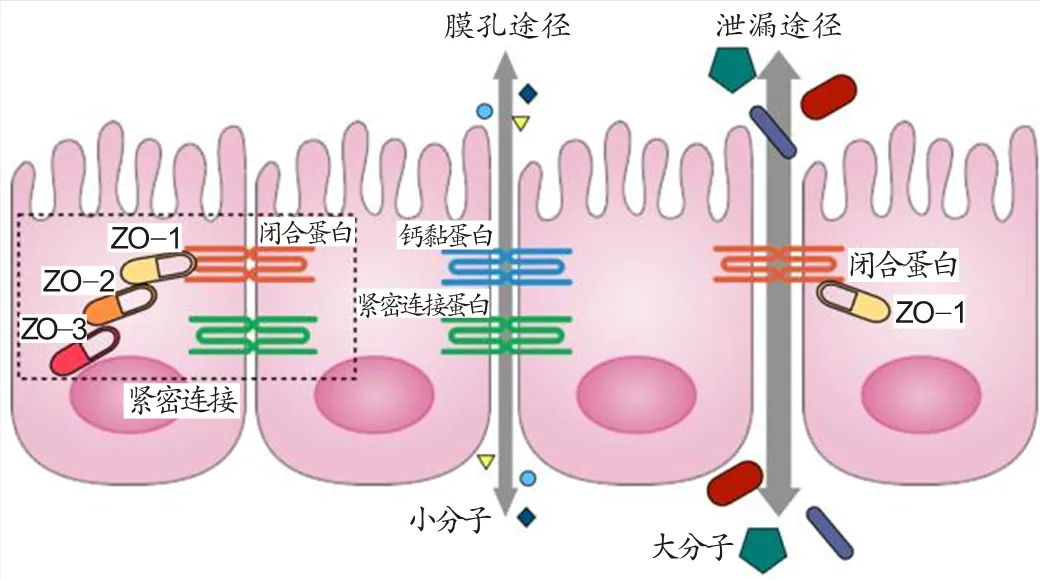
图2 肠上皮细胞间的紧密连接
Figure2. The Intestinal Epithelium and Tight Junction
肠上皮细胞中的模式识别受体(pattern recognition receptor,PRR)可识别病原微生物的病原相关分子模式(pathogen-associated molecular patterns,PAMP),区分外来的病原微生物和宿主共生菌[45]。当机体检测到病原相关分子模式,模式识别受体与病原相关分子模式相互作用可迅速激活效应细胞,促进对外来病原微生物的清除。在肠黏膜免疫系统中的模式识别受体主要为肠上皮细胞表面的Toll样受体(Toll-like receptors)[68]和胞浆中的Nod样受体(Nod-like receptors)。模式识别受体可使机体在肠黏膜屏障发生广泛的感染之前,快速检测识别病原微生物[12,15,34,84,88],对肠黏膜屏障的免疫监视和局部稳态维持起着重要的作用。
分泌型免疫球蛋白A(secretory immunoglobulin A,SIgA)也是肠黏膜屏障免疫机能的重要组成部分。免疫球蛋白A(immunoglobulin A,IgA)由固有层中的B细胞合成[60],与肠上皮细胞表面的分泌成分(secretory component,SC)结合为SIgA。SIgA与聚合免疫球蛋白受体(polymeric immunoglobulin receptor,pIgR)结合,经转胞吞作用转运至黏液层,促进黏液层清除病原微生物的能力,防止病原微生物的入侵[33, 39]。Sait L C等[71]通过敲除SIgA受体的小鼠模型发现,由于SIgA受体敲除小鼠肠道产生的IgA无法转运至肠腔,肠系膜淋巴结培养的细菌数量上升,血清型IgA和免疫球蛋白G(Immunoglobulin G,IgG)水平升高,表现出肠黏膜屏障功能受损,说明SIgA能够限制肠道细菌易位,降低肠道细菌入侵引起的全身性免疫反应。
2 运动对肠黏膜免疫的影响

多项研究证明,运动过程中机体的血液重新分配,影响肠黏膜屏障免疫稳态。由于肠腔内有大量厌氧菌群,固有层中存在丰富的血管和充沛的血液供应,肠黏膜从隐窝至绒毛尖端存在急剧变化的氧梯度,形成生理性低氧(physiological hypoxia)[25];运动可引起肠黏膜血流灌注减少和代谢变化,导致肠道氧分压(partial pressure of oxygen,PO2)下降。通过多普勒超声监测到,中等强度运动导致肠道血流量下降43%[67];采用黏膜二氧化碳张力计(Tonometry)测定运动员功率自行车运动时胃肠道二氧化碳分压(partial pressure of carbon dioxide,PCO2)发现,运动员胃肠道中PCO2随着运动强度的增大显著升高,在进行最大强度运动时均出现胃-动脉和空肠-动脉的PCO2梯度增加、胃和空肠缺血的现象[86]。
本文将从中小强度和大强度两个方面阐述运动对肠黏膜屏障免疫稳态的影响。
2.1 中小强度运动对肠黏膜屏障免疫稳态的影响
中小强度运动可适度增强肠黏膜屏障的免疫机能,抵御病原微生物入侵。在动物实验中,评价肠黏膜屏障功能主要通过检测肠组织形态结构、SIgA和肠免疫机能相关的细胞因子水平。研究发现,中等强度跑台运动使小鼠肠黏膜增厚,而黏液素分泌未受影响[62]。8周龄雄性Balb/c小鼠进行18周中等强度游泳运动后,与安静对照小鼠相比,同时都进行5.5 h一次性大强度游泳运动后,中等强度训练小鼠小肠近端SIgA和pIgR水平较高,小肠远端SIgA和浆细胞IgA+水平较高,TNF-α和IFN-γ mRNA表达较低[30]。Campos-Rodriguez R等[10]通过小鼠伤寒模型发现,中等强度游泳运动通过增加肠道SIgA水平,减少侵入的伤寒杆菌,降低细菌感染的炎症病灶数量。中等强度游泳运动还能够升高小鼠十二指肠和远端回肠中α链、J链及pIgR的mRNA水平和蛋白水平,并能降低肠道固有层中淋巴细胞的数量,增加小肠白细胞介素(interleukin,IL)-4、IL-6、IL-10、IL-12、TNF-α、TGF-β的mRNA和蛋白水平[97]。此外,研究还发现,中等强度游泳运动对大鼠肠道屏障紧密连接相关基因表达的影响有区域性差异,如显著上调十二指肠中表达,并下调结肠中这些基因的表达[85]。与此同时,小鼠炎症性肠病模型的中小强度运动干预研究中,存在一些相悖的结论。Saxena A等[72]发现,为期4周的中等强度跑台运动干预降低了小鼠结肠中促炎细胞因子IL-6、TNF-α和IL-1β水平,减轻了肠道炎症反应,同时上调趋化因子CCL6的mRNA表达,促进DC和巨噬细胞向炎症部位的迁移。但是,Cook M D等[17]通过对结肠炎小鼠进行6周中等强度跑台运动研究发现,运动后结肠IL-1β、IL-6、IL-10、IL-17、TNF-α以及CXCL1 mRNA表达水平升高,表明跑台运动加重了肠道炎症反应。这种差异可能与建模手段、运动干预剂量或者小鼠品系等相关。
由于肠道炎症反应的发生发展与肠神经活动及心理应激密切相关[3,11],自主运动和强迫运动干预可能起到截然不同的效果。在Packer N等[64]研究中,为期4个月自由转轮运动降低小鼠肠淋巴细胞的TNF-α表达水平,保护肠黏膜屏障功能,同样,Cook M D等[17]发现,30天自由转轮运动干预,下调结肠中IL-1β、IL-6、IL-10、IL-17、TNF-α以及CXCL1 mRNA表达水平。Allen J等[4]将运动或安静小鼠的肠道菌群分别移植入无菌(germ-free,GF)小鼠模型,结果显示,运动组和安静组小鼠的肠道菌群存在较大差异,6周自由转轮运动增加肠道菌群丰度(community richness),并在将运动训练或安静小鼠的肠道菌群分别移植到无菌小鼠后,进一步诱导小鼠急性结肠炎,结果显示,接受运动组小鼠菌群的受体小鼠结肠中炎性细胞浸润程度以及IL-1β、IL-23、IDO1和TGF-β的mRNA水平更低,即运动引起肠道菌群的改变可减轻肠道炎症反应。
连蛋白(zonulin)是可调节紧密连接、可逆调控肠通透性的蛋白,在病原微生物的刺激下,zonulin在肠腔中与受体结合发生信号转导,使肠道紧密连接开放,肠通透性增加[21,22]。乳果糖(lactulose)是一种异构化乳糖,口服后在肠道内几乎不被吸收,仅可能通过紧密连接进入血液循环;鼠李糖(rhamnose)是单糖,可直接通过肠上皮细胞被吸收。乳果糖和鼠李糖溶液可作为口服探针联合使用[65]。肠脂肪酸结合蛋白(Intestinal fatty acid binding protein,I-FABP)是参与长链脂肪酸从肠腔吸收和转运的蛋白,存在于肠上皮细胞中。当肠黏膜屏障损伤时,I-FABP在小肠绒毛中表达并迅速通过损伤的细胞膜,进入血液,在血浆中含量迅速升高。因此,可将I-FABP作为肠黏膜屏障损伤早期的标志物[27]。在人体实验中,通常根据血浆或尿液中zonulin含量、乳果糖/鼠李糖(L/R)比值或I-FABP含量评估肠道的通透性。研究发现,长期中小强度训练可缓解一次大强度运动导致的肠黏膜屏障稳态变化及炎症反应。一次90 min的大强度递增负荷自行车运动后,运动员粪便中zonulin表达水平低于未训练受试者,说明规律中小强度训练通过调节zonulin表达,缓解大强度运动导致的肠道通透性增加[48,49]。研究发现,中等强度运动后血浆L/R比值无明显变化,血浆I-FABP2未升高,证明中等强度运动不会使肠黏膜屏障通透性升高[38]。
因此,目前的研究普遍认为,中小强度运动通过紧密连接蛋白改善肠黏膜屏障通透性,同时改变肠道菌群的组成,并通过上调SIgA、下调促炎细胞因子等维持肠黏膜免疫的稳态(图3)。
2.2 大强度运动对肠黏膜免疫稳态的影响


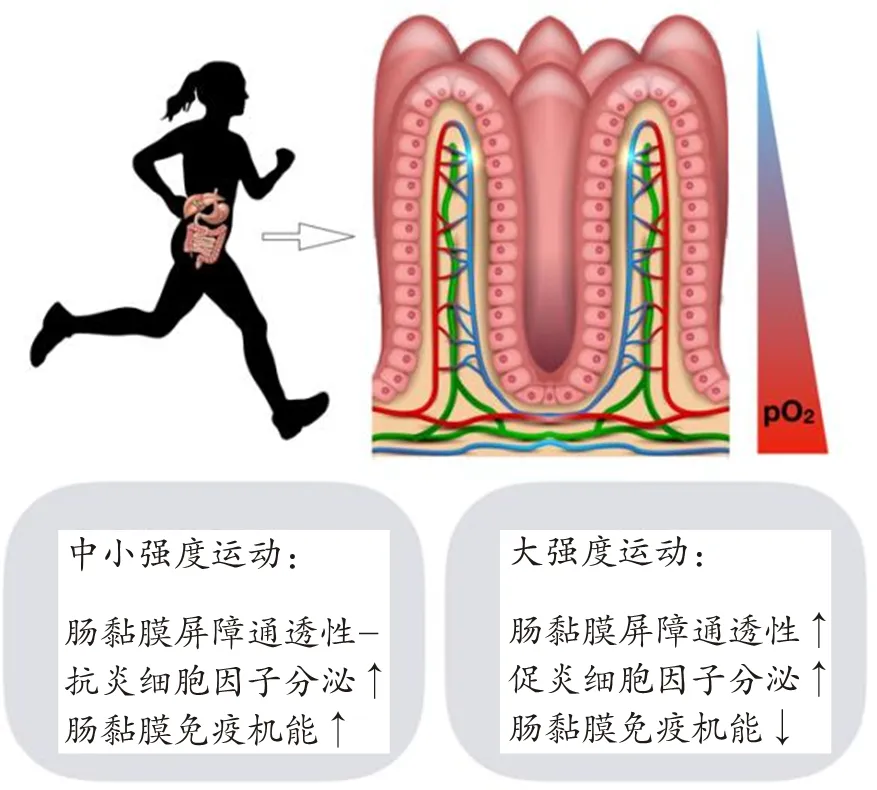
图3 运动对肠黏膜屏障免疫稳态的影响
Figure3. Effect of Exercise on Intestinal Barrier and Mucosal Immunity Homeostasis
由以上的研究报道可以看出,大强度运动致肠黏膜缺血、缺氧程度加重,引起肠黏膜屏障功能受损,促进促炎细胞因子分泌和肠道炎症反应,导致机体运动免疫机能失衡(图3)。
2.3 HIF在运动对肠黏膜免疫稳态影响中的作用机制
HIF是在低氧环境中调节细胞内氧稳态的关键转录因子[37,41,73],转录介导缺氧相关信号途径。HIF蛋白主要由α和β亚基构成,β亚基在组织中稳定表达,不受氧分压变化的影响[74],α亚基在常氧环境中经脯氨酰羟化酶(prolyl hydroxylase, PHD)[41]羟化其脯氨酸残基,进而经泛素/蛋白酶体途径降解。当氧分压降低时,PHD活性被抑制,HIF-1α羟基化修饰水平降低,HIF-1α和HIF-1β结合,进入胞核,与低氧反应元件(hypoxia response elements,HRE)结合,启动一系列靶基因的转录调控[79]。HIF在肠黏膜中表达,在常氧条件下可被激活,HIF-1α、HIF-2α和HIF-3α3种主要的HIF亚型均存在于肠中[55],在组织缺氧之前能够快速启动炎症反应。
正常情况下,肠黏膜免疫系统时刻接受大量微生物性抗原及食物抗原等的刺激,持续发生免疫应答和免疫耐受等免疫反应,呈现低度生理性炎症(physiological inflammation)[24]。运动引起肠道局部缺血,导致运动中腹痛、腹泻和胃肠道出血的发生,其发生机制可能与HIF密切相关。当肠黏膜受损,局部炎症反应加重,一方面,炎症部位代谢增强,需要合成酶与细胞因子,导致氧需求增加;另一方面,炎症部位血管功能障碍、血液灌注减少,不能维持免疫细胞浸润的黏膜组织充足的氧供应,使肠黏膜屏障持续处于低氧水平,活化的中性粒细胞在呼吸爆发过程中消耗更多氧[9],可能使缺氧影响到包括肠黏膜下层等更大范围的组织。
HIF信号途径对肠黏膜屏障结构完整性有重要作用。HIF-1α可通过诱导MUC3和肠三叶因子(intestinal trefoil factor)等肠道屏障保护基因的转录[28,52],形成保护性黏液层。在Karhausen J等[42]的研究中,肠黏膜Hif1a突变,引起小鼠死亡率上升、体重减轻和结肠长度降低。敲除肠上皮细胞的HIF可显著降低紧密连接蛋白claudin-1[70]和Defb1等一系列抗菌肽基因[44]的表达水平,引起杯状细胞的结构异常,改变杯状细胞分泌的黏液素表达[99]。
同时,HIF也参与免疫细胞的激活和细胞因子的分泌,调控肠黏膜免疫稳态。在Campbell E L等[9]的研究中,稳定结肠炎小鼠肠黏膜中HIF-1α的表达,可防止细菌与肠上皮接触,抑制肠黏膜屏障通透性增加,降低IL-1β、IFN-γ和IL-12的分泌,促进IL-6和IL-10的分泌,有效缓解炎症反应。最近的研究表明,小鼠B细胞特异性敲除Hif1a可降低B细胞分泌抗炎细胞因子IL-10,导致炎症反应加重[59]。
由此可见,运动导致机体血液重新分配,内脏血液灌流减少,肠道中缺氧程度加剧,诱导HIF-1α稳定表达,在这一过程中,HIF-1α对肠黏膜屏障有调节作用。中小强度运动中,HIF-1α可能通过调节肠黏膜屏障紧密连接,避免肠黏膜屏障通透性增加;而大强度运动导致肠黏膜屏障结构受损后,HIF-1α可能通过参与免疫细胞的激活,启动炎症反应相关通路,调节细胞因子的分泌,缓解肠黏膜屏障通透性增加,减轻炎症反应。
3 小结与展望
肠黏膜免疫系统是全身免疫系统的重要组成部分,肠黏膜屏障直接与外界抗原接触,是机体抵抗病原微生物入侵机体的主要途径。随着运动强度增加,内脏血流量急剧下降,肠黏膜缺氧程度加剧。大强度运动可能造成肠黏膜免疫稳态被打破,肠道屏障通透性增加,病原微生物入侵,启动炎症反应;中小强度运动对肠道屏障通透性和肠黏膜免疫稳态可能有改善作用,这些现象与运动过程中HIF-1α表达的上调有着密切联系。中小强度运动中,肠道HIF稳定表达可改善肠道屏障通透性;大强度运动后,肠道HIF稳定表达可避免肠道屏障组织结构与黏膜免疫功能的进一步破坏。
然而,在不同强度运动中,HIF-1α对肠黏膜屏障的具体作用机制仍不明确,在今后的研究中,需要通过合适的运动模型,阐明不同强度运动与肠道屏障中HIF-1α上调幅度的关系,运动过程中HIF介导的信号途径对肠黏膜屏障代谢的影响,运动后HIF-1α对肠黏膜免疫稳态的维持和保护作用是否具有时效性和适应性等问题。这些新的研究成果将为合理的运动训练保护和改善肠黏膜屏障免疫功能的新思路和方法提供强有力的理论与实验依据,为运动免疫研究领域提供重要的研究方向。
[1] 刘霞, 金其贯, 金爱娜. 低氧训练对大鼠小肠黏膜屏障功能的影响及其机制 [J].中国运动医学杂志, 2017, 36(4): 312-319.
[2] 罗贝贝, 陈佩杰. 运动应激与肠道黏膜屏障 [J]. 中国运动医学杂志, 2013, 32(5): 462-465.
[3] 张磊, 宋军, 侯晓华. 脑-肠轴失调在肠易激综合征发病中作用的研究进展 [J]. 胃肠病学, 2014, 19(11): 688-691.
[4] ALLEN J, MAILING L, COHRS J,Exercise training-induced modification of the gut microbiota persists after microbiota colonization and attenuates the response to chemically-induced colitis in gnotobiotic mice [J]. Gut Microbes, 2017:1-16.
[5] ANDERSON J M, VAN ITALLIE C M. Physiology and function of the tight junction [J]. Cold Spring Harbor Perspectives Biol, 2009, 1(2): a002584.
[6] BARBERIO M, ELMER D, LAIRD R,. Systemic LPS and inflammatory response during consecutive days of exercise in heat [J]. Int J Sports Med, 2015, 36(3): 262-270.
[7] BELLINGER A M, REIKEN S, DURA M,Remodeling of ryanodine receptor complex causes “leaky” channels: a molecular mechanism for decreased exercise capacity [J]. Proceed National Academy Sci, 2008, 105(6): 2198-2202.
[8] BERGMANN K R, LIU S X, TIAN R,Bifidobacteria stabilize claudins at tight junctions and prevent intestinal barrier dysfunction in mouse necrotizing enterocolitis [J]. Am J Pathol, 2013, 182(5): 1595-1606.
[9] CAMPBELL E L, BRUYNINCKX W J, KELLY C J,Transmigrating neutrophils shape the mucosal microenvironment through localized oxygen depletion to influence resolution of inflammation [J]. Imm, 2014, 40(1): 66-77.
[10] CAMPOS-RODRIGUEZ R, GODINEZ-VICTORIA M, ARCIN-IEGA-MARTÍNEZ I,. Protective effect of moderate exercise for BALB/c mice with salmonella typhimurium infection [J]. Int J Sports Med, 2016, 37(1): 63-70.
[11] CARABOTTI M, SCIROCCO A, MASELLI M A,The gut-brain axis: interactions between enteric microbiota, central and enteric nervous systems [J]. Ann Gastroenterology: Publicat Hellenic Soc Gastroenterol, 2015, 28(2): 203-209.
[12] CARIO E, GERKEN G, PODOLSKY D K. Toll-like receptor 2 enhances ZO-1-associated intestinal epithelial barrier integrity via protein kinase C [J]. Gastroenterol, 2004, 127(1): 224-238.
[13] CASEY E, MISTRY D J, MACKNIGHT J M. Training room management of medical conditions: sports gastroenterology [J]. Clin Sports Med, 2005, 24(3): 525-540.
[14] CECHELLA J L, LEITE M R, DOBRACHINSKI F,Moderate swimming exercise and caffeine supplementation reduce the levels of inflammatory cytokines without causing oxidative stress in tissues of middle-aged rats [J]. Amino Acids, 2014, 46(5): 1187-1195.
[15] CHAMAILLARD M, GIRARDIN S E, VIALA J,Nods, Nalps and Naip: Intracellular regulators of bacterial‐induced inflammation [J]. Cell Microbiol, 2003, 5(9): 581-592.
[16] CLEVERS H C, BEVINS C L. Paneth cells: maestros of the small intestinal crypts [J]. Ann Rev Physiol, 2013, 75(1): 289-311.
[17] COOK M D, MARTIN S A, WILLIAMS C,Forced treadmill exercise training exacerbates inflammation and causes mortality while voluntary wheel training is protective in a mouse model of colitis [J]. Brain Behavior Imm, 2013, 33(5): 46-56.
[18] DAVISON G, MARCHBANK T, MARCH D S,Zinc carnosine works with bovine colostrum in truncating heavy exercise–induced increase in gut permeability in healthy volunteers [J]. Am J Clin Nut, 2016, 104(2): 526-536.
[19] DUERKOP B A, VAISHNAVA S, HOOPER L V. Immune responses to the microbiota at the intestinal mucosal surface [J]. Imm, 2009, 31(3): 368-376.
[20] ELTZSCHIG H K, BRATTON D L, COLGAN S P. Targeting hypoxia signalling for the treatment of ischaemic and inflammat-ory diseases [J]. Nature Rev Drug Discovery, 2014, 13(11): 852.
[21] FASANO A. Intestinal permeability and its regulation by zonulin: diagnostic and therapeutic implications [J]. Clin Gastroenterol Hepatol, 2012, 10(10): 1096-1100.
[22] FASANO A. Zonulin and its regulation of intestinal barrier function: the biological door to inflammation, autoimmunity, and cancer [J]. Physiol Rev, 2011, 91(1): 151-175.
[23] FASANO A, SHEA-DONOHUE T. Mechanisms of disease: the role of intestinal barrier function in the pathogenesis of gastrointestinal autoimmune diseases [J]. Nature Clini Practice Gastroenterol Hepatol, 2005, 2(9): 416-422.
[24] FIOCCHI C. What is “physiological” intestinal inflammation and how does it differ from “pathological” inflammation? [J]. Inflam-mat Bowel Diseases, 2008, 14(S2): S77-S78.
[25] FISHER E M, KHAN M, SALISBURY R,Noninvasive monitoring of small intestinal oxygen in a rat model of chronic mesenteric ischemia [J]. Cell Biochem Biophysics, 2013, 67(2): 451-459.
[26] FOGOROS R N. Gastrointestinal disturbances in runners [J]. JAMA, 1980, 243(17): 1743-1744.
[27] FUNAOKA H, KANDA T, KAJIURA S,Development of a high-specificity sandwich ELISA system for the quantification of human intestinal fatty acid-binding protein (I-FABP) concentrati-ons [J]. Imm Investigat, 2011, 40(3): 223-242.
[28] FURUTA G T, TURNER J R, TAYLOR C T,. Hypoxia-inducible factor 1–dependent induction of intestinal trefoil factor protects barrier function during hypoxia [J]. J Exper Med, 2001, 193(9): 1027-1034.
[29] GARBER C E, BLISSMER B, DESCHENES M R,. Quantity and quality of exercise for developing and maintaining cardioresp-iratory, musculoskeletal, and neuromotor fitness in apparently healthy adults: guidance for prescribing exercise [J]. Med Sci Sports Exe, 2011, 43(7): 1334-1359.
[30] GODÍNEZ-VICTORIA M, DRAGO-SERRANO M E, REYNA-GARFIAS H,. Effects on secretory IgA levels in small intestine of mice that underwent moderate exercise training followed by a bout of strenuous swimming exercise [J]. Brain Behavior Imm, 2012, 26(8): 1300-1309.
[31] GOMES J, FREITAS J, GRASSIOLLI S. Effects of physical exercise on the intestinal mucosa of rats submitted to a hypothala-mic obesity condition [J]. Anatomical Rec, 2016, 299(10): 1389-1396.
[32] GUTEKUNST K, KRÜGER K, AUGUST C,. Acute exercises induce disorders of the gastrointestinal integrity in a murine model [J]. Eur J Appl Physiol, 2014, 114(3): 609-617.
[33] HARRIMAN G R, BOGUE M, ROGERS P,. Targeted deletion of the IgA constant region in mice leads to IgA deficiency with alterations in expression of other Ig isotypes [J]. J Imm, 1999, 162(5): 2521-2529.
[34] HAYASHI F, SMITH K D, OZINSKY A,The innate immune response to bacterial flagellin is mediated by Toll-like receptor 5 [J]. Nature, 2001, 410(6832): 1099-1103.
[35] HOLLAND A M, HYATT H W, SMUDER A J,. Influence of endurance exercise training on antioxidant enzymes, tight junction proteins, and inflammatory markers in the rat ileum [J]. BMC Res Notes, 2015, 8(1): 514-522.
[36] HOOPER L V, MACPHERSON A J. Immune adaptations that maintain homeostasis with the intestinal microbiota [J]. Nature Rev Imm, 2010, 10(3): 159-169.
[37] IYER N V, KOTCH L E, AGANI F,. Cellular and developmental control of O2 homeostasis by hypoxia-inducible factor 1α [J]. Genes Develop, 1998, 12(2): 149-162.
[38] JANSSENDUIJGHUIJSEN L M, MENSINK M, LENAERTS K,. The effect of endurance exercise on intestinal integrity in well-trained healthy men [J]. Physiol Reports, 2016, 4(20): e12994.
[39] JOHANSEN F-E, PEKNA M, NORDERHAUG I N,. Absence of epithelial immunoglobulin a transport, with increased mucosal leakiness, in polymeric immunoglobulin receptor/ secretory component–deficient mice [J]. J Exper Med, 1999, 190 (7): 915-922.
[40] JOHANSSON M E, AMBORT D, PELASEYED T,. Composition and functional role of the mucus layers in the intestine [J]. Cell Molecular Life Sci, 2011, 68(22): 3635-3641.
[41] KAELIN W G, RATCLIFFE P J. Oxygen sensing by metazoans: the central role of the HIF hydroxylase pathway [J]. Molecular Cell, 2008, 30(4): 393-402.
[42] KARHAUSEN J, FURUTA G T, TOMASZEWSKI J E,. Epithelial hypoxia-inducible factor-1 is protective in murine experimental colitis [J]. J Clin Investigat, 2004, 114(8): 1098-1106.
[43] KARHAUSEN J, HAASE V H, COLGAN S P. Inflammatory hypoxia: role of hypoxia-inducible factor [J]. Cell Cycle, 2005, 4(2): 255-257.
[44] KELLY C J, GLOVER L E, CAMPBELL E L,. Fundamental role for HIF-1α in constitutive expression of human β defensin-1 [J]. Mucosal Imm, 2013, 6(6): 1110-1118.
[45] KHOR B, GARDET A, XAVIER R J. Genetics and pathogenesis of inflammatory bowel disease [J]. Nature, 2011, 474(7351): 307-317.
[46] KIM Y S, HO S B. Intestinal goblet cells and mucins in health and disease: recent insights and progress [J]. Current Gastroenterol Reports, 2010, 12(5): 319-330.
[47] LAMOUREUX E V, GRANDY S A, LANGILLE M G. Moderate exercise has limited but distinguishable effects on the mouse microbiome [J]. MSystems, 2017, 2(4): e00006-17.
[48] LAMPRECHT M, BOGNER S, SCHIPPINGER G,Probiotic supplementation affects markers of intestinal barrier, oxidation, and inflammation in trained men; a randomized, double-blinded, placebo-controlled trial [J]. J Int Soc Sports Nut, 2012, 9(1): 45-57.
[49] LAMPRECHT M, BOGNER S, STEINBAUER K,Effects of zeolite supplementation on parameters of intestinal barrier integrity, inflammation, redoxbiology and performance in aerobic-ally trained subjects [J]. J Int Soc Sports Nut, 2015, 12(1): 40-50.
[50] LARA-PADILLA E, GODÍNEZ-VICTORIA M, DRAGO-SERRANO M E,Intermittent fasting modulates IgA levels in the small intestine under intense stress: A mouse model [J]. J Neuroimmunol, 2015, 285: 22-30.
[51] LIANG G H, WEBER C R. Molecular aspects of tight junction barrier function [J]. Current Opinion Pharmacol, 2014, 19(19C): 84-89.
[52] LOUIS N A, HAMILTON K E, CANNY G,. Selective induction of mucin‐3 by hypoxia in intestinal epithelia [J]. J Cellular Biochem, 2006, 99(6): 1616-1627.
[53] LUO B, XIANG D, NIEMAN D C,The effects of moderate exercise on chronic stress-induced intestinal barrier dysfunction and antimicrobial defense [J]. Brain Behavior Imm, 2014, 39 (4): 99-106.
[54] LUTGENDORFF F. Defending the barrier: Effects of probiotics on endogenous defense mechanisms [D]. Utrecht University, 2009.
[55] MA X, ZHANG H, XUE X,. Hypoxia-inducible factor 2α (HIF-2α) promotes colon cancer growth by potentiating Yes-associated protein 1 (YAP1) activity [J]. J Biol Chem, 2017, 292(41): 17046-17056.
[56] MARCHBANK T, DAVISON G, OAKES J R,. The nutriceutical bovine colostrum truncates the increase in gut permeability caused by heavy exercise in athletes [J]. Am J Physiol-Gastrointestinal Liver Physiol, 2011, 300(3): G477-G484.
[57] MÁRQUEZ M, DEL ÁLAMO C F G, GIRÓN-GONZÁLEZ J A. Gut epithelial barrier dysfunction in human immunodeficiency virus-hepatitis C virus coinfected patients: Influence on innate and acquired immunity [J]. World J Gastroenterol, 2016, 22(4): 1433-1448.
[58] MCGUCKIN M A, LINDÉN S K, SUTTON P,. Mucin dynamics and enteric pathogens [J]. Nature Rev Microbiol, 2011, 9(4): 265-278.
[59] MENG X, GRÖTSCH B, LUO Y,. Hypoxia-inducible factor-1α is a critical transcription factor for IL-10-producing B cells in autoimmune disease [J]. Nature Communicat, 2018, 9(1): 251-267.
[60] MORA J R, IWATA M, EKSTEEN B,Generation of gut-homing IgA-secreting B cells by intestinal dendritic cells [J]. Science, 2006, 314(5802): 1157-1160.
[61] MOREIRA N M, DE MORAES S M F, DALÁLIO M,. Moderate physical exercise protects myenteric metabolically more active neurons in mice infected with Trypanosoma cruzi [J]. Digestive Diseases Sci, 2014, 59(2): 307-314.
[62] MOREIRA N M, SANTOS F D N, TOLEDO M J D O,. Moderate physical exercise reduces parasitaemia and protects colonic myenteric neurons in mice infected with Trypanosoma cruzi [J]. Int J Exper Pathol, 2013, 94(6): 426-435.
[63] NIEMAN D, SIMPSON-WESTERBERG L B M, ARABATZIS K,Effects of long-endurance running on immune system parameters and lymphocyte function in experienced [J]. Int J Sports Med, 1989, 10(05): 317-323.
[64] PACKER N, HOFFMAN-GOETZ L. Exercise training reduces inflammatory mediators in the intestinal tract of healthy older adult mice [J]. Can J Aging/La Revue canadienne du vieillisse-ment, 2012, 31(2): 161-171.
[65] PALS K L, CHANG R-T, RYAN A J,. Effect of running intensity on intestinal permeability [J]. J Appl Physiol, 1997, 82(2): 571-576.
[66] PEDERSEN B, TVEDE N, KLARLUND K,Indometacin in vitro and in vivo abolishes post-exercise suppression of natural killer cell activity in peripheral blood [J]. Int J Sports Med, 1990, 11(2): 127-131.
[67] QAMAR M, READ A. Effects of exercise on mesenteric blood flow in man [J]. Gut, 1987, 28(5): 583-587.
[68] RAKOFF-NAHOUM S, PAGLINO J, ESLAMI-VARZANEH F,. Recognition of commensal microflora by toll-like receptors is required for intestinal homeostasis [J]. Cell, 2004, 118(2): 229-241.
[69] ROSA E F, FREYMÜLLER E, IHARA S S,. Damaging effects of intense repetitive treadmill running on murine intestinal musculature [J]. J Appl Physiol, 2008, 104(5): 1410-1417.
[70] SAEEDI B J, KAO D J, KITZENBERG D A,. HIF-dependent regulation of claudin-1 is central to intestinal epithelial tight junction integrity [J]. Molecular Bio Cell, 2015, 26(12): 2252-2262.
[71] SAIT L C, GALIC M, PRICE J D,Secretory antibodies reduce systemic antibody responses against the gastrointestinal commensal flora [J]. Int Imm, 2007, 19(3): 257-265.
[72] SAXENA A, LARSEN B, FLETCHER E,Effect of exercise on chemically-induced colitis in adiponectin deficient mice [J]. J Inflammat, 2012, 9(1): 30-41.
[73] SCHOFIELD C J, RATCLIFFE P J. Oxygen sensing by HIF hydroxylases [J]. Nature Rev Molecular Cell Biol, 2004, 5(5): 343-354.
[74] SCHULER B, LUNDBY C, GASSMANN M. HIF-1 and the adaptation of man to high altitude [J]. Schweizerische Zeitschrift Fur Sportmedizin Sporttraumatologie, 2005, 53(2): 82.
[75] SCHULTZ A, MENDONCA L S, AGUILA M B,. Swimming training beneficial effects in a mice model of nonalcoholic fatty liver disease [J]. Exper Tox Pathol, 2012, 64(4): 273-282.
[76] SCOMPARIN D X, GRASSIOLLI S, GOMES R M,Low-Intensity swimming training after weaning improves glucose and lipid homeostasis in MSG hypothalamic obese mice [J]. Endocrine Res, 2011, 36(2): 83-90.
[77] SEMENZA G L. Hypoxia-inducible factor 1 (HIF-1) pathway [J]. Science’s Stke Signal Transduction Knowledge Environment, 2007, 2007(407): cm8.
[78] SEMENZA G L. HIF-1, O2, and the 3 PHDs: how animal cells signal hypoxia to the nucleus [J]. Cell, 2001, 107(1): 1-3.
[79] SEMENZA G L. Hypoxia-inducible factor 1: master regulator of O 2 homeostasis [J]. Current Opinion Genetics Develop, 1998, 8(5): 588-594.
[80] SEMENZA G L. HIF-1: mediator of physiological and pathophysiological responses to hypoxia [J]. J Appl Physiol, 2000, 88(4): 1474-1480.
[81] SHANAHAN F. The host–microbe interface within the gut [J]. Best Practice Res Clin Gastroenterol, 2002, 16(6): 915-931.
[82] SHIBUYA T, KABURAGI T, NAGAI R,. The effects of moderate exercise on secretory IgA production in mice depends on dietary carbohydrate intake [J]. J Clin Biochem Nut, 2015, 57(1): 44-49.
[83] TADAISHI M, MIURA S, KAI Y,. Effect of exercise intensity and AICAR on isoform-specific expressions of murine skeletal muscle PGC-1α mRNA: a role of β2-adrenergic receptor activation [J]. Am J Physiol-Endocrinol Metabol, 2011, 300(2): E341-E349.
[84] TAKEDA K, KAISHO T, AKIRA S. Toll-like receptors [J]. Ann Rev Imm, 2003, 21(1): 335-376.
[85] TEERAPORNPUNTAKIT J, DORKKAM N, WONGDEE K,Endurance swimming stimulates transepithelial calcium transport and alters the expression of genes related to calcium absorption in the intestine of rats [J]. Am J Physiol-Endocrin Metabol, 2009, 296(4): E775-E786.
[86] TER STEEGE R W, GEELKERKEN R H, HUISMAN A B,. Abdominal symptoms during physical exercise and the role of gastrointestinal ischaemia: a study in 12 symptomatic athletes [J]. Br J Sports Med, 2012, 46(13): 931-935.
[87] UCHIDA M, OYANAGI E, KAWANISHI N,Exhaustive exercise increases the TNF-α production in response to flagellin via the upregulation of toll-like receptor 5 in the large intestine in mice [J]. Imm Letters, 2014, 158(1-2): 151-158.
[88] UEMATSU S, FUJIMOTO K, JANG M H,. Regulation of humoral and cellular gut immunity by lamina propria dendritic cells expressing Toll-like receptor 5 [J]. Nature Imm, 2008, 9(7): 769-776.
[89] ULLUWISHEWA D, ANDERSON R C, MCNABB W C,. Regulation of tight junction permeability by intestinal bacteria and dietary components [J]. JNut, 2011, 141(5): 769-776.
[90] VAISHNAVA S, YAMAMOTO M, SEVERSON K M,. The antibacterial lectin RegIIIγ promotes the spatial segregation of microbiota and host in the intestine [J]. Sci, 2011, 334(6053): 255-258.
[91] VAN DER SLUIS M, DE KONING B A, DE BRUIJN A C,Muc2-deficient mice spontaneously develop colitis, indicating that MUC2 is critical for colonic protection [J]. Gastroenterol, 2006, 131(1): 117-129.
[92] VAN ITALLIE C M, FANNING A S, BRIDGES A,. ZO-1 stabilizes the tight junction solute barrier through coupling to the perijunctional cytoskeleton [J]. Molecular Bio Cell, 2009, 20(17): 3930-3940.
[93] VAN ITALLIE C M, HOLMES J, BRIDGES A,The density of small tight junction pores varies among cell types and is increased by expression of claudin-2 [J]. J Cell Sci, 2008, 121(3): 298-305.
[94] VAN NIEUWENHOVEN M A, BROUNS F, BRUMMER R-J M. Gastrointestinal profile of symptomatic athletes at rest and during physical exercise [J]. Eur J Appli Physiol, 2004, 91(4): 429-434.
[95] VAN WIJCK K, LENAERTS K, VAN LOON L J,Exercise-induced splanchnic hypoperfusion results in gut dysfunction in healthy men [J]. PloS One, 2011, 6(7): e22366.
[96] VAN WIJCK K, PENNINGS B, VAN BIJNEN A A,. Dietary protein digestion and absorption are impaired during acute postexercise recovery in young men [J]. Am J Physiol-Regulatory, Integrative Comparative Physiol, 2013, 304(5): R356-R361.
[97] VILORIA M, LARA-PADILLA E, CAMPOS-RODRÍGUEZ R,Effect of moderate exercise on IgA levels and lymphocyte count in mouse intestine [J]. Imm Investigat, 2011, 40(6): 640-656.
[98] WATSON C J, HOARE C J, GARROD D R,Interferon-γ selectively increases epithelial permeability to large molecules by activating different populations of paracellular pores [J]. J Cell Sci, 2005, 118(22): 5221-5230.
[99] WHITNEY A K, SCHWISOW K D, BAYLESS A J,Role of Hypoxia-Inducible Factor (HIF) on colonic goblet cell differentiation and function during mucosal healing [J]. FASEB J, 2016, 30(1 Supplement): 305-310.
[100] ZUHL M, SCHNEIDER S, LANPHERE K,. Exercise regulation of intestinal tight junction proteins [J]. Bri J Sports Med, 2012, 48(12): 980-986.
Effect of Exercise on Intestinal Barrier Function and Mucosal Immunity Homeostasis
WU Die, CHEN Pei-jie, LUO Bei-bei
Shanghai University of Sport, Shanghai 200438, China.
Intestinal barrier and mucosal immunity homeostasis are the vital defense against pathogen invasion. Exercise induces tissue blood flow redistribution, leading to hypoxia in gastrointestinal tract. Under hypoxic condition, hypoxia-inducible transcription factor (HIF)-1α is phosphorylated and stabilized, regulating intestinal mucosal immune homeostasis. Related studies have revealed that moderate exercise maintains mucosal immunity homeostasis, while heavy exercise causes intestinal barrier dysfunction. Therefore, we briefly introduce the intestinal barrier structure and mucosal immune system; review the effects of different intensities of exercise on intestinal barrier function and mucosal immunity homeostasis; further discuss the possible protective mechanism of HIF-1α. This review provides a new insight for exercise training, intestinal barrier function and mucosal immunity related research areas.
G804.5
A
1000-0000(2018)06-0067-09
10.16469/j.css.201806008
2017-12-30;
2018-05-02
国家自然科学基金资助项目(31471135); 上海市教委晨光计划(16CG57); 上海市科委扬帆计划(17YF1418000);运动健身科技省部共建教育部重点实验室。
吴嵽,女,在读博士研究生,主要研究方向为运动免疫学, E-mail:dale_wo@163.com; 陈佩杰,男,教授,博士,博士研究生导师,主要研究方向为运动免疫学与青少年体质,E-mail: chenpeijie@sus.edu.cn; 罗贝贝,女,讲师,博士,硕士研究生导师,主要研究方向为运动对消化系统的影响,E-mail:lbb0220@126.com。
