Solid phase microextraction chemical biopsy tool for monitoring of doxorubicin residue during in vivo lung chemo-perfusion
2021-03-22BrbrBojkoNikitLoobyMriolOlkowiczAnnRoszkowskBogumiKupcwiczPdroRckdosSntosKhldRmdnShKshvjThomsWddllGrmnomzosMrcosTsconKrzysztoGorynskiMrcloCyplJnuszPwliszyn
Brbr Bojko , Nikit Looby , Mriol Olkowicz , Ann Roszkowsk ,Bogumił Kupcwicz , Pdro Rck dos Sntos , Khld Rmdn , Sh Kshvj ,Thoms K. Wddll , Grmn G′omz-Ríos , Mrcos Tscon , Krzyszto Gory′nski ,b,Mrclo Cypl ,**, Jnusz Pwliszyn ,*
a Department of Chemistry, University of Waterloo, Waterloo, ON M1B 6G3, Canada
b Department of Pharmacodynamics and Molecular Pharmacology,Faculty of Pharmacy,Collegium Medicum in Bydgoszcz,Nicolaus Copernicus University in Torun, 85-089, Bydgoszcz, Poland
c Jagiellonian Centre for Experimental Therapeutics (JCET), Jagiellonian University, 30-348 Krakow, Poland
d Department of Pharmaceutical Chemistry, Medical University of Gdansk, 80-416, Gdansk, Poland
e Department of Inorganic and Analytical Chemistry, Faculty of Pharmacy, Collegium Medicum in Bydgoszcz, Nicolaus Copernicus University in Torun, 85-089, Bydgoszcz, Poland
f University Health Network -TGH, Toronto, ON M5G 2C4, Canada
Keywords:In vivo solid phase microextraction Metabolite profiling Spatial resolution Therapeutic drug monitoring Tissue analysis
ABSTRACT Development of a novel in vivo lung perfusion (IVLP) procedure allows localized delivery of high-dose doxorubicin (DOX) for targeting residual micrometastatic disease in the lungs. However, DOX delivery via IVLP requires careful monitoring of drug level to ensure tissue concentrations of this agent remain in the therapeutic window. A small dimension nitinol wire coated with a sorbent of biocompatible morphology (Bio-SPME) has been clinically evaluated for in vivo lung tissue extraction and determination of DOX and its key metabolites. The in vivo Bio-SPME-IVLP experiments were performed on pig model over various(150 and 225 mg/m2)drug doses,and during human clinical trial.Two patients with metastatic osteosarcoma were treated with a single 5 and 7 μg/mL(respectively)dose of DOX during a 3-h IVLP.In both pig and human cases,DOX tissue levels presented similar trends during IVLP.Human lung tissue concentrations of drug ranged between 15 and 293 μg/g over the course of the IVLP procedure.In addition to DOX levels, Bio-SPME followed by liquid chromatography-mass spectrometry analysis generated 64 metabolic features during endogenous metabolite screening, providing information about lung status during drug administration. Real-time monitoring of DOX levels in the lungs can be performed effectively throughout the IVLP procedure by in vivo Bio-SPME chemical biopsy approach. Bio-SPME also extracted various endogenous molecules, thus providing a real-time snapshot of the physiology of the cells, which might assist in the tailoring of personalized treatment strategy.
1. Introduction
Pulmonary metastases are known to develop in approximately 30%of patients with malignant tumors,most frequently in patients suffering from sarcomas. Although surgical resection is a widely accepted treatment for pulmonary metastases, a 5-year survival rate of about 20% after complete resection remains disappointing[1]. Unfortunately, most patients develop recurrent metastasis,probably as a result of micrometastatic disease present at the time of the initial operation [2]. Most recurrences occur in the lungs themselves,suggesting that the lung is the major reservoir of occult metastatic burden (Fig.1). Therefore, a treatment capable of specifically targeting the lung to eradicate micrometastatic disease would be highly beneficial for this patient population. By building upon our experience with ex vivo lung perfusion for repair of donor lungs for transplantation [3,4], our group recently developed and described a technique for isolated in vivo lung perfusion (IVLP) to facilitate localized drug delivery to the lungs during surgical resection[2,5].Based on these pre-clinical studies,a phase I clinical trial (NCT02811523) using doxorubicin (DOX) during IVLP for treatment of lung sarcoma metastases is underway.
Direct determination of DOX in different compartments of lung tissue is desired during IVLP as it would allow to monitor drug level and its biodistribution within particular lung lobes. Traditionally,accurate determination of DOX in the lungs requires collection of tissue via biopsy, an invasive process that can only be performed from peripheral locations of the organ[6].In addition,such method requires lengthy sample processing and analysis, thus preventing real-time determinations of concentration and distribution of DOX in lung tissue over the course of IVLP. The absence of such information during this procedure, in turn, introduces potential for administration of subtherapeutic or toxic tissue levels of DOX.Therefore,a minimally invasive method capable of monitoring DOX levels in tissue in close to real-time conditions would allow for safe,effective, and accurate administration of DOX in the lungs during IVLP. Certainly, implementation of such a monitoring tool during IVLP would be of critical therapeutic benefit, as such a measure would optimize treatment while preventing adverse effects associated with toxic levels of DOX in the target organ. Additionally,analysis of other molecular indicators such as metabolites could provide insights into ongoing biochemical processes during IVLP,a result of the drug activity or organ stress demonstrating also individual patient’s response [7]. Access to such information would be useful for effective tailoring of the treatment,as it would enable dose adjustments based on evaluation of patient response rather than just DOX concentration levels.
Herein, biocompatible solid phase microextraction (Bio-SPME)is proposed as a new analytical strategy to assist in real-time measurements of DOX in tissue and perfusate during IVLP. Bio-SPME has already been reported for analysis of tissues [8,9]. This chemical biopsy procedure involves the extraction of small molecules such as xenobiotics and endogenous molecules from a biological system by directly inserting a sharpened microprobe the size of an acupuncture needle(ca.0.3 mm diameter)into the tissue(Fig. 2) without removal of any tissue residue [10,11]. The probe consists of a nitinol (Ni/Ti) alloy wire coated with a matrixcompatible phase; in this case, a crosslinked slurry composed of micron-sized sorbent particles suspended in a biocompatible oligomer binder.The crosslinked polyacrylonitrile oligomer forms a smooth surface characterized by small size pores,which exclusively allow the diffusion of small molecules from tissue to dispersed sorbent particles while preventing the adsorption of tissue components such as proteins, red blood cells, and other endogenous macromolecules onto the surface of the coating [11]. Recovery of these small compounds is obtained via their desorption from the coating into a small amount of organic solvent, which is then subjected to instrumental analysis.
Quantitative analysis can be performed since Bio-SPME is conveniently amenable to a range of separation and detection techniques, including high performance liquid chromatography(HPLC) coupled to tandem mass spectrometry (MS/MS) - the standard method for bioanalytical determinations[12].Qualitative analysis by way of untargeted metabolomics can also be performed via HPLC coupled to a high-resolution mass spectrometer (HRMS)[13]. SPME has shown a great potential in the field of clinical metabolomics due to its extractive capabilities for a broad range of both polar and non-polar small molecules (100-1200 Da) [11,14].The advantage of carrying out a chemical biopsy via SPME in comparison to several other approaches is that Bio-SPME could be used to perform in vivo metabolomics[14-17].Given its minimally invasive design, non-depletive extraction principle, and biocompatibility,information regarding the metabolites present within the system at a given time can thus be easily obtained without disrupting molecular binding or equilibrium within the system, nor damaging the microprobe itself [15]. In addition, Bio-SPME can be hyphenated to stand-alone mass spectrometer (MS) instruments,resulting in shorter analysis time and being closer to real-time in vivo monitoring of DOX levels [15]. The herein proposed approach certainly fits within the growing trend of incorporating intraoperative real-time analysis of small molecules in animal and human studies via automated MS-based diagnostic protocols. Devices such as intelligent Knife (iKnife), MasSpec Pen, and Spider-Mass are among modern biocompatible tools that enable easy and fast introduction of extracted analytes to MS instrumentation,thus providing access to relevant information regarding the status of tissue during surgery [18-20]. In line with these trends, the development of sampling/sample preparation techniques based on microdialysis (MD) and SPME for such applications has observed increased demands in recent years, as such techniques facilitate reliable in vivo measurements of drug concentration and biodistribution within selected organs [10,11,21].
Here, Bio-SPME is used to monitor DOX and its metabolites, as well as small endogenous molecules, present within the system over the course of IVLP. The current work presents the determination and quantification of DOX in the following settings: (i) an in vivo pig model over various drug doses as a proof-of-concept and(ii)in vivo IVLP in human clinical trial.The work also demonstrates that the SPME chemical biopsy captures other low molecularweight compounds, such as potential markers of drug toxicity or organ stress over the course of IVLP.

Fig.1. Pre-operative lung computed tomography scan for 1st patient subjected to in vivo lung perfusion showing numerous bilateral pulmonary sarcoma metastases as denoted by arrows.

Fig. 2. Biocompatible solid phase microextraction (Bio-SPME) microprobe.
2. Materials and methods
2.1. Experimental procedures
The experiments were performed on(1)in vivo pig model over various (150 and 225 mg/m2) drug doses and (2) during in vivo clinical IVLP. Two patients with metastatic osteosarcoma (the first patient underwent bilateral pulmonary metastasectomy) were treated with 5 and 7 μg/mL(respectively)dose of DOX during a 3-h IVLP.Concomitantly,perfusate samples were collected at the same time points as in vivo SPME sampling time points in order to analyze the level of DOX during IVLP. The assessment of DOX concentration was performed with the use of previously optimized method [22]. In addition, we investigated in vivo profile of small endogenous molecules in human lung tissue over the course of IVLP-DOX administration. For metabolomics data analysis, Least Absolute Shrinkage and Selection Operator(LASSO)was applied for selection of variables prior to chemometric analysis.IVLP procedure and Bio-SPME lung sampling were conducted at Toronto General Hospital.Bio-SPME extraction of perfusate samples was performed on-site at Toronto General Hospital and in the analytical laboratory at University of Waterloo, Canada. HPLC-MS/MS and HPLC-HRMS analyses were performed in the analytical laboratory at University of Waterloo.For the studies the prototype SPME-biocompatible mixed-mode (MM) fibers with C8+benzenesulfonic acid particles were used as in-kind contribution of Supelco(Bellefonte,PA,USA).
2.2. In vivo animal study
Two Yorkshire pigs weighing approximately 35 kg were used.A porcine left lung IVLP model was used to model delivery of high doses of DOX during IVLP. The IVLP model procedure has been described elsewhere[2,5].The doses of DOX administered were 150 and 225 mg/m2. SPME sampling was performed from the lower lobe of the left lung before perfusion (t0) and 30, 90, 150, and 210 min after the start of the IVLP procedure, using the protocol described below. The study was approved by the Animal Care Research Committee at Toronto General Research Institute.
2.3. In vivo human study
The clinical trial was registered on www.clinicaltrials.gov and institutional research ethics board approval was obtained. Two individuals were enrolled in the clinical study:22-year old and 66-year-old males with bilateral recurrent pulmonary metastases secondary to osteogenic sarcoma (Fig. 1). All lung lesions treated with DOX using the IVLP platform were resectable with wedge or segmental resections. The first patient had previous systemic exposure to DOX(<450 mg)and had previously received a bilateral metastasectomy. The technical details of the procedure have been described elsewhere [5]. Briefly, a clam-shell thoracotomy was performed and the left pulmonary hilum was dissected. The pulmonary artery and upper and lower pulmonary veins were dissected and subsequently cannulated and attached to the IVLP perfusion system. Lung isolation was performed using vascular clamps, and IVLP was initiated and run for 3 h. DOX was added to the IVLP perfusion solution at a concentration of 5 and 7 μg/mL for the first and second patient, respectively.
For Bio-SPME sampling of the lungs, three fibers (Fig. 2) were placed in different areas of the lung (upper, middle, and lower sections) at predetermined time points over the course of the surgery.The lung was sampled prior to drug administration,hourly during IVLP, and once after reperfusion. Extractions were carried out for 20 min under static conditions. For Bio-SPME sampling of perfusate,three fibers were used for on-site extraction of collected perfusion fluid.A 20-min static extraction was performed on these samples as well.For both Bio-SPME sampling cases,the fibers were subsequently rinsed manually in water for 5 s, wiped with a Kimwipe,and immediately snap frozen in dry ice.Additional perfusate samples were also collected and snap frozen in dry ice until further analysis in the laboratory. DOX concentrations in the lung and in perfusate were determined using a validated SPME-HPLC-MS/MS method after minor modifications [22]. In addition, the analytical method to identify and quantify DOX key metabolites (doxorubicinol (DOX-ol), doxorubicinone (DOX-one), and doxorubicinolone (DOX-olone)) has been developed. A brief description of analytical conditions employed (during the drug and its metabolites’ determination) together with materials used has been included in the Supplementary Material.
2.4. Statistical analysis
The metabolomics experiments were performed separately for both cases because of the long-time interval between the recruitment of the patients. The first dataset was used for selection of important endogenous features,while the second dataset was used for confirmation of the initial findings. In metabolomics experiments, the number of features detected is typically much larger than the number of samples studied.In our study,the first dataset consisted of 12 samples (n) and 138 variables (p). Selection of variables prior to chemometric analysis was carried out using the LASSO method. LASSO applies a regularization process where it penalizes the coefficients of regression variables, forcing certain coefficients to be set to zero. Thus, LASSO removes variables that are redundant by effectively providing a simpler model that does not contain those coefficients. The number of variables selected using the LASSO method has to be smaller or equal to the number of samples. Moreover, LASSO will often select only a single feature from a set of highly correlated variables[23].Principal component analysis (PCA) was applied to the selected data after autoscaling(Unit-Variance scaling). PCA employed internal cross-validation using the Leave-one-out (LOOCV) method. The validation metrics root mean squares errors of calibration (RMSEC) and crossvalidation (RMSECV) were calculated.

Fig. 3. In vivo sampling of pig lungs during IVLP. (A) Triplicate of Bio-SPME probes during sampling; (B) the same location of lung after removal of SPME probes.(Reprinted from Ref. [9] with permission).
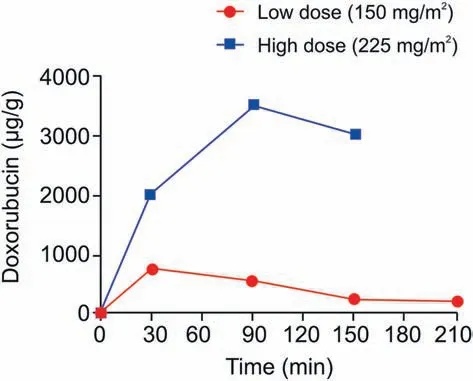
Fig. 4. Concentration profile of DOX in lower lobe of the lung during IVLP (sampling points 30, 90, 150, and 210 min). Red circles and blue squares: levels of DOX after administration with 150 and 225 mg/m2, respectively.
3. Results and discussion
3.1. Determination and quantification of DOX during IVLP
As a proof-of-concept for the sampling method, initial experiments were carried out using pigs as the experimental model. No bleeding or adverse effects were observed in any of the sampled areas of the lung (Fig. 3) [9]. Stability and biocompatibility of the probes were proven previously on several in vivo animal models like fish, mice, rats, pigs, and monkeys [15]. The organ samplings included liver, lung, kidney, brain and muscle, i.e., tissues of different texture, density, resistance and other properties. As mentioned before,the core of the probes is made of nickel-titanium alloy, which gives them a shape-memory and prevents breakage during tissue penetration, while the extractive phase particles are effectively immobilized on the mentioned metal support by polyacrylonitrile. The only modification made in the present samplings is the angle of cutting the tip of the fiber.The blunt end can easily penetrate soft tissues like liver or brain, but in case of more resistant tissues like muscle or lung (presence of pleura), prepiercing with needle or use of pointed tip or guide cannula is needed. Single Bio-SPME probe from each time point was used to assess lung tissue concentration of DOX.Lung tissue concentrations of DOX were successfully measured during two separate IVLP procedures, where DOX doses of either 150 mg/m2or 225 mg/m2were administered (Fig. 4). As expected, sampling before DOX administration (time zero, pre-IVLP) did not show the presence of the chemotherapeutic agent in lung tissue. Subsequent samplings exhibited a trend of a quick peak of drug, followed by steady decline. For the lung perfused with a drug dose of 150 mg/m2, a maximum measured concentration of 723 μg/g DOX was noted at the 30 min mark after IVLP was initiated.For the lung perfused with a dose of 225 mg/m2DOX,a maximum measured concentration of 3521 μg/g was reached after 90 min of IVLP.In the case of the high dose(225 mg/m2DOX),the whole procedure ceased at 150 min,as the animal did not tolerate the injury caused by the high dose being administered.
In the human study, two patients, one aged 22 and the other aged 66, underwent the IVLP procedure. There were no intraoperative complications and use of Bio-SPME probes did not cause lung injury. For the first patient (22-year-old), wedge resection was performed for 6 lesions on the right lung and 4 lesions on the left lung.Total operative time was 7 h and 19 min in the case of the 22-year-old patient and 6 h and 47 min in the case of the 66-year-old patient. Both patients tolerated the procedure well and did not require Intensive Care Unit admission. Each patient was discharged home on postoperative day 7 (POD7). They had an uneventful post-operative period and returned to regular activity after 1 month.At 6 months,the first patient’s forced expiratory volume in 1 s (FEV1) decreased from 4.7 L (82% predicted) to 2.9 L (50%predicted)and his forced vital capacity(FVC)decreased from 5.6 L(80% predicted) to 3.4 L (48% predicted). The patient developed bilateral lung recurrences after 6 months of follow-up, which progressed despite further treatment. He remained alive until 1 year of follow-up. The second patient remained in overall good health at 6 months of follow-up.
For the 22-year old patient, concentrations of DOX in the upper,middle, and lower regions of the perfused lung showed similar trends during IVLP (Fig. 5A). Expectedly, before perfusion started(pre-IVLP), lung tissue was free of DOX. Maximum lung tissue concentrations of drug measured by Bio-SPME-LC-MS/MS method were reached at the first hour of IVLP,and were 200,36,and 166 μg/g for the upper,middle,and lower sections of the lung,respectively.Lung tissue DOX concentration dropped steadily over the course of IVLP and reached a minimum of 14,8, and 9 μg/g for the upper, middle,and lower areas of the lung, respectively, by 30 min post blood reperfusion(after IVLP termination).The hospital assay(current gold standard technique done in tissue biopsies to measure DOX),which consisted of protein precipitation (PPT) followed by HPLC with fluorescence detection (HPLC-FLD),showed a DOX concentration of 7.0 μg/g in the biopsy tissue 30 min post-reperfusion.Further details on the hospital assay employed have been published elsewhere[6].For the 66-year old patient,the highest tissue concentrations of DOX estimated with Bio-SPME probes were also noticed in the first hour of lung perfusion, and were 179, 69, and 293 μg/g for the upper,middle, and lower sections of the lung, respectively (Fig. 5C). Similarly to the first human case, lung tissue concentrations of DOX exhibited a decreasing trend throughout the procedure reaching the levels of 30,18 and 28 μg/g for the upper,middle,and lower areas of the lung, respectively 30 min post blood reperfusion. The hospital assay utilizing collected biopsy samples predictably revealed no drug at the stage of pre-perfusion and its very low content during pulmonary reperfusion(Fig.5C).
In addition to in vivo tissue measurements of DOX and its metabolites,the drug was also extracted by SPME probes on-site as well as in the analytical laboratory from perfusate samples collected at the same time points used for the in vivo lung sampling. For the 22-year old patient, the total drug dose given to the perfusion circuit was 5.0 μg/mL. Concentration of DOX in the perfusate decreased steadily from the beginning of the IVLP procedure (Fig. 5B). SPME-HPLC-MS/MS analysis revealed that after 30 min,the DOX concentration in the perfusate was 2.4 μg/mL and that over the course of the procedure this value decreased to a minimum of 0.3 μg/mL by the end of the perfusion. PPT-HPLC/FLD analysis of perfusate samples performed by the clinical laboratory showed a concentration of 7.2 μg/mL after 30 min, and a nadir of 1.0 μg/mL by the end of the perfusion. DOX concentration measurements of frozen perfusate samples analyzed by SPME-HPLCMS/MS after transport to an off-site location showed no statistically significant differences from perfusate samples analyzed by SPME in the operating room (P = 0.75) (Table S1). During 2nd clinical IVLP, the dose of the drug administered into the perfusion circuit was 7 μg/mL and detected concentration of DOX in the perfusate samples in the first hour of the procedure was 2.7 μg/mL.In next hours of IVLP,DOX level decreased to 1.2 μg/mL at the 3rd h of perfusion (Fig. 5D). For the 66-year old patient, a similar decreasing trend in the terms of the drug level in perfusate samples over the course of IVLP was also observed with the standard approach employing PPT as a streamlined sample preparation method (Fig. 5D). However, using the same instrumental method and analytical conditions, the results received with two different strategies were even more comparable. Figs. 5B and D present a direct comparison of SPME results with standard assays showing relatively good match between the data.In case of Fig.5B the PPTHPLC/FLD assay was performed by external clinical lab for single samples at each time point, while in Fig. 5D the PPT-LC/MS assay was performed on triplicate samples,similar as SPME.It should be noted that the estimated precision obtained for SPME (RSD below 5%) was much better than PPT-LC/MS (RSD below 15%).

Fig. 5. Concentration of doxorubicin measured by Bio-SPME-HPLC-MS/MS in lung tissue during first (A) and second (C) clinical IVLP. Time points include 1 h pre-IVLP,1, 2, 3 h during IVLP, and 30 min post-IVLP during lung reperfusion. Green, blue, and red dots represent the three locations of the fiber in the lung during sampling. The green triangle designates the upper section of the lung, the blue square designates a section between the upper and lower lobes (middle) of the lung, and the red circle designates the lower section of the lung.Black diamonds show when hospital biopsies were taken.(B and D)Concentration of doxorubicin in perfusate as measured by Bio-SPME-HPLC-MS/MS and the standard approach(hospital assay-PPT-HPLC/FLD or PPT-LC/MS)during human IVLP,represented by the blue and red diamonds,respectively.Results are presented as mean±SD.Statistical significance was determined by a two-tailed t-test; ns, *, **, and *** indicate not significant, P <0.05, P <0.01, P <0.001, respectively.
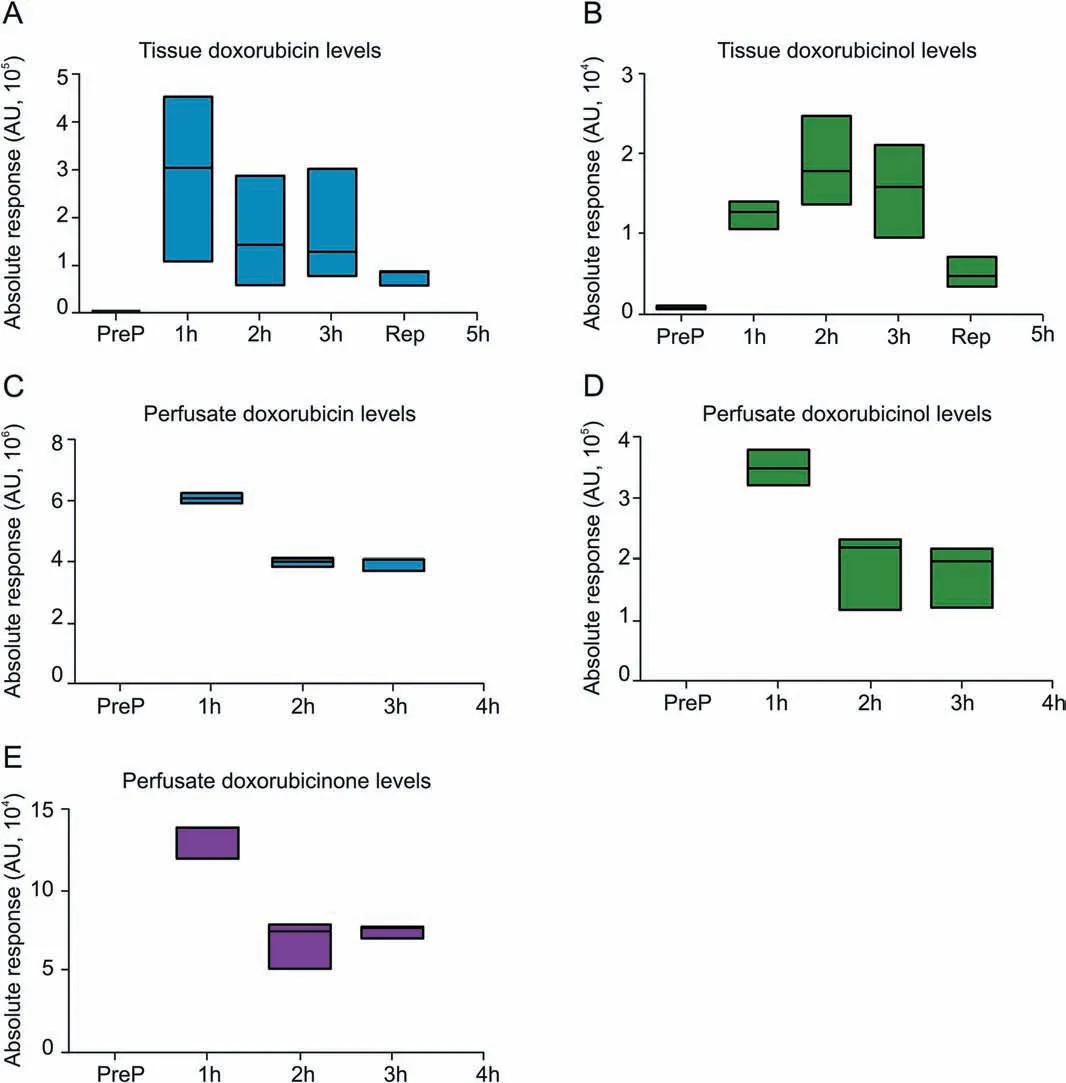
Fig. 6. Floating bars presenting levels of doxorubicin and its key metabolites(doxorubicinol and doxorubicinone) over the course of IVLP procedure. The detection range together with median values for each compound analyzed in relevant matrices(lung tissue(A and B)or perfusate samples(C-E))have been denoted.As indicated above,the intact drug was a predominant one detected in investigated systems with some amounts of metabolites resulting from its biotransformation but contributing no more than 5%to the resultant pool of analyzed compounds.
Furthermore, the identification and quantification of key metabolites of DOX, namely, DOX-ol, DOX-one, and DOX-olone, were performed for both clinical cases.However,the results showed that DOX metabolites were present at trace levels in the tested matrices(Fig. 6). Only DOX-ol, formed in the major metabolic route of DOX metabolism, was detectable at visible level,but its content in lung tissue did not exceed 5% of the intact drug, and therefore was considered as insignificant.
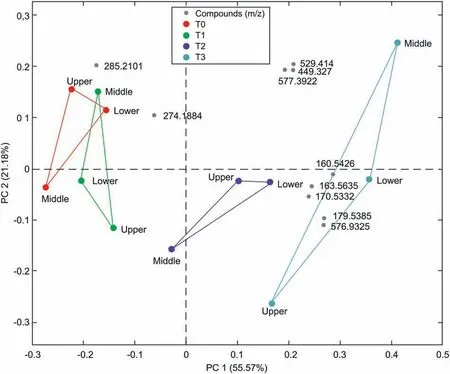
Fig. 7. Principal component analysis showing separation of the data obtained at subsequent time points for different sections of the lung; red circles denote baseline sampling,before IVLP; green, blue, and turquoise circles denote 1 h and 2 h after the start of IVLP and 30 min after the start of blood reperfusion (RMSEC = 0.462, RMSECV = 0.838),respectively. The biplot reflects the distribution of molecular features (compounds), denoted as grey points, selected with LASSO with respect to subsequent data points.
The current study demonstrates that the developed method based on SPME enabled quantitative monitoring of DOX from lung tissue in a safe and minimally invasive manner.Multiple insertions of Bio-SPME chemical biopsy probes did not cause tissue injury during in vivo administration in either pig or human lungs. Each patient recovered uneventfully and had no complications from the SPME fiber insertion or the IVLP-DOX administration. Bio-SPME sampling was successfully performed on-site (in vivo and in situ),but analysis and data processing were done in the analytical lab.No difference was observed among perfusate results for analyses carried out on-site or in-lab; however, the availability of MS instruments within or near the operating room would offer the possibility of more rapid target drug concentration determinations.A fully on-site setup is feasible since Bio-SPME probes can be directly coupled to a mass spectrometer (SPME-MS), thereby providing results within seconds[15,24,25].More recently we have also developed a portable fluorometer coupled to a laptop designed to be deployed in a surgery room for fast and direct quantification of DOX in the tissue via detection of this compound in the SPME coating [26]. Fluorescence readout, however, can only provide the information about DOX concentration, but not about its metabolites or endogenous compounds; hence the information about organ condition in respect to biochemical alterations still requires the application of MS analysis.
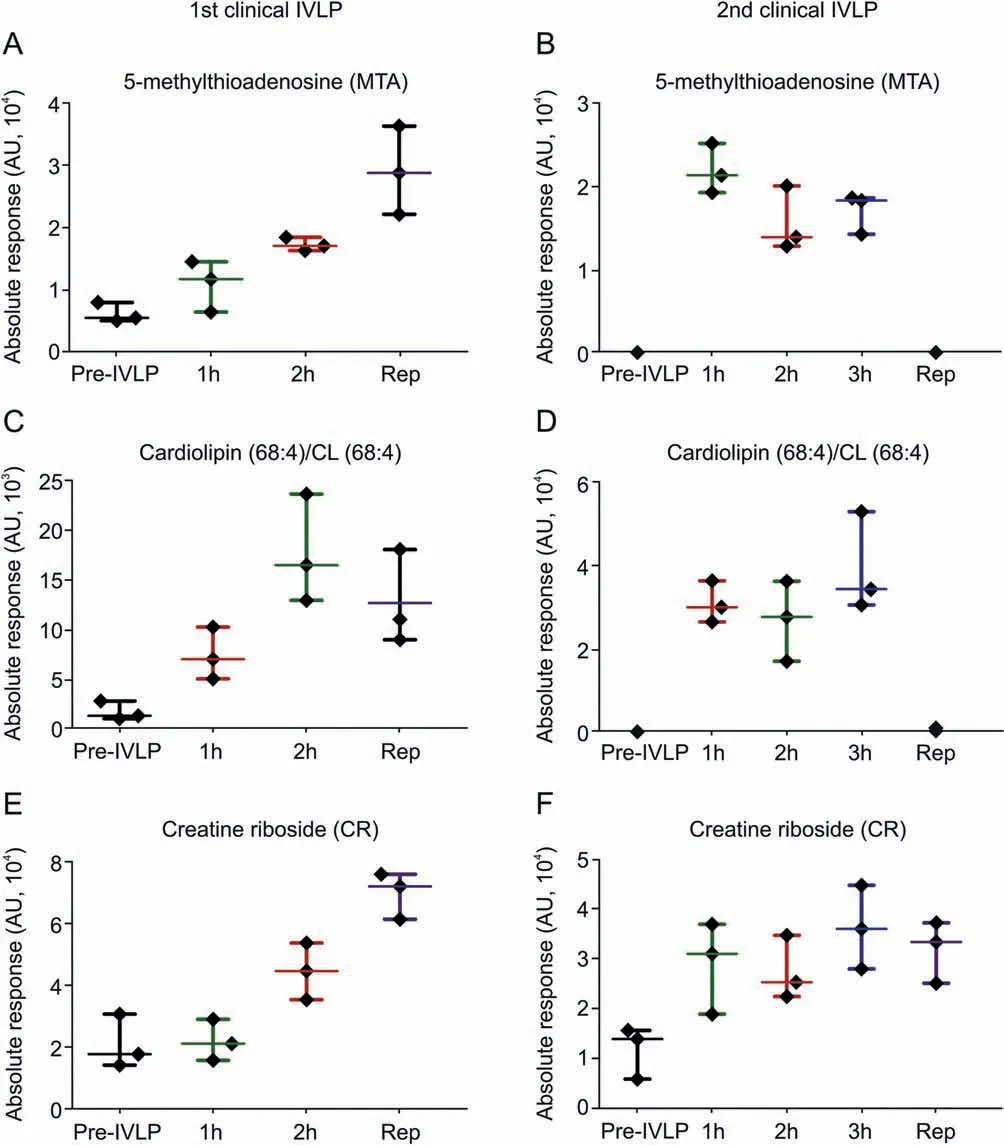
Fig.8. Scatter plots showing relative intensities of selected features/metabolites primarily dysregulated upon one-time chemotherapy treatment for an individual patient.Each dot in the scatter plots represents an individual metabolite in each sample.1st Clinical IVLP refers to the 22-year old patient; 2nd Clinical IVLP refers to the 66-year old patient.
In both pig and human cases, DOX tissue levels presented similar trends during IVLP, wherein drug concentrations peaked between 60 to 90 min after the start of IVLP and subsequently dropped.This is more evident in the clinical trial since all sampled areas of the lung-upper,middle,and lower-exhibited the same pattern. As there were no measured leaks in the IVLP circuit, two potential reasons may explain this observed pattern: a) biotransformation of DOX into metabolites, leading to less intact drug available for extraction (that was rather not supported by our results,indicating the formation of negligible amount of metabolites)or b)diffusion of DOX into cells.Bio-SPME probe extracts only free concentration of compounds, and therefore will not remove DOX bound to the intracellular components [17]. The binding or partitioning of DOX into cells,therefore,decreases its free concentration in the extracellular environment of the lung where the microprobe performs sampling, which is reflected in the trend of decreasing DOX concentration.The results of DOX level obtained by Bio-SPMEHPLC-MS/MS for the lower lobe of the lung after drug washout(9 μg/g for first human case and 28 μg/g for second human case)were comparable to results obtained on tissue biopsies employing the standard assay, further confirming the reliability of the approach proposed. Furthermore, the results from this study indicate that perfusion of the lung does not necessarily provide homogenous drug distribution,which may also account for the slight difference between the results provided by each assay, as the samples,although derived from the same lobes,were not obtained from the exact same location.It is important to emphasize that the differences in sample preparation fundamentals between SPME and PPT as well as much broader calibration range used in detection method by the clinical lab contributed to some differences in the results gathered for perfusate samples employing these two methods. Furthermore, Bio-SPME has the additional advantage of providing information about the total as well as free drug concentrations, the latter offering the drug fraction immediately available for tissue binding [15,17]. Despite some differences in absolute measured DOX concentrations between the SPME-HPLCMS/MS and standard assays, more evident with PPT-HPLC/FLD method and less pronounced with PPT-LC/MS method (and comparable calibration curve range),as seen in Figs.5B and D,there was a coherent decreasing trend observed by both techniques,showing reliability and relative agreement of the methods. In addition,although the developed Bio-SPME-LC/MS method also offers a possibility to monitor DOX key metabolites (DOX-ol, DOX-one and DOX-olone), none of them were found at significant level in the lung tissue during DOX administration via IVLP. This finding is in line with generally accepted assumption that intact drug rather than its metabolites is responsible for pharmacological activity and anticancer effects [27,28].
3.2. Screening of small molecules in lung tissue during IVLP-DOX administration
As demonstrated in previous studies, SPME is suitable for extraction and monitoring of a broad range of metabolites (from highly hydrophilic to hydrophobic ones) in different biomatrices[29], including unstable compounds, like oxylipins, which was recently reported in brain studies[30].The same Bio-SPME extracts used to perform targeted therapeutic analysis of DOX via HPLC-MS/MS analysis were also used for screening other low molecular weight compounds via HPLC-HRMS. Fig. 7 shows the principal component analysis (PCA) conducted on samples obtained for the 1st clinical case at different time points for the duration of IVLP.It is evident that samples collected from each time point group relatively well together,with separation seen as a transitionary pattern from the left to the right part of the plot. It was even more pronounced for 2nd clinical case pointing at a clear discrimination among metabolomic patterns for samples collected before commencing perfusion, during IVLP, and reperfusion (Fig. S1).
Introduction of LASSO analysis enabled selection of the features(compounds) of the highest importance in the separation among subsequent time points of the experiments: T0, T1, T2, and T3, or between T0 and T3 only (before IVLP and after reperfusion). It should be emphasized that after the IVLP procedure,the lungs were flushed with perfusate not containing DOX(reperfusion)in order to remove any remaining drug from the isolated organ, thereby avoiding systemic side effects. As expected, among the features differentiating all four time points were doxorubicin and several other molecules of possible endogenous or exogenous origin. The biplot in Fig.7 presents the relationship between the data collected at given time points during IVLP for all three sections of the lung,and masses of importance selected by LASSO.Penalized regression methods such as the LASSO have proven useful for variable selection in many fields,especially for situations with high-dimensional data where the number of variables far exceed the number of samples. LASSO identifies a set of metabolites that are relatively important in relation to a given outcome. Indeed, there are some drawbacks of LASSO. For example, it may not recover the full sparsity pattern when p ≫n and is sensitive for the highly correlated variables.Despite the presence of collinearity,LASSO can still distinguish between important variables. The size of the coefficients for the variables selected essentially represents the maximum effect size of the cluster of correlated predictors. Moreover, in many high-dimensional metabolomic datasets, it is reasonable to assume that a small number of compounds are relevant for the outcome [31,32].
Additionally, correlation analysis performed between all detected features and DOX lung concentration and distribution resulted in a number of metabolites exhibiting statistical significance. Cases where the statistical significance was observed in at least two lung sections were subjected to further identification and biological interpretation.The list of selected masses and the biplot plotted for the aforementioned metabolites can be found in Supplementary Material (Table S2 and Fig. S2, respectively). Putative identification of the presented features enabled selection of those of important biological significance,namely,compounds related to lung toxicity or injury. One of the identified endogenous compounds was 5′-methylthioadenosine (MTA), which showed a negative correlation with DOX(Fig.S2)over the course of IVLP.MTA levels were found to increase throughout IVLP and after reperfusion(Fig. 8A). Another interesting feature identified in SPME extracts from different sampling time points during IVLP was creatine riboside (CR) (Figs. 8E and F). Similar to MTA, CR also shows a negative correlation with DOX level. In addition, the Bio-SPME approach was capable of detecting alterations in several lipid classes. Among them, cardiolipin (CL) correlated negatively with the level of DOX at different time points of the IVLP(Figs.S2 and 8C and D).
Changes in the profiles of endogenous compounds corresponding to the administration of DOX during IVLP were monitored to verify whether the analytical strategy proposed for drug monitoring could also be adopted for screening of known markers of drug activity. As a proof of concept, compounds that showed correlation with DOX activity including MTA and CL as well as the biomarker of lung cancer, CR, were putatively identified in sample extracts.Presence of the metabolites was reported in the first case and further confirmed in the second one. It has been previously reported that MTA exhibits proapoptotic effects on different cancer cells [33,34].On the other hand,based on a study where DOX was used as a model drug,MTA was proposed to provide mechanisms to evade drug-induced apoptosis in tumor cells [35]. It has been suggested that MTA may have a dual role in the regulation of cellular homeostasis; therefore, further investigation of the relationship between MTA-DOX during IVLP should be performed on a larger cohort of patients/cases. DOX has also been previously shown to exhibit a strong correlation with lipid metabolism. In a rodent model, DOX led to elevated serum triglyceride and blood glucose levels through the down-regulation of peroxisome proliferator-activated receptor γ (PPARγ) [36]. An in vitro study with parental (MCF-7/S) and DOX-resistant (MCF-7/Dox) human breast cancer cells demonstrated that lipid profile changes can mediate the modulation of membrane fluidity in drug resistant cells[37].The authors observed an increase in the levels of several lipids,including CL,in resistant cells.Interestingly,DOX was already described as a specific probe for transversal distribution of CL[38,39]. As it can be seen in Fig. 8, MTA and CL (68:4) levels were elevated during the IVLP, while CR was found in the extract obtained at all time points, i.e., it was independent on DOX administration(Fig.8).CR is considered a product of high creatine levels and high phosphate flux due to the higher energy demands of rapidly dividing tumor cells [40]. The level of this compound has been found to be significantly higher in the urine of patients with non-small cell lung cancer(NSCLC)than that of healthy individuals[41,42]. Moreover, it has been previously reported that increased levels of CR are associated with increased tumor size and poor prognosis. Therefore, it has been speculated that this compound may not only be useful as a marker of early detection of lung cancer,but also aid in the provision of more accurate prognoses[41].Based on our finding, it can be postulated that MTA, CL and CR may be promising targets for further study on SPME-based DOX activity evaluation during IVLP. Moreover, as the first attempt at real-time profiling of metabolites in human lung tissue exposed to chemotherapy, this case study provides insights into the correlation between DOX and other endogenous molecules in the living system.
However, the results of metabolite screening, although promising, contain limited data to draw any concluding remarks as the analyses in this case study were carried out in extracts specifically obtained for DOX analysis, and most of the small molecules described in this paper had low sensitivity.Therefore,as a next step,an optimum sample preparation protocol should be implemented to improve recovery of compounds of interest other than DOX.This will facilitate more in-depth studies of organ-specific mechanisms as well as the adverse effects of DOX action, as such analyses are usually conducted on a large number of samples to account for biological variability and improve reliability of significant findings.
4. Conclusions
This work presents the first successful clinical use of the in vivo Bio-SPME chemical biopsy approach. The sampling/enrichment tool was used to reliably measure drug concentrations in lung tissue during a clinical case of localized DOX delivery. At the same time, a wide range of other molecules were extracted for metabolomic analysis, demonstrating the potential of the technique for real-time monitoring and early indication of lung toxicity and injury during the procedure. Based on these results,Bio-SPME can be used as a tool to assist in the safe and efficacious implementation of IVLP treatment for local chemotherapy by offering temporal and spatial monitoring of the drug concentration in individual patients.The Bio-SPME chemical biopsy tool provides significant advantages as a novel approach for tissue sampling since it imparts real-time information about both the levels of the administered chemotherapy and its effect and toxicity, as presented by the changing metabolomic landscape in the organ. Bio-SPME also shows potential as a complementary technique to aid in the elucidation of pathways affected or mechanisms of action of targeted therapies.
Declaration of competing interest
The authors declare that there are no conflicts of interests.Acknowledgments
We are grateful to the Canadian Cancer Society for its support in the development of IVLP via innovation grants,as well as its further support in the clinical trial with DOX IVLP as part of the innovationto-impact grant. We are also grateful to the Canadian Institute of Health Research (CIHR) - Natural Sciences and Engineering Research Council(NSERC)of Canada Collaborative Health Research Projects program for the financial support (Grant No. 355935 entitled “Supervised in vivo lung perfusion strategy for treatment of cancer metastases to the lungs. Real-time monitoring of chemotherapy by on-site analytical platform”) and the Natural Sciences and Engineering Research Council of Canada Industrial Research Chair (IRC) program.
We would like to thank our collaborators at Millipore/Sigma for kindly providing us with the fibers and chromatographic columns employed for this investigation. We would also like to thank Thermo Fisher Scientific for lending us the triple quadrupole mass spectrometer that was used in this work(TSQ Quantiva).
Appendix A. Supplementary data
Supplementary data to this article can be found online at https://doi.org/10.1016/j.jpha.2020.08.011.
杂志排行
Journal of Pharmaceutical Analysis的其它文章
- Comprehensive metabolic profiling of Alismatis Rhizoma triterpenes in rats based on characteristic ions and a triterpene database
- Evaluation of apoptotic effects of mPEG-b-PLGA coated iron oxide nanoparticles as a eupatorin carrier on DU-145 and LNCaP human prostate cancer cell lines
- Development of an analytical method for multi-residue quantification of 18 anthelmintics in various animal-based food products using liquid chromatography-tandem mass spectrometry
- Drug target discovery by magnetic nanoparticles coupled mass spectrometry
- Analytical methodologies for sensing catechol-O-methyltransferase activity and their applications
- Applications and challenges of low temperature plasma in pharmaceutical field
