Mesenchymal stem cells and COVID-19: What they do and what they can do
2021-10-11EjlalAbuElRubRamadaKhasawnehFatimahAlmahasnehZaidAltaanyNesreenBatainehHanaZegallaiSaravananSekaran
Ejlal Abu-El-Rub, Ramada R Khasawneh, Fatimah Almahasneh, Zaid Altaany, Nesreen Bataineh, Hana Zegallai, Saravanan Sekaran
Ejlal Abu-El-Rub, Department of Physiology and Pathophysiology, University of Manitoba, Winnipeg R2H2A6, Canada
Ejlal Abu-El-Rub, Department of Physiology and Pathophysiology, Basic Medical Sciences, Yarmouk University, IRBID 21163, Jordan
Ramada R Khasawneh, Department of Anatomy and Histology, Basic Medical Sciences, Yarmouk University, IRBID 21163, Jordan
Fatimah Almahasneh, Department of Physiology and Pharmacology, Basic Medical Sciences, Yarmouk University, IRBID 21163, Jordan
Zaid Altaany, Department of Biochemistry and Genetics, Basic Medical Sciences, Yarmouk University, IRBID 21163, Jordan
Nesreen Bataineh, Department of Pathology, Basic Medical Sciences, Yarmouk University, IRBID 21163, Jordan
Hana Zegallai, Department of Pharmacology and Therapeutics, University of Manitoba, Winnipeg R2H2A6, Canada
Saravanan Sekaran, Department of Pharmacology, Saveetha Dental College and Hospitals to be University, Chennai 600077, India
Abstract The severe acute respiratory syndrome coronavirus 2 (SARS-CoV-2) or coronavirus disease 2019 (COVID-19) pandemic has exhausted the health systems in many countries with thousands cases diagnosed daily. The currently used treatment guideline is to manage the common symptoms like fever and cough, but doesn’t target the virus itself or halts serious complications arising from this viral infection. Currently, SARS-CoV-2 exhibits many genetic modulations which have been associated with the appearance of highly contagious strains. The number of critical cases of COVID-19 increases markedly, and many of the infected people die as a result of respiratory failure and multiple organ dysfunction. The regenerative potential of mesenchymal stem cells (MSCs) has been extensively studied and confirmed. The impressive immunomodulation and antiinflammatory activity of MSCs have been recognized as a golden opportunity for the treatment of COVID-19 and its associated complications. Moreover, MSCs regenerative and repairing abilities have been corroborated by many studies with positive outcomes and high recovery rates. Based on that, MSCs infusion could be an effective mechanism in managing and stemming the serious complications and multiple organ failure associated with COVID-19. In the present review, we discuss the commonly reported complications of COVID-19 viral infection and the established and anticipated role of MSCs in managing these complications.
Key Words: SARS-CoV-2; COVID-19; Mesenchymal stem cells; Inflammation; Acute respiratory distress syndrome; Pulmonary fibrosis; Pneumonia; Renal injury; Cardiovascular diseases; Regeneration; Anti-inflammatory
INTRODUCTION
The 2020 severe acute respiratory syndrome coronavirus 2 (SARS-CoV-2), coronavirus disease 2019 (COVID-19) pandemic catastrophe continues to strike many countries severely with million cases and thousands of death reported daily by the World Health Organization. This disease urged many clinicians and researchers to sprint to find effective treatments so as to control and manage the rampant spread of COVID-19 virus worldwide. Despite the fact that COVID-19 is primarily a pulmonary disease, it can hit other organs leading to hematological, hepatic, neurological, cardiac and renal complications[1]. There are no approved remedy to treat COVID-19 and its associated complications. There are many treatment recommendations that are considered as a “leap-of-faith” approach to save the life of many COVID-19 patients[2]. The most commonly used medications to treat symptomatic COVID-19 patients are oxygen, corticosteroids, remdesivir, chloroquine, hydroxychloroquine, lopinavir/ritonavir, nitazoxanide, vitamin super B-complex, zinc and vitamin D[2]. All these suggested medications have many limitations, including: (1) The lack of information regarding the initiation and duration of treatment regimen; (2) Many studies that have been conducted to evaluate the efficacy of these treatments recruited young patients who had relatively asymptomatic disease; (3) The long-run side effects have not been addressed yet; and (4) There is a dearth of data regarding the effect of these treatments in preventing post-infection complications which can be serious and life-threatening[2,3]. Recently, mesenchymal stem cells (MSCs) have been used as one of the promising therapeutic strategies for COVID-19 symptoms and complications[4]. MSCs are one of the most trustworthy stem cells in regenerating and repairing the damages of many organs. MSCs can be isolated and transplanted even in unrelated donor-recipient due to their hypoimmunogenic characteristics[5]. MSCs can repair and restore the function of many injured tissues and improve the quality of life for patients with chronic disorders[5]. MSCs exert their therapeutic effectsviadifferent mechanisms[5]. MSCs have the ability to subdue severe inflammation by releasing anti-inflammatory factors such as interleukin-10 (IL-10), IL-4, Indoleamine 2, 3-dioxygenase (IDO), prostaglandin E2 (PGE2), and Heme oxygenase-1 (HO-1)[6]. Furthermore, MSCs release many antifibrotic factors, including Transforming growth factor beta-1 (TGF-β1), matrix metalloprotein (MMP) 1, 2 and 9, tissue inhibitor of metalloproteinases-1 (TIMP1) and Collagen Type III Alpha 1 Chain (COL3A1)[7]. MSCs can also promote the angiogenesis and proliferation of cells mediated by releasing many growth factors such as vascular endothelial growth factor (VEGF), Insulin-like growth factor 1 (IGF-1), hepatocyte growth factor (HGF), and platelet-derived growth factor (PDGF)[8]. As MSCs are multipotent stem cells, they can differentiate into many cell types, including cardiomyocytes-like cells, adipocytes, chondrocytes, Clara cells or alveolar cells, endothelial and epithelial cells, which can restore the normal function of impaired organs[9]. The encouraging therapeutic outcomes of MSCs transplantation are expected to be effective in treating COVID-19 complications and decreasing the death rate even in seriously ill patients. Numerous preclinical and clinical trials have been carried out to study the role of MSCs in alleviating COVID-19 symptoms and complications, including acute respiratory distress syndrome (ARDS), pneumonia, pulmonary fibrosis, systemic inflammation, hypercoagulability, and cardiovascular complications. The search for effective COVID-19 management strategies continues to evolve, therefore, in the current review article, we discussed the research outcomes and therapeutic potential of MSCs for COVID-19 patients to provide an updated reference for many clinicians and scientific researchers.
COVID-19 INFLAMMATORY CASCADE AND THE ANTI-INFLAMMATORY ROLE OF MSCS
Current understanding of SARS-CoV-2 mechanisms suggests a central role for exaggerated activation of the innate immune system as an important contributor to COVID-19 adverse outcomes. COVID-19 progression can be divided into three distinct phases, including: (1) Early infection phase, wherein the virus infiltrates host cells in the lung parenchyma; (2) Pulmonary phase, in which viral propagation causes lung tissue injury as the host immune response is activated; and (3) The inflammatory cascade which is triggered by pathogen-associated molecular patterns (i.e., viral RNA) and damage-associated molecular patterns (DAMPs,i.e., cellular debris is released during pyroptosis) are exposed during active viral replication and release[10]. The inflammatory response plays a crucial role in the clinical manifestations and subsequent complications of COVID-19. Post SARS-CoV-2 entry, host factors trigger an immune response against the virus, which, if it is left uncontrolled, may result in pulmonary tissue damage, pulmonary functional impairment, and reduced lung capacity[11]. In addition to respiratory failure, other complicated feature among patients with severe COVID-19 infection is a sudden decline of the patient’s health status approximately two weeks after onset. Continuous infiltration of monocytes and macrophages accompanied by extremely high levels of inflammatory response leads to atrophy of the spleen and lymph nodes, along with reduced lymphocytes in lymphoid organs, hypercoagulability, thrombosis, and multiple organ damage[12]. Cytokine storm syndrome (CSS) has been proposed as underlying the etiology of respiratory failure and multiple organ impairment in patients with COVID-19[13]. CSS is delineated as a significant pro-inflammatory cytokines gush that leads to inflammatory cells recruitment and widespread tissue damage. High levels of the cytokines have been reported, IL-2, IL-7, IL-10, granulocyte colony-stimulating factor, interferon γ-induced protein 10, monocyte chemoattractant protein 1 (MCP1)[13]. The disproportionately high levels of these cytokines were found to be associated with more severe forms of COVID-19 and increased need for intensive care units (ICU) admission.
MSCs remarkable immunomodulatory capacity is one of the most important therapeutic mechanism elicited by MSCs[14]. MSCs immunomodulation abilities can be effective through controlling both innate and adaptive immune systems[14]. Therefore, MSCs have the aptitude to “turn on and turn off” the immune system based on the body needs. Numerous studies on the immune regulation potential of MSCs reported a potent ability of MSCs to control and regulate the functions of different types of immune cells, including B lymphocytes, natural killer (NK) cells, dendritic cells, macrophages, and T lymphocytes by secreting many immunomodulatory factors, primarily TGF-β, HGF, IL-10, IDO, and PGE2[15-17]. Many studies verified the usefulness of MSCs to treat autoimmune diseases and inflammation. There is a lot of preclinical and clinical evidence for the anti-inflammatory effect of MSCs in mitigating virally engendered lung injury and mortality in mice. Several studies have illustrated that MSCs are capable of significantly reducing acute lung injury (ALI) supervening the infection of H9N2 and H5N1 viruses by decreasing the levels of pro-inflammatory cytokines and chemokines, as well as lessening the recruitment of inflammatory cells into the lungs[18]. Applying MSCs to interfere in endotoxin liposaccharide (LPS)-induced ALI mouse model proved that MSCs can remarkably lead to reduction of inflammatory cell infiltration in lung tissue, alleviate inflammation, and regenerate the damaged lung tissues[18]. To date, there is limited published literature regarding the potential efficacy of MSCs in COVID-19 disease. In February 2020, a clinical study from China has been published in which they evaluated a single dose infusion of bone marrow (BM) MSCs (obtained commercially) in seven COVID-19 patients aged 45 to 65[17]. The recruited patients, who were either in critical condition or had mild disease, did not improve with the treatment protocol. At 14 d post-infusion, all critically ill patients were weaned off mechanical ventilation. These positive outcomes regarding the clinical efficacy of infused MSCs were also accompanied by marked decrease in the levels of C-reactive protein, NK cells, T-cells, and tumor necrosis factor-α (TNF-α). Over-activated cytokine-secreting immune cells, CXCR3+CD4+ T cells, CXCR3+CD8+ T cells, and CXCR3+ NK cells were not existent after 3-6 Dy postinfusion[17]. In May 2020, the results of a clinical trial conducted in Liaocheng People’s Hospital in China, using human umbilical cord (UC) Wharton’s jelly-derived MSCs was published[19]. In the trial, a single female patient was involved who tested positive for COVID-19. With standard treatment, the patient vital physical signs improved substantially, but after that the patient took a turn for the worse. Considering the severe organ injury caused by an inflammatory response, human UC Wharton’s jelly-derived MSCs from a healthy donor was intravenously transfused to the patient. Two days after the infusion, the pulmonary function and symptoms of the patient with COVID-19 pneumonia significantly improved, and chest computerized tomography (CT) imaging showed a great amelioration. The patient recovered rapidly and was discharged 7 days after treatment[19]. The results showed that the frequency of CD3+ T, CD4+ T and CD8+ T cells increased, and serum C-reactive protein, IL-6 and TNF-α levels were reduced. Six days after MSC treatment, the patient became negative for SARS-CoV-2. Therefore, therapy based on the use of Wharton’s jellyderived MSCs may be effective in the treatment of patients with COVID-19[19].
Lianget al[20] and colleagues reported that transplantation of human UC-derived MSCs could modulate the immune response and promote the functional recovery in a 65-year-old female patient with critically ill COVID-19 and severe complications such as respiratory failure and multiple organ failure. The patient received three doses of allogeneic UC-MSCs intravenously at day 9 of infection which were followed by two infusions with an interval of three days. Following the second dose, the vital signs were improved, and she did not require the ventilator. Two days after the third dose, she was transferred out of the ICU[20]. After the administration of UC-MSCs, many clinical indexes and symptoms of the patient were improved. The counts of CD3+ T cell, CD4+ T cell, and CD8+ T cell remarkably increased to the normal level indicating the reversal of lymphopenia, which is a common feature of the COVID-19 patients and is associated with disease severity and mortality. The patient showed a significant decreased level of C-reaction protein, white blood, neutrophil, and alanine aminotransferase/aspartate aminotransferase along with increased level of total lymphocyte count. The study also suggested that the infusion of thymosin α1 will greatly enhance the immunomodulation potential of MSCs.
The safety of human UC-derived MSCs infusions for the treatment of patients with moderate and severe COVID-19 pulmonary disease was evaluated. A total of 18 patients were enrolled in clinical trial, nine of whom (five with moderate disease and four with severe disease) received three cycles of UC-derived MSCs treatment. The patients in the MSCs group displayed a reduction of serum IL-6. Moreover, this trial demonstrated that transplantation of MSCs in patients with COVID-19 was safe, and no serious adverse events were reported[21]. The data show that intravenous UCderived MSCs infusion in patients with moderate and severe COVID-19 is safe and well tolerated.
The published clinical study and case report have demonstrated that intravenous transplantation of MSCs was safe and effective in treating COVID-19, and remarkably, MSCs may be resistant to virus infection, whereas more clinical trials with larger samples are warranted for more convincing evidence.
COVID-19 RESPIRATORY COMPLICATIONS AND MSCS
COVID-19 pneumonia and MSCs
The most common manifestations of COVID-19 primarily target the respiratory system. According to a large cohort study from China, 81% of COVID-19 patients showed mild to moderate symptoms, including dry cough and moderate pneumonia, 14% had severe presentation, such as dyspnea, hypoxia or more than 50% lung involvement on imaging, and 5% were in critical condition manifested by having respiratory failure, shock, or multi-organ system dysfunction[22]. Besides being the first target for SARS-CoV-2 virus, the respiratory system is also involved in the transmission of SARS-CoV-2 virus, which occurs through respiratory droplets emittedviasneezing and coughing. SARS-CoV-2 virus was found to be stable in aerosols and different surface materials, and was detected in stool, tears and conjunctival secretions[23]. These can add to certain extent to its transmission. To enter the host organism, the SARS-CoV-2 virus passes through the mucous membranes of the nose and larynx, gaining access to the respiratory tract and eventually resides in the lungs. The lung alveolar epithelial cells are considered the site where the spike proteins of the virus bind to the cell surface angiotensin converting enzyme 2 (ACE2) receptors[23]. This leads to the activation of various proteases, which cleave the spike proteins at a site close to the S1/S2 subunit boundary, inducing viral fusion and subsequent internalization by endocytosis[24]. Some patients with COVID-19 pneumonia rapidly progress to critical illness and ARDS[25]. ARDS is the most severe form of ALI which is a form of diffuse alveolar injury. ARDS is defined as “an acute condition characterized by bilateral pulmonary infiltrates and severe hypoxemia (PaO2/FiO2ratio < 200) in the absence of evidence for cardiogenic pulmonary edema”[26].
COVID-19 ARDS shares mechanistic and pathophysiologic properties with influenza A virus-induced pneumonia. The presence of viral RNA in the cytoplasm of host epithelial cells stimulates antiviral pathways which then activate a number of molecular and cellular mechanisms of immunity[27]. This includes elevated serum cytokines (especially IL-1β, IL-6 and TNF-α), impaired interferon I and III responses, cytokines storm and peripheral lymphopenia[28,29]. Although these immune processes eliminate the virus, they also cause prolonged inflammation and destruction of the host alveolar epithelial cells, which result in severe lung injury and ARDS.
Compared to ARDS caused by other pathologies, the ARDS induced by SARS-CoV-2 seems to be caused by an injury to the alveolar epithelial cells with almost no effect is exerted on endothelial cells. This produces less exudation and, hence, dry cough. Clinical symptoms are often unpredictable and inconsistent with the severity of laboratory and imaging findings. The time of the onset of ARDS in COVID-19 cases is 8-12 d, which is longer compared to other respiratory conditions (up to 7 d)[25]. Mortality associated with COVID-19 is attributable to the development of pneumoniainduced ARDS, consequently, it would be of paramount importance to find effective prevention and treatment strategies for pneumonia-induced ARDS complication in COVID-19 patients[27]. MSCs are known to be an excellent candidate for cell therapy that targets lung diseases. MSCs had been successfully tested in the setting of different lung disease where they regulated the immune response by suppressing infiltrated cells and decreasing edema[30]. Plenty of studies have investigated the effects of MSCs in animal models of infection-induced ALI/ARDS. A recently published meta-analysis found that MSCs can reduce lung injury score of ALI, improve animal’s survival, decrease lung edema and improve lung compliance. In addition, MSCs reduce levels of pro-inflammatory factors such as IL-1β, IL-6 and TNF-α, while increasing the level of anti-inflammatory IL-10[31]. Comparable results were observed in human clinical trials. In an open-label study in 2013, administration of allogeneic menstrual-bloodderived MSCs lowered the death rate in patients with influenza H7N9-induced ARDS, with no significant undesirable effects were reported in the treated patients over a five-year follow-up period[32]. Other clinical trials[33-35] failed to prove the therapeutic effectiveness of MSCs in ARDS due to the limited number of recruited patients or inappropriate dose of MSCs. However, they confirmed the safety of such therapy; as no MSC-related hemodynamic, respiratory adverse events, or infusion toxicities were reported. Also, there were no significant differences in the overall number of adverse events between the treatment and control groups.
Due to the lack of effective therapies against COVID-19 virus complications, and based on the promising results of many preclinical and clinical studies of MSCs in the setting of ARDS, a number of patients with severe COVID-19 were empirically treated with MSCs and their cases were reported and published[19,20,36-41]. The majority of these cases involved male patients older than 50 and were reported in China. The source of MSCs injected included Wharton’s jelly, UC blood, and BM. The route of MSCs administration was intravenous infusions, and in one of the cases having brain manifestations, MSCs were delivered intrathecally[36]. Indeed, IV infusion is the preferred route of administration of UC-MSCs since most of the infused cells will be trapped in the lungs, the organ most affected by COVID-19[4]. All patients received other recommended lines of therapy, such as antivirals, antibiotics and/or corticosteroids. There was one patient who was treated with both convalescent plasma and MSCs[39]. In all reported cases, MSCs therapy caused no adverse effects, and patients showed complete resolving of COVID-19 symptoms.
The database clinicaltrials.gov lists 71 clinical trials of MSCs in COVID-19, which are summarized in Table 1. Eight of these trials were completed and their results disclosed. The outcome of one clinical trial has been reported on the same website[42], while the results of a few others have been published. Meng and colleagues, for example, reported the findings of a non-randomized phase 1 clinical trial which enrolled 18 patients with moderate to severe COVID-19. The patients were divided into a control and an UC-MSCs treatment group. No serious adverse events were reported, although two patients receiving UC-MSCs developed transient facial flushing and fever, and one developed transient hypoxia. Treatment with UC-MSCs decreased the need for mechanical ventilation, repaired lung pathological changes and improved pulmonary functions[21]. Lanzoniet al[43] and colleagues conducted a double-blind, phase 1/2a, controlled trial that recruited 24 patients with COVID-19-induced ARDS. The study was randomized to either the UC-MSCs treatment or the control group. UC-MSCs infusions were found to be safe and remarkably improved the patients’ survival and shortened the recovery time[43]. An open-label, individually randomized, controlled trial was carried out by Shuet al[44] which included 12 patients in the treatment group (UC-MSCs) and 29 in the control group. The treatment group had significantly shorter recovery time, rapid symptomatic improvement, faster alleviation of lung inflammation, and without any safety issues. In Spain, 13 patients with severe COVID-19 on mechanical ventilation were treated with adipose tissuederived MSCs (AD-MSCs). The therapy was associated with clinical, radiological and ventilation improvements[45]. Lenget al[17] studied the therapeutic effects of injecting ACE2- MSCs in 7 patients with severe COVID-19 pneumonia. The patients exhibited significant clinical improvements without observed adverse effects.
Other studies have addressed the therapeutic use of MSCs-derived components, such as exosomes, in COVID-19 patients. Sengupta and colleagues run a nonrandomized open-label cohort study to assess the safety and efficacy of exosomes derived from allogeneic BM-MSCs for the treatment of severe COVID-19 in 24 patients. No adverse effects related to infusion were reported. Treatment was associated with better clinical status and oxygenation, improvement in laboratory absolute neutrophils count, and a decline in acute phase reactants[46]. The abovementioned results indicate that MSCs may play a pivotal role in the treatment of COVID-19 related respiratory complications due to their excellent safety profile and a wide range of therapeutic outcomes as improvement in the clinical, radiological and immunological status of COVID-19 concomitant pneumonia.
COVID-19-ARDS and MSCs
COVID-19, that is concomitant with ARDS, emerged as a primary concern all over the world. ARDS is a life-threatening severe lung condition that causes damage to the pulmonary capillary endothelium and alveolar epithelium. The ARDS, which is caused by infection and other mechanical stimulation, allows fluid to leak into the lungs. As a result, breathing becomes difficult as the lungs turn non-functional and cannot supply the body's vital organs with enough oxygen. Most people who get ARDS are already at the hospital for trauma or illness[47]. The Spike proteins (S proteins) of SARS-CoV-bind the ACE2 in the alveolar cells which is catalyzed by a transmembrane protease serine 2 (TMPRSS2) protease in type II alveolar epithelial cells[48,49]. The high binding affinity of SARS-CoV-2 Spike proteins for ACE2 is the main determinant of the replication rate of SARS-CoV-2 and the cause of the severity of COVID-19 disease[50].
Various animal studies and early Phase I/II clinical trials have espoused the tremendous potential of MSCs therapy in treating the ARDS. SARS-CoV-2 infects type II alveolar epithelial cells or other target cells that express ACE2. The keratinocyte growth factor (KGF), which is secreted by MSCs, can decrease injury and improve the proliferation and regeneration of alveolar epithelial cells by rising surface-active substances, including MMP-9, IL-1Ra, and granulocyte-macrophage colonystimulating factor (GM-CSF)[51]. In addition, it has been reported that MSCs can improve gas exchange and reduce the levels of bronchoalveolar lavage fluid’ chemokines and cytokines, including GM-CSF, MIG, IL-1α, IFN-γ, IL-6, and TNF-α[18]. Researching experts elucidated that MSCs treatment expresses vital anti-inflammatory cytokines like IL-1RA, IL-8, and IL-10 and an array of bioactive molecules that stimulate local tissue regeneration by buffering the cytokine and chemokine storm provoked by COVID-19 virus[52]. A published clinical study conducted in Beijing Youan Hospital in China, showed that the transplantation of MSCs improved the clinical outcomes in all recruited COVID-19 elderly patients at day 4 post-MSCs injection without serious adverse effects[17]. Cytokine-secreting immune cells, including CXCR3+CD4+ T cells, CXCR3+CD8+ T cells, and CXCR3+ NK cells, eradicated within 1 wk of MSCs transplantation with a significant reduction in the inflammation biomarkers[17]. MSCs treatment in COVID-19 patients having ARDS were able to subside the over-activated immune system and promote endogenous repair by improving the lung microenvironment[53]. Taking into regard the positive results that have been revealed in some published phase I clinical trials, MSCs will be probably effective in reducing the risk of cytokine storms which cause ARDS and organs failure in patients with severe COVID-19 disease.
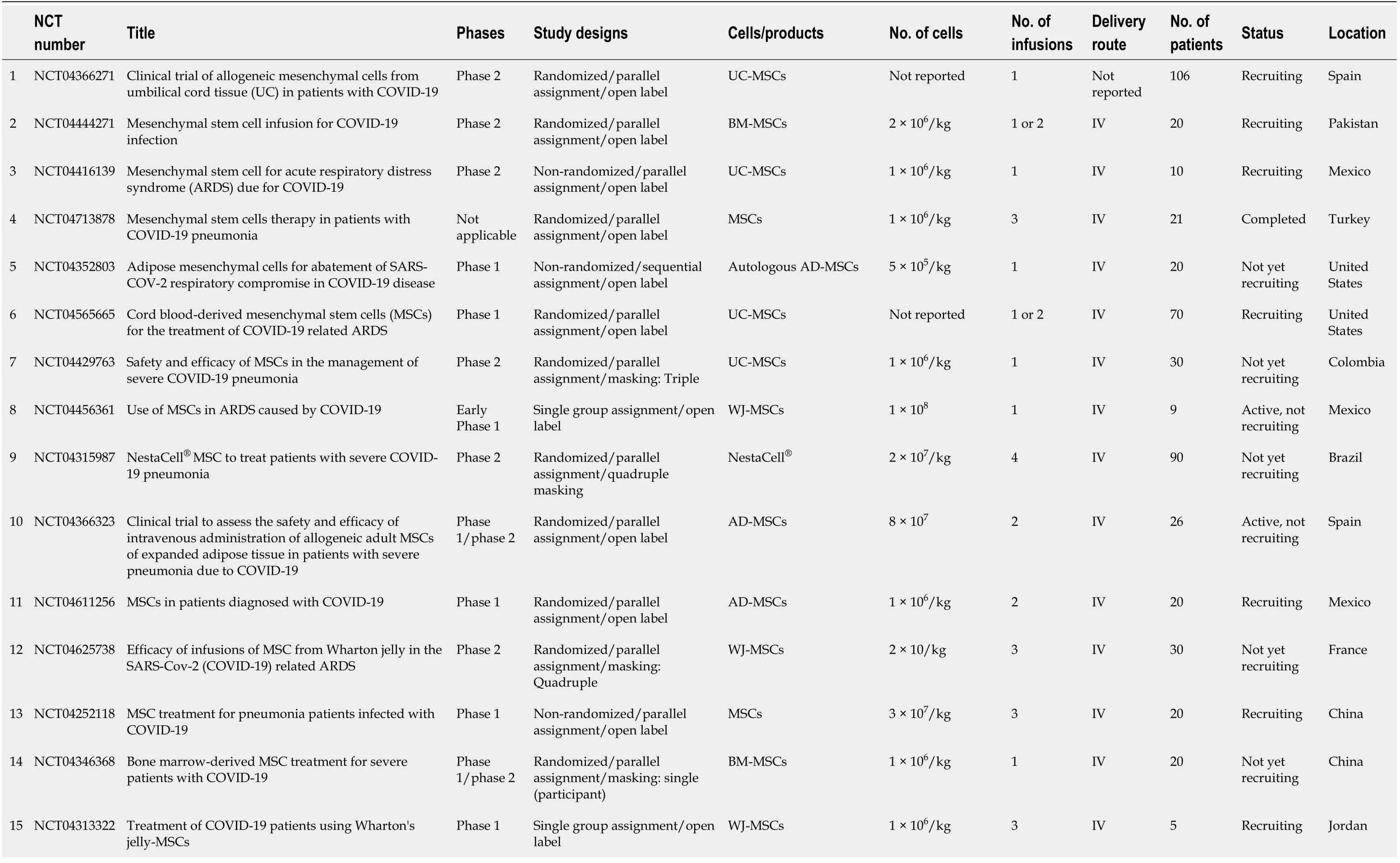
Table 1 Summary of clinical trials listed in clinicaltrials.org involving mesenchymal stem cells in the treatment of coronavirus disease 2019
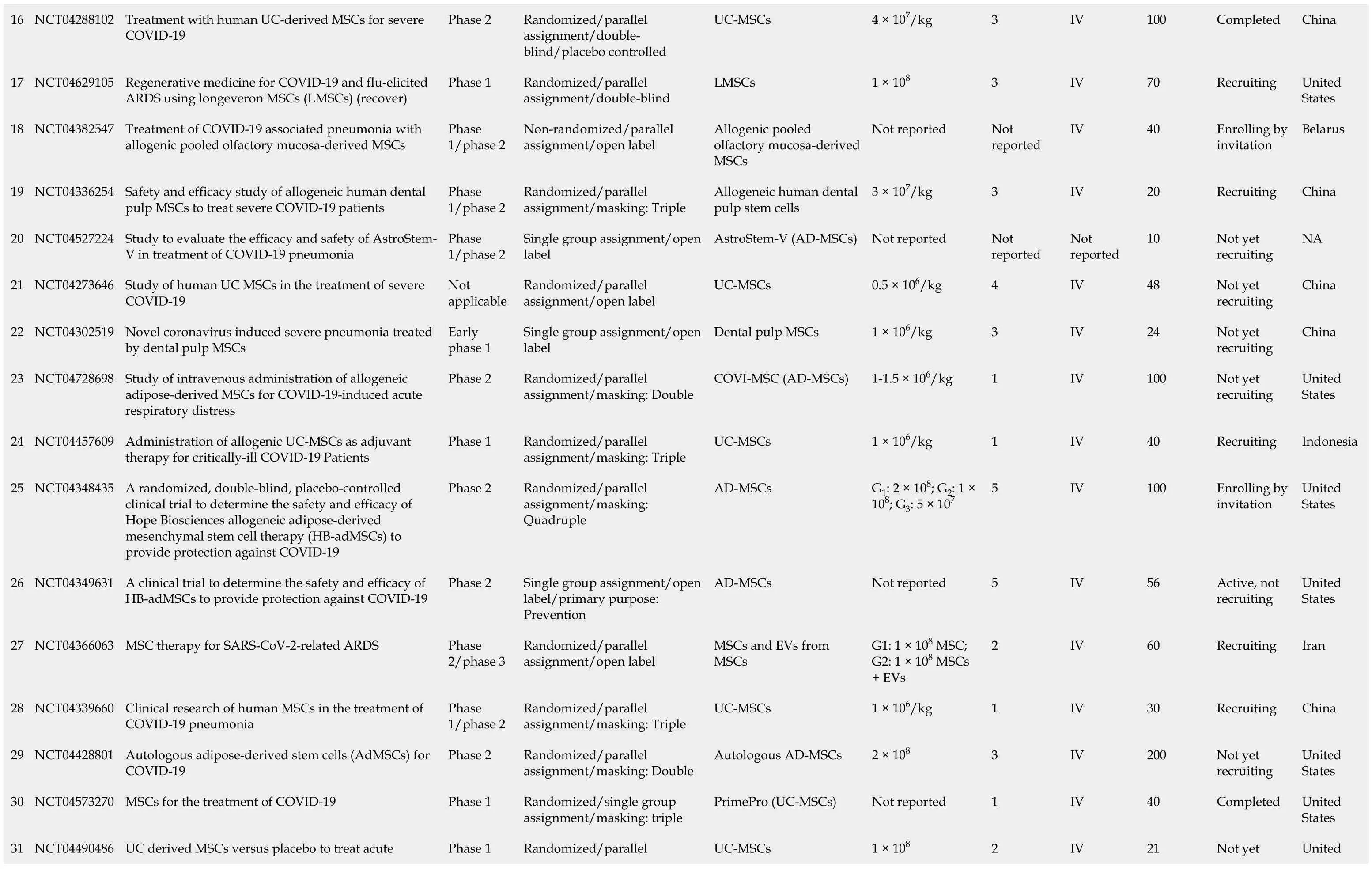
16 NCT04288102 Treatment with human UC-derived MSCs for severe COVID-19 Phase 2 Randomized/parallel assignment/doubleblind/placebo controlled UC-MSCs 4 × 107/kg 3 IV 100 Completed China 17 NCT04629105 Regenerative medicine for COVID-19 and flu-elicited ARDS using longeveron MSCs (LMSCs) (recover)Phase 1 Randomized/parallel assignment/double-blind LMSCs 1 × 108 3 IV 70 Recruiting United States 18 NCT04382547 Treatment of COVID-19 associated pneumonia with allogenic pooled olfactory mucosa-derived MSCs Phase 1/phase 2 Non-randomized/parallel assignment/open label Allogenic pooled olfactory mucosa-derived MSCs Not reported Not reported IV 40 Enrolling by invitation Belarus 19 NCT04336254 Safety and efficacy study of allogeneic human dental pulp MSCs to treat severe COVID-19 patients Phase 1/phase 2 Randomized/parallel assignment/masking: Triple Allogeneic human dental pulp stem cells 3 × 107/kg 3 IV 20 Recruiting China 20 NCT04527224 Study to evaluate the efficacy and safety of AstroStem-V in treatment of COVID-19 pneumonia Phase 1/phase 2 Single group assignment/open label AstroStem-V (AD-MSCs)Not reported Not reported Not reported 10 Not yet recruiting NA 21 NCT04273646 Study of human UC MSCs in the treatment of severe COVID-19 Not applicable Randomized/parallel assignment/open label UC-MSCs 0.5 × 106/kg 4 IV 48 Not yet recruiting China 22 NCT04302519 Novel coronavirus induced severe pneumonia treated by dental pulp MSCs Early phase 1 Single group assignment/open label Dental pulp MSCs 1 × 106/kg 3 IV 24 Not yet recruiting China 23 NCT04728698 Study of intravenous administration of allogeneic adipose-derived MSCs for COVID-19-induced acute respiratory distress Phase 2 Randomized/parallel assignment/masking: Double COVI-MSC (AD-MSCs)1-1.5 × 106/kg 1 IV 100 Not yet recruiting United States 24 NCT04457609 Administration of allogenic UC-MSCs as adjuvant therapy for critically-ill COVID-19 Patients Phase 1 Randomized/parallel assignment/masking: Triple UC-MSCs 1 × 106/kg 1 IV 40 Recruiting Indonesia 25 NCT04348435 A randomized, double-blind, placebo-controlled clinical trial to determine the safety and efficacy of Hope Biosciences allogeneic adipose-derived mesenchymal stem cell therapy (HB-adMSCs) to provide protection against COVID-19 Phase 2 Randomized/parallel assignment/masking: Quadruple AD-MSCs G1: 2 × 108; G2: 1 × 108; G3: 5 × 107 5 IV 100 Enrolling by invitation United States 26 NCT04349631 A clinical trial to determine the safety and efficacy of HB-adMSCs to provide protection against COVID-19 Phase 2 Single group assignment/open label/primary purpose: Prevention AD-MSCs Not reported 5 IV 56 Active, not recruiting United States 27 NCT04366063 MSC therapy for SARS-CoV-2-related ARDS Phase 2/phase 3 Randomized/parallel assignment/open label MSCs and EVs from MSCs G1: 1 × 108 MSC; G2: 1 × 108 MSCs + EVs 2 IV 60 Recruiting Iran 28 NCT04339660 Clinical research of human MSCs in the treatment of COVID-19 pneumonia Phase 1/phase 2 Randomized/parallel assignment/masking: Triple UC-MSCs 1 × 106/kg 1 IV 30 Recruiting China 29 NCT04428801 Autologous adipose-derived stem cells (AdMSCs) for COVID-19 Phase 2 Randomized/parallel assignment/masking: Double Autologous AD-MSCs 2 × 108 3 IV 200 Not yet recruiting United States 30 NCT04573270 MSCs for the treatment of COVID-19 Phase 1 Randomized/single group assignment/masking: triple PrimePro (UC-MSCs)Not reported 1 IV 40 Completed United States UC derived MSCs versus placebo to treat acute Randomized/parallel Not yet United 31 NCT04490486 Phase 1 UC-MSCs 1 × 108 2 IV 21

pulmonary inflammation due to COVID-19 assignment/masking: Double recruiting States 32 NCT04355728 Use of UC-MSCs for COVID-19 patients Phase 1/Phase 2 Randomized/parallel assignment/masking: triple UC-MSCs + heparin 1 × 108 2 IV 24 Completed United States 33 NCT04371601 Safety and effectiveness of MSCs in the treatment of pneumonia of COVID-19 Early Phase 1 Randomized/parallel assignment/open label UC-MSCs 1 × 106/kg 4 IV 60 Active, not recruiting China 34 NCT04522986 An exploratory study of adr-001 in patients with severe pneumonia caused by SARS-CoV-2 Infection Phase 1 Single group assignment/open label AD-MSCs (ADR-001)1 × 108 4 IV 6 Not yet recruiting Japan 35 NCT04390152 Safety and efficacy of intravenous W harton's jelly derived MSCs in ARDS due to COVID 19 Phase 1/Phase 2 Randomized/parallel assignment/masking: Quadruple WJ-MSCs 5 × 107 2 IV 40 Recruiting Colombia 36 NCT04461925 Treatment of coronavirus COVID-19 pneumonia (pathogen SARS-CoV-2) with cryopreserved allogeneic P_MMSCs and UC-MMSCs Phase 1/phase 2 Non-randomized/parallel assignment/open label Placenta-derived MSCs 1 × 106/kg 3 IV 30 Recruiting Ukraine 37 NCT04348461 BAttLe against COVID-19 using mesenchymal stromal cells Phase 2 Randomized/parallel assignment/masking: Quadruple Allogeneic and expanded AD-MSCs 1.5 × 106/kg 2 IV 100 Not yet recruiting Spain 38 NCT04535856 Therapeutic study to evaluate the safety and efficacy of DW-MSC in COVID-19 patients Phase 1 Randomized/parallel assignment/masking: Quadruple DW -MSCs G1: 5 × 107; G2: 10 × 107 1 IV 9 Completed Indonesia 39 NCT04362189 Efficacy and safety study of allogeneic HB-adMSCs for the treatment of COVID-19 Phase 2 Randomized/parallel assignment/masking: Quadruple AD-MSCs 1 × 108 4 IV 100 Active, not recruiting United States 40 NCT04494386 UC lining stem cells (ULSC) in patients with COVID-19 ARDS Phase 1/Phase 2 Randomized/parallel assignment/masking: Triple UC-MSCs 1 × 108 1 or 2 IV 60 Recruiting United States 41 NCT04397796 Study of the safety of therapeutic tx with immunomodulatory MSC in adults with COVID-19 infection requiring mechanical ventilation Phase 1 Randomized/parallel assignment/masking: Quadruple BM-MSCs Not reported Not reported Not reported 45 Recruiting United States 42 NCT04345601 Mesenchymal stromal cells for the treatment of SARSCoV-2 induced acute respiratory failure (COVID-19 Disease)Phase 1/Phase 2 Randomized/parallel assignment/open label MSCs 1 × 108/kg Up to 2 IV 30 Recruiting United States 43 NCT04452097 Use of hUC-MSC product (BX-U001) for the treatment of COVID-19 with ARDS Phase 1/phase 2 Non-randomized/sequential assignment/open label UC-MSCs G1: 0.5 × 106; G2: 1 × 106; G3: 1.5 × 106 1 IV 39 Not yet recruiting United States 44 NCT04492501 Investigational treatments for COVID-19 in tertiary care hospital of Pakistan Not applicable Non-randomized/factorial assignment/open label BM-MSCs (+ TPE and convalescent plasma)2 × 106/kg 1 IV 600 Completed Pakistan 45 NCT04377334 MSCs in inflammation-resolution programs of COVID-19 induced ARDS Phase 2 Randomized/parallel assignment/open label BM-MSCs Not reported Not reported IV 40 Not yet recruiting Germany 46 NCT04390139 Efficacy and safety evaluation of MSCs for the treatment of patients with respiratory distress due to COVID-19 Phase 1/phase 2 Randomized/parallel assignment/masking: Quadruple WJ-MSCs 1 × 106/kg 2 IV 30 Recruiting Spain
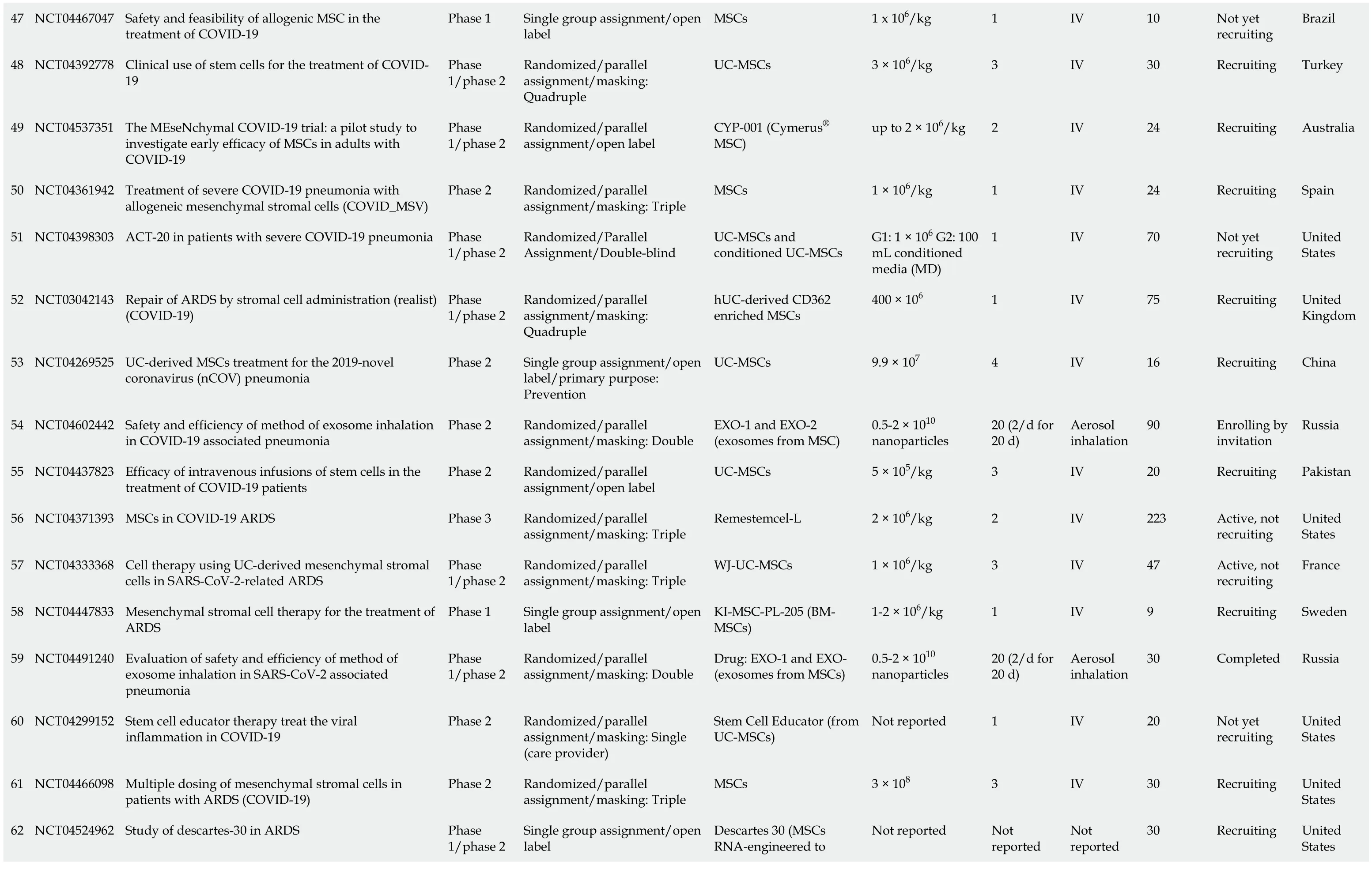
47 NCT04467047 Safety and feasibility of allogenic MSC in the treatment of COVID-19 Phase 1 Single group assignment/open label MSCs 1 x 106/kg 1 IV 10 Not yet recruiting Brazil 48 NCT04392778 Clinical use of stem cells for the treatment of COVID-19 Phase 1/phase 2 Randomized/parallel assignment/masking: Quadruple UC-MSCs 3 × 106/kg 3 IV 30 Recruiting Turkey 49 NCT04537351 The MEseNchymal COVID-19 trial: a pilot study to investigate early efficacy of MSCs in adults with COVID-19 Phase 1/phase 2 Randomized/parallel assignment/open label CYP-001 (Cymerus® MSC)up to 2 × 106/kg 2 IV 24 Recruiting Australia 50 NCT04361942 Treatment of severe COVID-19 pneumonia with allogeneic mesenchymal stromal cells (COVID_MSV)Phase 2 Randomized/parallel assignment/masking: Triple MSCs 1 × 106/kg 1 IV 24 Recruiting Spain 51 NCT04398303 ACT-20 in patients with severe COVID-19 pneumonia Phase 1/phase 2 Randomized/Parallel Assignment/Double-blind UC-MSCs and conditioned UC-MSCs G1: 1 × 106 G2: 100 mL conditioned media (MD)1 IV 70 Not yet recruiting United States 52 NCT03042143 Repair of ARDS by stromal cell administration (realist) (COVID-19)Phase 1/phase 2 Randomized/parallel assignment/masking: Quadruple hUC-derived CD362 enriched MSCs 400 × 106 1 IV 75 Recruiting United Kingdom 53 NCT04269525 UC-derived MSCs treatment for the 2019-novel coronavirus (nCOV) pneumonia Phase 2 Single group assignment/open label/primary purpose: Prevention UC-MSCs 9.9 × 107 4 IV 16 Recruiting China 54 NCT04602442 Safety and efficiency of method of exosome inhalation in COVID-19 associated pneumonia Phase 2 Randomized/parallel assignment/masking: Double EXO-1 and EXO-2 (exosomes from MSC)0.5-2 × 1010 nanoparticles 20 (2/d for 20 d)Aerosol inhalation 90 Enrolling by invitation Russia 55 NCT04437823 Efficacy of intravenous infusions of stem cells in the treatment of COVID-19 patients Phase 2 Randomized/parallel assignment/open label UC-MSCs 5 × 105/kg 3 IV 20 Recruiting Pakistan 56 NCT04371393 MSCs in COVID-19 ARDS Phase 3 Randomized/parallel assignment/masking: Triple Remestemcel-L 2 × 106/kg 2 IV 223 Active, not recruiting United States 57 NCT04333368 Cell therapy using UC-derived mesenchymal stromal cells in SARS-CoV-2-related ARDS Phase 1/phase 2 Randomized/parallel assignment/masking: Triple WJ-UC-MSCs 1 × 106/kg 3 IV 47 Active, not recruiting France 58 NCT04447833 Mesenchymal stromal cell therapy for the treatment of ARDS Phase 1 Single group assignment/open label KI-MSC-PL-205 (BMMSCs)1-2 × 106/kg 1 IV 9 Recruiting Sweden 59 NCT04491240 Evaluation of safety and efficiency of method of exosome inhalation in SARS-CoV-2 associated pneumonia Phase 1/phase 2 Randomized/parallel assignment/masking: Double Drug: EXO-1 and EXO- (exosomes from MSCs)0.5-2 × 1010 nanoparticles 20 (2/d for 20 d)Aerosol inhalation 30 Completed Russia 60 NCT04299152 Stem cell educator therapy treat the viral inflammation in COVID-19 Phase 2 Randomized/parallel assignment/masking: Single (care provider)Stem Cell Educator (from UC-MSCs)Not reported 1 IV 20 Not yet recruiting United States 61 NCT04466098 Multiple dosing of mesenchymal stromal cells in patients with ARDS (COVID-19)Phase 2 Randomized/parallel assignment/masking: Triple MSCs 3 × 108 3 IV 30 Recruiting United States Descartes 30 (MSCs RNA-engineered to 62 NCT04524962 Study of descartes-30 in ARDS Phase 1/phase 2 Single group assignment/open label Not reported Not reported Not reported 30 Recruiting United States
COVID-19: Coronavirus disease 2019; SARS-CoV-2: Severe acute respiratory syndrome coronavirus 2; MSCs: Mesenchymal stem cells; UC-MSCs: Umbilical cord mesenchymal stem cells; BM-MSCs: Bone marrow mesenchymal stem cells; AD-MSCs: Adipose tissue mesenchymal stem cells; WJ-MSCs: Wharton’s jelly mesenchymal stem cells; TPE: Total plasma exchange; NA: Not available.
COVID-19 pulmonary fibrosis and MSCs
Many studies interestingly revealed that COVID-19 patients who develop ARDS can survive and might eventually be discharged. On the other hand, a large proportion of COVID-19 patients cannot survive once they develop idiopathic pulmonary fibrosis[54]. Idiopathic pulmonary fibrosis is a progressive lung disease manifested by compromised lung functions with extensive damage to the alveolar cells and capillaries. Patients with severe idiopathic pulmonary fibrosis need lung transplantation to improve pulmonary functions and save their lives[55]. Cytokine storm, which causes oxygen stress and the hyperactive inflammation presenting in the severe stages of COVID-19 infection, is probably the main cause of pulmonary fibrosis. Elevated levels of cytokines such as IL1-β, IL-7, IL-8, IL-9, IL-10, GM-CSF, IFN-γ, MCP1, TNF-α and VEGF have been reported in COVID-19 patients[56-58]. The levels of above-mentioned cytokines with high neutrophil counts are typically more pronounced in patients with severe stages of COVID-19 disease. The development of pulmonary fibrosis in severe cases of COVID-19 is due to the induction of oxidative stress, excessive production of reactive oxygen species (ROS), and the presence of high levels of fibrotic factors mainly TGF-b, FGF, and PDGF[59,60].
MSCs have been introduced as one of the therapeutic approaches in the treatment of COVID-19 pulmonary fibrosis[61]. Golchinet al[61] reported that using MSCs immunomodulatory and differentiation abilities can counteract the cytokine storm and prevent lung tissues death and pulmonary fibrosis by regenerating and reconstructing damaged tissues. Similarly, Yanget al[62] showed that MSCs transplantation in ALI mouse model significantly reduced lung permeability, attenuated lung epithelium apoptosis, facilitated vascular endothelium (VE)-cadherin recovery, and reduced lung injury. These beneficial outcomes were mediated by VEGF released from the transplanted MSCs. Moreover, Junget al[63] reported that injecting MSCs in a LPSinduced ARDS mouse model resulted in the diminution of neutrophil infiltration, collagen deposition and lung fibrosis. MSCs have specific cytokines counteracting potentials to oppose the viral infection and reduce the spread of pulmonary fibrosis. MSCs secrete anti-inflammatory chemokines and cytokines such as IL-10, and PGE2 which help in preventing pulmonary fibrosis[64]. Preclinical studies on animal models with COVID-19 indicated that MSCs considerably improve the disease symptoms, including pulmonary fibrosis. MSCs can secrete a variety of growth factors, including epidermal growth factor (EGF), KGF, PDGF, fibroblast growth factor (FGF), HGF, and VEGF[65]. These factors can recuperate the inflammatory microenvironment of the lung tissues and promote the endogenous lung repair mechanisms and differentiation. Furthermore, MSCs prevent myofibroblast growth by reducing the levels of TGF-β, TNF-α, type I collagen, type III collagen, hydroxyproline, and serum ceruloplasmin in lung tissues, thereby alleviating pulmonary fibrosis[66]. Based on the findings of previous studies, the administration of MSCs in severe cases of COVID-19 appears to be beneficial in resolving and reducing the mortality rate of life-threatening pulmonary fibrosis.
COVID-19 RENAL COMPLICATIONS AND MSCs
Various renal complications have been reported in hospitalized COVID-19 patients, mainly the electrolyte disturbance and acute kidney injury (AKI)[67]. The incidence of renal complications in COVID-19 patients increases if there is an existing kidney diseases,e.g., chronic kidney disease, or a risk factors for kidney complications, including diabetes mellitus[68]. The presence of AKI in COVID-19 patients is associated with worse prognosis and high death rate[69]. In Europe, the death rate in COVID-19 older than 60 years patients was found to be higher when renal complications existed[70]. In severe cases of COVID-19, the rate of continuous renal replacement therapy was significantly higher in North America and Europe compared to other regions[71]. The proposed mechanism of how COVID-19 can induce renal injury is supposed to be through ACE2[72]. It is already known that SARS-CoV-2 (COVID-19) virus attacks the host cells and bindsviaspike proteins (S-protein) to the extracellular enzymatic domain of ACE2 causing its internalization and downregulation. Renal tissues contain ample amount of Ang (1-7) peptides that are produced by ACE2 mediated hydrolysis of AngII[73]. A significant decrease in ACE2 and Ang (1-7) has been found in many kidney diseases, including AKI[73]. Additionally, it has been reported that deficiency of ACE2 by either pharmacological inhibition or genetic knocking out of ACE2 is correlated to the onset of diabetic nephropathy which is manifested by albuminuria and glomerulosclerosis. SARS-CoV-2 mediated down-regulation of ACE2 causes the loss of the beneficial and protective Ang (1-7) peptides and accumulates the AngII[73]. This molecular disturbance in the RAS system by SARS-CoV-2 induces many pathological changes in the kidney, including tubular necrosis and multiple inflammatory infiltrates, endothelial injury and dysfunction, and erythrocytes aggregation which obstructs the capillary lumina (microthrombus formation)[73]. In addition to direct SARS-CoV-2 mediated pathological changes in the kidneys, SARS-CoV-2 invades the alveolar cells and enkindles the activation of many immune cells. These recruited immune cells secrete massive amount of cytokines that circulate systematically inducing endothelial dysfunction and promoting the coagulation and micro thrombus formation resulting in multiple organ failure and septic shock[68]. AKI can occur in severe cases of COVID-19 as a result of this cytokines storm. Interestingly, a study examined SARSCoV-2 viral load in 22 patients who died from COVID-19. It has been reported that 17 out of 22 autopsies showed high load of SARS-CoV-2 in the kidney indicating high mortality rate when COVID-19 was complicated with AKI[74]. Moreover, the coexpression of ACE2 and protease TMPRSS2 in the podocytes and proximal convoluted tubules increases the amplification rate of SARS-CoV-2 in the kidney cells[74]. MSCs have been corroborated by many studies as an effective treatment option for AKI, especially when they are injected as early as possible. Zhouet al[75] found that the transplantation of erythropoietin pretreated BM MSCs in AKI rat model significantly reduced the inflammation and improved the kidney function parameters. Similarly, Zhouet al[75] reported that the infusion of human UC MSC-derived exosomes (HucMSC-Ex) in a mouse model of sepsis-associated AKI was able to restore the renal function parameters and decrease inflammationviathe inhibition of NF-κB activity. HucMSC-Ex transplantation was effective in ameliorating the apoptosis in renal tubular cells and improving survival in mice with sepsis. Caoet al[76] reported that the extracellular vesicles derived from MSCs were capable of stimulating mitochondrial antioxidant defense and ATP production in renal ischemia-reperfusion (I/R) mouse model resulting in recovery of kidney functionviaactivating the Keap1-Nrf2 signaling pathway. Koet al[77] used a hyperbaric oxygen and autologous adipose-derived MSC combined therapy in SD rat model of renal I/R injury. They found that this combination was effective in reducing the expression of inflammatory markers (MMP-9/TNF-α/NF-κB/ICAM-1) and apoptotic markers (mitochondrial-Bax/cleavedcaspase3/PARP), ameliorating oxidative damage and promoting angiogenesis in renal tubular cells which ultimately improved the kidney injury score and restored many renal function parameters, including the blood urea nitrogen and creatinine. Based on the proved potential of MSCs in alleviating AKI and renal I/R injury, we can anticipate that they might exert comparable therapeutic effects in COVID-19 patients with AKI and other renal complications[77]. Future studies should investigate the effectiveness of MSCs-based cell therapy in COVID-19 patients who developed renal complications. Such therapy could be a worthwhile approach in restoring their kidney functions and decreasing the death rate in this group of patients.
COVID-19 CARDIOVASCULAR COMPLICATIONS AND MSCS
Many patients with COVID-19 were reported to develop acute cardiac injury during the course of the illness. Concomitant presence of cardiac events and COVID-19 increases the mortality rate in COVID-19 patients. COVID-19 cardiovascular complications (CV) include acute myocardial injury, heart failure, pericardial effusion with or without tamponade, arrhythmias, sudden cardiac death, and thrombosis of small and large blood vessels. The clinical reports regarding the severity of COVID-19 were noticeable in patients with previous medical history of underlying CV diseases. Furthermore, many patients infected with COVID-19 developed severe myocardial injury[78], including myocardial dysfunction, cardiomyopathy, arrhythmias and heart failure during the course of critical illness[79-84]. Clinical reports revealed that 38% of patients testing positive for COVID-19 display high levels of cardiac damage biomarkers, such as cardiac troponin and creatinine kinase with significant electrocardiographic changes. Data acquired from multiple clinical centers showed conspicuous correlation between plasma lactate dehydrogenase and chronic kidney levels with COVID-19 severity and the rate of ICU[85].
The proposed mechanisms of the deleterious effect of COVID-19 on the heart are intriguing many clinicians and researchers. Acute myocardial injury caused by SARSCoV-2 infection could be due to its high affinity for ACE2 which is extensively expressed in the heart, and is associated with direct myocardial injury and remodeling[22,86]. Another proposed mechanism is linked to cytokine storm. Cytokine storm is triggered by an excessive response of type 1 and type 2 T-helper cells, sympathetic hyperactivity, anemia, and hypoxemic myocardial cells damage caused by respiratory dysfunction [type 2 myocardial infarction (MI)][86]. Myocardial oxygen supply/ demand mismatch as a result of severe pneumonia or ARDS can lead to inadequate supply of oxygen and subsequent myocardial damage[87]. The current treatment regimen, including antiviral medications, corticosteroids, and immunological agents; can also potentiate the risk of developing CV complications. The disturbance of electrolytes can occur in any critical systemic illness, including COVID-19 and can trigger life-threatening arrhythmias. There is a particular concern about the risk of hypokalemia in COVID-19 patients as a result of the interaction between SARS-CoV2 with the RAS system. Hypokalemia is a well-known electrolyte disturbance that is associated with serious arrhythmias[87]. Several studies have demonstrated the possible role of MSCs in CV diseases in non-COVID-19 patients. MSCs can offer the potential as regenerative cells for the CV system where through a paracrine mechanism, they activate endogenous repair mechanisms leading to blood vessel growthviaangiogenesis, improve cardiomyocytes survival, and reduce cardiomyocytes reactive hypertrophy and fibrosis[88]. Treatment with MSCs may give a clinical benefit to patients due to their regenerative and reparative potential if there are significant myocardial injury and myocardial cell death.
The infusion and transplantation of MSCs have been deemed safe for treating MI patients[89,90]. A study was carried out to evaluate the safety and feasibility of intramyocardial MSCs injection in patients, shortly after AMI during short-term and 5-year follow-up, a safety analysis demonstrated one transient ischemic attack without any adverse events related to MSCs treatment[90].
Furthermore, MSCs injection successfully improves some cardiac functional measures post-MI[91,92]. MSCs were shown to restore endothelial function by increasing endothelial progenitor cells function and regain flow-mediated vasodilation (FMD which is clinically significant for heart failure patients[93].
There is study that has suggested a potential effect for in situ myocardial regeneration in ischemic heart failure by delivering allogeneic mesenchymal precursor cells called immunomodulatory progenitor cells (iMP)viaintramyocardial route[94]. Intramyocardial implantation of iMP cells with CABG was safe with preliminary evidence of efficacy of improved myocardial contractility and perfusion of nonrevascularized territories resulting in a significant reduction in left ventricular scar area at 12 mo after treatment. Clinical improvement was associated with a significant improvement in quality of life at 6 mo post-treatment in all patients[94].
A study focused on dose comparison of allogeneic MSCs in patients with ischemic cardiomyopathy demonstrated that the optimal dose of MSCs was 100 million cells, and this optimal dose increases the ejection fraction in ischemic patients[95].
A meta-analysis of 52 pre-clinical animal studies performed on different animal species such as pig, dog and sheep investigated the effect of cardiac stem cells therapy in ischemic cardiomyopathy (52 studies;n= 888 animals). This study reported that MSCs therapy is safe and associated with significant approximately 7.5% improvements in left ventricular ejection fraction (LVEF), without any increased mortality due to MSCs administration[96].
The use of MSCs to treat CV dysfunction and damage in COVID-19 patients has yet to be fully elucidated. The studies over the past decade provide good preliminary evidence for researchers and clinicians alike to further investigate the use of this MSCs therapy in COVID-19 patient cohorts.
CONCLUSION
As the number of COVID-19 cases upsurges rampantly with many patients suffering from life-threatening multiple organ failure, finding a curative way to save the life of critically-ill patients and improve the clinical outcomes is urgently needed. As mentioned above, there are no specific therapeutic medications for COVID-19 serious symptoms and complications, and the applied therapeutic guidelines in the clinical settings are non-specific and based on recommendations. At present, cell therapy is a highly sophisticated approach for treating many diseases, and a large number of studies have been carried out in recent months to treat the SARS-CoV-2 virus using stem cells, particularly MSCs. The safety and therapeutic role of MSCs for ARDS and organ dysfunction that are caused by SARS-CoV-2 infection have been validated by many studies. MSCs can secrete a cocktail of factors that modulate the lung microenvironment, subdue the immune system over-activation, potentiate tissue repair machinery, invigorate alveolar epithelial cells, and inhibit pulmonary remodeling and fibrosis that result in massive improvement in the lung functions. MSCs can regenerate other damaged organs which are associated with COVID-19. Many issues related to the application of MSCs, including the ideal dose and optimum timing of MSCs delivery should be further explored with the objective of enhancing the clinical outcomes in COVID-19 critically-ill patients.
ACKNOWLEDGEMENTS
We would like to thank Mr. Abu-El-Rub M for the time he spent in proof reading and editing of the review manuscript.
杂志排行
World Journal of Stem Cells的其它文章
- Effects of living and metabolically inactive mesenchymal stromal cells and their derivatives on monocytes and macrophages
- Stem cells' centrosomes: How can organelles identified 130 years ago contribute to the future of regenerative medicine?
- Effects of storage media, supplements and cryopreservation methods on quality of stem cells
- Recent advances in stem cell therapy for neurodegenerative disease: Three dimensional tracing and its emerging use
- Stem cell therapies in cardiac diseases: Current status and future possibilities
- Current evidence on potential of adipose derived stem cells to enhance bone regeneration and future projection
