Recent trends in stem cell-based therapies and applications of artificial intelligence in regenerative medicine
2021-07-24SayaliMukherjeeGarimaYadavRajnishKumar
Sayali Mukherjee, Garima Yadav, Rajnish Kumar
Sayali Mukherjee, Garima Yadav, Rajnish Kumar, Amity Institute of Biotechnology, Amity University Uttar Pradesh, Lucknow 226028, Uttar Pradesh, India
Abstract Stem cells are undifferentiated cells that can self-renew and differentiate into diverse types of mature and functional cells while maintaining their original identity.This profound potential of stem cells has been thoroughly investigated for its significance in regenerative medicine and has laid the foundation for cellbased therapies.Regenerative medicine is rapidly progressing in healthcare with the prospect of repair and restoration of specific organs or tissue injuries or chronic disease conditions where the body’s regenerative process is not sufficient to heal.In this review, the recent advances in stem cell-based therapies in regenerative medicine are discussed, emphasizing mesenchymal stem cell-based therapies as these cells have been extensively studied for clinical use.Recent applications of artificial intelligence algorithms in stem cell-based therapies, their limitation, and future prospects are highlighted.
Key Words: Artificial intelligence; Machine learning; Mesenchymal stem cells; Regenerative medicine; Stem cells; Therapy
INTRODUCTION
Stem cells hold great potential in regenerative medicine as these cells are endorsed with indefinite self-renewal characteristics and can be differentiated into any cells of the body.These cells express specific markers and are karyotypically normal.Based on the differentiation potential, stem cells can be categorized as totipotent, pluripotent, multipotent, or unipotent[1,2].Totipotent cells are those that can form the entire organism.In animals, only the zygote is totipotent, and therefore, cannot be used for therapeutic purposes.The pluripotent embryonic stem cells (ESCs) are isolated from the inner cell mass (ICM) of the embryo's blastocyst.These pluripotent cells can differentiate into all cells, including germ cells but not the entire organism.The multipotent cells can generate a definite group of cells,e.g., hematopoietic stem cells (HSCs) or mesenchymal stem cells (MSCs).The unipotent stem cells have restricted potential and can differentiate into a single cell type,e.g., a neuronal stem cell.Stem cells have been classified according to the origin into ESCs and tissue-derived stem cells[3,4].The tissue-derived stem cells may be adult stem cells (ASCs) isolated either from bone marrow, peripheral blood[5], adipose tissue[6], dental pulp[7], skeletal muscle[8], skin[9], neural tissue[10], liver[11], heart[12], pancreas[13] or intestine[14].ASCs have reduced potency compared to ESCs.They are still preferred over ESCs for regenerative medicine because of ethical issues associated with ESCs.The fetal stem cells can be isolated from either fetal tissue like blood, spleen, liver, kidney, or extraembryonic sources like an amnion, amniotic fluid, umbilical cord, Wharton's jelly, and placenta.The HSCs and MSCs isolated from fetal tissues or extraembryonic sources have greater potential than their adult counterparts.Therefore, these cells are considered a "halfway house" between the ESCs and ASCs in terms of plasticity[15,16].Induced pluripotent stem cells (iPSCs) are produced by reprogramming differentiated cells into an undifferentiated state[17].These reprogrammed stem cells are artificially produced cells with properties of ESCs.Induced tissue-specific stem (iTS) cells have recently been produced by incomplete reprogramming and tissue-specific selection[18,19].
Cell therapy applies the biochemical and biophysical properties of stem cells to generate healthy tissues and repair damaged organs, preventing them from further damage[20].The bone marrow serves as a source of stem cells from where HSCs, neural stem cells, and MSCs can be derived for therapeutic purposes[21].These cells (termed adult cells) are non-pluripotent, limiting them to proliferate in only those types of tissues from which they have been isolated.Further advancement in stem cell therapy happened with identifying pluripotency of ESCs in 1998[22].As the generation of ESCs involves the exploitation of embryos, this raised major ethical issues and limited the scope of stem cell-based therapies yet again until the creation of iPSCs in 2007[23-25].Stem cell therapy holds the solid potential for combat transplantation-related issues such as graft rejection or tissue insufficiency and opens the door for precision medicines.Figure 1 depicts regenerative strategies, MSC differentiation, iPSCs, and the role of artificial intelligence (AI) in regenerative medicines.
The non-pluripotent nature of available ASCs has restrained the scope of cell therapy in regenerative medicine.The ESCs have their share of ethical and fewer availability issues.These limitations called for an advancement in cell therapy and led to the creation of iPSCs.The genetic reprogramming of ASCs has imparted them with ESC-like functional similarity and pluripotency, generating iPSCs[26,27].iPSCs have gained popularity in multiple facets of cell-based therapies by serving as an unlimited source of any cell type of interest[28].It has enabled the employment of iPSCs as novel human disease models[29,30], which have been applied to drug discovery as well as the fields of precision medicine and regenerative medicine[31].Several preclinical studies in animal models have established MSC suitability for regenerative medicine[32].Although several clinical trials have been carried out with stem cells to treat genetic disorders, autoimmune diseases, or degenerative disorders, several challenges and limitations are encountered in human clinical translation, which are being addressed to improve the regenerative potential of these cells.
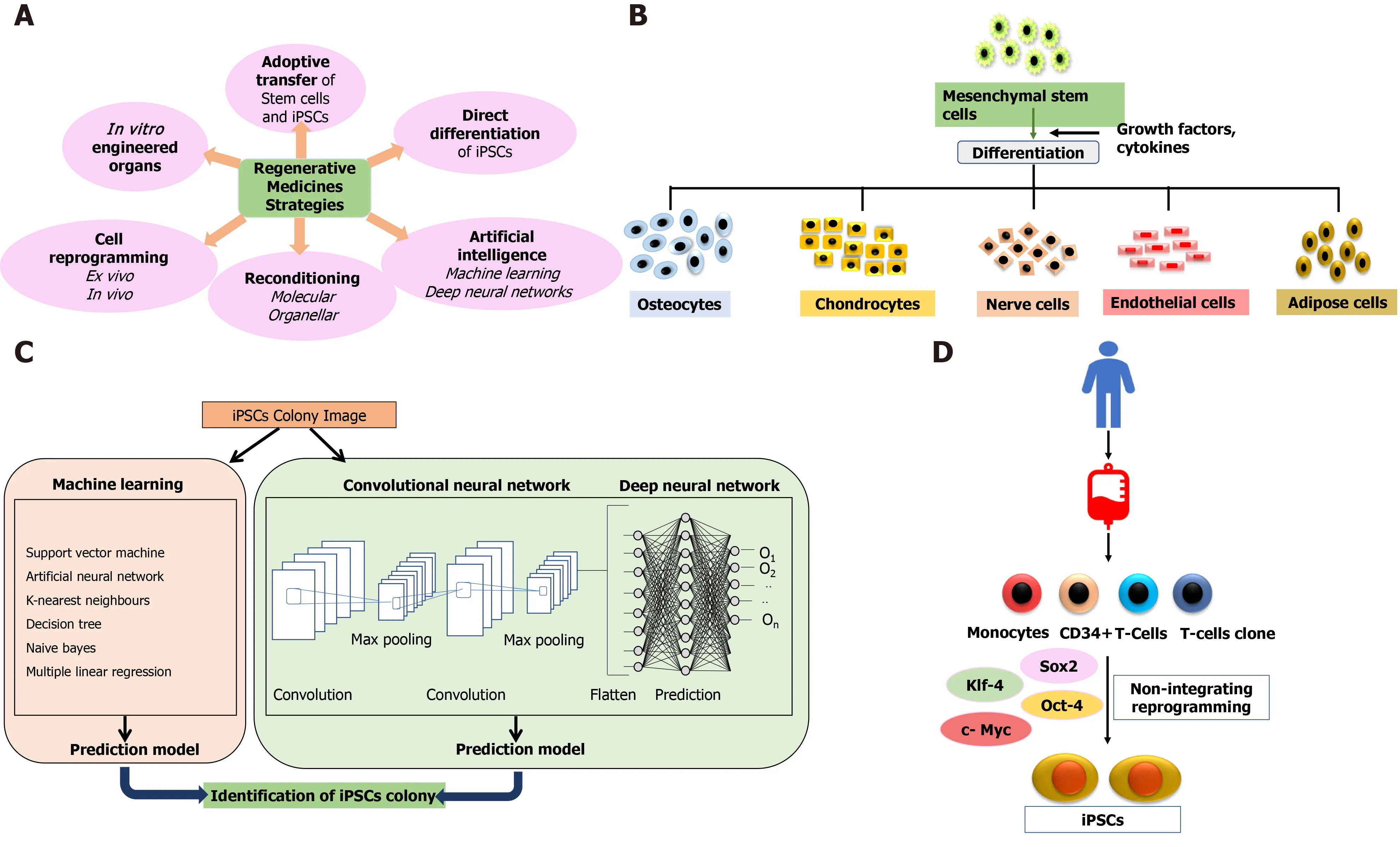
Figure 1 Current strategies and approaches in regenerative medicine.
CURRENT TRENDS IN THE THERAPEUTIC USE OF STEM CELLS
ESCs
ESCs exhibit pluripotency and differentiate into the three germ layers: ectoderm, mesoderm, and endoderm.These cells are derived from the ICM layer of the embryo's blastocyst.Mouse ESC was first derived in 1981 by Evans and Kaufman[33] in the United Kingdom and Martin[34] in the United States from the ICM of the blastocyst (d2.5).Human ESCs (hESCs) were derived by Thomson and colleagues isolated from preimplantation blastocysts[22].hESCs have been an excellent source of pluripotent cells for therapeutic use[35].Derivation of pluripotent ESCs from the blastocyst’s ICM layer is usually done by a standardized immunosurgery technique[36].The ESCs are isolated and seeded on feeder layers in culture plates.The cells may be characterized for pluripotency markers by immunostaining using specific antibodies to octamerbinding transcription factor ¾ (Oct3/4), stage-specific embryonic antigen 3 (SSEA-3), SSEA-4, TRA-1-60, and TRA-1-81 or by assessing alkaline phosphatase activity.ESCs are karyotypically normal and possess a high telomerase activity[37,38].However, there are severe concerns in using ESC in regenerative medicine despite being a promising candidate.Severe ethical concerns prevail in using human embryos for the isolation of hESCs[39].Different legal guidelines are governing ESC research in various countries.In the United States, the destruction of human embryos for any form of research is banned.According to the guidelines, hESC-lines derived before August 9, 2001 can be used for research.The development of hESC therapies is restricted, and most research studies are focused on animals[40].In the United Kingdom, research using hESCs derived from discarded embryos in in vitro fertilization clinics is allowed; however, hESC research is prohibited in Italy[41-43].
It is also essential to analyze the safety issues associated with ESCs in regenerative medicine.The ESCs can differentiate into any cell type of the body.However, when these undifferentiated cells are implantedin vivo, this plasticity poses the risk of developing teratomas and tumors[44-47].The alternative method is to differentiate the undifferentiated ESCsin vitrointo a specific cell type along a lineage and then transplant the differentiated cellin vivo.hESC-derived cardiomyocytes, when transplanted in mice, did not result in teratoma[48].However, in certain instances, the transplanted progenitor cells continue to proliferate, as, for example, nestin+ dopaminergic neurons derived from hESCs continue to proliferate in the striatum[49].The screening of the undifferentiated cells by specific markers and their subsequent purification before transplantation may solve the problem.
There are some reported methods to overcome the risk of tumorigenesis, such that ESCs can be used for regenerative therapies[50].One of the recent findings involves cluster of differentiation 133 (CD133) (prominin 1), a transmembrane protein generally expressed on cancer stem cells is highly expressed on hESCs.CD133-deficient knockout hESC line retained the capacity to differentiate into the three embryonic germ layersin vivo.Still, the proliferating potential is reduced and results in reduced teratoma formation[51].Therefore, CD133 may be used to sort ESCs for transplantation[52].
Despite the safety issues, hESC-derived progenitor cells are still considered promising candidates in regenerative medicine under controlled conditions[53].The first approval of the hESC trial for spinal cord injury was received in 2009, in which hESC-derived oligodendrocytes progenitor cells were used[54].The hESC-based clinical trials have been performed for the treatment of macular degeneration with some positive results in follow-up studies[55-58], diabetes mellitus[59], and ischemic heart disease[60].One clinical trial has been approved for Type 1 diabetes produced by the company ViaCyte (ClinicalTrials.gov Identifier: NCT03163511, NCT02239354)[61].These pancreatic progenitor cells are produced from human pluripotent stem cellsin vitroand differentiate into beta cells after transplantation in an immune isolation devicein vivo[62].Clinical trials for Parkinson’s disease with hESCs are being conducted in Australia (NCT02452723) and China (NCT03119636)[63].
iPSCs
iPSCs were first successfully generated by Takahashi and Yamanaka[64] in 2006 by inserting the reprogramming factors known as “Yamanaka factors,” Oct4/3, Sox2, Klf4, and c-Myc in mouse fibroblast cells.Initially, Yamanaka and his group (Okitaet al[65]) started transducing mouse fibroblasts with a recombinant retrovirus carrying 24 genes responsible for maintaining the ESC characteristics.The mouse fibroblasts were selected by antibiotic-resistant gene cassette under the promoter, Fbx15, which is active only in the ESCs.The number of genes was narrowed down to ten and finally to four genes, Oct3/4, c-Myc, Sox2, and Klf4.The first generation induced pluripotent cells selected by Fbx15 possessed unlimited self-renewal and differentiation capacity, produced embryoid bodies and fetal chimeras but failed to produce adult chimeras.However, the DNA methylation pattern, post-translational modifications, and epigenetic changes revealed that the generated iPSCs were intermediate between fibroblasts and ESCs.Yamanaka and his group further generated second-generation iPSCs using the selection for Nanog instead of Fbx15 selection.The second-generation iPSCs showed greater ES cell-like characteristics, DNA methylation pattern, and germline competence[65].However, 20% of the chimeric mice developed cancer as two of the genes, c-Myc, and Klf4, are oncogenic.Human-induced pluripotent cells were generated from somatic cells in 2007 by two independent groups simultaneously by introducing Oct3/4 and Sox2 with either Klf4 and c-Myc or Nanog and Lin28[66].The latter group has shown that reprogramming of human somatic cells is possible even when the reprogramming factors are not integrated into the genome.The use of nonintegrated episomal vectors makes these cells more suitable for clinical use[67].iPSCs have also been derived from peripheral blood mononuclear cells[68].Adenovirus has also been used vector for the delivery of reprogramming factors.
However, virus-mediated delivery systems sometimes threaten iPSCs' clinical use due to insertional mutagenesis, mainly caused by cMyc[69-71].RNA, proteins, and small molecules enhance iPSCs' efficiency and safety.Reprogramming of somatic cells by mRNA or microRNA became very successful and effective[72-75].It has been reported that activation of the innate immune system enhances the efficiency of induced pluripotent cells by mRNA transfection[76].Insertion of recombinant proteins such as Oct 4, sex-determining region Y-box 2 (Sox2), Kruppel-like factor 4 (Klf4), and cMyc can be used to reprogram somatic cells to induce pluripotent cells[77-79].The protein-based approach has been used to generate dopaminergic neurons from iPSCs to treat Parkinson’s disease in rats[80].
The iPSCs have potential similar to that of ESCs, and additionally overcome the ethical concerns associated with ESC research and clinical use.The iPSCs have gained much attention in recent years because of their advancement in regenerative medicine, organoid formation, and scope for personalized therapies.The first human clinical trial with iPSCs was done for macular degeneration in which retinal pigmented epithelial cells generated from autologous iPSCs were transplanted into a patient[81].There have been severalin vitro[82,83] andin vivopreclinical studies investigating the safety and efficacy of iPSCs[84-87].A human clinical trial was recently conducted to treat Parkinson’s disease in Kyoto, Japan, by transplanting dopaminergic neurons generated from iPSCs.The clinical-grade iPSCs are produced from cells taken from healthy volunteers[88].Autologous iPSC-derived dopaminergic neurons transplanted in a patient with Parkinson’s disease retained the function until 2 years without any adverse effect[89].Autologous iPSCs are more advantageous to avoid immunological rejection, but the development of autologous iPSC is time-consuming and costly.
The iPSC technology has enormous potential in regenerative medicine.However, more interventional studies[90] must be conducted to address the challenges of routine clinical applications of these cellse.g., genomic instability[91], carcinogenicity[92], immunological rejection[93].Humanized mouse models,e.g., a mouse with human immune cells, may be developed in the future to investigate the immunogenicity of human pluripotent stem cells[94].
iTS
iTS are produced by incomplete reprogramming of somatic cells by transient overexpression of reprogramming factors by plasmids and performing a tissue-specific selection.These cells have the potential to self-renew but also express tissue-specific markers.iTS have been produced from mouse pancreatic cells, which can self-renewal and express pancreatic tissue-specific transcription factor, Pdx1[13].These generated iTS can differentiate into insulin-producing cells more efficiently than ESCs or iPSCs and, probably, can be utilized to treat diabetes.iTS with neural stem cell-like characteristics have also been reported[95-98].Teratoma formation is not reported when iTS are transplanted in nude mice.In this respect, iTS are advantageous over the ESCs or iPSCs in terms of clinical applications due to the risk of tumorigenicity associated with the use of pluripotent stem cells.These cells are “incompletely reprogrammed” cells and have different methylation patterns than ESCs or iPSCs.The iTS retain the donor tissue’s epigenetic memory and can be explored as a potential candidate for cell replacement therapy.
The wine that she had given to John in the castle of the goblins, the bouquet of immortelles, and the scarf, all had the power of producing sleep like death
Fetal stem cells and ASCs
Fetal stem cells are collected from aborted fetal tissues and extraembryonic structures like amniotic fluid, umbilical cord, Wharton’s jelly, and placenta[99].The fetal stem cells are multipotent HSCs or MSCs.HSCs collected from fetal bone marrow or umbilical cord express CD34 and CD45 Like adult HSCs but show greater proliferating capacity, low immunogenicity, and lower risk of graftvshost disease (GvHD) compared to adult HSCs.Fetal MSCs can be isolated from fetal blood, bone marrow, liver, lung, or pancreas.These cells have more differentiation capacity than the adult MSCs.Fetal MSCs have active telomerase and express low levels of human leukocyte antigen (HLA) I and lack intracellular HLA II[100].First trimester fetal MSCs express baseline levels of pluripotent stem cell markers such as Oct4, Nanog, Rex1 SSEA3, SSEA4, Tra-1-60, and Tra-1-81.Umbilical cord blood MSCs are easy to harvest and can be stored under controlled conditions for longer periods for future clinical use[101].Although the ethical concerns associated with fetal stem cells are minimal, there are several reports on the adverse effects of fetal stem cellsin vitroin animals and humans[102].Thromboembolism has been reported in patients who received transplantation of umbilical cord blood MSCs[103].Placental-derived MSCs express higher levels of tissue factor[104,105], which aggravates the thrombotic events in patients infused with these MSCs for treatment of Crohn’s disease[106].The ASCs are less prevalent and undifferentiated cells present in various adult tissues with a primary role of repair and maintenance of residing tissues.
HSCs
ASCs are derived from different tissues and have limited potency compared to the ESCs or iPSCs.ASCs are named depending on the tissue of origin,e.g., HSCs, pancreatic stem cells, corneal stem cells,etc.HSCs were the first multipotent ASCs isolated from the bone marrow[107].HSC transplantation is used as therapy for several malignant and non-malignant disorders and autoimmune diseases.These cells are also used for the recovery of patients undergoing chemotherapy and radiotherapy[108].The allogeneic transplantation requires matching HLA, Class I, and Class II between donor and recipient[109]; however, there are risks of GvHD[110].The emergence of more modern and less toxic methods of treatments replaces HSC transplantation in hematologic malignancies.Recent reports suggest that risks of bloodstream infections caused by Gram-negative bacteria are associated with allogeneic hematopoietic transplantation[111,112].Hemorrhagic cystitis is another complication that has been reported in patients post-HSC transplantation[113].
MSCs
Cell-based therapy using MSCs is currently an essential domain of research.MSCs may be isolated from various sources, including bone marrow, adipose tissues, dental pulp, peripheral blood, synovium, and extraembryonic sources, as described earlier.These cells are plastic adherent and multipotential and can differentiate into bones, cartilage, fat tissues, muscles, and tendons[114].The International Society for Cellular Therapy defined MSCs as plastic adherent cells expressing CD73, CD105, CD90 (≥ 90%), and not expressing hematopoietic markers CD34, CD45, CD14, CD19, and HLADR (≤ 2%) and have the potential of multilineage differentiation to osteogenic, adipogenic and chondrogenic lineage[115].There is, however, no single marker to identify MSCs from various sources[116-118].Figure 2 shows immune-modulatory characteristics and the ability of differentiation of MSCs.
The three primary characteristics of MSCs are: Differentiation into specific cell types and their incorporation into tissues that make it suitable for regenerative medicine; secretion of cytokines and exosomes that stimulate cell growth and proliferation and modulate inflammation; and direct contact with the host tissue and regulate effector function[119,120].MSCs are therapeutically more successful due to the multipotentiality, immunomodulatory, anti-inflammatory, efficient homing capacity to injured sites, and minimum ethical issues[121-123].However, there are disparities in MSC potency and pharmacological functionality, depending on tissue sources, cell handling, method of harvest, cultural expansion, dose, and route of delivery[124].
The most important sources of MSCs for clinical trials are bone marrow and adipose tissues[125].Although MSCs isolated from the bone marrow have been extensively studied.However, there are several challenges in the clinical use of these cells.Cells collected from bone marrow are contaminated with HSCs, and a very small fraction (0.001%-0.01%) of MSCs are harvested from bone marrow.The cells isolated also show early signs of senescence during culture[126].Although the collection of bone marrow aspirate is considered safe, certain complications and morbidity have been reported during collection from the sternum and posterior iliac crest[127].Adipose tissuederived stem cells are another potential source of MSCs by less invasive procedures[128].Adipose tissue is abundant in the body and can be easily collected by liposuction, and the yield of stem cells is comparatively greater without any adverse effects.Adipose tissue-derived stem cells have more stability in the culture and have greater differentiation potential to osteocytes, chondrocytes, adipocytes, cardiomyocytes, and neurocytes[129].Several studies have reported adipose tissuederived MCSs' efficacy and safety for regenerative medicine[130,131].The other sources include dental pulp, tendon, or from the perivascular fraction of any tissue[132].
MSCs have immunomodulatory properties and can reduce the inflammatory response.These cells can modulate the function of the innate and adaptive immune response.MSCs have very low levels of major histocompatibility complex and reduced expression of FasL (Fas Ligand) or costimulatory signals like B7, CD40, or CD40L[133].MSCs secrete extracellular vesicles containing various growth factors and cytokines that suppress B lymphocyte and T lymphocyte function and maturation of dendritic cells while activating T regulatory cells.MSCs also secrete angiogenic, antiapoptotic, and antioxidative effects[134].Table 1 summarizes the discovery time, sources, advantages, disadvantages, current clinical applications and prospects of various stem cells.
TRENDS IN APPLICATIONS OF MSC-BASED THERAPIES IN REGENERATIVE MEDICINE
MSCs have been utilized for preclinical and clinical studies for a wide range of diseases owing to multipotentiality, immunomodulation, and regeneration.The first clinical trials involved the infusion of MSCs post high dose chemotherapy and reversing GvHD, which is resistant to steroids[135].To date, numerous clinical trials have been undertaken using MSCs for various diseases like myocardial infarction, Crohn’s disease, multiple sclerosis, diabetes, GvHD, amyotrophic lateral sclerosis, arthritis, neurodegenerative disorders, trauma, coronavirus disease 2019, and many more[136] (Table 2).According to recent reports, 10000 patients have undergone treatment with MSCs for different diseases, and 1094 clinical trials are registered at present in different phases[137].Due to the relative ease of isolation and efficacy, the most prevalent source of MSCs for therapeutic purposes remains bone marrow, followed by the umbilical cord and then adipose tissue.Placental MSCs represent less than 2% of all clinical trials, and then MSCs from other sources are utilized[138].
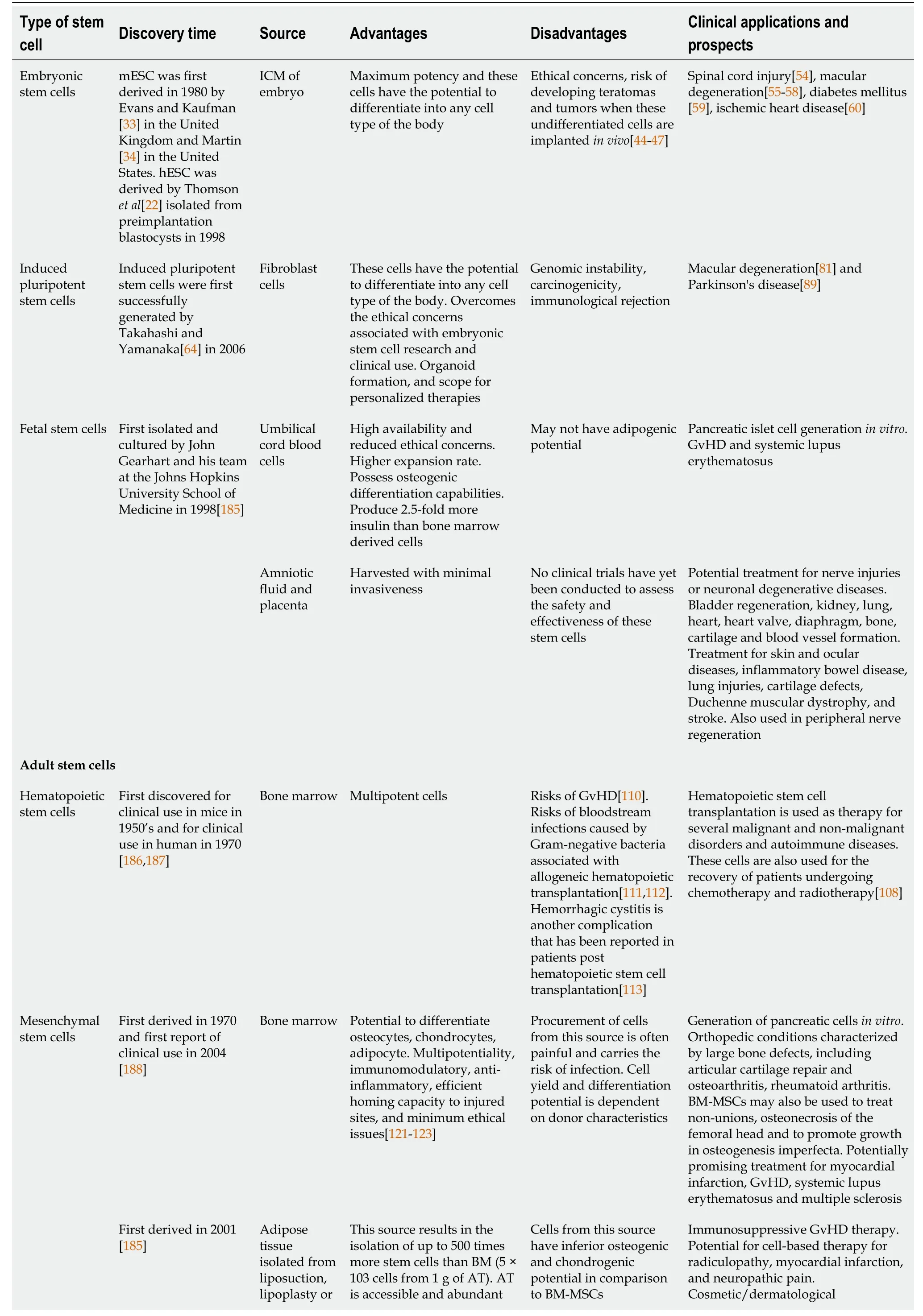
Table 1 Summary of the clinical applications of different types of stem cells

AT: Adipose tissue; AT-MSCs: Adipose-tissue derived mesenchymal stem cells; BM-MSCs: Bone marrow derived mesenchymal stem cells; GvDH: Graft vs host disease; hESC: Human embryonic stem cell; ICM: Inner cell mass; mESC: Mouse embryonic stem cell.

Table 2 Status of mesenchymal cell-based therapies for different diseases
RECENT AI-BASED MODELS FOR STEM CELLS THERAPY
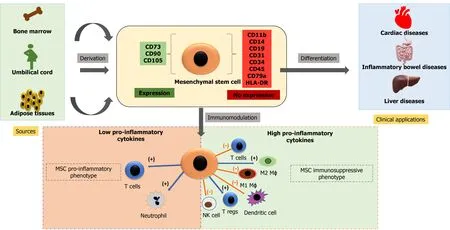
Figure 2 Characteristics and therapeutic potential of mesenchymal stem cells.
The conventional approach of regenerative medicine and cell-based therapies faced multiple challenges related to the enormous amount of data analysis, rising quantity and complexity, standardization methods, manual errors, and extremely difficult data handling.This further complicates the process of conclusion derivation and decisionmaking practices without the risk of errors involved.The implementation of iPSCs in different experimental and therapeutic approaches requires identifying iPSC-derived cells, a thorough estimation of iPSC quality, and characterization of the cell type.Evaluation of colony morphology through a manual approach is a tedious and errorprone process, and it is not feasible for large-scale cultures.Various study groups have implemented different AI algorithms to install an automated approach for accurate segmentation and colony quality estimation to overcome this limitation.
The two AI algorithms which are most widely adopted in the healthcare industry are machine learning (ML) and deep learning (DL) (Figure 3)[160,161].ML refers to a data-based algorithm that allows the software to learn from first-hand information and become "intelligent" enough to perform predictive analysis and classification without being programmed for it[162].The ML learning methods have been categorized into three types: supervised, unsupervised, and reinforcement learning.Supervised learning holds most of the ML tasks, and its training dataset consists of class labels.The label is a specific known outcome of interest, and the ML algorithm finds the best way to predict that outcome[163].Unsupervised learning predicts the outcome without any prespecified labels and predicts the unknown patterns within the data.The supervised ML algorithms used in the field of medicine commonly include artificial neural network (ANN), support vector machine (SVM), naïve bayes, random forest (RF), k-Nearest Neighbors, decision tree (DT), and adaptive boosting (Adaboost),etc.[164].Among these, ANN is the most widely used ML algorithm alongside SVM.ANN is a simulation of the human neuronal structure where neurons serve as the basic unit for communication and are arranged in sequential layers, with varying strength connections between layers.The input layer receives the training signal in pixel series or a speech series, relayed through the hidden layer where the data is analyzed.The concepts within the data are extracted to make the predictions and passed on to the output layer.The output layer further refines the data by performing classification/regression tasks[165].DL's revolutionary concept emerged with an increased number of hidden layers, making it a more complex and, subsequently, even more, advanced concept of AI.DL techniques utilize these massive hidden neuronal units to automatically learn the complex relationship among the raw and noisy data, eliminating the tedious manual feature extraction required in ML algorithms[166].Convolutional neural network (CNN) is a DL network robustly implemented in medical-imaging to perform image-classification and disease diagnosis tasks[167].Figure 4 illustrates the general schema for AI-based prediction model development.
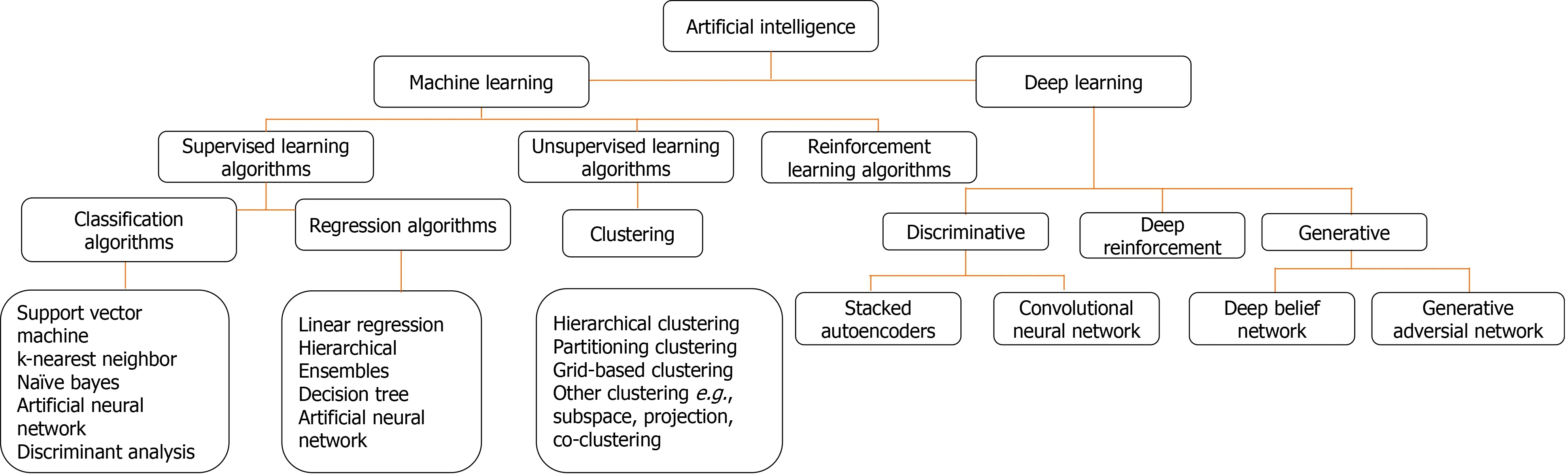
Figure 3 Commonly used machine learning and deep learning algorithms.
Joutsijokiet al[168] conducted a study focusing on an automated approach to identify iPSC colony images with 1608 × 1208 resolution by using ML classifiers- SVMs and k-NN.The authors also used scale-invariant feature transform descriptors in feature extraction.The k-NN classifier with Euclidean measure and equal weighting yielded the best result with an accuracy of 62.4%, which was a measured value compared to previous studies.
Kavithaet al[169] conducted a study to evaluate different automated texture features extracted from the segmented colony sections of iPSCs and use ML techniques to confirm their potential for characterization of colonies.They quantified 151 features obtained from the iPSCs images from phase-contrast microscope using moment-based, shape-based, spectral texture feature and statistical groups.A forward stepwise regression model was used to select the most relevant features for categorizing the colonies.Five ML classifiers- SVM, RF, MLP, Adaboost, and DT were used with 10-fold cross-validation to estimate the texture features within each texture-feature group and fused-feature groups to characterize diseased and healthy iPSC colonies.Based on one of their findings, SVM, RF, and Adaboost classifiers were concluded to exhibit superior performances compared to MLP and DT.
Several studies have reported the implementation of the DL algorithm through CNNs in iPSC studies.Kavithaet al[170], in their study, developed a vector-based CNN (V-CNN) using extracted features of the iPSC colony for finding out colony characteristics.They compared the V-CNN model with an SVM classifier using textural, morphological, and combined features.The study applied 5-fold crossvalidation to examine the V-CNN performance.The precision, recall, and F-measure values were comparatively much higher than SVM (87%-93%).The V-CNN model was also subjected to determine the colony quality where the accuracy values on textural (91.0%), morphological (95.5%), and combined features (93.2%) bases were also found to be much higher than those of SVM values, which were 83.3%, 87.6%, and 83.4% respectively.In another study by Kusumotoet al[171], CNN's were utilized to identify iPSC-derived endothelial cells without immunostaining or lineage tracing.They obtained a dataset of 200 images from four experimental setups, of which 64 were applied for training alongside 160 for testing purposes.iPSC-derived endothelial cells' morphological descriptors (Phase-contrast images-based) were used to train the network.Its prediction validation was done by comparing with immunofluorescence staining for CD31, which is an endothelial cells marker.The method parameters were iteratively and automatically improved to obtain an error-free prediction.It was found that prediction accuracy was a function of the pixel size of the images and network depth in question.The k-fold cross validation also suggested that morphological features alone could be enough for optimizing CNNs, and they can deliver a highvalue prediction.The next year, Waismanet al[172] used CNNs to separate pluripotent cells from initial differentiating cells.The authors used light microscopic images of PSCs to train the CNN model.Images of mouse-embryonic cells induced to epiblastlike cells were taken at different intervals after induction.The findings suggested that CNN can be trained to distinguish among differentiated and undifferentiated cells with an accuracy of 99%.

Figure 4 General schema for artificial intelligence-based prediction models development.
The well-established role of iPSCs in disease modeling and AI in therapeutics has also been exploited in several studies.Juholaet al[173] used iPSC-derived cardiomyocytes to study drug effects and their calcium transient signals with ML.Six iPSC-lines containing various catecholaminergic polymorphic ventricular tachycardia causing mutations were used to assess the drug-effect.The drug being studied was dantrolene after adrenaline stimulation by ML analysis of Ca2+signals.
They identified the beats of transient signals with a previously proposed analytical algorithm[174], which recognizes signal abnormality depending upon whether the assessed cell signal has at least one abnormal transient peak-based on characteristics of a single peak.They computed 12 peak-variables for all identified signal-peaks.These data were used to classify the signals into various classes that correspond to those influenced by dantrolene or adrenaline.The algorithm's best classification accuracy was found to be nearly 79% suggesting a significant role of ML in the analysis of iPSCcardiomyocytes drug effects.
In a similar study, Hwanget al[175] employed advanced ML techniques with an Analytical Algorithm to build an analytical pipeline for automatic evaluation of Ca2+transient anomaly in cardiomyocytes.The pipeline was made up of peak detection, peak and signal abnormality assessment, and peak and signal variable detection.A peak-level SVM classifier was trained by using manual expertise.Two hundred cells were used as training data to train the SVM (cell-level), and other datasets of 54 cells were used to test the accuracy.The training and test accuracies were found to be 88% and 87%, respectively.
In a recent study, Nishinoet al[176] developed a linear classification learning model to differentiate between iPSCs, ESCs, somatic cells, and embryonal carcinoma cells based on DNA methylation profiles.The accuracy of the ML model in identifying various cell types was found to be 94.23%.Furthermore, component analysis of the learned models identified the distinct epigenetic signatures of the iPSCs.Studies about recent AI-based stem cell therapies are summarized in Table 3.
Apart from the above-mentioned studies that demonstrate applications of AI in cell culture stages, sufficient evidences are available that AI could play a significant role in predicting the MSC’s therapeutic outcomes[177-179].Precise therapeutic outcome prediction of the MSC therapy could provide vital information for clinicians to assist them in decision support and decide the optimized treatment strategies.AI algorithms could be applied to optimize the clinical trials of innovative stem cell therapies for various diseases by precise treatment-planning for patients, clinical outcomes prediction, and patient recruitment, thereby reducing the complexity of the study and overall costs[180].Machine and human intelligence together could have an exponen-tially high impact on the continual progress of stem cell-based therapy.
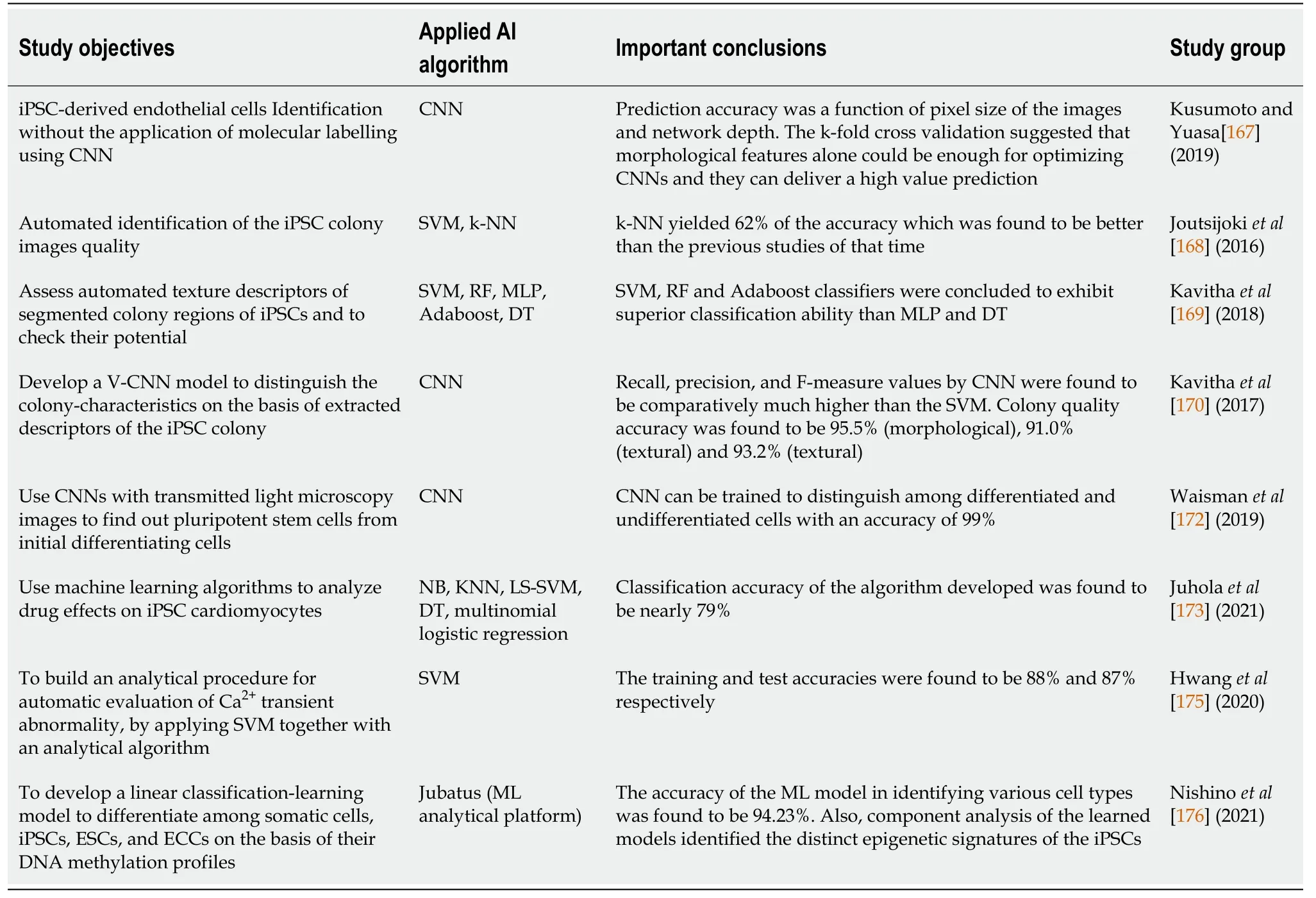
Table 3 Summary of recent artificial intelligence-based stem cells therapies
CONCLUSION
Research gaps and future prospect
Regenerative medicines offer enormous potentials for better treatment of patients and quick recovery.However, there are certain drawbacks due to inefficient production, lengthy and complex processes, and human errors due to excessive human efforts.Understanding genetic components that influence the development of shape, size and orientation of an organ is extremely vital.Although the mechanism of most of the regenerative models is available with diagrams depicting the gene regulations, however, the stepwise dynamics to produce a particular shape of an organ are lacking.The AI-driven models and constructive algorithms could be a powerful solution for a deeper understanding of such mechanisms.These models could make the development of regenerative medicines automated and minimize human error factors.
The regenerative medicines' manufacturing has its own set of challenges, viz.efficient, cost-effective, large-scale production, lack of automation and quality control systems, and absence of closed and modular systems[181].
A vast number of datasets are published every year for experimental regenerative biology, but there is no international guideline for standard and high-quality datasets for regenerative medicines.Also, the tools for analyzing available datasets to get deeper insights and meaningful patterns are lacking.There have been some limited non-AI-based computational methods, platforms, and tools available to assist regenerative therapies.In an effort to derive such computational methods, Lobo and Levin developed a computational method to understand the physiological controls in planarian regeneration[182].The exceptional ability of planaria to regenerate its body parts could be a potential model for leading regenerative medicine research.Another computational platform, KeyGene[183], can predict the tissue origin of various cell types.It could find out the equivalent stage of human PSC differentiation products along with the identification of stem cell derivatives.The KeyGene algorithm applies next-generation sequencing and microarray datasets and could be used to predict human adult tissue identity.This tool also helps monitor the cell-differentiation conditions and evaluate in-vitro cell-differentiation efficacy, thereby fetching improved protocol outcomes.CellNet[184] is among the recent computational tools that provide cell identity parameters, evaluate cell fate conversions, and rank suitable candidates for future interventions.However, it cannot differentiate between cell subtypes and cell heterogenicity, which remains a challenge to solve to date.
Nevertheless, AI-driven methods have emerged as an important component of stem cell research.Over the last decade, AI algorithms have advanced very rapidly, and along with enormous progress, methods to apply them have also enhanced subsequently.Many algorithms and tools can be expected in the recent future, which could efficiently assist stem cell-based regenerative medicines development, outcome prediction, and decision support to healthcare providers.
杂志排行
World Journal of Stem Cells的其它文章
- Genome engineering and disease modeling via programmable nucleases for insulin gene therapy; promises of CRISPR/Cas9 technology
- Immunotherapy in the treatment of lymphoma
- Epigenetic regulation of autophagy: A key modification in cancer cells and cancer stem cells
- Review of the potential of mesenchymal stem cells for the treatment of infectious diseases
- Growing and aging of hematopoietic stem cells
- Therapeutic potential of periodontal ligament stem cells
