Syntheses,Crystal Structures and Characterization of Two Polymeric Cadmium(Ⅱ)Complexes Based on 2-Methyl-5-(2-pyridyl)-1,3,4-oxadiazole
2020-10-12SUNAiJingBUDeYanZHANGHuiWANGZuoXiang
SUN Ai-Jing BU De-Yan ZHANG HuiWANG Zuo-Xiang
(School of Chemistry and Chemical Engineering,Southeast University,Nanjing 211189,China)
Abstract:Two polymeric cadmium(Ⅱ) complexes derived from 2-methyl-5-(2-pyridyl)-1,3,4-oxadiazole(L),[Cd2(L)2(μ-SCN)4]n(1)and[Cd2(L)2(μ-Cl)4]n(2),were synthesized and characterized by single crystal X-ray diffraction,UV-visible spectra,fluorescence and thermogravimetric analysis(TGA).Complexes 1 and 2 crystallize in triclinic and monoclinic system,respectively.The space groups of complexes 1 and 2 are and P21/n,respectively.Complex 1 is a double-bridged cadmium(Ⅱ) polymer bridged by two thiocyanate anions,and the central Cd1(Ⅱ) ions have distorted octahedral geometries[CdN4S2].Complex 2 is a double-bridged cadmium(Ⅱ)polymer bridged by two chloride anions,and the central cadmium(Ⅱ)ions have distorted octahedral geometries[CdCl4N2].Both complexes 1 and 2 have fluorescence properties.CCDC:1945282,1;1945285,2.
Keywords:1,3,4-oxadiazole;synthesis;crystal structure;cadmium(Ⅱ)complex;fluorescence
0 Introduction
1,3,4-Oxadiazole and its derivatives are important heterocyclic complexs,which play a significant role in the fields of medicine,pesticides and chemistry due to their extensive biological activities.Some 1,3,4-oxadiazole derivatives have important physiological activities,such as antibacterial[1],antiinflammatory[2],anticonvulsive[3],hypoglycemic[4],analgesic[5],anticancer[6],antidepressant[7],anti-tumor[8],insecticidal[9],antioxidant[10],antiviral[11]and anesthetic[12].The 1,3,4-oxadiazole ring can be introduced into organic complexes to synthesize special materials,such as electron transporting materials[13],corrosion inhibitors of metals[14-16]and stabilizers for polymer[17].
1,3,4-Oxadiazole ring possesses a planar structure and has aromatic properties.The nitrogen atoms on the 1,3,4-oxadiazole ring have strong electron-donating capacities,which can coordinate to transition metal ions and form stable complexes[18-21].Some metal complexes of 1,3,4-oxadiazole derivatives exhibit good biological activities[22-24].Khler and Rentschler reported[25]the first 1,3,4-oxadiazole based dinuclear iron(Ⅱ)complex which shows spin crossover behavior in 2016.Although metal complexes with 1,3,4-oxadiazole derivatives have been reported,few transition metal complexes with pyridine-substituted 1,3,4-oxadiazole complex have been reported so far.To continue our work[26],we reported here two polymeric cadmium(Ⅱ)complexes,bis(2-methyl-5-(2-pyridyl)-1,3,4-oxadiazole-κ2N,N′)tetra(μ-thiocyanato-κ2N,S)biscadmium(Ⅱ) polymer,[Cd2(L)2(μ-SCN)4]n(1),and bis(2-methyl-5-(2-pyridyl)-1,3,4-oxadiazole-κ2N,N′)tetra( μ-chloro)biscadmium (Ⅱ)polymer,[Cd2(L)2(μ-Cl)4]n(2),where L=2-methyl-5-(2-pyridyl)-1,3,4-oxadiazole.Their crystal structures,spectral properties and thermal stabilities were studied.
1 Experimental
1.1 Materials and measurements
2-Methyl-5-(2-pyridyl)-1,3,4-oxadiazole(L)was synthesized according to the literature method[26].All other chemicals were analytical grade and used without further purification.The UV-Vis spectra were recorded on UV-3900 UV spectrophotometer.Fluorescence spectral data were obtained on F-2500 fluorescence spectrometer.Single crystal data were collected on a Bruker Smart APEXⅡCCD single crystal X-ray diffractometer.Thermogravimetric analysis(TGA)was performed on a NETZSCH TG209C thermal analyzer in nitrogen atmosphere at a heating rate of 10 K·min-1.
1.2 Synthesis of complex 1
Ligand L(0.097 g,0.6 mmol)was dissolved in 20 mL warm distilled water,then 0.093 g Cd(NO3)2·4H2O(0.3 mmol)and 0.058 g KSCN(0.6 mmol)were added to the warm solution with stirring,and a white precipitate formed.The solution became clear after 14 mL distilled water was added,then it was filtered,and the filtrate was left to stand at room temperature for slowly evaporating.Several days later white crystals were collected(0.084 g,yield:71.8%).A single crystals suitable for X-ray diffraction was picked.Anal.Calcd.for C10H7CdN5OS2(%):C 30.82,H 1.81,N 17.97;Found(%):C 30.71,H 1.76,N 17.84.UV-Vis(thin acetonitrile, λ/nm):235.66 (εmax=3.084×103L·mol-1·cm-1),276.30(εmax=1.682×104L·mol-1·cm-1).
1.3 Synthesis of complex 2
Ligand L(0.161 g,1 mmol)was dissolved in 30 mL warm acetonitrile,then 0.114 g CdCl2·2.5H2O(0.5 mmol)dissolved in 5.0 mL distilled water was added with stirring,and the solution became turbid.The solution became clear after 12 mL distilled water was added,then it was filtered,and the filtrate was left to stand at room temperature for slowly evaporating.Several days later,white crystals were obtained(0.120 g,yield:69.8%).A single crystal suitable for X-ray diffraction was picked.Anal.Calcd.for C8H7CdCl2N3O(%):C 27.89,H 2.05,N 12.20;Found(%):C 27.70,H 1.89,N 12.06.UV-Vis(thin acetonitrile,λ/nm):238.68(εmax=1.970×103L·mol-1·cm-1),275.74(εmax=1.343×103L·mol-1·cm-1).
1.4 Crystal structure determination
Selected single crystals of 1 and 2 were mounted on a Bruker SMART APEXⅡCCD X-ray diffractometer with a detector distance of 5 cm and frame exposure time of 10 s using graphite-monochromated Mo Kα (λ=0.071 073 nm)radiation at 293(2)K.The structures were solved by direct methods and refined on F2by fullmatrix least squares procedures using SHELXTL-2014 software[27].All non-hydrogen atoms were anisotropically refined,and all hydrogen atoms on carbon atoms were generated geometrically on the parent atoms,but not refined.Crystal data and structure refinements for complex 1 and 2 are listed in Table 1.Selected bond lengths and angles for 1 and 2 are listed in Table 2.
CCDC:1945282,1;1945285,2.

Table 1 Crystal data and structure refinement for 1 and 2
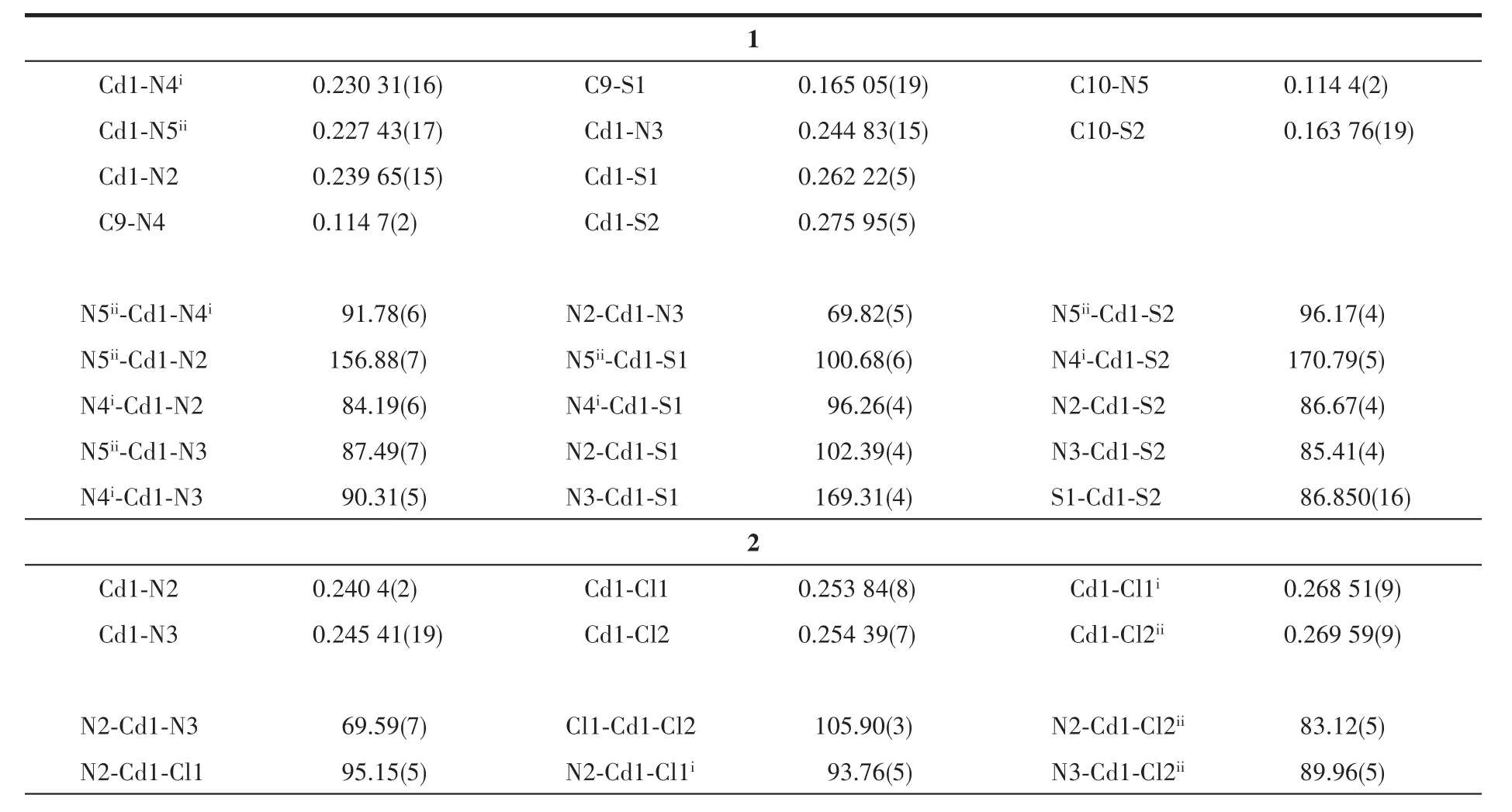
Table 2 Selected bond lengths(nm)and bond angles(°)for 1 and 2

Continued Table 2
2 Results and discussion
2.1 Crystal structural analyses
2.1.1 Crystal structure of 1
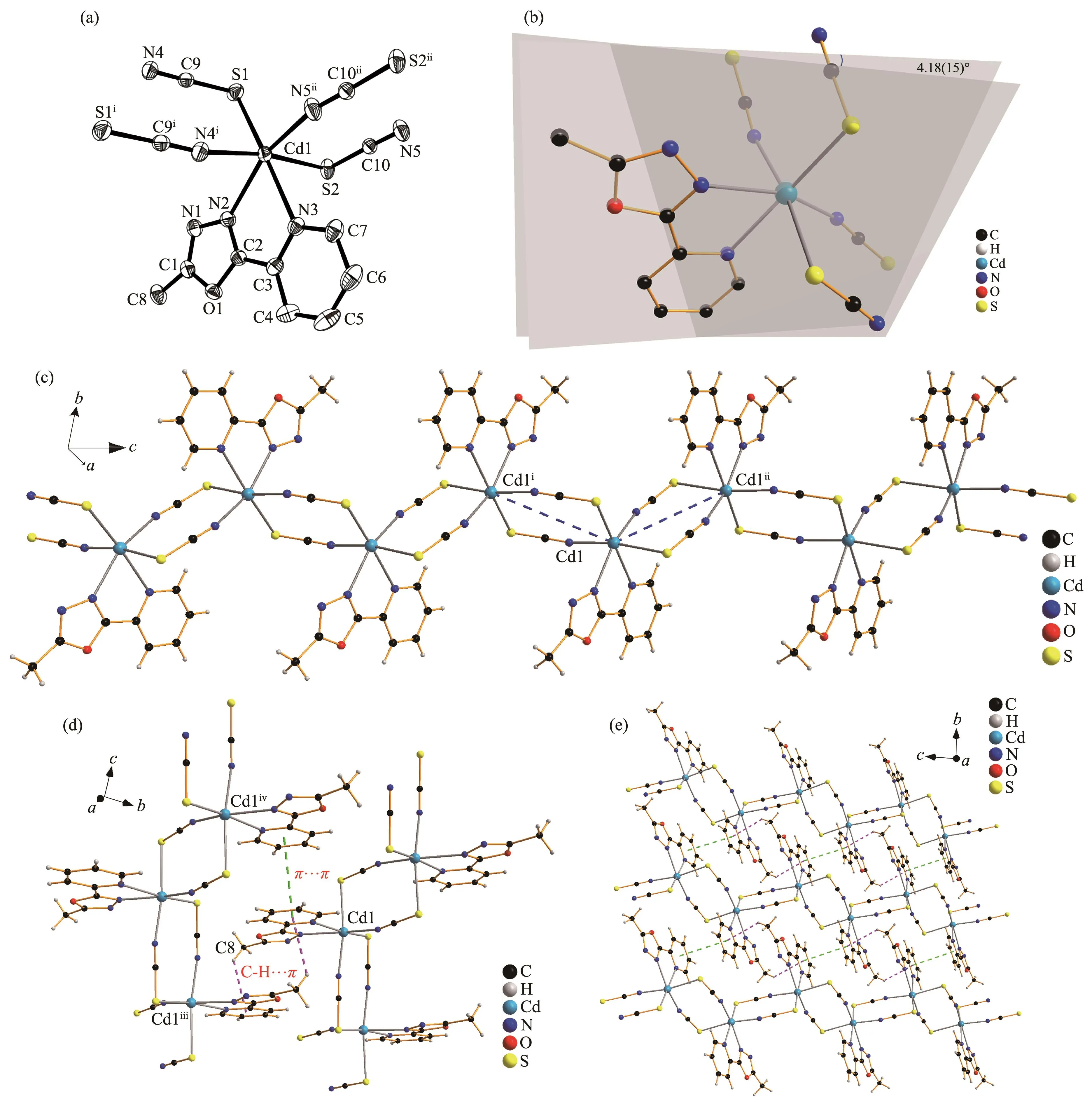
Fig.1 (a)Structure of 1 with ellipsoids at 30% level and the hydrogen atoms omitted for clarity,symmetry codes:i1-x,1-y,-z;ii1-x,1-y,1-z;(b)Diagram of 1 showing the dihedral angle between the oxadiazole and pyridine rings;(c)Segment of polymeric chain of 1;(d)Non-covalent interactions in 1,symmetry codes:iii2-x,-y,-z;iv2-x,-y,1-z;(e)2D structure of 1 linked through π…π and C-H…π interactions
Complex 1 crystallizes in triclinic system with space group P1(Fig.1a).It is a polymeric cadmium(Ⅱ)complex bridged by two thiocyanate anions(Fig.1c).The distances between the adjacent cadmium ions Cd1-Cd1iand Cd1-Cd1iiare 0.579 24(2)and 0.572 98(2)nm,respectively.In complex 1,the central Cd1(Ⅱ)ions are coordinated by four thiocyanate ions through nitrogen or sulfur atoms(N4i,N5ii,S1 and S2).The central Cd1(Ⅱ)ions are also coordinated by two nitrogen atoms from L(N2 and N3),and every central Cd1(Ⅱ)ion possesses a distorted octahedral geometry[CdN4S2].The bond lengths of Cd1-S1,Cd1-S2,Cd1-N4iand Cd1-N5iiare 0.262 22(5),0.275 95(5),0.230 31(16)and 0.227 43(17)nm,respectively.The bond lengths of Cd1-N2 and Cd1-N3 are 0.239 65(15)and 0.244 83(15)nm,respectively.
In complex 1,the 1,3,4-oxadiazole ring and pyridine ring are coplanar with a dihedral angle of 4.18(15)°(Fig.1b).There is a weak face-to-face π…π interaction(Cg3…Cg3iv)between pyridine rings with a centroidcentroid distance of 0.389 39(12)nm.In addition,the pyridine ring involves an edge-to-face C-H…π interaction(C8-H8B…Cg3iii).The polymeric chains of 1 are linked together through π…π and C-H…π interactions to form a two-dimensional network structure[28](Fig.1d and 1e,Table 3).

Table 3 C-H…π and π…π interactions for 1
2.1.2 Crystal structure of 2
Complex 2 crystallizes in monoclinic system with space group P21/n.It is a polymeric cadmium(Ⅱ)complex bridged by two chloride anions(Fig.2a,2b and 2c).The distances between the adjacent cadmium ions Cd1-Cd1iand Cd1-Cd1iiare 0.384 19(10)and 0.388 5(1)nm,respectively.The Cd1(Ⅱ)ions are coordinated by four bridged chlorine atoms(Cl1,Cl1i,Cl2 and Cl2ii)and two nitrogen atoms(N2 and N3)from the ligand of L,and the central Cd1(Ⅱ)ions have distorted octahedral geometries[CdCl4N2].The bond lengths of Cd1-Cl1,Cd1-Cl1i,Cd1-Cl2 and Cd1-Cl2iiare 0.253 84(8),0.268 51(9),0.254 39(7)and 0.269 59(9)nm,respectively.The bond lengths of Cd1-N2 and Cd1-N3 are 0.240 4(2)and 0.245 41(19)nm,respectively.
In complex 2,the 1,3,4-oxadiazole ring and pyridine ring are coplanar with a dihedral angle of 5.33(11)°(Fig.2b).One non-classical hydrogen bond of C4-H4…Cl2iii(Symmetry code:x,-1+y,z)exists between the C4 and Cl2 atoms(Table 4 and Fig.2d).There is also a weak face-to-face π…π interaction(Cg4…Cg5v)between the 1,3,4-oxadiazole ring and pyridine ring with a centroid-centroid distance of 0.378 18(18)nm.In addition,an edge-to-face C-H…π interaction involving the pyridine ring,C8-H8C…Cg5iv(Symmetry code:1-x,1-y,-z).The polymeric chains of 2 are linked together through hydrogenbonding,π…π and C-H…π interactions to form a two-dimensional network structure[28](Fig.2e).

Table 4 Hydrogen bonds,C-H…π and π…π interactions for 2
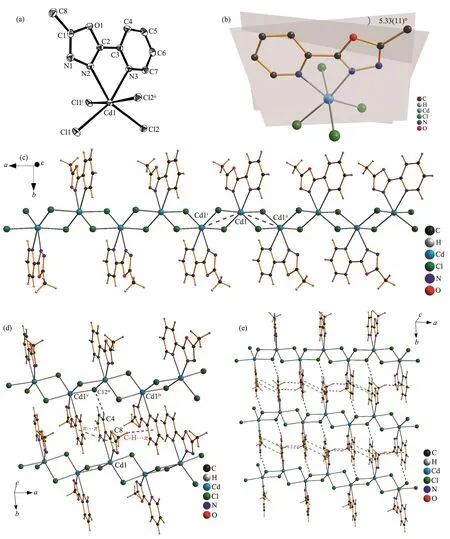
Fig.2 (a)Structure of 2 with ellipsoids at 30% level and the hydrogen atoms are omitted for clarity,symmetry codes:i1-x,2-y,-z;ii-x,2-y,-z;(b)Diagram of 2 showing the dihedral angle between oxadiazole and pyridine rings;(c)Segment of the polymeric chain;(d)Non-covalent interactions in 2,symmetry codes:iiix,-1+y,z;iv1-x,1-y,-z;v-x,1-y,-z;(e)2D structure of 2 linked through H-bonding,π…π and C-H…π interactions
2.2 Spectral characterizations
The UV-Vis spectra of 1 and 2 in acetonitrile solution are shown in Fig.3a.The maximum absorption peaks of complex 1 appeared at 235.66 nm(ɛmax=3.084×103L·mol-1·cm-1)and 276.30 nm(ɛmax=1.682×104L·mol-1·cm-1),and those for 2 appeared at 238.68 nm(ɛmax=1.970×103L·mol-1·cm-1)and 275.74 nm(ɛmax=1.343×103L·mol-1·cm-1).All these are attributed to the π-π*transition of ligand L.
The fluorescence properties of 1 and 2 were investigated in acetonitrile solution at room temperature(Fig.3b).The maximum emission wavelength of com-plexes 1 and 2 were 357 nm(λmax,ex=274.5 nm)and 358 nm(λmax,ex=274 nm)with the fluorescence quantum yields of 0.22 and 0.54,respectively.All these are attributed to the π-π*transitions of ligand L.The fluorescence quantum yields of 1 and 2 were calculated using 2-amino-pyridine(Φ=0.60 in H2SO4,0.05 mol·L-1)as the standard according to the following equation:
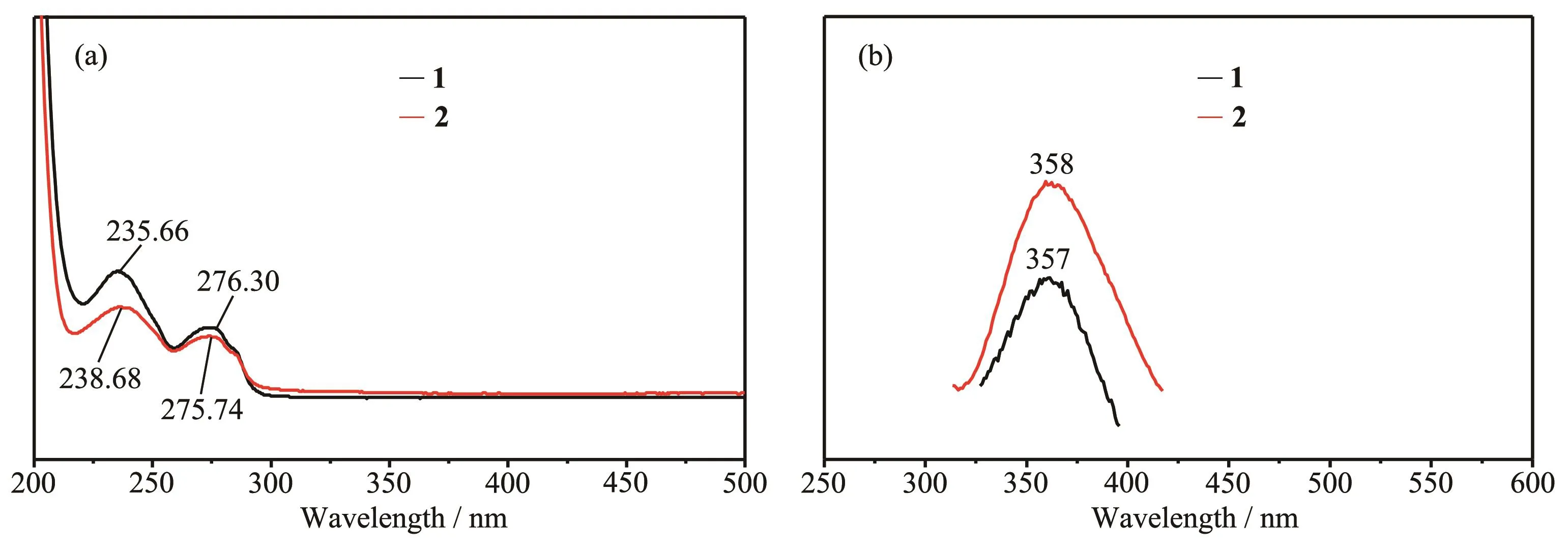
Fig.3 UV-Vis(a)and fluorescence(b)spectra for 1 and 2 in acetonitrile solution

The sample and std represent the sample and reference sample of 2-amino-pyridine,respectively;I represents the fluorescence integral strength;A represents the absorbance;n represents refractive index of the solvent.
2.3 Thermal gravimetric analyses
Thermalgravimetric analyses(TGA)of complexes 1 and 2 were performed on a NETZSCH TG209C thermal analyzer in nitrogen atmosphere with a heating rate of 10 K·min-1.The final residues of complex 1 and 2 were determined by X-ray energy dispersive spectroscopy(EDS)analyses on EDAX ELEMENT.
The TGA curves of complexes 1 and 2 are presented in Fig.4.Complex 1 was stable below 150℃and decomposed sharply between 220 and 320℃.Total weight loss is equal to 49.52% at 650℃.EDS analyses show that the residues are Cd(Fig.5a)and Cd2S(Fig.5b).
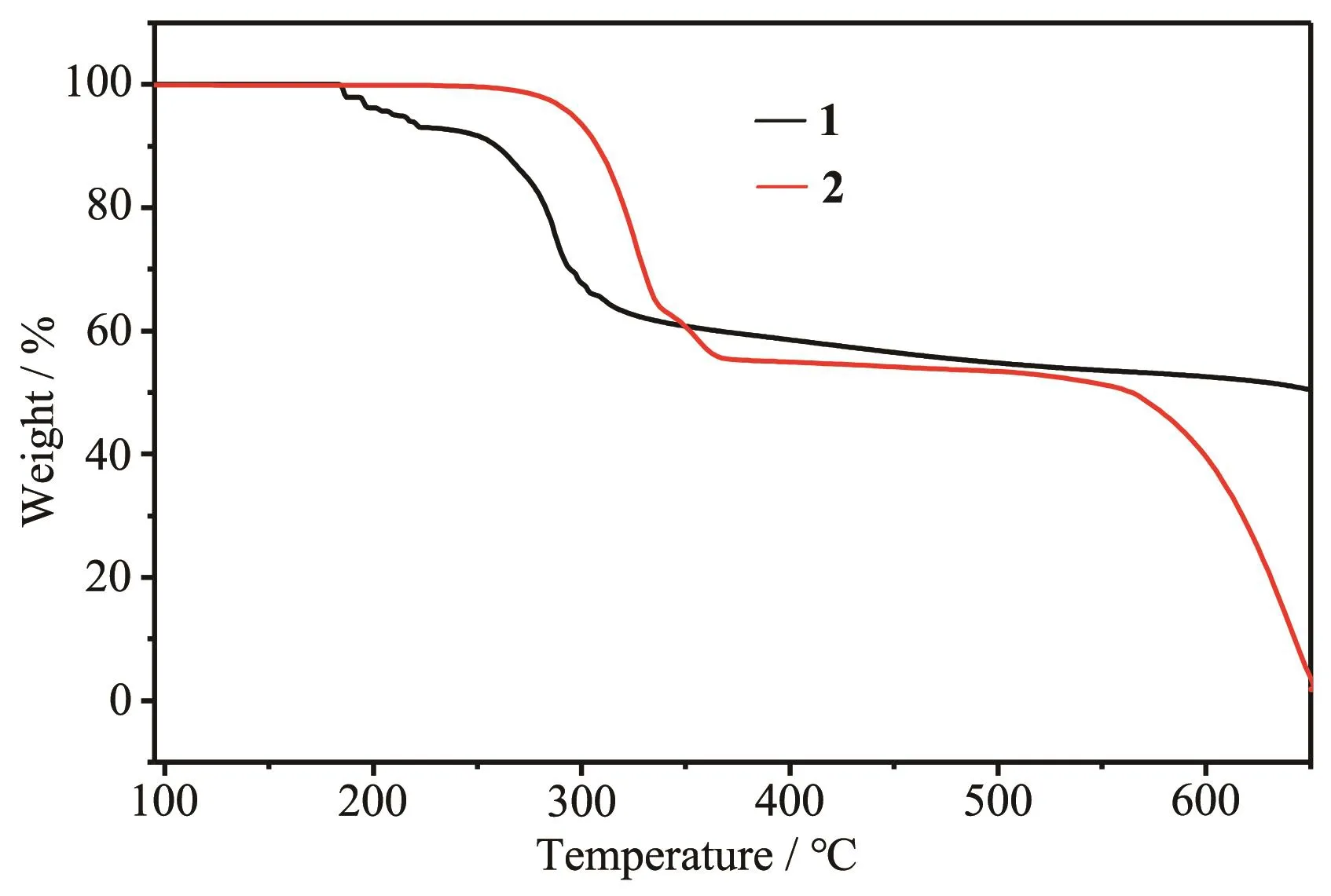
Fig.4 TGA curves for complexes 1 and 2

Fig.5 Scanning electron microscopy(SEM)images and EDS spectra of the residues of 1
Complex 2 was stable below 270℃and decom-posed sharply between 280 and 360℃.When the temperature was 500℃,the total weight loss was equal to 46.57%.EDS analyses show that the residues at 500℃are CdCl2with a little CdO(Fig.6).When the temperature continued to rise,almost no residue was left and observed at 650℃(the total weight loss was close to 98.05%).

Fig.6 SEM image and EDS spectrum of the residue of 2
3 Conclusions
Two polymeric cadmium(Ⅱ) complexes of[Cd2L2(μ-SCN)4]n(1)and[Cd2L2(μ-Cl)4]n(2)were synthesized and characterized.Complex 1 is a double-bridged cadmium(Ⅱ)complex through two thiocyanate anions,and the central cadmium(Ⅱ) ions have distorted octahedral geometries[CdN4S2].Complex 2 is a double-bridged cadmium(Ⅱ)complex through two chloride anions,and the central cadmium(Ⅱ)ions have distorted octahedral geometries[CdCl4N2].Both complexes of 1 and 2 have fluorescence properties.
