Radix Ilicis Pubescentis total flavonoids combined with mobilization of bone marrow stem cells to protect against cerebral ischemia/reperfusion injury
2016-12-02MingsanMiaoLinGuoRuiqiLiXiaoMa
Ming-san Miao, Lin Guo, Rui-qi Li, Xiao Ma
Henan University of Traditional Chinese Medicine, Zhengzhou, Henan Province, China
RESEARCH
Radix Ilicis Pubescentis total flavonoids combined with mobilization of bone marrow stem cells to protect against cerebral ischemia/reperfusion injury
Ming-san Miao*, Lin Guo, Rui-qi Li, Xiao Ma
Henan University of Traditional Chinese Medicine, Zhengzhou, Henan Province, China
Graphical Abstract

orcid: 0000-0003-2681-4291 (Ming-san miao)
Previous studies have shown that Radix Ilicis Pubescentis total flavonoids have a neuroprotective effect, but it remains unclear whether Radix Ilicis Pubescentis total flavonoids have a synergistic effect with the recombinant human granulocyte colony stimulating factor-mobilized bone marrow stem cell transplantation on cerebral ischemia/reperfusion injury. Rat ischemia models were administered 0.3, 0.15 and 0.075 g/kg Radix Ilicis Pubescentis total flavonoids from 3 days before modeling to 2 days after injury. Results showed that Radix Ilicis Pubescentis total flavonoids could reduce pathological injury in rats with cerebral ischemia/reperfusion injury. The number of Nissl bodies increased, Bax protein expression decreased, Bcl-2 protein expression increased and the number of CD34-positive cells increased. Therefore, Radix Ilicis Pubescentis total flavonoids can improve the bone marrow stem cell mobilization effect, enhance the anti-apoptotic ability of nerve cells, and have a neuroprotective effect on cerebral ischemia/reperfusion injury in rats.
nerve regeneration; Radix Ilicis Pubescentis total flavonoids; bone marrow stem cells; mobilization; cerebral ischemia/reperfusion; Nissl bodies; Bax; Bcl-2; CD34; neurons; neural regeneration
Introduction
Bone marrow stem cells stimulate endogenous neural cell regeneration and inhibit apoptosis (Isele et al., 2007), repair and replace nerve cells after cerebral ischemia, promote or participate in angiogenesis in the ischemic region (Qiao and Cao, 2014). In conjunction with a mobilizing agent, they improve the survival rate of rats with cerebral ischemia by increasing the number of CD34-positive cells and the expression of Bcl-2 protein at the edges of the infarct, reducing the area of cerebral infarction (Yanqing et al., 2006). Studies have shown that bone marrow stem cell transplantation can improve the expression of Bcl-2 after cerebral ischemia reperfusion in rats and decrease the expression of Bax. Thus it reduces the cerebral ischemia/ reperfusion injury-induced apoptosis and improves the neurological status in rats with focal cerebral ischemia (Liu et al., 2005; Huang et al., 2006). Bone marrow stem cell mobilization has a protective effect on cerebral ischemia (Chen et al., 2012). Granulocyte colony stimulating factor is a strong bone marrow stem cell mobilizing agent and has been widely applied clinically. Granulocyte colony stimulating factor can mobilize bone mar-row stem cell release into the peripheral blood and help them reach the ischemic region. Here they protect injured neurons, improve the blood supply and promote the recovery of neurological function (Zhang and Lei, 2014).
Promotion of circulation and removing blood stasis method can improve microcirculation, encourage bone marrow stem cells to move from the bone marrow into the blood circulation and so exert the mobilization effect. These stem cells can easily traverse blood vessels and reach the ischemic region (Zhang and Zhang, 2014). Radix Ilicis Pubescentis, a commonly used Chinese herbal medicine that can remove blood stasis, alter cerebral blood flow, and assist in the prevention and treatment of cerebral vascular disease (Cai et al., 2001). Radix Ilicis Pubescentis total flavonoids (RIPTFs) is one of the effective ingredients extracted from the dried root of the holly, Ilicis Pubescentis (Liu et al., 2014). Our previous studies found that RIPTFs had an anti-ischemic effect (Zhang et al., 2012a, b). In this study, we used different concentrations of RIPTFs to treat rat models of cerebral ischemia/reperfusion injury after they received transplants of bone marrow stem cells that were mobilized by recombinant human granulocyte colony stimulating factor. We investigated the mechanism of RIPTFs on enhancing bone marrow stem cell mobilization.
Materials and Methods
Preparation of rat models of cerebral ischemia/ reperfusion
A total of 96 healthy, clean, male Sprague-Dawley rats aged 6-7 weeks and weighing 280-300 g were provided by the Hebei Provincial Experimental Animal Center of China (license SCXK (Ji) 2003-1-003). The rats were housed at 25 ± 3°C and humidity of 55 ± 10%, and were allowed free access to food and water. The experiment was approved by the Animal Ethics Committee of Henan University of Traditional Chinese Medicine in China. The 96 rats were equally and randomly divided into sham operation, model, mobilization, high-, moderate-, low-dose RIPTFs groups.
In all groups, except the sham operation group, rats were intraperitoneally anesthetized with 10% chloral hydrate 0.3 mL/100 g (Tianjin Municipality Kemi’ou Chemical Reagents Development Center, Tianjin, China) after fasting for 12 hours. The rats were fixed on the operation table in a supine position. A median incision was made on the neck. According to the method of Kitagawa et al. (1991), a nylon thread (approximately 0.30 mm in diameter) coated with 1 mm of silicone rubber on the head end was inserted into proximal end of the middle cerebral artery through the left common carotid artery to a depth of 18-20 mm to block the blood supply of the middle cerebral artery for 2 hours. The nylon thread was then taken out, and the left middle cerebral artery was occluded. Intraoperative temperature was maintained at 23-24°C. In the sham operation group, middle cerebral artery was exposed under anesthesia but had no embolism. Rats were scored at 24 hours after ischemia and reperfusion in accordance with the Longa’s method (Longa et al., 1989): 0 points, no nerve damage symptoms; 1 point, cannot fully extend right forepaw; 2 points, circling to the right; 3 points, dumping to the opposite side; 4 points, cannot walk by itself, loss of consciousness; 5 points, death. The rats scoring 1 point or above were considered successful ischemic models.
Drug intervention and bone marrow stem cell mobilization
Radix Ilicis Pubescentis extract was provided by the Laboratory of Analytical Chemistry, Henan University of Traditional Chinese Medicine, China. The RIPTF content was 52%. Radix Ilicis Pubescentis is the root of the plant Ilex pubescens Hook.et Arn, belongs to Ilex L. (Aquifoliaceae), and was found in Anhui Province of China, and identified by Professor Cheng-ming Dong from Henan University of Traditional Chinese Medicine, China. Radix Ilicis Pubescentis coarse powder was taken and soaked with 10 times the amount of 70% ethanol for 0.5 hour, followed by 2 reflux extractions, the first time for 1.5 hours and the second time for 1 hour. After filtration, the two filtrates were combined and subjected to vacuum ethanol recovery until no alcohol smell was detected. The Radix Ilicis Pubescentis extract obtained was put on an AB-8 macroporous resin column (Tianjin Haiguang Chemical Co., Ltd., Tianjin, China). The concentration of RIPTF in the sample liquid was 0.4 g/mL; the sample liquid pH was 4.5; the ratio of the crude drug to macroporous resin was 1:8; the ratio of the diameter to the height of the macroporous resin column was 1:15. Total saponins were eluted by 12 bed volume water and 10 bed volume 30% ethanol. The 30% ethanol eluent was collected, followed by vacuum ethanol recovery, and then dried at 50°C, to obtain the RIPTFs (Feng et al., 2012b). Ultraviolet spectrophotometry was used and rutin was used as the control. The content of RIPTFs was determined (Xu et al., 2011). RIPTFs were dissolved in 0.5% sodium carboxymethyl cellulose (Tianjin Fuchen Chemical Reagent Factory, Tianjin, China) to make 1.5, 0.75, and 0.375 mg/ mL suspensions.
In accordance with Miao’s method (Miao, 2003), rats were intragastrically administered 0.2 mL/10 g drug solutions, once a day, from 3 days before the modeling to the 2 days after the modeling. The rats in the sham operation, model and mobilization groups were given 0.5% sodium carboxymethyl cellulose. The rats in the high-, moderate- and low-dose RIPTFs groups received 0.3, 0.15 and 0.075 g/kg body weight of RIPTFs solutions, respectively.
From 3 days before the ischemic modeling, rats in the mobilization group, high-, moderate- and low-dose RIPTFs groups were also given normal saline-diluted recombinant human granulocyte colony stimulating factor (Approval No. GYZZ S19990049; Shandong Qilu Pharmaceutical Co., Ltd., Jinan, Shandong Province, China) by subcutaneous injection (10 µg/ kg daily), until the second day after injury for a total of 5 days. Rats in the model and sham operation groups were injected with an equal volume of normal saline. At 1 day after the ischemic injury, rats were intraperitoneally injected with 50 mg/ kg BrdU (Shanghai Hao Ran Biological Co., Ltd., Shanghai, China) twice, with an interval of 2 hours, so as to mark the dividing bone marrow stem cells.
A total of 35 rats died from poor drug tolerance and severe reaction to ischemic modeling during the experiment. The data from the 61 remaining rats were used in the analysis, including 16 in the sham operation group, 7 in the model group, 9 inthe mobilization group, 12 in the high-dose RIPTFs group, 8 in the moderate-dose RIPTFs group and 9 in the low-dose RIPTFs group.
Sample collection
In each group, the rats were sacrificed at 1 hour after administration on day 5. The brain was immediately separated on an ice plate. In accordance with a previous method (Paxinos and Watson, 2005), coronary sections were obtained at the optic chiasm and 4 mm posterior to the optic chiasm, fixed with 10% formaldehyde solution, and embedded with paraffin. Subsequently, samples were sliced into 5 µm thick serial sections, pasted on the polylysine-treated glass slides, and stored at room temperature.
Pathological observation of brain tissue
Sections of the left hemisphere underwent hematoxylin-eosin staining (Sun et al., 2014) and Nissl staining (Wu et al., 2015). Under a light microscope at 400× magnification (Olympus, Japan), nerve cells and Nissl bodies were observed in the brain. According to the results of hematoxylin-eosin staining, pathological change of nerve cells could be divided into four levels:“-” nerve cells, cytoplasm and nucleus were normal; “+”most of nerve cells, cytoplasm and nucleus were normal, but a few nerve cells were atrophied; “++” a small number of nerve cells were atrophied, and the majority of nerve cells, cytoplasm and nucleus were normal; “+++” most of nerve cells were atrophied, cytoplasm was apparently reduced, their nuclei and nucleoli were blurred and indistinct. According to the results of Nissl staining, pathological change of nerve cells can be divided into four levels: “-” abundant Nissl bodies in the cytoplasm; “+” less than 2/10 Nissl bodies had decreased or disappeared; “++” 3/10-6/10 Nissl bodies had decreased or disappeared; “+++” 7/10-10/10 Nissl bodies had decreased or disappeared.
Bcl-2, Bax, CD34 and BrdU immunoreactivities in the brain tissue as detected by immunohistochemical method
Sections of ischemic brain tissue in each group were attached to the glass slide coated with polylysine, dewaxed, hydrated, incubated with 3% H2O2for 10 minutes, and washed twice with PBS each for 5 minutes. Antigen was retrieved by exposure to microwave radiation in an oven. Following two washes with PBS each for 3 minutes, sections were blocked with normal goat serum at 37°C in a wet box for 20 minutes. Redundant liquid was drawn off. Sections were incubated with rabbit anti-rat BrdU monoclonal antibody (Boster, Wuhan, China), rabbit anti-CD34 polyclonal antibody (1:200) (Boster), rabbit anti-Bax polyclonal antibody (1:200) (Boster), and rabbit anti-Bcl-2 polyclonal antibody (1:200) (Boster) at 4 ° C overnight. After two washes with PBS each for 3 minutes, sections were incubated with biotinylated goat anti-rabbit IgG (1:100) (Boster) at 37°C for 20 minutes in a wet box, washed with PBS (3 minutes × 2), incubated with streptavidin biotin peroxidase complex at 37°C for 20 minutes in a wet box, washed with PBS (5 minutes × 4), and visualized with 3,3′-diaminobenzidine. Visualization was terminated with PBS. Subsequently, sections were counterstained with hematoxylin and mounted with a neutral resin. Bcl-2 and Bax expression and the number of CD34- and BrdU-positive cells were observed under 400× light microscope (Olympus). According to results of staining, the expression of different proteins in nerve cell membrane and cytoplasm was graded: “-”negative expression of Bax; “+” weakly positive expression of Bax; “++” positive expression of Bax; “+++” strongly positive expression of Bax.
Statistical analysis
Data were analyzed using SPSS 13.0 for windows medical statistical package (SPSS, Chicago, IL, USA). Ranked data were measured using a Ridit test. The difference between groups was compared using t-test. A value of P < 0.05 was considered statistically significant.
Results
RIPTFs improved the effects of bone marrow stem cell mobilization on pathological changes in rat brain tissue after cerebral ischemia/reperfusion injury
Hematoxylin-eosin staining showed that, compared with the sham operation group, nerve cell atrophy and cytoplasmic volume shrinkage were observed and nuclei and nucleoli were indistinct. Compared with the model group, nerve cells, cytoplasm, nucleus were normal in the mobilization group and high-dose RIPTFs group. In most nerve cells, cytoplasm and nucleus were normal in the moderate-dose RIPTFs group, but some nerve cells were atrophic, cytoplasm volume was clearly reduced, and nuclei and nucleoli were indistinct. In the lowdose RIPTFs group, most nerve cells were atrophic, cytoplasm had shrunk and the nuclei and nucleoli were indistinct, and some nerve cells, cytoplasm, nucleus were normal (Figure 1, Table 1).
RIPTFs improved the effects of bone marrow stem cell mobilization on Nissl body in rat brain tissue with cerebral ischemia/reperfusion injury
Nissl staining results show that compared with the sham operation group, neuronal cell edema and vacuolar degeneration were observed in the experimental groups. Nissl bodies disappeared in the model group. Compared with the model group, a large number of Nissl bodies were found in the cytoplasm of most neuronal cells, and Nissl bodies disappeared in a few cells in the mobilization, high- and moderate-dose RIPTFs groups. Nissl bodies disappeared in most neurons, and Nissl bodies could be seen in the cytoplasm of some cells (Figure 1, Table 2).
RIPTFs elevated the effect of bone marrow stem cell mobilization on Bax immunoreactivity in rat brain tissue after cerebral ischemia/reperfusion injury
Immunohistochemical staining showed that no Bax immunoreactivity was detected in the sham operation group. Compared with the sham operation group, Bax immunoreactivity did not obviously alter in nerve cells of rats from the mobilization group (P > 0.05), but Bax expression was weakly positive in the model group (P < 0.05). Compared with the model group, Bax immunoreactivity became weaker in the high-dose RIPTFs group(P < 0.01), but no significant changes in Bax immunoreactivity were detected in the moderate- and low-dose RIPTFs groups (P> 0.05; Figure 2, Table 3).
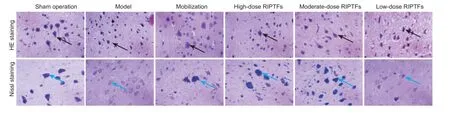
Figure 1 RIPTFs effect on pathological changes and Nissl bodies in rat brain tissue with cerebral ischemia/reperfusion injury after bone marrow stem cell mobilization (× 400).
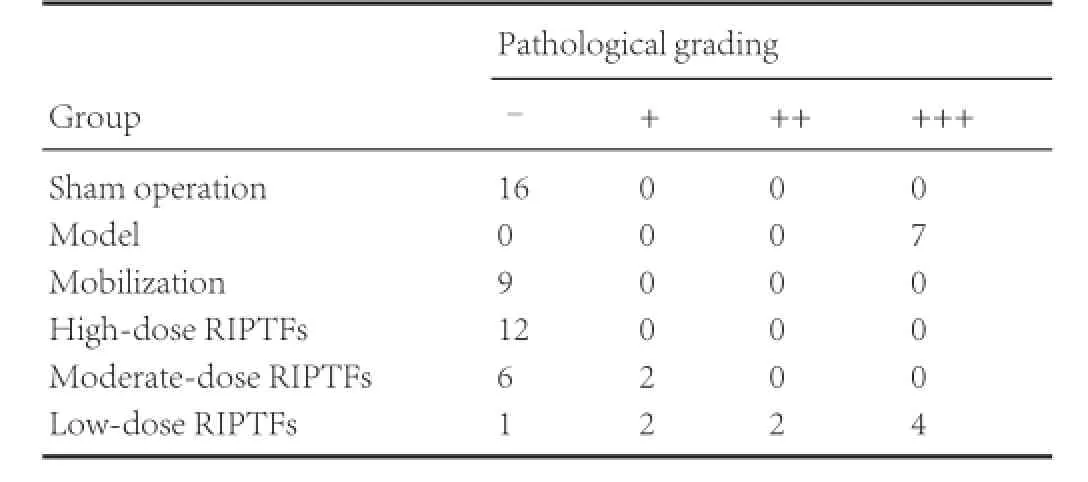
Table 1 RIPTFs improved the effect of bone marrow stem cell mobilization on pathological changes in rat brain tissue with cerebral ischemia/reperfusion injury
RIPTFs elevated the effect of bone marrow stem cell mobilization on Bcl-2 immunoreactivity in rat brain tissue after cerebral ischemia/reperfusion injury
Immunohistochemical staining showed that Bcl-2 was not detected in the sham operation and model groups. Compared with the sham operation group, Bcl-2 immunoreactivity was significantly greater in the mobilization, high- and moderate-dose RIPTFs groups (P < 0.01). No significant changes were detectable in Bcl-2 immunoreactivity in the low-dose RIPTFs group (P > 0.05). Compared with the model group, Bcl-2 immunoreactivity was significantly higher in the mobilization and high-dose RIPTFs groups (P < 0.05). No significant changes in Bcl-2 immunoreactivity were found in the moderate- and low-dose RIPTFs groups (P > 0.05; Figure 2, Table 4).
RIPTFs elevated the effect of bone marrow stem cell mobilization on CD34 immunoreactivity in rat braintissue after cerebral ischemia/reperfusion injury

Table 2 RIPTFs improved the effects of bone marrow stem cell mobilization on Nissl bodies in rat brain tissue after cerebral ischemia/ reperfusion injury
Immunohistochemical staining showed that, compared with the sham operation group, CD34 immunoreactivity was significantly greater in the mobilization, high- and moderate-dose RIPTFs groups (P < 0.01). No significant changes in CD34 immunoreactivity were detected in the ischemic region in the model and low-dose RIPTFs groups (P > 0.05). Compared with the model group, CD34 immunoreactivity in the ischemic region was significantly higher in the high-dose RIPTFs group (P < 0.05). No significant changes in CD34 immunoreactivity were found in the ischemic region of the mobilization, moderate- and low-dose RIPTFs groups (P > 0.05; Figure 2, Table 5).
RIPTFs elevated the effect of bone marrow stem cell mobilization on BrdU immunoreactivity in rat brain tissue after cerebral ischemia/reperfusion injury

Figure 2 RIPTFs elevated the effect of bone marrow stem cell mobilization on Bax, Bcl-2, CD34 and BrdU immunoreactivities in rat brain tissue after cerebral ischemia/ reperfusion injury (immunohistochemical staining, × 400).
Immunohistochemical staining showed that there was no BrdU immunoreactivity in the model group. Compared with the sham operation group, BrdU immunoreactivity was significantly greater in the high-dose RIPTFs group (P < 0.05). No significant change in BrdU immunoreactivity was detected in the mobilization, moderate- and low-dose RIPTFs groups (P > 0.05). BrdU immunoreactivity was significantly higher in the high-, moderate- and low-dose RIPTFs groups than in the model group (P < 0.01). BrdU immunoreactivity was significantly higher in the mobilization group than in the model group (P < 0.05; Figure 2, Table 6).
Discussion
Ischemic stroke is the most common cerebrovascular disease, and cerebral ischemia is frequently detected in the middle cerebral artery (Zhu et al., 2015). The methods of inducing animal models of focal cerebral ischemia include the thread embolism method, micro-embolism method, and chemical stimulus-induced thrombotic occlusion method (Chen et al., 2015). The thread embolism method is recognized as the ideal method for modeling of focal cerebral ischemia as there is no craniotomy, little trauma and good repeatability (Fan et al., 2014). We chose this model as it is similar to the clinical conditions. Nissl body is a morphological index of nerve cell viability (Zeng et al., 2015) as its function was strongly related to neuronal function. Therefore, it is often used to judge the pathological changes of nerve cells. Moreover, the changes in Nissl bodies can reflect nerve cell changes in the hippocampus. Nissl bodies are very sensitive to ischemia and hypoxia in nerve cells. When axons are damaged, Nissl bodies of nerve cells tend to dissolve or disappear (Zhong et al., 2011). This study demonstrated that the number of Nissl bodies in the cytoplasm of nerve cells in the hippocampus of the rats in the model group clearly diminished after cerebral ischemia/reperfusion injury. The decrease in the number of Nissl bodies was lessened by drug administration following ischemia. The use of the RIPTFs drug increased the number of Nissl bodies, promoted axonal regeneration and lengthened the nerve growth cone, indicating that RIPTFs protected nerve cells of rats against ischemia/reperfusion injury.
Nerve cell apoptosis is strongly correlated with cerebral ischemia and is regulated by apoptosis-related genes (Luo et al., 2005). Bcl-2 is an apoptosis-inhibiting gene, and Bax protein is a pro-apoptotic gene; both are considered the most important genes to suppress and promote apoptosis (Banjerdpongchai et al., 2011; Song et al., 2011). Our experimental results showed that RIPTFs suppressed neuronal apoptosis caused by cerebral ischemia by decreasing the expression of Bax and enhancing the expression of Bcl-2. CD34 cells can migrate to the ischemic region or injury site and participate in local angiogenesis (Mu et al., 2013). Immunohistochemical staining can be used to measure the microvessel density and area of the ischemic tissue using a CD34 antibody so as to determine neovascularization in the early stage (Zhu et al., 2011). This study confirmed that RIPTFs effectively improved capillary blood flow and oxygensupply and reduced the degree of damage to capillary endothelial cells in the hypoxic and ischemic brain. BrdU as an analogue of thymidine can compete with endogenous thymidine and integrate into the nucleus during the DNA synthesis of S phase of the cell cycle. BrdU as a marker of cell proliferation has been widely used in the field of neural stem cells (Seaberg and van der Kooy, 2002). This study verified that, after cerebral ischemia, BrdU expression was apparently stronger in the RIPTFs groups than that of the model group, and BrdU expression was higher in the mobilization group than in the model group, which suggested that RIPTFs and bone marrow mobilizing agent (recombinant human granulocyte colony stimulating factor) may have a synergistic effect, to help improve blood circulation in the ischemic region, and promote the reconstruction of neural circuits.
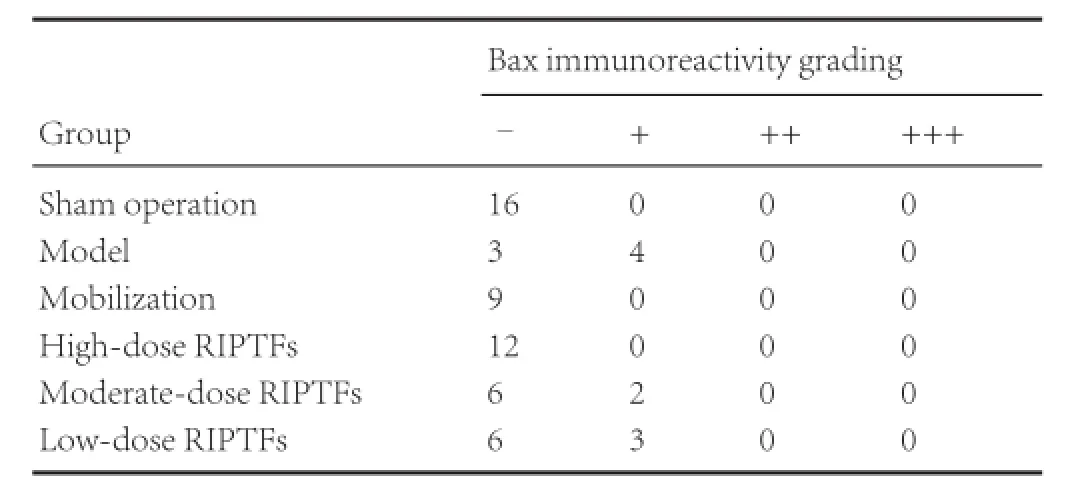
Table 3 RIPTFs elevated the effect of bone marrow stem cell mobilization on Bax immunoreactivity in rat brain tissue after cerebral ischemia/ reperfusion injury
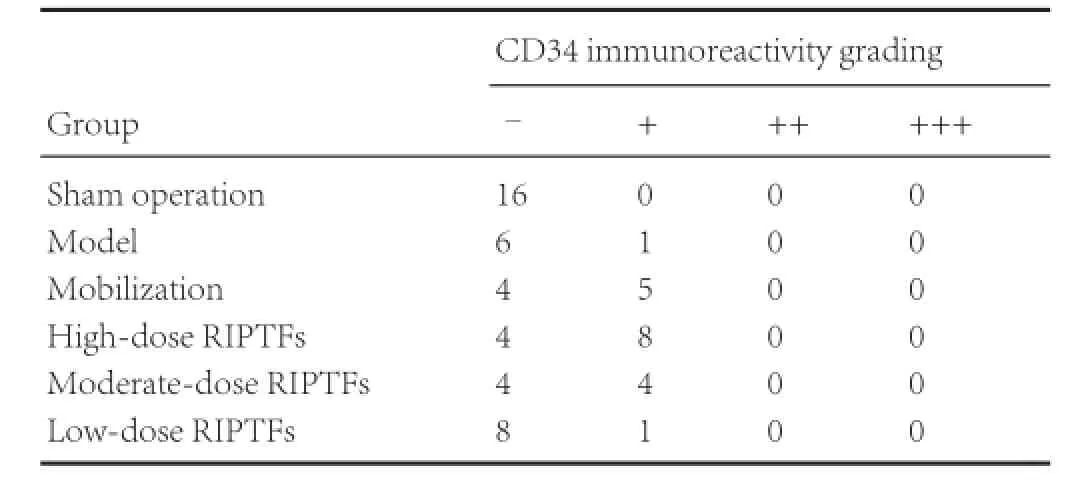
Table 5 RIPTFs elevated the effect of bone marrow stem cell mobilization on CD34 immunoreactivity in rat brain tissue following cerebral ischemia/ reperfusion injury
The present study verified that RIPTFs improved the mobilization of bone marrow stem cells, reduced the apoptosis of nerve cells in brain tissue, could prevent and repair damage from cere-bral ischemia, and had synergistic effect on bone marrow stem cell mobilizing agent in rat models of cerebral ischemia/reperfusion injury. However, a more comprehensive dose-effect relationship of RIPTFs should be investigated. Despite the simplicity of the mobilization of bone marrow stem cells and that immunological rejection of allogeneic transplantation can be avoided so it is an effective treatment for cerebral ischemia (Rüster et al., 2006; Dietz et al., 2007; Wang et al., 2007), its efficacy and safety need further evaluation. The honing of concordant remedies for the mobilization of bone marrow stem cells towards ischemic tissue, and the precise mechanism of the differentiation into functional cells should be clarified. New drugs to enhance the targeting and effectiveness of bone marrow stem cell mobilization should be developed. It would be beneficial to establish a complete technology platform for animal models, and explore the possibility of clinical application of the various technologies. Thus, bone marrow stem cell mobilization co-medication to improve cerebral ischemia/reperfusion strategy will become a new means ofcell transplantation therapy incorporating traditional medicine, hyperacute thrombolytic therapy and stem cell therapy.
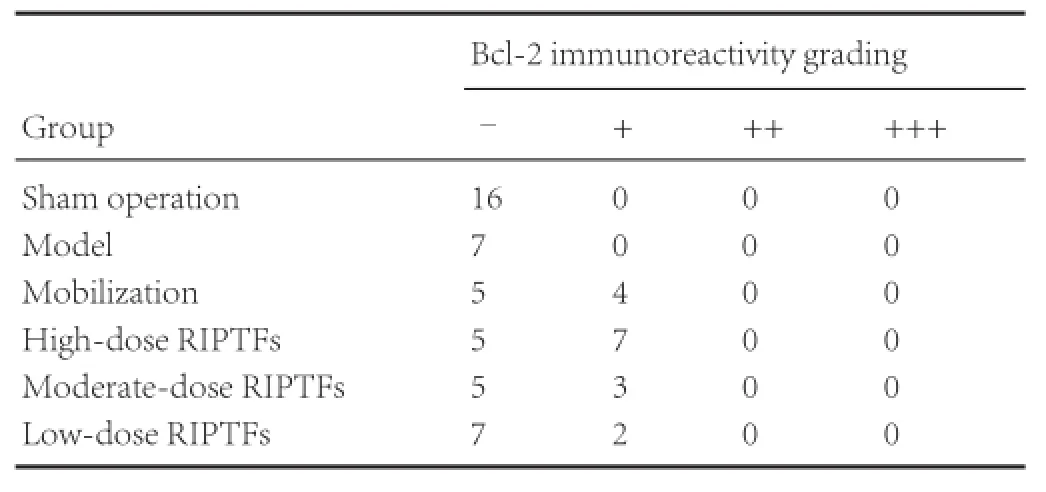
Table 4 RIPTFs elevated the effect of bone marrow stem cell mobilization on Bcl-2 immunoreactivity in rat brain tissue after cerebral ischemia/ reperfusion injury

Table 6 RIPTFs elevated the effect of bone marrow stem cell mobilization on BrdU immunoreactivity in rat brain tissue after cerebral ischemia/ reperfusion injury
Acknowledgments: We are very grateful to Professor Jian-guo Li from Henan University of Traditional Chinese Medicine, China for assistance in the implementation of pathology.
Author contributions: MSM designed this experiment and was responsible for the paper authorization. LG, RQL and XM performed experiments and collected the data. MSM and LG estimated experiments. LG wrote the manuscript. All authors approved the final version of the paper.
Conflicts of interest: None declared.
Plagiarism check: This paper was screened twice using Cross-Check to verify originality before publication.
Peer review: This paper was double-blinded and stringently reviewed by international expert reviewers.
Banjerdpongchai R, Punyati P, Nakrob A, Pompimon W, Kongtawelert P (2011) 4′-Hydroxycinnamaldehyde from Alpinia galanga (Linn.) induces human leukemic cell apoptosis via mitochondrial and endoplasmic reticulum stress pathways. Asian Pac J Cancer Prev 12:593-598.
Cai X, Liu ZQ, Zhu CC, Wang PX, Liu L (2001) Progress of chemical constituents, pharmacological action and clinical application of Ilex pubescens. Guangdong Yaoxue 11:4-6.
Chen HX, Li YH, Pei HT (2012) Stem cell mobilization in the treatment of ischemic disease. Zhongguo Jichu Kexue 14:2-8.
Chen XJ, Xiao ZY, Pan Q, Wang L, Hou Q, Lei YC, Wu SZ (2015) Modified establishment of Wistar rats ischemia-reperfusion injury model. Zhongguo Yixue Chuangxin 12:24-27.
Dietz AB, Padley DJ, Gastineau DA (2007) Infrastructure development for human cell therapy translation. Clin Pharmacol Ther 82:320-324.
Fan RJ, Luo YF, Sun YF (2014) A new thread embolism method to establish the rat model of focal cerebral ischemia reperfusion. Zhongguo Minzu Minjian Yiyao 23:26-27.
Feng SX, Miao MS, Miao JX, Xu P, Shao S, Hou CJ (2012) Simultaneous separation and purification technology of total flavonoids and total saponins from Llex pubescens by AB-8 macroporous resin. Zhongguo Shiyan Fangji Xue Zazhi 18:5-8.
Huang Y, Xu YM, Song B, Fang SY (2006) Effects of mesenchymal stem cell transplantation on neural cell apoptosis and expressions of Bcl-2, Bax in rat cerebral ischemia-reperfusion tissue. Zhengzhou Daxue Xuebao: Yixue Ban 41:862-864.
Isele NB, Lee HS, Landshamer S, Straube A, Padovan CS, Plesnila N, Culmsee C (2007) Bone marrow stromal cells mediate protection through stimulation of PI3-K/Akt and MAPK signaling in neurons. Neurochem Int 50:243-250.
Kitagawa K, Matsumoto M, Kuwabara K, Tagaya M, Ohtsuki T, Hata R, Ueda H, Handa N, Kimura K, Kamada T (1991) ‘Ischemic tolerance’phenomenon detected in various brain regions. Brain Res 561:203-211.
Liu YH, Song YC, Zhao LH, Shen X (2005) Effects of bone marrow stromal cells derived neural stem cells on the changes of apoptosis and correlative proteins in rats after focal cerebral ischemia and reperfusion. Zhionghua Qiguan Yizhi Zazhi 26:716-718.
Liu ZG, Li XL, Li SS, Liu L, Weng LD, Liu Q, Huang DH, Huang ZG (2014) Effects of common pharmaceutical excipients on moisture absorption of total flavonoids from Ilex pubescens. Zhongguo Shiyan Fangji Xue Zazhi 20:24-27.
Longa EZ, Weinstein PR, Carlson S, Cummins R (1989) Reversible middle cerebral artery occlusion without craniectomy in rats. Stroke 20:84-91.
Luo T, Xia Z, Ansley DM, Ouyang J, Granville DJ, Li Y, Xia ZY, Zhou QS, Liu XY (2005) Propofol dose-dependently reduces tumor necrosis factor-alpha-induced human umbilical vein endothelial cell apoptosis: effects on Bcl-2 and Bax expression and nitric oxide generation. Anesth Analg 100:1653-1659.
Miao MS (2003) Experimental Animal and Animal Experiment Technology. Beijing: China Press of Traditional Chinese Medicine.
Mu GP, Zeng YC, Chen PD, Luo WS, Ma XM, Xu SG, Liu XL, Liu WH (2013) Influence of conception and governor vessel acupuncture on vascular endothelial growth factor and CD34 protein expressions in rats with cerebral ischemia-reperfusion injury. Zhongguo Yiyao Daobao 10:18-22.
Paxinos G, Watson C (2005) The Rat Brain in Stereotaxic Coordinates. London: Academic Press.
Qiao HZ, Cao Y (2014) Research progress of acupuncture on bone marrow stemcell mobilization in cerebral infarction patients. Zhongyiyao Linchuang Zazhi 56:206-208.
Rüster B, Göttig S, Ludwig RJ, Bistrian R, Müller S, Seifried E, Gille J, Henschler R (2006) Mesenchymal stem cells display coordinated rolling and adhesion behavior on endothelial cells. Blood 108:3938-3944.
Seaberg RM, van der Kooy D (2002) Adult rodent neurogenic regions: the ventricular subependyma contains neural stem cells, but the dentate gyrus contains restricted progenitors. J Neurosci 22:1784-1793.
Song XD, Zhang JJ, Wang MR, Liu WB, Gu XB, Lv CJ (2011) Astaxanthin induces mitochondria-mediated apoptosis in rat hepatocellular carcinoma CBRH-7919 cells. Biol Pharm Bull 34:839-844.
Sun L, Wang L, Li Y, Zhang J, Fang L (2014) The protective effect and mechanism of astragaloside in cerebral ischemia reperfusion in rats. Zhongguo Linchuang Shenjing Kexue 22:43-49.
Wang DC, Liu JX, Zhang XF, Tian YS, Li JS (2007) Bone marrow stem cell mobilization in the treatment of cerebral ischemia injury. Zhongguo Zuzhi Gongcheng Yanjiu yu Linchuang Kangfu 11:9365-9368.
Wu YN, Zhang Y, Lin RH, Chen LD, Tao J (2015) Effects of electroacupuncture on learning and memory ability and RhoA expression in rats after cerebral ischemic-reperfusion. Zhongguo Kangfu Lilun yu Shijian 21:17-21.
Xu YH, Miao MS, Xu P, Feng SX (2011) Determination on content of total flavonoids in the effective fraction of llex pubescens hook. Zhongyi Xuebao 26:826-827.
Yanqing Z, Yu-Min L, Jian Q, Bao-Guo X, Chuan-Zhen L (2006) Fibronectin and neuroprotective effect of granulocyte colony-stimulating factor in focal cerebral ischemia. Brain Res 1098:161-169.
Zeng GF, Zhang ZY, Lu L, Xiao DQ, Zong SH, He JM (2015) Influence of ginger extract onbehavior function of Alzheimer’s disease rats. Zhongguo Laonian Xue Zazhi 356:160-161.
Zhang F, Zhang XL, Miao MS (2012a) Effect on total flavonoids of Radix Ilicis pubescentis in mice models of cerebral ischemia. Zhongyao Xinyao yu Linchuang Yaoli 23:409-412.
Zhang F, Zhang XL, Miao MS (2012b) Effect of flavonoids of Ilex pubescens on blood stasis with cerebral ischemia. Zhongguo Shiyan Fangji Xue Zazhi 18:187-191.
Zhang J, Zhang EH (2014) Mobilizing effects of salviol A and paeonol combination on bone marrow stem cells in cerebral ischemia injury of rats. Zhongguo Yiyuan Yaoxue Zazhi 34:90-94.
Zhang SS, Lei P (2014) Research progress of granulocyte colony-stimulating factor on cerebral protective effect in ischemic stroke. Zhongxiyi Jiehe Xinnao Xueguan Bing Zazhi:1557-1559.
Zhong ZD, Xu YZ, Zhong H, Yang L, Li Y, Wang HX (2011) Effect of nimotop vial on cerebral ischemia-reperfusion injury of MCAO rat in morphology. Sichuan Shengli Kexue Zazhi 33:57-60.
Zhu HB, Chen Y, Tan H, Ge JW (2011) Effects of Naotai recipe extracts on cerebral CD34 expression in rats with focal cerebral ischemia. Zhongyao Xinyao yu Linchuang Yaoli 22:141-144.
Zhu XQ, Han J, Hu J, Pang WS (2015) The protection and mechanism of Buyang huanwu tang on focal cerebral ischemia-reperfusion in rats. Zhongguo Minzu Minjian Yiyao:42-43.
Copyedited by Dawes EA, Pack M, Yu J, Qiu Y, Li CH, Song LP, Zhao M
10.4103/1673-5374.177736 http://www.nrronline.org/
How to cite this article: Miao MS, Guo L, Li RQ, Ma X (2016) Radix Ilicis Pubescentis total flavonoids combined with mobilization of bone marrow stem cells to protect against cerebral ischemia/reperfusion injury. Neural Regen Res 11(2):278-284.
Funding: This study was supported by a grant from the State “Major New Drug Creation” Science and Technology Major Project in China, No. 2009ZX09103-324; a grant from the Henan Provincial Science and Technology Innovation Team in University in China, No. 2012IRTSTHN011.
Accepted: 2015-07-08
*Correspondence to: Ming-san Miao, M.D., miaomingsan@126.com.
杂志排行
中国神经再生研究(英文版)的其它文章
- Tissue-type plasminogen activator is a modulator of the synaptic vesicle cycle
- Impaired consciousness caused by injury of the lower ascending reticular activating system: evaluation by diffusion tensor tractography
- Considering calcium-binding proteins in invertebrates: multi-functional proteins that shape neuronal growth
- Cardiovascular dysfunction following spinal cord injury
- Practical application of the neuroregenerative properties of ketamine: real world treatment experience
- Exergames: neuroplastic hypothesis about cognitive improvement and biological effects on physical function of institutionalized older persons
