Screening and Identification of Strains Converting Trans-anethole to Anisic Acid
2021-07-08JianglinYINXuejianLIXueyingZHENGYuyanFENGJianZHANG
Jianglin YIN Xuejian LI Xueying ZHENG Yuyan FENG Jian ZHANG

Abstract [Objectives]This study was conducted to isolate and screen the bacteria that can convert trans-anethole to anisic acid from star anise and its environmental samples, and identify the bacteria. [Methods]According to the traditional microbial culture method, with trans-anethole as the sole carbon source, through enrichment culture and separation and purification, preliminary screening by thin layer chromatography and re-screening by high-performance liquid chromatography, strains that degraded trans-anethole to produce anisic acid were obtained, and 16S rDNA sequencing and phylogenetic tree construction were performed for genetic analysis. [Results]Eleven strains that degraded trans-anethole to produce anisic acid were obtained, among which strain NT2 that produced anisic acid with a relatively high efficiency was initially identified as Pseudomonas sp. The strains trans-anethole degradation rate was 45.41%, and the molar production rate and cumulative concentration of anisic acid were 21.80% and 1.96 g/L, respectively. [Conclusions]Strain NT2 has a strong ability to degrade trans-anethole to produce anisic acid, and can enrich strain resources for degradation of trans-anethole to anisic acid through microbial conversion.
Key words Trans-anethole; Anisic acid; Strain screening; Microbial conversion
Star anise is the dried and mature fruit of Illicium verum of Magnoliaceae, which is a characteristic Chinese medicine and spice resource in southern China. It has important economic value and is mainly distributed in Guangxi, Guangdong, Yunnan and other places[1]. The chemical name of trans-anethole is trans-1-methoxy-4-(1-propenyl) benzene, which is the main component of star anise oil extracted from star anise fruit[2]. Anisic acid with high added value can be obtained through oxidization of trans-anethole[3]. The chemical name of anisic acid is 4-methoxybenzoic acid, which is an intermediate of various food spices, flavors and medicines[4]. Methyl anisate obtained by esterification of anisic acid with methanol is a spice used in wines and cosmetics; ethyl anisate obtained by esterification with ethanol can be used to formulate perfumes, flavors and food additives, etc.[5-6]; and the p-methoxybenzoyl chloride synthesized with anisic acid as a raw material can be used for the synthesis of amiodarone, aniracetam and benzbromarone and other drugs[7-9].
At present, the preparation methods of anisic acid mainly include chemical oxidation method[10], indirect electrolysis method[11], and chemical synthesis method[12]. These preparation methods are costly, harsh in process conditions, and easy to produce waste liquid to pollute the environment. The microbial conversion method is an efficient, economical, and environmentally friendly preparation method, which is widely used in the fields of food processing, biopharmaceuticals, cosmetics research and development, and new energy development[13]. At present, there are few domestic and foreign research reports on the degradation of trans-anethole to produce anisic acid by microorganism conversion. Shimoni et al.[14-15]isolated the Arthrobacter bacterium TA13, which can use trans-anethole as the sole carbon source, from soil, and initially explored the degradation pathway of trans-anethole. Ryu et al.[16]detected anisic acid, anethole epoxy compounds, and p-hydroxybenzoic acid from the trans-anethole conversion fluid of Pseudomonas putida JYR-1, and initially explored the metabolic pathways of its mutants to degrade anethole. Related research at home mainly focuses on the selection of strains degrading trans-anethole and fermentation optimization to explore new ways for deep processing of trans-anethole, so as to increase the economic value of anise. Su et al.[17-18]selected Pseudomonas sp. BT-13, which can degrade trans-anethole to produce anisaldehyde and anisic acid. After investigating the single factors one by one, a medium formula and fermentation conditions that can increase the production rate of anisic acid were obtained. Pseudomonas putida WGBF9 screened by Wang et al.[19]has high tolerance to trans-anethole and can transform trans-anethole into anisic acid. Shen et al.[4]and Zeng et al.[20]significantly increased the production rate of anisic acid combining the single factor method and response surface method. Lu[21]screened and obtained a mold ZZ-1 with high trans-anethole degradation rate, but its molar production rate of anisic acid was relatively low, only 9.21%.
In this study, through the traditional microbial culture method, the strains that can degrade trans-anethole and produce anisic acid were selected from the star anise and its environmental samples in the Gaofeng Forest Farm in Guangxi. This study is of great significance to the deep processing of trans-anethole.
Materials and Methods
Experimental materials
Main reagents
Standard trans-anethole (99%+, chromatographically pure); anisic acid (99%+, chromatographically pure); anisaldehyde (99%+, chromatographically pure), purchased from Shanghai Yuanye Biotechnology Co., Ltd.; substrate trans-anethole (99%), purchased from Macklin; acetonitrile and methanol (chromatographically pure), purchased from Thermo Fisher Scientific; anhydrous ethanol (analytical pure), purchased from Tianjin Fuyu Fine Chemical Co., Ltd.; other chemical reagents, all of which were domestic reagents, analytically pure or chemically pure.
Main instruments
Constant temperature incubator (Shanghai Jinghong Experimental Equipment Co., Ltd.); PCR instrument (Bio-Rad, USA); Waters e2695 high performance liquid chromatograph (Waters); SW-CJ-2F clean bench (Airtech); HVA-110 fully automatic sterilization pot (Japan Hirayama Company).
Medium
Modified M9 medium [15]: Na2HPO4·12H2O 17.14 g, KH2PO4 3 g, NH4Cl 1 g, NaCl 0.5 g, MgSO4·7H2O 1 g, FeSO4·7H2O 0.02 g, CaCl2 0.02 g, distilled water 1 000 ml. Enrichment medium: modified M9 medium+a certain amount of trans-anethole. The solid media were added with 1.5 g/L agar powder. All media were sterilized by autoclaving at 0.1 MPa for 20 min. The three components of MgSO4·7H2O, FeSO4·7H2O and CaCl2 in the media were sterilized by filtration and added separately.
Experimental methods
Sample collection and pretreatment
The five-point sampling method was used in the star anise planting area of the Jiepai branch of Gaofeng Forest Farm in Guangxi to collect 5 parts of star anise tree root soil, decayed root silt, and bark, respectively. The samples were stored in storage bags, and taken back to the laboratory. After mixing and weighing, three groups of samples of soil, silt and bark each of which was 30 g, were obtained.
Enrichment culture and separation and purification
The silt, soil, and bark samples with the same amount were separately added to the first enrichment medium (contained 0.3% trans-anethole) and cultured at 30 ℃ and 200 r/min in a shaker for 48 h. Each group of samples was set up to 3 replicates. The bacterial suspension was transferred to the second enrichment medium (contained 0.5% trans-anethole) at 1% inoculum amount, followed by 48 h of culture. After enriched by the third concentration gradient (1%) of trans-anethole, the bacterial suspension was diluted into 10-5, 10-6 and 10-7 with gradient sterile water. The three dilutions were coated on the solid modified M9 medium, and cultured at 30 ℃ for 3 to 5 d. A great number of starting strains were observed, and different single colonies were picked out. The single colonies were separated and purified by streak inoculation until pure single colonies were obtained.
Thin layer chromatography (TLC) preliminary screening
Inoculating loops were used to pick single colonies and inoculated them into the modified M9 medium, followed by culture in a shaker at 30 ℃ and 200 r/min. After 12 h, 1% of trans-anethole in volume fraction was added, and the fermentation was continued for 36 h. After the culture was completed, anhydrous ethanol 2 times of the volume was added for extraction, followed by shaking and mixing well. Then, 15 μl of the conversion liquid was doted on the activated GF254 silica gel plate, and developed in a developing solvent (petroleum ether∶chloroform∶ethyl acetate∶formic acid = 25∶10∶3∶0.2, v/v/v/v) for 18 min. Detection was performed with ultraviolet light, and the spot sizes of trans-anethole, anisic acid and anisaldehyde were recorded. The strains with larger anisic acid spots were selected and preserved.
High performance liquid chromatography (HPLC) re-screening
High performance liquid chromatography conditions: Referring to the method of Su et al.[17], the Agilent reverse phase C18 column (4.6 mm×250 mm, 5 μm) was selected to perform isocratic elution. The HPLC was carried out with acetonitrile∶water∶glacial acetic acid=70∶30∶0.02 (v/v/v) as the mobile phase at a flow rate of 0.8 ml/min under the injection volume of 5 μl, and the detection wavelength was 260 nm.
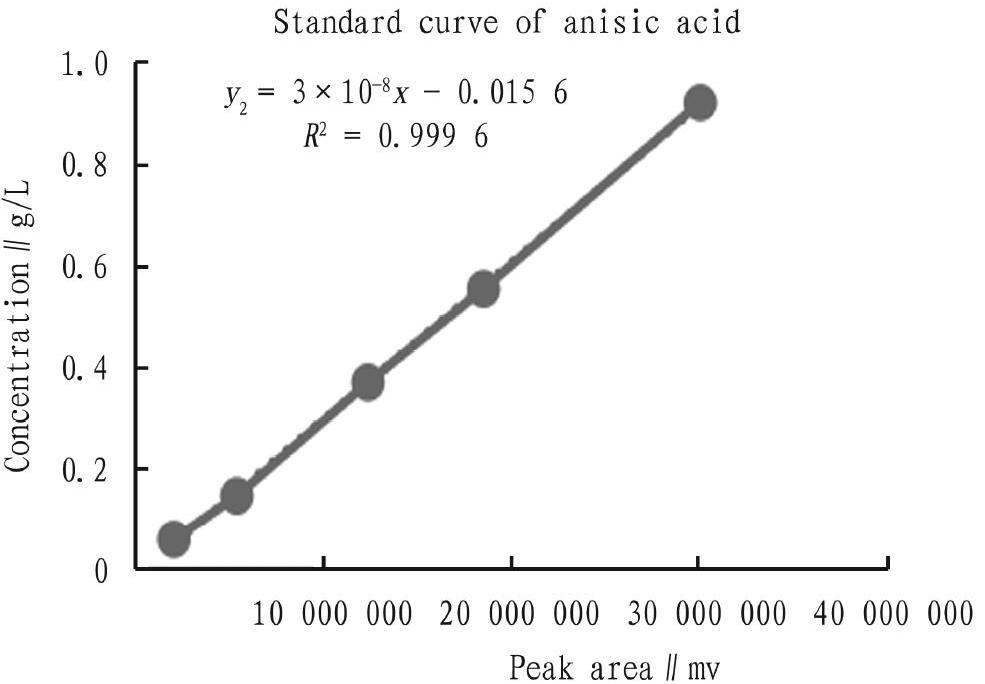
Standard curve determination: The above chromatographic conditions were adopted to explore the linear relationships of different concentrations of trans-anethole and anisic acid with the peak area of high performance liquid chromatography. The trans-anethole standard product was accurately weighed and prepared to 1.0, 0.59, 0.385, 0.168 and 0.078 g/L solutions with anhydrous ethanol as the solvent, and the anisic acid standard was accurately weighed and prepared to solutions with concentrations of 0.92, 0.552, 0.368, 0.144, and 0.057 6 g/L. With the substance concentration as the ordinate and the peak area as the abscissa, the standard curves and linear regression equations of trans-anethole and anisic acid were made. According to the TLC preliminary screening results, the conversion liquids of the strains with a better conversion effect were extracted with 2 times volume of anhydrous ethanol, and the extracts were diluted 3 times and filtered with 0.45 um microporous membranes. The concentrations of trans-anethole and anisic acid in the conversion liquids were detected by HPLC.
The calculation formulas of conversion efficiency and degradation rate were as follows:
Molar formation rate of anisic acid (%) = Molar amount of anisic acid produced/Molar amount of trans-anethole added×100%;
Degradation rate of trans-anethole (%) = Molar amount of trans-anethole consumed/Molar amount of trans-anethole added×100%.
Strain identification
Observation of morphological characteristics
The observation of morphological features was performed through plate colony morphology observation, Gram staining, scanning electron microscope, etc.
16S rDNA sequence analysis
Bacterial genome extraction: The Chelex-100 method[22]was used to extract total bacterial DNA, and using the total DNA as a template, 16S rRNA gene amplification was performed with 27F (5′-AGAGTTTGATCCTGGCTCAG-3′) and 1492R (5′-GGTTACCTTGTTACGACTT-3′) as primers. The PCR reaction was started with pre-denaturation at 94 ℃ for 5 min, followed by 34 cycles of denaturation at 94 ℃ for 30 s, annealing at 60 ℃ for 30 s and extension at 72 ℃ for 1 min, and competed with final extension at 72 ℃ for 10 min. The PCR product was detected by 1% agarose gel electrophoresis, and the result of the electrophoresis was observed by a Bio-RAD gel imaging system. After the band was qualified, the PCR product was sent to Majorbio Guangzhou Branch for sequencing. The sequencing results were processed by the DNA Star software and used for sequence alignment using the database EzBioCloud (https:∥www.ezbiocloud.net/). The Neighbor-Joining method of MEGA6.0 software was used to construct a phylogenetic tree. Combining with the morphological characteristics and 16S rDNA sequence analysis, the taxonomic status of the strains was determined.
Results and Analysis
Determination of standard curve of anisic acid and trans-anethole
The standard curves of the concentrations of trans-anethole and anisic acid to the peak area x detected by HPLC are shown in Fig. 1. The linear regression equations were:
y1=3×10-8x-0.069 4 (R2=0.999 0)
y2=3×10-8x-0.015 6 (R2=0.999 6)
Screen of strains
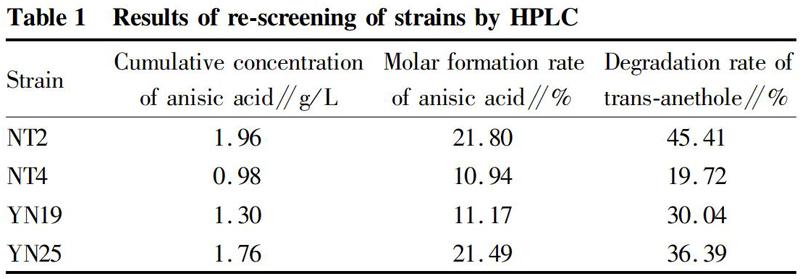
After enrichment culture and separation and purification, a total of 63 bacterial strains were obtained, of which 11 strains that could degrade trans-anethole to produce anisic acid were preliminarily determined by thin layer chromatography (see Fig. 2 for some results). According to the size and color intensity of anisic acid spots in thin layer chromatography, 4 strains YN19, NT4, NT2, YN25 with better conversion effects were selected for the re-screening by high performance liquid chromatography. The results are shown in Table 1. Strain NT2 had the best conversion effect, and showed a trans-anethole degradation rate of 45.41%, an anisic acid molar production rate of 21.8%, and an anisic acid cumulative concentration of 1.96 g/L. The detection result of strain NT2 by high performance liquid chromatography (Fig. 3) showed that the main product was anisic acid, and the specificity was stronger. Therefore, strain NT2 was selected for the next step in this study.
Identification of strain NT2
Morphological identification
The colonial morphology of strain NT2 is shown in Fig. 4. The colonies were milky white, round, with neat edges, and a protrusion in the middle, smooth and moist. The results of Gram staining are shown in Fig. 5. The bacteria were red after Gram staining and were Gram-negative bacteria (G-). The scanning electron microscopy results showed that the cells were short rod-shaped (Fig. 6).
16S rDNA sequence alignment and phylogenetic tree analysis
The 16S rDNA sequence of strain NT2 was compared in the database EzBioCloud. The sequence shared a high degree of homology with Pseudomonas, and the sequence similarity with Pseudomonas alloputida was 100%. The phylogenetic tree constructed by MEGA6.0 is shown in Fig. 7.
Conclusions and Discussion
In this study, 63 strains that could degrade trans-anethole and produce anisic acid were isolated from the environmental samples of star anise trees. After preliminary screening by thin layer chromatography and re-screening by high performance liquid chromatography, a strain NT2 that could convert trans-anethole to produceanisic acid more efficiently was obtained. After 16S rDNA sequence alignment analysis, it was preliminarily identified as Pseudomonas sp. Some reported strains that could degrade trans-anethole were mainly Pseudomonas sp., Burkholderia sp., Arthrobacter sp., and mold. Compared with them, according to molecular identification and phylogenetic tree analysis, strain NT2 in this study was another Pseudomonas sp. So this study provides a new strain for the microbial conversion strain resource bank.
Fermentation of strain NT2 at 30 ℃ and 200 r/min for 48 h produced anisic acid from 1% of trans-anethole, and the molar production rate of anisic acid was 21.8%, which was better than strains WGBF9 (17.9%)[19]and WGB31 (10.25%)[20]. Compared with BT13[17], the molar production rate of anisic acid is similar, but the degradation rate of trans-anethole is reduced by nearly 40%, indicating that strain NT2 has a greater conversion potential and can produce more anisic acid after optimization of fermentation conditions.
References
[1]WANG Q, JIANG L, WEN QB. Advances in studies on Illicium verum[J]. Cereals & Oils, 2005(5): 42-44. (in Chinese)
[2]LI ZG, XU DQ, XU ZY. Study on the aroma compounds released from star anise fruits[J]. China Condiment, 2003(10): 13-15, 20. (in Chinese)
[3]HE CM. Properties, production and uses of anethole[J]. Chemistry & Industry of Forest Products, 2004(1): 31-35. (in Chinese)
[4]SHEN P, SONG Z, ZHANG Z, et al. Screening of Burkholderia sp. WGB31 producing anisic acid from anethole and optimization of fermentation conditions[J]. Journal of Basic Microbiology, 2014, 54(11): 1251-1257.
[5]ZHOU GH, PENG SS. Synthesis of methyl anisate[J]. Journal of Guangxi Normal University: Natural science edition, 1995(2): 81-83. (in Chinese)
[6]ZHOU GH, PENG SS. Synthesis of ethyl anisate[J]. Journal of Guangxi Normal University: Natural science edition, 1996(4): 42-45. (in Chinese)
[7]ELSTON TW, PANDIAN A, SMITH GD, et al. Aniracetam does not alter cognitive and affective behavior in adult C57BL/6J mice[J]. PLoS One, 2014, 9(8): e104443.
[8]HU SH. Clinical effect analysis of amiodaron in the treatment of cardiomyopathy and heart failure[J]. China Modern Medcine, 2013, 20(1): 89-90. (in Chinese)
[9]AKKASILPA S, OSIRI M, DEESOMCHOK U, et al. The efficacy of combined low dose of allopurinol and benzbromarone compared to standard dose of allopurinol in hyperuricemia[J]. Journal-medical Association of Thailand, 2004, 87(9): 1087-1091.
[10]YU WX. Anisic acid prepared from star anise oil[J]. Chemical World, 1952(3): 23-24. (in Chinese)
[11]TANG Q. Preparation of anisic acid by indirect electrolysis[J]. Guangxi Chemical Industry, 1988(1): 20-22. (in Chinese)
[12]MENG XJ, ZHANG BF, GE X, et al. Preparation of p-methoxybenzoic acid[J]. Chemical World, 1995(2): 83-84. (in Chinese)
[13]CAO SF, LI WC. Research and application of microbial conversion[J]. Guangdong Canye, 2012,46(2): 45-50. (in Chinese)
[14]SHIMONI E, RAVID U, BAASOV T, et al. The trans-anethole degradation pathway in an Arthrobacter sp.[J]. Journal of Biological Chemistry, 2002, 277(14): 11866-11872.
[15]SHIMONI E, BAASOV T, RAVID U, et al. Biotransformations of propenylbenzenes by an Arthrobacter sp. and its t-anethole blocked mutants[J]. Journal of Biotechnology, 2003, 105(1-2): 61-70.
[16]RYU J, SEO J, LEE Y, et al. Identification of syn-and anti-anethole-2, 3-epoxides in the metabolism of trans-anethole by the newly isolated bacterium Pseudomonas putida JYR-1[J]. Journal of agricultural and food chemistry, 2005, 53(15): 5954-5958.
[17]SU GJ, LIU XM, ZHU P, et al. Isolation of trans-anethole degrading bacterial strains and biotransforrnation of trans-anethole[J]. Chemical Industry and Engineering Progress, 2009, 28(1): 141-144, 172. (in Chinese)
[18]SU GJ, LIU XM, LI WG, et al. Optimization of biotransformation conditions for transformation of trans-anethole to anisic acid by Pseudomonas sp. BT-13[J]. Journal of Chemical Engineering of Chinese Universities, 2011, 25(4): 643-649. (in Chinese)
[19]WANG XY, SHEN PH, HUA YF, et al. Isolation and identification of a strain capable of transforming trans-anethole to anisic acid[J]. Journal of Guangxi Normal University: Natural Science Edition, 2016, 34(4): 121-128. (in Chinese)
[20]ZENG HH. Isolation and characterization of strains about bioconversion of anethole generate anisic acid and optimization of fermentation conditions[D]. Nanning: Guangxi University, 2012. (in Chinese)
[21]LU ZQ. Study on the process of biosynthesis of anisic acid with trans-anethole[D]. Nanning: Guangxi University, 2013. (in Chinese)
[22]ZHOU SQ, HUANG XL, HUANG DY, et al. A rapid method for extracting DNA from actinomycetes by Chelex-100[J]. Biotechnology Bulletin, 2010, 26(2): 123-125. (in Chinese)
杂志排行
农业生物技术(英文版)的其它文章
- Review on Effects of Sunlight on the Internal Quality of Peach Fruit
- Research Progress on Genetic Breeding of Sweet Sorghum Related to Sugar Traits
- Screening of Red-flesh Small Watermelon Varieties for Substrate Cultivation in Spring Greenhouses
- Planting Techniques of Pennisetum giganteum in Huanghuai Area
- Bibliometric Analysis of Status Quo and Trend of the Research on Duck Based on the Web of Science Database
- Preparation and Insecticidal Activity of Sea Anemone Peptide AP-GI from Aiptasia pallida
