Composite bioabsorbable vascular stents via 3D bio-printingand electrospinning for treating stenotic vessels
2015-05-08LiuYuanyuanXiangKeLiYuChenHaipingHuQingxi
Liu Yuanyuan Xiang Ke Li Yu Chen Haiping Hu Qingxi
(1Rapid Manufacturing Engineering Center, Shanghai University, Shanghai 200444, China)(2Shanghai Key Laboratory of Intelligent Manufacturing and Roboties, Shanghai University, Shanghai 200072, China)
Composite bioabsorbable vascular stents via 3D bio-printingand electrospinning for treating stenotic vessels
Liu Yuanyuan1,2Xiang Ke1Li Yu1Chen Haiping1Hu Qingxi1,2
(1Rapid Manufacturing Engineering Center, Shanghai University, Shanghai 200444, China)(2Shanghai Key Laboratory of Intelligent Manufacturing and Roboties, Shanghai University, Shanghai 200072, China)
A new type of vascular stent is designed for treating stenotic vessels. Aiming at overcoming the shortcomings of existing equipment and technology for preparing a bioabsorbable vascular stent (BVS), a new method which combines 3D bio-printing and electrospinning to prepare the composite bioabsorbable vascular stent (CBVS) is proposed. The inner layer of the CBVS can be obtained through 3D bio-printing using poly-p-dioxanone (PPDO). The thin nanofiber film that serves as the outer layer can be built through electrospinning using mixtures of chitosan-PVA (poly (vinyl alcohol)). Tests of mechanical properties show that the stent prepared through 3D bio-printing combined with electrospinning is better than that prepared through 3D bio-printing alone. Cells cultivated on the CBVS adhere and proliferate better due to the natural, biological chitosan in the outer layer. The proposed complex process and method can provide a good basis for preparing a controllable drug-carrying vascular stent. Overall, the CBVS can be a good candidate for treating stenotic vessels.
3D (three-dimensional) bio-printing; bioabsorbable vascular stent (BVS); electrospinning; cell proliferation; composite forming
Cardiovascular diseases (CVD) are one of the leading causes of death across the globe (accounting for 48% of all deaths in Europe alone)[1]. Intravascular interventions (PCI) with intravessel stenting are nowadays a golden standard in cardiology[2]. Metal stents have been widely used in clinics[3], but they are usually not biodegradable. The implantation will set a fixed diameter for blood vessels, which is not suitable for children’s vascular growth and may affect the further development of blood vessels[4-5]. In addition, the stenosis rate and the thrombosis rate using metal stents are also high[6]. Due to degradation, bioabsorbable vascular stents (BVS) can dissolve gradually, and, therefore, can avoid the occurrence of the above mentioned phenomenon[7]. Moreover, BVS is a novel approach for treating coronary lesions and it can provide transient vessel support and drug delivery to the vessel wall without the potential long-term limitations of conventional metallic drug-eluting stents, such as stent thrombosis and prevention of future surgical revascularization[8]. Therefore, BVS is ideal for pediatric patients.
At present, the preparation methods of the bioabsorbable polymer vascular stent include 3D bio-printing, weaving, laser engraving and coating. BVS processed by the weaving method and coating method have a low efficiency and poor flexibility in terms of structural change, while the usage of 3D bio-printing can overcome the above-mentioned disadvantages. 3D bio-printing has received much attention in the biomedical field in recent years for its many potentially useful applications[9]. It has obvious advantages of high flexibility and controllability which, compared with other traditional methods, are unparalleled.
However, the stent prepared only through 3D bio-printing has a poor mechanical property, and it cannot effectively carry the required drug and provide a good microenvironment for cells’ growth[10]. When implanted into the animals’ body, thrombi can easily be formed. In order to solve these problems, nanofibers should be used for constructing the suitable microenvironment of cell growth. Electrospinning has important applications in biomedicine. Nanofibers have received considerable attention in the tissue engineering field due to their distinctive properties, including a high surface-area-to-volume ratio, biomimicry of the structure and functions of the extracellular matrix of human body tissues, etc[11]. Electrospun polymer nanofibers are also used in drug delivery[12].
In order to offset the defects of the stent prepared through 3D bio-printing alone, the complex forming platform was developed by our research team. Through this platform, the inner layer of the stent can be obtained by 3D bio-printing and with electrospinning, and the outer layer can also be built. Nanofibers were added to stents’surface by electrospinning to increase the effect of resilience, which can inhibit the proliferation of vascular endothelial abnormalities in the stents. Moreover, laying micro or nano scaled structures prepared through electrospinning on the outside of stent can not only embellish the outside of stent prepared by 3D bio-printing, but also can meet cells’ growth requirements. In addition, it is also an effective method for carrying drugs via the stent. Therefore, the complex process and method proposed in this paper can provide a good basis for preparing a controllable drug-carrying vascular stent, thus making effectively solving stenotic vessels diseases possible.
1 Test Platform for the Fabrication of CBVS
In order to increase the effect of combining 3D bio-printing and electrospinning, the proposed composite forming system consists of a motion platform subsystem, a multi-temperature field controlling subsystem, a high voltage electric field controlling subsystem, a Taylor cone monitoring subsystem, a 3D bio-printing feeding subsystem, an electrospinning feeding subsystem, and an electrospinning collection device subsystem, as shown in Fig.1.
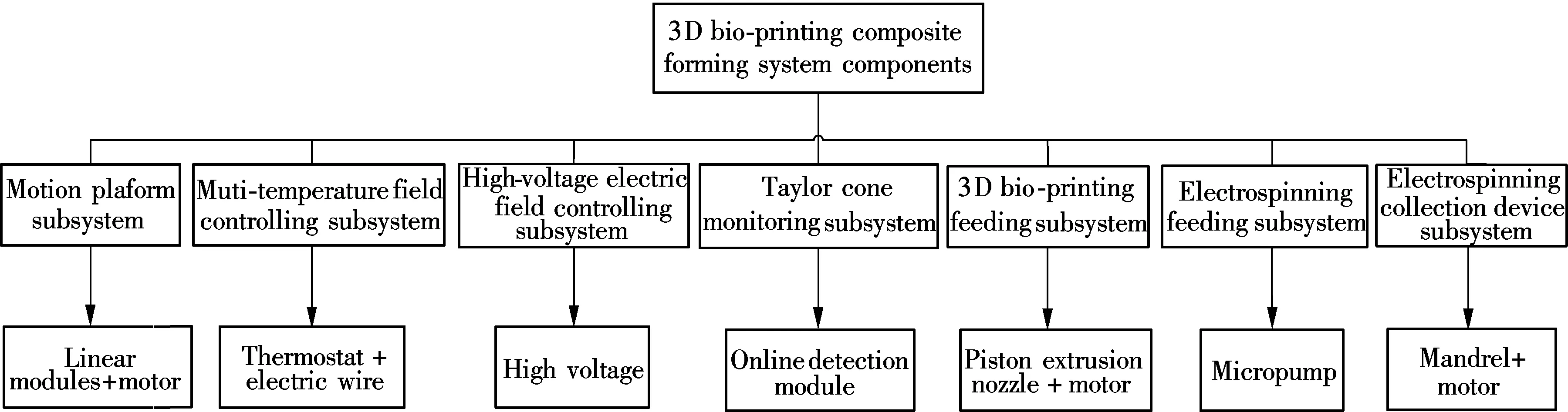
Fig.1 Components of 3D bio-printing composite forming system
1.1 Workstation for 3D bio-printing
This workstation for 3D bio-printing includes a motion platform subsystem, a PC, a PMAC controller, a thermostat, a receiving board and 3D bio-printing nozzle (see Fig.2). The motion platform subsystem consists of four axes (X-axis,Y-axis,Z-axis, andU-axis), which moves through the PMAC motion control card driving the motors. The pre-processing module extract the coordinate information of STL files and the post-processing module translate it to the G code which can be read by the Turbo PMAC. The final G code program can allow the Turbo PMAC to realize the processing path, making the automation of the entire preparation process possible.
1.2 Workstation for electrospinning
This workstation for electrospinning includes a high speed CCD camera, eletrospinning nozzle, the inner layer of the stent, micropump, electrospinning collection device and high-voltage electric field control subsystem (see Fig.3). The electric field control subsystem includes a high-voltage DC power supply (DW-P503-1AC, Dong Wen, Tianjin), which provides 0 to 50 kV voltage. The Taylor cone monitoring subsystem is used to monitor the entire electrospinning process and it includes a high speed CCD camera (CMLN-13S2M, PointGrey), a light source and custom detection software. The electrospinning feeding subsystem is a syringe pump (TJ-3A/W0109-1B, USA Lange). The movement, voltage, feeding and online monitoring system are automatically controlled by custom PC software[13].

Fig.3 Workstation for electrospinning
2 Experimental
2.1 Inner layer of the stent via 3D bio-printing
Granular PPDO is an aliphatic polyester-polyether with excellent biodegradability, biocompatibility and bioabsorbability. It has been approved by the US FDA for use as a base material for absorbable sutures (trade name PDS) and also has potential applications in orthopedic and drug delivery[14]. PPDO is chemically stable, histocompatible, safe, pliable, and exhibits an intermediate rate of degradation[15]. In addition, much independent research indicates that in vitro the degradation time of PPDO is about 3 to 4 months[16]and it accords with the ideal time for supporting self repair of the blood vessel[17]. Therefore, PPDO is adopted for preparing BVS.
The test platform of the 3D bio-printing complex forming CBVS as shown in Fig.2 is used for preparing the inner layer of the stent. Briefly, PPDO was inserted into a stainless steel syringe and heated using an electric wire. When the polymer reached the molten phase, PPDO was extruded through a nozzle and deposited on a continuously moving platform controlled by a computer. The inner layer of the stent was fabricated by depositing PPDO fibers along the predefined path (see Fig.4)[18]. Finally,the inner layer of the stent was fabricated by the 3D bio-printing, as shown in Fig.5.

1—Framework; 2—Barbs; 3—Lamellar mesh structure

Fig.5 The inner layer of the stent
2.2 Outer layer of the stent through electrospinning
PVA (grades JP233, degree of polymerization 3 500, alcoholysis degree of 88%, Kuraray Company of Japan, Ltd.) was dissolved in hot water with a mass fraction of 8%.This solution was heated to boiling point on a magnetic stirrer and stirred until the PVA was completely dissolved. Chitosan (viscosity-average molecular weight of 112×105, degree of deacetylation of 82.5%, Zhejiang Golden-Shell Biochemical Co., Ltd.) was dissolved in a 10% acetic acid solution. The PVA and chitosan solutions were then mixed with a respective volume ratio of 2∶1 and stirred[19].
The 3D bio-printed stent was fixed onto the rotating collection device shown in Fig.3. To fabricate the outer layer of the stent, the mixed solution of chitosan-PVA was placed in the injector and the outer layer was electrospun with 3 mL of mixed solution. The solutions were placed in 5 mL plastic syringes with a blunt-ended needle, and the syringe was attached to a micropump that dispensed the solution at a rate of 20 μL/min. A voltage of 15 kV was applied across the needle and the grounded collector (a 8-mm-diameter mandrel with a rotating speed of 300 r/min). After spinning for 3 h, the outer layer of the stent can be obtained. Thus, the two-layers-structured composite stent can be built in this way, and named “Type Ⅰ stents”, as shown in Fig.6. The ones from the control group which are prepared through 3D bio-printing alone, are named “Type Ⅱ stents”, as shown in Fig.5.

Fig.6 Type Ⅰ stent
2.3 CBVS handing and cell seeding
The CBVS was soaked in alcohol for 1 h and then washed three times with PBS to remove the alcohol from the CBVS. The excessive solution left in the stent was removed by extensive suction. Rat fibroblast cells were harvested and suspended in the culture medium at a density of 5.0×107cells/mL. Then, 0.05 mL cell suspensions were respectively seeded into CBVS to form cellstent constructs. The cellstent constructs placed in culture dishes were added with 8 mL of the growth medium after being incubated for 4 h to allow cells to adhere. The cellstent constructs in media were cultured in vitro, respectively, with the media changed twice a week[20].
3 Results and Discussion
3.1 Morphology of CBVS
The morphology of the final products was observed by scanning electron microscopy (SEM SU1510, Analysis and Testing Center, Shanghai University). As shown in Fig.7, the outer layer consists of evenly distributed chitosan-PVA fibers with uniform diameters and good porosity. This simulated extracellular matrix pattern works well for cell growth. A SEM micrograph of a cross-section of the two-layered composite is shown in Fig.8. The inner layer is approximately 0.25 mm in thickness and the outer layer is approximately 0.03 mm in thickness. The specimen thicknesses are measured using a micrometer with a precision of ±0.01 mm. Overall, this composite stent consists of the two-layered structure and meets the requirements of good mechanical properties and biocompatibility.

Fig.7 SEM micrograph of chitosan-PVA nanofibers in two-layered CBVS

Fig.8 SEM micrograph of cross-section of two-layered CBVS
3.2 Mechanical properties
Based on data from 10 samples of each group, the tensile strength was measured for each group, and the average values were calculated. The tensile strength of Type Ⅰ stents is (18.5±1.5) MPa, and the tensile strength of Type Ⅱ stents is (16.7±1.2) MPa (Model 5542, Instron, Canton, USA). Clearly, the Type Ⅰ stents have greater tensile strength than the Type Ⅱ stents. The tensile strength is defined as the maximum stress during the tensile test until fracture[21].
The radial strength of the stent is the resistance of the stent towards radial outer pressure and it is one of the most important technical indicators of a stent. The experiment of the stents is conducted on a radial strength tester, RX550 (Instron company). The test chamber temperature is 37 ℃; the compression ratio is 50%; and the compression speed is 0.1 mm/s. According to Ref.[10], when a stent is compressed to 88% of the diameter, the radial strength is the maximum value of the support stent. Based on data from the same 10 samples, the radial strength of Type Ⅰ stents is (127±11.6) kPa and the radial strength of Type Ⅱ stents is (122±13.7) kPa. Similarly to the tensile strength, the Type Ⅰ stents have greater radial strength than the Type Ⅱ stents. Test results on mechanical propensities of two groups of stents are listed in Tab.1.

Tab.1 Mechanical propensities for two groups of stents
3.3 Morphology and proliferation of cell
The CBVS is designed to provide a suitable environment for cell growth, proliferation and differentiation. Since fibroblasts can be easily cultivated and grow quickly, they are good candidates for evaluating stent biocompatibility. Therefore, rat fibroblast cells were seeded to evaluate cell attachment and proliferation on the outer layers of the CBVSs. Fig.9 is a SEM micrograph acquired after 7 d of cell growth.

Fig.9 SEM micrograph of rat fibroblast cell on the CBVS after 7 d
Cells on the CBVS proliferated promptly, most likely because of biological functional groups introduced by the chitosan. The results also suggest that natural hydrophilic surfaces may be more suitable for cell growth, which is consistent with the good biocompatibility of the CVBS.
4 Conclusion
A CBVS was prepared for the first time through 3D bio-printing and electrospinning techniques. It consisted of a chitosan-PVA nanofibers outer layer and a PPDO inner layer. This procedure for composite fabrication can avoid defects that result from single processes. This CBVS has good mechanical properties and biocompatibility. In addition, its outer layer can even be used for drug delivery. Therefore, the CBVS can be a better candidate for treating stenotic vessels.
[1] Thipparaboina R, Khan W, Domb A J. Eluting combination drugs from stents [J].InternationalJournalofPharmaceutics, 2013, 454(1): 4-10.

[3] Nef H M, Möllmann H, Weber M. Cobalt-chrome multi-link visionTM—stent implantation in diabetics and complex lesions: results from the DaVinci-Registry [J].ClinResCardio, 2011, 98(11): 731-737.
[4] König A, Schiele T M, Rieber J,et al. Influence of stent design and deployment technique on neointima formation and vascular remodeling [J].ZKardiol, 2002, 91(Sup 3): 98-102.
[5] Waksman R. Update on Bioabsorbable stents: from bench to Clinical [J].IntervenCardiol, 2006, 19(5): 414-421.
[6] Stepak B, Antonczak A J, Bartkowiak-Jowsa M, et al. Fabrication of a polymer-based biodegradable stent using a CO2laser [J].Archivesofcivilandmechanicalengineering, 2014, 14(2): 317-326.
[7] Ghimire G, Spiro J, Kharbanda R. Initial evidence for the return of coronary vasoreactivity following the absorption of bioabsorbable magnesium alloy coronary stents [J].EuroIntervention, 2009, 4(4): 481-484.
[8] Muramatsu T, Onuma Y, García-García H M, et al. Incidence and short-term clinical outcomes of small side branch occlusion after implantation of an everolimus-elutingbioresorbable vascular scaffold [J].CardiovascularInterventions, 2013, 6(3): 247-257.
[9] Pati F, Shim J-H, Lee J-S, et al. 3D printing of cell-laden constructs for heterogeneous tissue regeneration [J].SocietyofManufacturingEngineers, 2013, 1(1): 49-53.
[10] Feng Q, Jiang W, Sun K, et al. Mechanical properties and in vivo performance of a novel sliding-lock bioabsorbable poly-p-dioxanone stent [J].JMaterSci:MaterMed, 2011, 21(10): 2319-2327.
[11] Tong H-W, Zhang X, Wang M. A new nanofiber fabrication technique based on coaxial electrospinning [J].MaterialsLetters, 2012, 66(1): 257-260.
[12] Hu X L, Liu S, Zhou G Y, et al. Electrospinning of polymeric nanofibers for drug delivery applications [J].JournalofControlledRelease, 2014, 185(10): 12-21.
[13] Yan F F, Liu Y Y, Chen H P, et al. A multi-scale controlled tissue engineering scaffold prepared by 3D printing and NFES technology [J].AIPAdvances, 2014, 4(3): 031321-1-031321-8.
[14] Yang K K, Wang Y Z. A recyclable and biodegrable polymer: poly(p-dioxanone) [J].MaterialsChina, 2011, 30(8): 25-34. (in Chinese)
[15] Sabino M A, Feijoe J L, Muller A J, et al. Crystallisation and morphology of poly(p-dioxanone) [J].MacromolChemPhys, 2000, 201(18): 2687-2698.
[16] Petrtyl J, Bruha R, Horak L, et al. Management of benign intrahepatic bile duct strictures: initial experience with polydioxanone biodegradable stents [J].Endoscopy, 2010, 42(Sup 2): 89-90.
[17] Alt E, Haehnel J, Beilharz C, et al. Inhibition of neo-intima formation after experimental coronary artery stenting: a new biodegradable stent coating releasing hirudin and the prostacyclin analogue iloprest [J].Circulation, 2000, 101(12): 1453-58.
[18] Sun K, Sun K. A novel bioabsorbable sliding-lock stents: China ZL201010111299[P]. 2011-08-24. (in Chinese)
[19] Yan F F, Chen H P, Zheng L L, et al. The controllable PVA-chitosan fiber prepared by the near-field electrospinning for tissue engineering [J].AdvanceJournalofFoodScienceandTechnology, 2013, 5(8): 1073-1078.
[20] Chen H P, Liu Y Y, Jiang Z L, et al. Cellscaffold interaction within engineered tissue [J].ExperimentalCellResearch, 2014, 323(2): 346-351.
[21] Han F X, Jia X L, Dai D D, et al. Performance of a multilayered small-diameter vascular scaffold dual-loaded with VEGF and PDGF [J].Biomaterials, 2013, 34(30): 7302-7313.
结合生物3D打印和静电纺丝制备复合生物可吸收血管支架用于血管狭窄治疗
刘媛媛1,2向 科1李 瑜1陈海萍1胡庆夕1,2
(1上海大学快速制造工程中心, 上海 200444)
(2上海大学上海市智能制造及机器人重点实验室, 上海 200072)
设计一种新型血管支架用于血管狭窄治疗.针对目前制备生物可吸收血管支架(BVS)在装备和技术上的不足,提出了结合生物3D打印和静电纺丝制备复合生物可吸收血管支架(CBVS)的新方法.首先,用PPDO材料通过3D打印制备支架内层;然后,配制壳聚糖和PVA混合溶液,通过静电纺丝制备支架外层.力学性能测试显示,结合3D打印和静电纺丝制备的支架要好于仅采用3D打印制备的支架.在支架上种植细胞试验表明,细胞在支架上有良好的粘附和增殖,因为外层含有天然生物材料壳聚糖.所提出的复合成形工艺和方法,为后续构建可控携载药物支架提供了很好的思路.该CBVS可用于血管狭窄治疗.
生物3D打印;生物可吸收血管支架;静电纺丝;细胞增殖;复合成形
TH16
Foundation items:The National Natural Science Foundation of China (No.51475281, 51375292), the National Natural Science Foundation for Young Scholar of China (No.51105239).
:Liu Yuanyuan, Xiang Ke, Li Yu, et al. Composite bioabsorbable vascular stents via 3D bio-printing and electrospinning for treating stenotic vessels[J].Journal of Southeast University (English Edition),2015,31(2):254-258.
10.3969/j.issn.1003-7985.2015.02.017
10.3969/j.issn.1003-7985.2015.02.017
Received 2014-09-24.
Biographies:Liu Yuanyuan (1979—), female, doctor, associate professor, yuanyuan_liu@shu.edu.cn.
猜你喜欢
杂志排行
Journal of Southeast University(English Edition)的其它文章
- Adaptive modulation in MIMO optical wireless communication systems
- An improving energy efficiency cooperation algorithm based on Nash bargaining solution in selfish user cooperative networks
- Performance analysis of an O2/CO2 power plantbased on chemical looping air separation
- Model of limestone calcination/sulfation under oxy-fuel fluidized bed combustion
- A novel carbon trap sampling systemfor coal-fired flue gas mercury measurement
- Applicability of Markov chain-based stochastic modelfor bubbling fluidized beds
