Upregulation of Nogo receptor expression induces apoptosis of retinal ganglion cells in diabetic rats
2014-03-24XuezhengLiuZhongfuZuoWanpengLiuZhiyunWangYangHouYunjieFuYuzhiHan
Xuezheng Liu, Zhongfu Zuo, Wanpeng Liu, Zhiyun Wang, Yang Hou, Yunjie Fu, Yuzhi Han
1 Department of Anatomy, Liaoning Medical University, Jinzhou, Liaoning Province, China
2 Scienti fi c Experimental Center, Liaoning Medical University, Jinzhou, Liaoning Province, China
Upregulation of Nogo receptor expression induces apoptosis of retinal ganglion cells in diabetic rats
Xuezheng Liu1, Zhongfu Zuo1, Wanpeng Liu1, Zhiyun Wang1, Yang Hou2, Yunjie Fu1, Yuzhi Han1
1 Department of Anatomy, Liaoning Medical University, Jinzhou, Liaoning Province, China
2 Scienti fi c Experimental Center, Liaoning Medical University, Jinzhou, Liaoning Province, China
Xuezheng Liu and Zhongfu Zuo
contributed equally to this study.
The Nogo receptor is an essential factor for neuronal apoptosis, but the changes in Nogo receptor expression in the retina and the effects of the Nogo receptor on retinal ganglion cell apoptosis in diabetes mellitus remain unclear. We found that Nogo receptor expression was mainly visible in retinal ganglion cells of a rat model of diabetes mellitus induced by streptozotocin. At 12 weeks after onset of diabetes mellitus, Nogo receptor and Rho kinase expression signi fi cantly increased in the retina, and retinal ganglion cell apoptosis was apparent. When RNA interference was used to suppress Nogo receptor expression in rat retina, Rho kinase expression was obviously inhibited, and retinal ganglion cell apoptosis was evidently reduced in rats with diabetes mellitus. These results indicate that upregulation of Nogo receptor expression is an important mechanism of retinal ganglion cell apoptosis in rats with diabetes mellitus.
nerve regeneration; diabetes mellitus; diabetic retinopathy; visual acuity; retinal ganglion cells; apoptosis; Nogo receptor; Rho kinase; myelin-associated protein; NSFC grant; neural regeneration
Funding: This study was supported by the National Natural Science Foundation of China, No. 31140072, 81300931; the Liaoning Provincial Department of Science and Technology Project, No. 2011225015; the Natural Science Foundation of Liaoning Province in China, No. 2013022055.
Liu XZ, Zuo ZF, Liu WP, Wang ZY, Hou Y, Fu YJ, Han YZ. Upregulation of Nogo receptor expression induces apoptosis of retinal ganglion cells in diabetic rats. Neural Regen Res. 2014;9(8):815-820.
Introduction
Diabetic retinopathy is a key complication of diabetes mellitus, but its pathogenesis remains unclear. Diabetic retinopathy lacks effective therapies and has been a key factor for blindness in adults (Silva et al., 2010). Nervous system lesions in the retina are a key reason for diabetic retinopathy and a main mechanism for diminution of vision (Robinson et al., 2012). Retinal ganglion cell axons are a major component of optic nerves. The injury, structural changes, hypofunction and apoptosis of retinal ganglion cells are major factors for decreased vision in patients with diabetic retinopathy (Lieth et al., 2000; Kim et al., 2003), but the mechanisms underlying retinal ganglion cell apoptosis remain poorly understood in patients with diabetes mellitus.
Nogo receptor (NgR) is a membrane receptor associated with neural regeneration, and can be combined with myelin-associated proteins, such as Nogo protein, myelin-associated glycoprotein and oligodendrocyte myelin gly-coprotein (Li et al., 2004). Activated NgR can suppress neural regeneration, leading to neuronal atrophy and apoptosis (Peng et al., 2010; Pernet et al., 2012). NgR is extensively expressed in the nervous system, but can only be observed in retinal ganglion cells of the retina (Yin et al., 2007). Upregulation of NgR expression could induce retinal ganglion cell apoptosis and diminution of vision after occurrence of glaucoma (Fu et al., 2011). Rho-associated protein kinase (ROCK), an NgR downstream molecule, was strongly associated with neuronal apoptosis (Tan et al., 2010; Wälchli et al., 2013). Retinal ganglion cell apoptosis is a crucial mechanism of diabetic retinopathy, but it remains unclear whether retinal ganglion cell apoptosis is associated with the alterations in NgR gene expression after diabetic retinopathy. We still do not know which molecular mechanisms are involved when NgR participates in retinal ganglion cell apoptosis. Therefore, this study sought to explore the changes in NgR expression in the diabetic retina, to investigate the effects and mechanisms of NgR in the occurrence and development of diabetic retinopathy, and to provide new ideas for clinical treatment of diabetic retinopathy.
Materials and Methods
Animals
A total of 40 clean adult Sprague-Dawley rats aged 3 months and weighing 220-250 g were enrolled in this study (Animal license No. SCXK (Liao) 20030007). Rats obtained from Liaoning Medical University in China were housed under controlled temperature and humidity with a 12-hour light/dark cycle (light on at 6:00 and off at 18:00). The investigation conformed to the Guide for the Care and Use of Laboratory Animals published by the US National Institutes of Health (NIH publication No. 85-23, revised 1996), and the protocol was approved by the Animal Care Committee from LiaoningMedical University, China.
Grouping
A total of 40 rats were randomly assigned to control, diabetes mellitus, siNgR and siRNA blank groups. Rat models of diabetes mellitus were induced by streptozotocin in the diabetes mellitus, siNgR and siRNA blank groups. Lentivirus-packed NgR antisense nucleotide sequence (10 μL) was injected into bilateral vitreous bodies of rats in the siNgR group. Lentivirus-packed silenced nucleotide sequence (10 μL) was administered in bilateral vitreous bodies of rats in the siRNA blank group. Preservation solution (10 μL), i.e., PBS supplemented with 10% glycerol, was injected into bilateral vitreous bodies of rats in the diabetes mellitus and control groups. 12 weeks later, morphological and molecular biological indexes were detected in each group.
Preparation of lentivirus-packed nucleotide sequence
The siNgR antisense oligonucleotide sequence was designed in accordance with a previous study (Fry et al., 2007). Nucleotide sequence was synthesized and lentivirus package was produced by Shanghai GenePharma Co., Ltd. (Shanghai, China). siNgR antisense oligonucleotide sequence was 5′-AAT GAC TCT CCA TTT GGG ACT-3′, whilst silenced nucleotide sequence was 5′-UUC UCC GAA CGU GUC ACG UTT-3′. Virus titer was 1 × 1012TU/mL.
Establishment of an animal model of diabetes mellitus
In accordance with a previously published method (Zuo et al., 2011), an animal model of diabetes mellitus was established. After 12-hour fasting, rats were intraperitoneally injected with streptozotocin 60 mg/kg (dissolved in pH 4.5 0.1 mol/L citrate buffer solution) for 48 hours. Blood was collected from the tail tip. Blood glucose concentration was measured using a Blood Glucose Meter (Roche Diagnostics, Mannheim, Germany). Blood glucose concentration >16.7 mmol/L served as a marker for successful establishment of the model. Rats with blood glucose lower than the above-mentioned criteria were excluded. Rats from the control group were intraperitoneally injected with citrate bu ff er solution without streptozotocin.
Preparation of retinal specimens
At 12 weeks after model induction, rats were intraperitoneally injected with 4% sodium pentobarbital (60 mg/kg). After opening the thoracic cavity, a perfusion cannula was inserted through the cardiac apex to the root of the ascending aorta. Blood was fl ushed away with 100 mL of physiological saline, and tissue was fi xed with 4% paraformaldehyde (pH 7.3, 500 mL) for 2 hours. Te eyes were removed and the anterior segment and vitreous body were peeled away. Te retina was dehydrated with ethanol, permeabilized with xylene, and sliced into 8 μm para ffi n sections.
Double-label fl uorescent immunohistochemistry for NgRand Brn3a expression in the rat retina
Sections were dewaxed and hydrated. Antibodies were diluted by diluent, containing 0.01 mol/L PBS, 0.1% Triton X-100 and 10% normal goat serum. Rabbit anti-rat NgR (1:200) was mixed with mouse anti-rat Brn3a (1:200; Sigma, St. Louis, MO, USA). Sections were incubated with the mixture at room temperature for 2 hours, then at 4°C overnight, followed by PBS washes (10 minutes × 3). Sections were then incubated with a mixture containing Alexa 488-labeled donkey anti-rabbit IgG and Alexa 594-labeled donkey anti-mouse IgG (1:500; Santa Cruz Biotechnology, Dallas, TX, USA) for 4 hours at room temperature, followed by PBS washes (10 minutes × 3). Tese sections were mounted, observed and photographed with a fluorescence microscope (CX31-32RFL; Olympus, Tokyo, Japan).
Fluorescence immunohistochemical detection for ROCK expression in the rat retina
Retinal sections were dewaxed, hydrated, and then incubated with 10% normal fetal bovine serum for 30 minutes at room temperature. This incubation was followed by exposure to mouse anti-rat ROCK (1:200; Sigma) at room temperature for 4 hours, and at 4°C overnight. After washes with PBS (10 minutes × 3), these sections were incubated with A488-labeled goat anti-mouse IgG (1:500; Sigma) for 4 hours at room temperature. Sections were then washed again and incubated with DAPI (1:1,000; Sigma) for 15 minutes, mounted, blocked, and observed with an inverted fluorescence microscope (Olympus). Four fi elds of each retina were randomly observed at 400 × magni fi cation. Te numbers of ROCK-positive cells and retinal ganglion cells were counted. Each fi eld was read three times, and the mean value was obtained. Te proportion of positive cells stained for ROCK in retinal ganglion cells was calculated.
Terminal deoxynucleotidyl transferase dUTP nick end labeling (TUNEL) assay for detection of apoptosis of retinal ganglion cells in the rat retina
TUNEL assay was used to detect the apoptosis of retinal ganglion cells in the rat retina (Jiang et al., 2011). Sections were dewaxed, hydrated and washed with PBS (3 × 10 minutes). Procedures were conducted in accordance with the instructions on the TUNEL kit (KeyGEN BioTECH, Nanjing, Jiangsu Province, China). Sections were visualized with 3, 3′-diaminobenzidine, washed with water for 30 minutes, and counterstained with hematoxylin for 5 minutes. Subsequently, sections were di ff erentiated with di ff erentiation medium for 3 seconds, washed for 15 minutes, permeabilized with xylene, and dehydrated through a graded alcohol series. Sections were fi nally mounted with neutral resin, and observed with an inverted microscope. Four fi elds of each retina were randomly read at 200 × magnification. The numbers of TUNEL-positive cells and retinal ganglion cells were counted. Each fi eld was counted three times, and the mean value was obtained. The proportion of positive TUNEL stained retinal ganglion cells was calculated.
Western blot assay for detection of NgR and ROCK protein expression in the rat retina
At 12 weeks after model induction, rats were intraperitoneally injected with 4% sodium pentobarbital (60 mg/kg). Eye-balls were obtained and retinas were removed. Samples were treated with lysate (Roche, Basal, Switzerland), triturated on ice, and centrifuged at 10,000 × g at 4°C for 20 minutes (Shanghai Anting Scientific Instrument Factory, Shanghai, China). Protein content of the supernatant was determined using a bicinchoninic acid protein assay kit (Pierce, Rockford, IL, USA). Protein was isolated using sodium dodecyl sulphate-polyacrylamide gel electrophoresis, and transferred onto polyvinylidene difluoride membrane (Millipore, Billerica, MA, USA). Te polyvinylidene di fl uoride membrane was blocked with Tris-bu ff ered saline containing Tween-20 at 37°C for 90 minutes (containing 50 g/L skimmed milk powder). Te sections were incubated with primary antibodies (goat anti-rat NgR antibody, 1:1,000, Sigma; goat anti-rat ROCK antibody, 1:1,000, Santa Cruz Biotechnology; goat anti-rat β-actin antibody, 1:1,000, Santa Cruz Biotechnology) at 4°C overnight. After three washes with Tris-bu ff ered saline containing Tween-20, each for 10 minutes, sections were incubated with horseradish peroxidase-labeled donkey anti-goat IgG (1:1,000; Santa Cruz Biotechnology) for 2 hours at room temperature. The samples were visualized with western blot substrate enhanced chemiluminescence kit (Abcam, Cambridge, UK). Absorbance of each band was scanned with a gel imaging system (Ultra-Violet Products Ltd., Cambridge, UK). Standardization to the expression of β-actin was then performed.
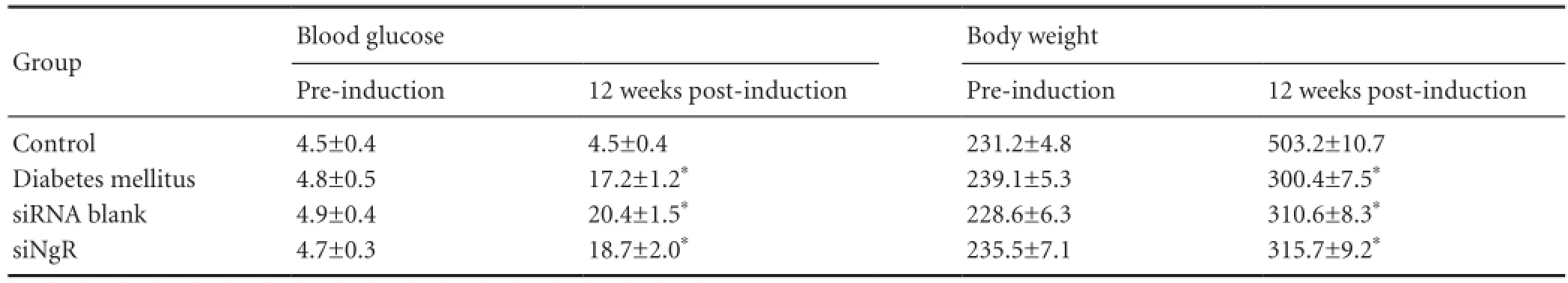
Table 1 Effects of intravitreal injection of siNgR nucleotide sequence on blood glucose (mmol/L) and body weight (g) of rats with diabetes mellitus
Statistical analysis
Data were analyzed using SPSS 16.0 software (SPSS, Chicago, IL, USA), and expressed as mean ± SEM. One-way analysis of variance was performed using a randomized block design. Paired comparison was completed using least signi fi cant difference test. A value of P < 0.05 was considered statistically signi fi cant.
Results
Intravitreal injection of siNgR nucleotide sequence did not signi fi cantly affect blood glucose and body weight in rats with diabetes mellitus
Before model induction, no significant difference in blood glucose concentration or body weight was detected in rats from each group (P > 0.05). At 12 weeks after model establishment, body weight was slightly heavier and blood glucose levels were higher compared with the control group (P < 0.01). Transfection with antisense nucleotide did not in fl uence rat body weight and blood glucose (P > 0.05; Table 1).
siNgR inhibited NgR and ROCK expression in the retina
Results of double-label fluorescent immunohistochemistry demonstrated that a large amount of NgR expression was detected in the ganglion cell layer of rat retina in the control group, and coexisted with Brn3a (Figure 1). Brn3a is a marker of retinal ganglion cells. The coexistence of NgR and Brn3a indicated that NgR is expressed in retinal ganglion cells and provides morphological evidence for NgR expression interfering and affecting apoptosis in retinal ganglion cells. Western blot assay results exhibited that at 12 weeks after model establishment, NgR and ROCK expression were higher in the diabetes mellitus and siRNA blank groups compared with the control group (P < 0.01). No signi fi cant difference in NgR and ROCK expression was detectable in the siNgR group (P > 0.05; Figure 2).
Inhibition of NgR expression suppressed ROCK expression in retinal ganglion cells
Immunohistochemical results revealed that 9.1±0.5% of cells were positively stained for ROCK in retinal ganglion cells from rats in the control group. At 12 weeks after model establishment, the proportion of ROCK stained cells was significantly higher in the diabetes mellitus and siRNA blank groups 35.2±1.7%, 30.6±2.1%, respectively than in the control group); P < 0.01. The number of ROCK-positive retinal ganglion cells was significantly lower in the siNgR group than that in the diabetes mellitus group (11.6±0.7%, P < 0.05). These results suggested that the inhibition of NgR expression could apparently downregulate ROCK protein expression in retinal ganglion cells of rats with diabetes mellitus (Figure 3).
Downregulation of NgR expression suppressed apoptosis in retinal ganglion cells in rats with diabetes mellitus
TUNEL-positive neurons were observed in the rat retina of the control group. At 12 weeks, a large number of TUNEL-positive retinal ganglion cells were detected in the diabetes mellitus, siRNA blank and siNgR groups, respectively (46.8±3.5%, 51.3±2.1%, and 18.7±1.5%; Figure 4). The proportion of TUNEL-positive cells in retinal ganglion was significantly lower in the siNgR group compared withthe diabetes mellitus group (P < 0.01). Results indicated that inhibition of NgR expression could evidently suppress apoptosis of retinal ganglion cells in the rat retina.
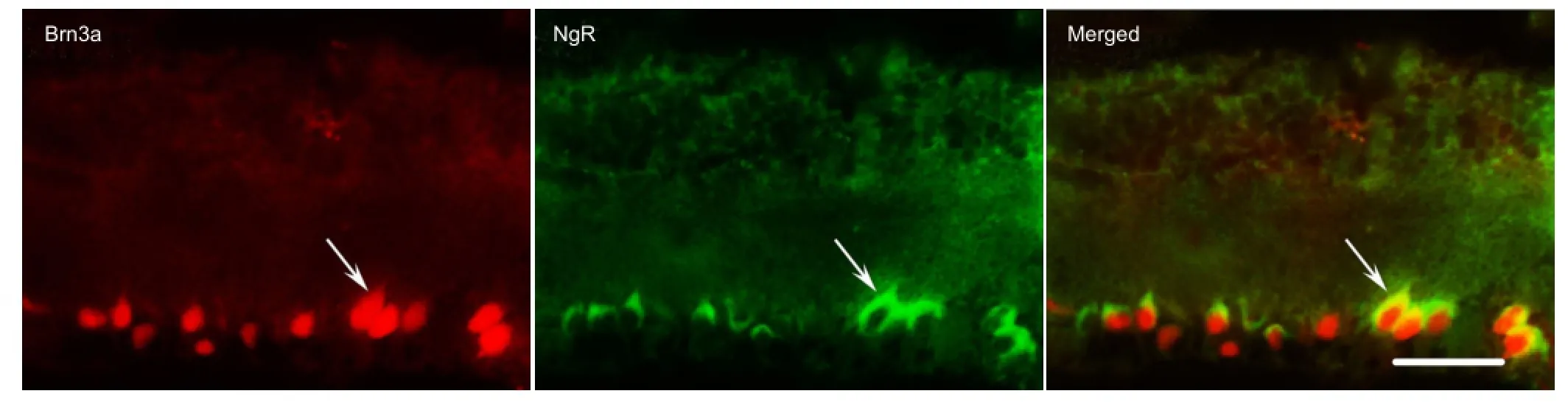
Figure 1 Coexistence of Brn3a and NgR in retinal ganglion cells (double-label fl uorescent immunohistochemistry).
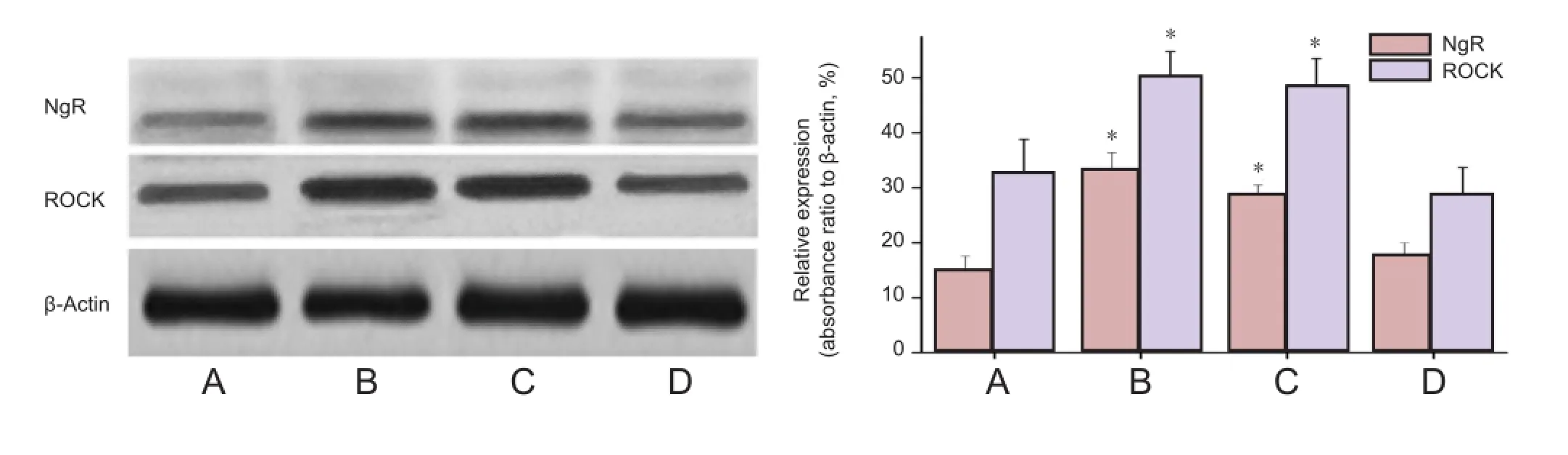
Figure 2 Western blot assay for siNgR effects on NgR and ROCK protein expression in the retina of rats with diabetes mellitus.
Discussion
Pathological changes in neurons directly cause a reduction in visual acuity in patients with diabetic retinopathy. Electroretinogram, contrast of visual acuity, range of visual fi eld and color sense all showed obvious abnormalities in these patients (Lieth et al., 2000; Zhang et al., 2013). Retinal ganglion cell axons are a major part of the optic nerve. Injured retinal ganglion cells are dif fi cult to regenerate, and retinal ganglion cell injury is a key factor for the decrease in visual acuity of diabetic retinopathy patients (Fischer et al., 2004; Harvey et al. 2006). This study also found that nervous system lesions in retina are an important pathological alteration in diabetic retinopathy. The number of TUNEL-positive retinal ganglion cells was apparently increased in rats with diabetes mellitus for 3 months, with the presence of apoptosis. The mechanisms of retinal ganglion cell apoptosis in the diabetic retina were not clear. Excitatory neurotransmitters such as glutamate, branched-chain amino acids and homocysteine, accumulated in the diabetic retina (Ola et al., 2013). The lacks of folic acid and vitamin B12 are important mechanisms in the apoptosis of retinal ganglion cells of the diabetic retina (Ola et al., 2013). Abnormal retinal metabolism is the key reason for the apoptosis of retinal ganglion cells, but the mechanisms underlying the apoptosis of retinal ganglion cells in the diabetic retina remain unclear. NgR is a myelin-associated protein, and can bind to Nogo protein, myelin-associated glucoprotein and oligodendrocyte myelin glycoprotein (Li et al., 2004). Activated NgR can suppress neural regeneration and promote neuronal apoptosis (Peng et al., 2010). NgR extensively expresses in nervous system, but only can be observed in retinal ganglion cells of the retina (Yin et al., 2007). The apoptosis of retinal ganglion cells is an essential mechanism of diabetic retinopathy, and strongly associated with NgR (Kern et al., 2008; Fu et al., 2011). To explore the effects and mechanisms of NgR in the apoptosis of retinal ganglion cells of diabetic retina, the present study detected the coexistence of NgR and Brn3a in the retina by double-label fluorescent immunohistochemistry (Badea and Nathans, 2011), and con fi rmed that NgR was only expressed in retinal ganglion cells. To investigate the effects of NgR on retinal ganglion cells of rats with diabetes mellitus, antisense oligonucleotide speci fi city was used to inhibit NgR expression in the diabetic retina. The downregulation of NgR expression in the diabetic retina evidently suppressed the apoptosis of retinal ganglion cells of rats with diabetes mellitus. Results indicated that the upregulation of NgRexpression was a key mechanism of the apoptosis in retinal ganglion cells from diabetic rats. A previous study showed that NgR expression apparently increased in retinal ganglion cells after glaucoma occurrence, so to inhibit NgR expression could suppress the apoptosis of retinal ganglion cells (Fu et al., 2011). These fi ndings were similar to results seen in this study. Therefore, increased NgR expression is an important mechanism underlying the apoptosis of retinal ganglion cells.
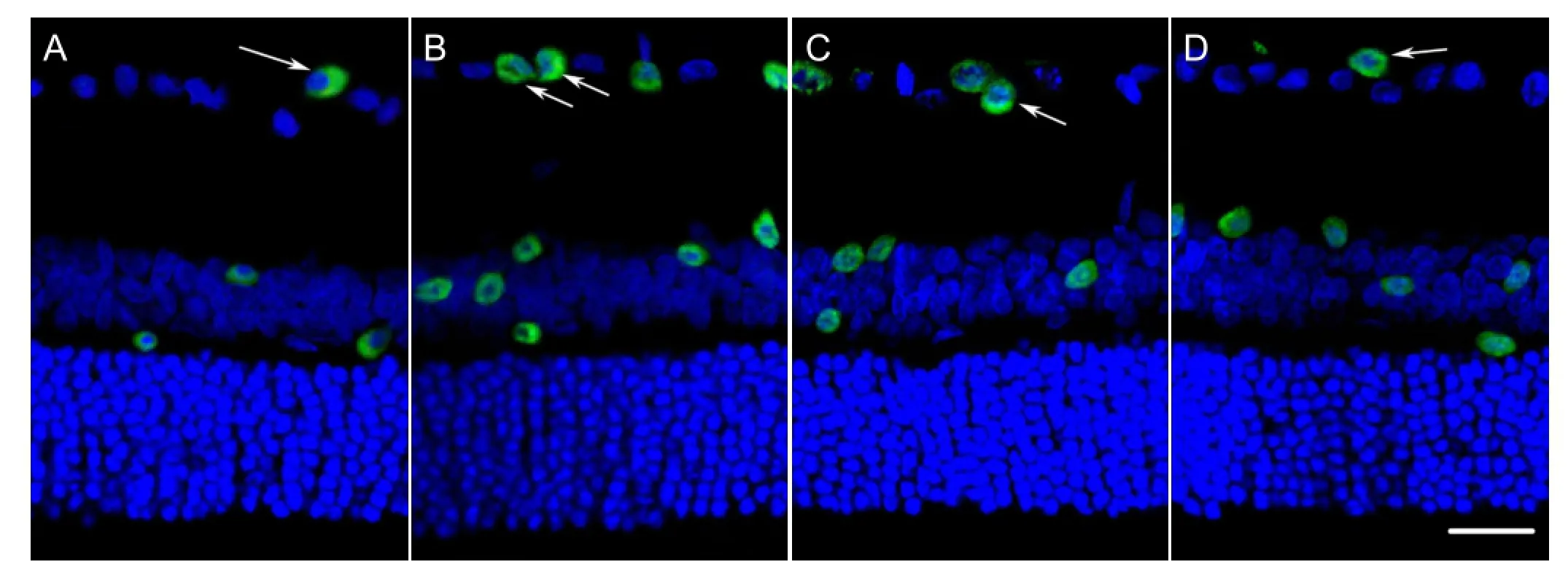
Figure 3 Inhibition of NgR expression affected the expression of ROCK in retinal ganglion cells from rat retina (immunohistochemical staining).
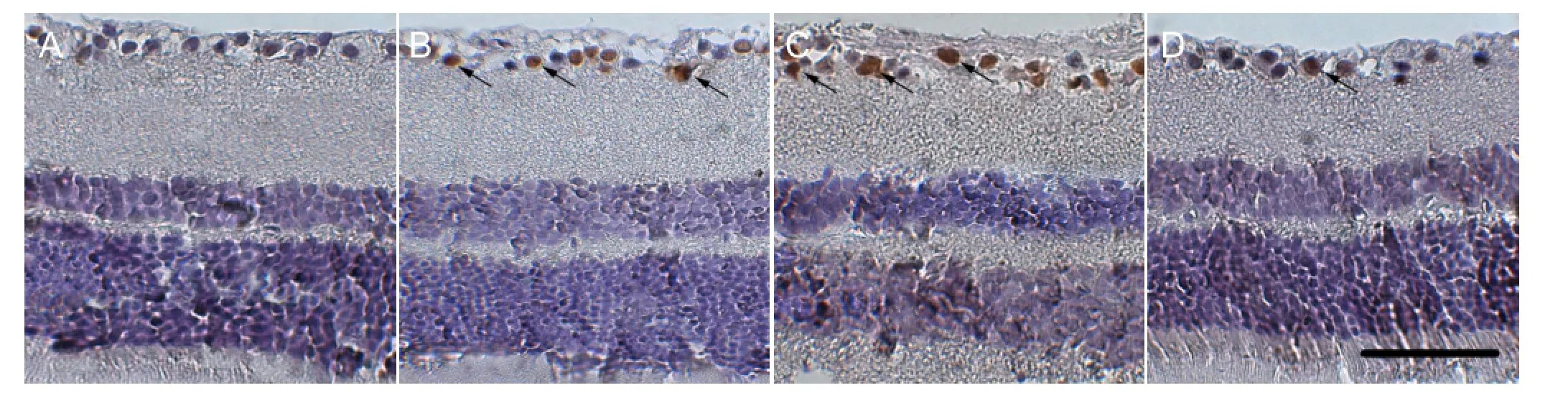
Figure 4 Downregulation of NgR expression suppressed apoptosis of retinal ganglion cells in rat retina (TUNEL staining, × 200).
ROCK, a serine-threonine kinase and an NgR downstream molecule, is strongly associated with retinal ganglion cell apoptosis. ROCK protein inhibitor Y-27632 promoted the regeneration of retinal ganglion cell axons, inhibited retinal ganglion cell apoptosis and contributed to neural regeneration in rats receiving optic neurotomy (Lingor et al., 2007; Lingor et al., 2008). Inhibiting NgR expression may downregulate ROCK protein expression in the retina of diabetic rats, but ROCK protein is extensively expressed in the retina (Li et al., 2013). To analyze the effects of downregulating NgR protein expression on ROCK protein expression in retinal ganglion cells of diabetic rats, this study detected ROCK protein expression in retinal ganglion cells using immunohistochemical methods, and detected ROCK changes in the retina using western blot assay, which supported immunohistochemical results. Results demonstrated that NgR regulated ROCK expression in the retina of diabetic rats. A large number of apoptotic retinal ganglion cells were visible when ROCK expression was increased. Moreover, to inhibit NgR expression and to downregulate ROCK expression could noticeably suppress the apoptosis of retinal ganglion cells of diabetic rats. These results suggest that ROCK protein is a key molecular mechanism for the regulatory effect of NgR on the apoptosis of retinal ganglion cells of diabetic rats.
This study verified that increased NgR expression is a key factor for the apoptosis of retinal ganglion cells, and its mechanism is probably associated with the upregulation of NgR on ROCK protein expression. However, this studyhas some limitations, and did not explore the downstream mechanism of the apoptosis of retinal ganglion cells of ROCK-induced diabetic retina. Moreover, NgR can induce apoptosis of retinal ganglion cells of diabetic rats through other pathways. As we have seen, there are no reports on NgR effects on the apoptosis of retinal ganglion cells of diabetic rats, or on the underlying mechanisms. This study investigated the effects and mechanisms of NgR on the apoptosis of retinal ganglion cells of diabetic rats, provided new ideas for revealing pathogenesis of diabetic retinopathy and clinical treatment of diabetic retinopathy.
Author contributions:Liu XZ participated in study design and technical support. Zuo ZF participated in morphological study and wrote the manuscript. Liu WP was in charge of morphological study. Wang ZY participated in statistical analysis. Hou Y detected molecular biology. Fu YJ and Han YZ established animal models. All authors approved the final version of the paper.
Con fl icts of interest:None declared.
Peer review:NgR has been paid great attention, due to its specific target molecule effects during signal transduction of myelin-inhibited axon regeneration. This study used animal models of diabetes mellitus, verified that NgR only expressed in retinal ganglion cells. NgR and ROCK expression significantly increased in retinal ganglion cells, and ganglion cell apoptosis was obvious. Moreover, to inhibit NgR expression could decrease the apoptosis of retinal ganglion cells of diabetic rats. Results confirmed that NgR extensively expressed in optic nerve and suppressed neural regeneration. This study provided an experimental basis for finding new blockers and drugs to promote optic nerve regeneration.
Badea TC, Nathans J (2011) Morphologies of mouse retinal ganglion cells expressing transcription factors Brn3a, Brn3b, and Brn3c: analysis of wild type and mutant cells using genetically-directed sparse labeling. Vision Res 51:269-279.
Fischer D, He Z, Benowitz LI (2004) Counteracting the Nogo receptor enhances optic nerve regeneration if retinal ganglion cells are in an active growth state. J Neurosci 24:1646-1651.
Fry EJ, Ho C, David S (2007) A role for Nogo receptor in macrophage clearance from injured peripheral nerve. Neuron 53:649-662.
Fu QL, Liao XX, Li X, Chen D, Shi J, Wen W, Lee DH, So KF (2011) Soluble Nogo-66 receptor prevents synaptic dysfunction and rescues retinal ganglion cell loss in chronic glaucoma. Invest Ophthalmol Vis Sci 52:8374-8380.
Harvey AR, Hu Y, Leaver SG, Mellough CB, Park K, Verhaagen J, Plant GW, Cui Q (2006) Gene therapy and transplantation in CNS repair: the visual system. Prog Retin Eye Res 25:449-489.
Jiang H, Fang J, Wu B, Yin G, Sun L, Qu J, Barger SW, Wu S (2011) Overexpression of serine racemase in retina and overproduction of D-serine in eyes of streptozotocin-induced diabetic retinopathy. J Neuroin fl ammation 8:119.
Kern TS, Barber AJ (2008) Retinal ganglion cells in diabetes. J Physiol 586:4401-4408.
Kim YS, Kim YH, Cheon EW, Park JM, Yoo JM, Kang SS, Cho GJ, Choi WS (2003) Retinal expression of clusterin in the streptozotocin-induced diabetic rat. Brain Res 976:53-59.
Li M, Yasumura D, Ma AA, Matthes MT, Yang H, Nielson G, Huang Y, Szoka FC, LaVail MM, Diamond MI (2013) Intravitreal administration of HA-1077, a ROCK inhibitor, improves retinal function in a mouse model of huntington disease. PLoS One 8:e56026.
Li S, Liu BP, Budel S, Li M, Ji B, Walus L, Li W, Jirik A, Rabacchi S, Choi E, Worley D, Sah DW, Pepinsky B, Lee D, Relton J, Strittmatter SM (2004) Blockade of Nogo-66, myelin-associated glycoprotein, and oligodendrocyte myelin glycoprotein by soluble Nogo-66 receptor promotes axonal sprouting and recovery after spinal injury. J Neurosci 24:10511-10520.
Lieth E, Gardner TW, Barber AJ, Antonetti DA (2000) Retinal neurodegeneration: early pathology in diabetes. Clin Experiment Ophthalmol 28:3-8.
Lingor P, Teusch N, Schwarz K, Mueller R, Mack H, Bähr M, Mueller BK (2007) Inhibition of Rho kinase (ROCK) increases neurite outgrowth on chondroitin sulphate proteoglycan in vitro and axonal regeneration in the adult optic nerve in vivo. J Neurochem 103:181-189.
Lingor P, Tönges L, Pieper N, Bermel C, Barski E, Planchamp V, Bähr M (2008) ROCK inhibition and CNTF interact on intrinsic signalling pathways and di ff erentially regulate survival and regeneration in retinal ganglion cells. Brain 131:250-263.
Ola MS, Nawaz MI, Khan HA, Alhomida AS (2013) Neurodegeneration and neuroprotection in diabetic retinopathy. Int J Mol Sci 14: 2559-2572.
Peng Y, Zhang QL, Xu D, Wang YP, Qin XY (2010) Small hairpin RNA interference of the Nogo receptor inhibits oxygen-glucose deprivation-induced damage in rat hippocampal slice cultures. Neuropathology 30:565-573.
Pernet V, Schwab ME (2012) Te role of Nogo-A in axonal plasticity, regrowth and repair. Cell Tissue Res 349:97-104.
Robinson R, Barathi VA, Chaurasia SS, Wong TY, Kern TS (2012) Update on animal models of diabetic retinopathy: from molecular approaches to mice and higher mammals. Dis Model Mech 5:444-456.
Silva PS, Cavallerano JD, Sun JK, Aiello LM, Aiello LP (2010) E ff ect of systemic medications on onset and progression of diabetic retinopathy. Nat Rev Endocrinol 6:494-508.
Tan HB, Zhong YS, Cheng Y, Shen X (2010) Rho/ROCK pathway and neural regeneration: a potential therapeutic target for central nervous system and optic nerve damage. Int J Ophthalmol 4:652-657.
Wälchli T, Pernet V, Weinmann O, Shiu JY, Guzik-Kornacka A, Decrey G, Yüksel D, Schneider H, Vogel J, Ingber DE, Vogel V, Frei K, Schwab ME (2013) Nogo-A is a negative regulator of CNS angiogenesis. Proc Natl Acad Sci U S A 110:E1943-1952.
Yin X, Chen C, Yuan R, Ye J (2007) An immunofluorescence-histochemistry study of the Nogo receptor in the rat retina during postnatal development. Ann Ophthalmol (Skokie) 39:140-144.
Zhang X, Wang N, Barile GR, Bao S, Gillies M (2013) Diabetic retinopathy: neuron protection as a therapeutic target. Int J Biochem Cell Biol 45:1525-1529.
Zuo ZF, Wang W, Niu L, Kou ZZ, Zhu C, Wang W, Zhao XH, Luo DS, Zhang T, Zhang FX, Liu XZ, Wu SX, Li YQ (2011) RU486 (mifepristone) ameliorates cognitive dysfunction and reverses the down-regulation of astrocytic N-myc downstream-regulated gene 2 in streptozotocin-induced type-1 diabetic rats. Neuroscience 190:156-165.
Copyedited by Apricò K, de Souza M, Yu J, Qiu Y, Li CH, Song LP, Zhao M
10.4103/1673-5374.131597
Xuezheng Liu, M.D., Department of
Anatomy, Liaoning Medical University, Jinzhou 121001, Liaoning Province,
China, Liuxuezheng168@vip.sina.com.
http://www.nrronline.org/
Accepted: 2014-03-28
杂志排行
中国神经再生研究(英文版)的其它文章
- The synthetic thyroid hormone, levothyroxine, protects cholinergic neurons in the hippocampus of naturally aged mice
- Citalopram increases the differentiation ef fi cacy of bone marrow mesenchymal stem cells into neuronal-like cells
- Fusion protein of single-chain variable domain fragments for treatment of myasthenia gravis
- Similar effects of substance P on learning and memory function between hippocampus and striatal marginal division
- Regulatory effects of anandamide on intracellular Ca2+concentration increase in trigeminal ganglion neurons
- Virtual reality interface devices in the reorganization of neural networks in the brain of patients with neurological diseases
