Acute and chronic toxicity studies of hydromethanol leaf extract of Helianthus annuus Linn. in rats
2018-10-16SamuelOnojaSamuelUdemAruhAnagaIsaacAsuzu
Samuel O. Onoja, Samuel C. Udem, Aruh O. Anaga, Isaac U. Asuzu
1Department of Veterinary Physiology and Pharmacology, Michael Okpara University of Agriculture, Umudike, PMB 7267 Umuahia Abia State, Nigeria
2 Department of Veterinary Physiology and Pharmacology, Faculty of Veterinary Medicine, University of Nigeria, Nsukka, 410001 Enugu State, Nigeria
Keywords:Helianthus annuus Toxicity Histopathology Serum biochemistry
ABSTRACT Objective: To investigate the safety levels of hydromethanol leaf extract of Helianthus annuus Linn. (H. annuus) in rat. Methods: Acute oral toxicity test of hydromethanol leaf extract of H.annuus was conducted through up and down method at 2.00 g/kg dose limit in rats. The chronic toxicity study was conducted by administering different concentrations (0.25, 0.50 and 1.00 g/kg) of hydromethanol extract of H. annuus in feed, for 90 consecutive days. On days 30, 60 and 90, blood samples were collected from the retro-orbital plexus of the rats for determination of serum biochemical parameters. Histopathological examination of the pancreas, livers, kidneys and testis were also conducted. Results: The LD50 of the hydromethanol extract of H. annuus was greater than 2.00 g/kg and it significantly (P < 0.05) reduced serum cholesterol. On days 60 and 90, the serum urea and creatinine levels of hydromethanol extract of H. annuus treated groups were elevated when compared with the control group. There were fibrosis in the kidneys and livers; degeneration and necrosis in the testis and significant dose-dependent increases in number and size of pancreatic islet of langerhans. Conclusions: The findings suggest that hydromethanol extract of H. annuus is tolerated in short term administration,but long term (up to 90 days) administration at high doses, may elicit hepatic, testicular and nephrotic disorder.
1. Introduction
Plant products form integral part of primary health-care system of many countries[1]. More than 80% of Asian and African population explore traditional medicines for their health-care needs[2]. Plant derived medicines are easily accessed, affordable, efficacious and culturally acceptable form of drug with reduced side-effects, which makes the combat of diseases possible in the midst of escalating cost of orthodox medicine and economic crunch[3]. As a result,the updated World Health Organization Traditional Medicine Strategies for 2014–2023 are to help member states in utilizing the contributions of traditional medicine to health and encourage the safe and judicious use of traditional medicine through regulation,evaluation and inclusion of traditional medicine products,practitioners and practice into health care systems[2,4]. Despite this,the professional health-care providers hesitate to recommend herbal products to their patients due to lack of information on the safety/toxicity profile of most herbal products[5].
Helianthus annuusLinn. (H. annuus, sunflower) is a member of the family Asteraceae and can be found in many countries and regions. It is an ornamental plant and the seed is a good source of vegetable oil as well as seed meal for farm animals[6].H. annuusleaves, decoctions and infusions are used in folkloric medicine as diuretic, antidiabetic, expectorant, gastrointestinal stimulant,antimicrobial, analgesic agentsetc.[7,8]. Poultices ofH. annuusleaves are applied on wound, snakebite and swelling[9]. The leaves ofH. annuusare endowed with pharmacological activities such as antispermatogenic, analgesic and anti-inflammatory, antidiabetic,antioxidant, antifertility, antidiarrheal, antihistaminic, antilithiatic and antimicrobial activities[9,10]. There is a dearth of information on the safety profile ofH. annuus. We thought it’s needful to investigate the safety level of the hydromethanol extract ofH. annuususing biochemical and histopathological examinations after prolonged exposure to rats.
2. Materials and methods
2.1. Extract preparation
The leaf ofH. annuuswas collected from the wild in Nsukka,Enugu state, Nigeria and authenticated by a plant taxonomist(Voucher number, UNN/VPP/2012/2). The leaves were dried under shed at ambient temperature (25–27) ℃. The extract ofH. annuuswas prepared using cold maceration method as described by Onoja and Anaga[11] and was referred to as hydromethanol leaf extract ofH. annuus.
2.2. Animals
Fifty six male albino Wistar rats were used for the study. They were housed in aluminum cages in a well-ventilated room at ambient temperature (25–27 ℃) and natural light/darkness cycle. The rats were acclimatized for 2 weeks and the ethical approval for this study(UNTH/CSA.329/VOL10) was provided by the Research Ethical Committee of University of Nigeria Teaching Hospitals, Enugu state,Nigeria, on 12 June 2016. The animals were humanely handled as stipulated in the Guide for the Care and Use of Laboratory Animals of National Research Council.
2.3. Feed formulation
The hydromethanol extract ofH. annuuswas incorporated in the feed at the rate of 0.25, 0.50 and 1.00 g/kg as described by Onojaet al.[9].
2.4. Acute toxicity
The up and down procedure with dose limit of 2.00 g/kg was adopted. Ten (10) rats were used for the study. Five rats were given 2.00 g/kg of hydromethanol extract ofH. annuusand the rest were given equal volume of 5% tween-20 orally. The rats received feed and waterad libitumand were monitored for signs of acute toxicity and death.
2.5. Experimental design
The rats were randomly divided into 4 groups (A–D,n=14). They were treated as follows: group A: Standard feed (control), and groups B-D: hydromethanol extract ofH. annuus0.25, 0.50 and 1.00 g/kg feed, respectively. The rats were fed (10% of their body weight daily) for 90 consecutive days[9]. Feed wastage was minimised by using deep feeding troughs. The rats were weighed on weekly basis and feeding was recalculated to accommodate the weight changes.Four selected rats from each group were bled from the retro orbital venous plexus with capillary tube, into plain sample bottles to obtain serum. The sera were stored in a refrigerator and the parameters were analyzed within 72 h.
2.5.1. Relative organ weights (ROW)
On days 30, 60 and 90, 4 rats were sacrificed from each group through cervical dislocation and vital organs; livers, kidneys, lungs,spleens, testis and hearts, were excised, weighed and observed for gross changes. The ROW were calculated using the formula:
ROW = weight of organ ÷÷weight of animal ×100%
2.5.2. Serum biochemical analysis
The serum total cholesterol (TC), triglycerides (TG), high density lipoprotein cholesterol (HDL-C), aspartate aminotransferase (AST),alanine aminotransferase (ALT), alkaline phosphatase (ALP), total protein, albumin, creatinine and urea levels were determined with commercially available diagnostic kit (Randox Diagnostic, United Kingdom). Serum low density lipoprotein cholesterol (LDL-C) and very low density lipoprotein-cholesterol (VLDL-C) were calculated using Friedewald’s equation as follows:

2.5.3. Histopathology
Sections of the pancreas, livers, kidneys and testis were prepared as described by Ezejaet al.[13]. Photomicrographs were captured at 400 × magnifications with an Olympus photomicroscope (Olympus Scientific Equipment, Ashburn, VA).
2.6. Data analysis
The data obtained were statistically evaluated using one-way analysis of variance (ANOVA), followed by least significant difference test with SPSS software version 20. The mean differences were considered significant at level ofP< 0.05.
3. Results
3.1. Acute toxicity
The rats tolerated the extract and no signs of toxicity, morbidity and mortality were recorded; the rats were healthy, well-fed and had normal feacal consistency. The LD50of the hydromethanol extract ofH. annuuswas greater than 2.00 g/kg.
3.2. Body weight and relative organ weight
The body weight of all groups increased (P< 0.05) from day 1 to day 90. The growth rate of the group treated with the hydromethanol extract ofH. annuus(0.50 g/kg feed) was higher (P<0.05) than other treatment groups, from week 5 till the end of the experiment(Figure 1). There was no significant (P>0.05) change in the ROW of the group treated with hydromethanol extract ofH. annuuswhen compared with the control group (Table 1). At days 60 and 90, the ROWs of testis of groups treated with hydromethanol extract ofH.annuuswere higher (P> 0.05) than the control group (Table 1).

Figure 1. Effects of chronic exposure of rats to hydromethanol extracts of H. annuus for 90 days on percentage weight gain.

Table 1 Relative organ weight (%).
3.3. Biochemical parameters
On day 30, hydromethanol extract ofH. annuusproduced significant (P< 0.05) decrease in the serum cholesterol in the treated groups when compared with the control group. The hydromethanol extract ofH. annuus0.50 and 1.00 g/kg feed reduced (P<0.05) serum cholesterol in treated groups when compared with the control group on days 90 and 60, respectively(Table 2). On days 30 and 90, the triglyceride levels of hydromethanol extract ofH. annuustreated groups were significant lower (Pall<0.05) when compared with the control group. On day 60, triglyceride levels of hydromethanol extract ofH. annuus(0.25 and 1.00 g/kg feed) treated groups were decreased (P<0.05)comparing with the control group (Pboth<0.05) (Table 2). The VLDL-C levels of hydromethanol extract ofH. annuus(0.25 and 0.50 g/kg feed) treated groups were lower (P<0.05) than the control group on day 30. On days 60 and 90, the VLDL-C levels of hydromethanol extract ofH. annuus(1.00 g/kg feed) treated group were low (P< 0.05) when compared with the control group (Table 2). However, the serum HDL-C levels of hydromethanol extract ofH. annuustreated groups were higher than the serum HDL-C level of the control group (Table 2). Hydromethanol extract ofH. annuus(0.50 and 1.00 g/kg feed) caused significant (P<0.05) decrease in the LDL-C levels of the treated group when compared with control group (Table 2).

Table 2 Effects of chronic exposure of rats to hydromethanol extracts of H.annuus for 90 days on serum lipid profile.
At days 30, 60 and 90, the ALP, AST and ALT activities of the hydromethanol extract ofH. annuustreated groups were not significant different from that of the control group (P>0.05) (Table 3). On day 30, serum urea and creatinine levels of hydromethanol extract ofH. annuustreated groups were not significant (P>0.05)when compared with the control group. But on day 60 and 90,the serum urea and creatinine levels of hydromethanol extract ofH. annuustreated groups were elevated when compared with the control group (Table 3).
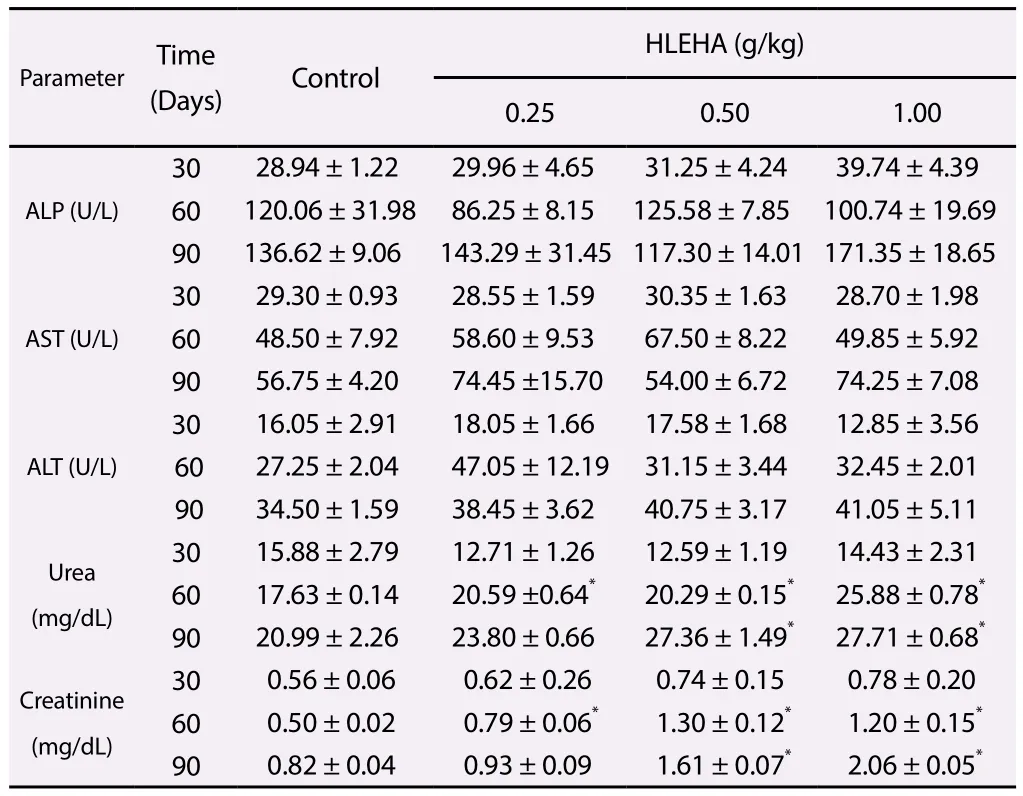
Table 3 Effects of chronic exposure of rats to hydromethanol leaf extracts of H.annuus for 90 days on serum enzyme markers of liver and kidney function.
3.4. Histopathology
The photomicrograph sections of the spleens, hearts and lungs of rats showed normal architecture. However, the treatment of the rats for 90 with hydromethanol extract ofH. annuuscaused histological changes in the livers, kidneys, testis and pancreas at varied periods and doses. In the pancreas, the hydromethanol extract ofH. annuustreatment caused dose- and time-dependent increases in number and size of pancreatic islets of Langerhans when compared with the control group (Figure 2). In the livers, hydromethanol extract ofH.annuus(0.50 and 1.00 g/kg feed) at day 60 caused congestion of the sinusoids, portal and central vein. At day 90, the extract caused dose-dependent hyperplasia of the bile duct and in addition to this, hydromethanol extract ofH. annuus0.50 and 1.00 g/kg feed,caused moderate fibrosis of the portal triad (Figure 3). At days 60 and 90, the hydromethanol extract ofH. annuustreatment caused congestion of renal blood vessel and peritubular fibroplasia in a dose dependent manner (Figure 4). In the testis, the hydromethanol extract ofH. annuusdid not cause any histopathological lesion on day 30 while on days 60 and 90, it produced degeneration and necrosis of the spermatogenic and sertoli cells. The lumen of the seminiferous tubule of the 1.00 g/kg extract feeded group was obliterate by necrotic debris on day 90, while the photomicrograph section of group B was normal on day 60 (Figure 5).

Figure 2. Photomicrograph of sections of the pancreas of experimental rats after 90 days (chronic study) treatment with hydromethanol leaf extract of H.annuus (H&E, ×400).
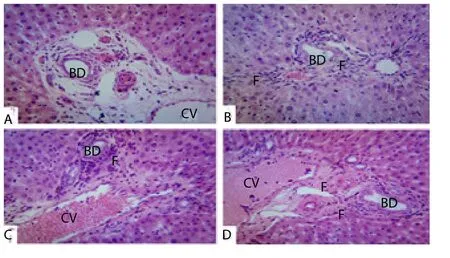
Figure 3. Photomicrograph of liver section (H&E, ×400) from experimental rats after 90 day (chronic study) treatment with HLEHA incorporated in feed.

Figure 4. Photomicrograph of kidney section (H&E, ×400) from experimental rats after 90 day (chronic study) treatment with HLEHA incorporated in feed.The ‘G’ indicates the glomerulus, ‘DN’ shows area of degeneration and necrosis; ‘F’ shows fibroblast cell and ‘H’ shows heamorrhage. A = control group; B = HLEHA 0.25 g/kg feed group; C = HLEHA 0.50 g/kg feed group; D = HLEHA 1.00 g/kg feed group.

Figure 5. Photomicrograph of testis section (H&E, ×400) from experimental rats after 90 day (chronic study) treatment with HLEHA incorporated in feed.
4. Discussion
The chronic exposure of the rats to hydromethanol extract ofH.annuuselicited hypolipidemic effects, increased the pancreatic islet size and mass, and as well caused hepatic fibrosis, testicular and kidney damage in the treated rats.
The chronic toxicity test investigated the safety levels of the hydromethanol extract ofH. annuusin rats. The study, which lasted for 90 days, was to ascertain if hydromethanol extract ofH. annuuswould cause toxic effects on chronic exposure. More so, the anti-diabetic potential of the extract makes it imperative to be used for long period of time[14]. Incorporation of the extract in feed was adopted to avoid the stress of daily handling and dosing associated with administering the extract by oral gavage. The rate of incorporation of the extract in the feed was based on the report of previous studies[15].
No signs of morbidity or mortality were noticed in the treated groups throughout the duration of the experiment, which indicates that the hydromethanol extract ofH. annuuswas tolerated by the rats.There was a progressive weight gain in all the treated animal groups but the group treated with hydromethanol extract ofH. annuus0.5 g/kg feed had more accelerated growth rate. The accelerated weight gain may be due to improved feed conversion efficiency which can be linked to the presence of some micronutrients, vitamins and phytochemicals in hydromethanol extract ofH. annuusthat may act as growth promoter[15]. The relative organ weight showed no significant change in all the organs (livers, kidneys, hearts, lungs,spleens) examined. This indicates that there was no adverse toxic effect on these organs as increase in ROW indicates marked organ degeneration[16].
The extract demonstrated good hypolipidemic effects which suggest that it might be useful in the management of hyperlipidemia-related diseases such as cardiovascular diseases and diabetes mellitus[17].Dyslipidemia causes arteriosclerosis, hypertension and may also lead to insulin resistance, which predisposes to diabetes mellitus[18,19].The mechanism of the hypolipidemic effects of hydromethanol extract ofH. annuusis not known but could be through the inhibition of intestinal absorption of cholesterol, increased activity of HMG-CoA reductase and lipase activities[20].H. annuuscontains phytosterol which interferes with intestinal cholesterol absorption by binding to the same receptor site[21,22].
The liver plays major role in nutrients and xenobiotics metabolism.As a result, it is usually affected by toxicants, which may lead to hepatocellular damage and reduce functional capacity. Hepatocellular damage and functional capacity of the liver are evaluated by determining the serum activities of some cytosolic enzymes (AST,ALT, ALP) and serum levels of proteins. The elevated activities of these cytosolic enzymes indicate hepatocyte or other cell-type damage[23]. The liver is also involved in the synthesis of proteins.Here, the total protein, albumin and globulin levels of the treated rats did not change significantly. This finding may suggests that protein metabolism was not affected[24]. The liver section showed mild congestion, hyperplasia of bile duct and fibrosis, the activities and level of biochemical markers of liver function were not significantly elevated. This indicates that the liver damage was not severe enough to cause a negative impact on its functional capacity. This is possibly due to reserved functional capacity of the liver; about 90% of the liver has to be damaged or compromised before it manifests signs of toxicity[25].
The functional capacity of the kidney was evaluatedviathe determination of the serum urea and creatinine levels. The observed degeneration, necrosis and fibrosis of the Bowman’s capsule and peritubular interstitium of the kidney corroborated the elevated serum urea and creatinine level on days 60 and 90. The serum urea and creatinine levels are elevated when about 75% of the kidney tissue has been damaged and this is due to reserved functional capacity of the kidney[26,27]. The kidney damage may be responsible forH. annuusinduced normocytic hypochromic anaemia reported by Onojaet al.[9].
The hydromethanol extract ofH. annuusproduced increased size and density of the pancreatic islet in treated rats. This suggests that hydromethanol extract ofH. annuuscould cause increased insulin production and this might be responsible for its antidiabetic potential[11,28].
The histopathologic examination of the testicle showed degeneration and necrosis of the spermatogenic and sertoli cells on days 60 and 90. This indicates that the extract might have caused reduced sperm count and testosterone level, which was not investigated in this study. The available literature have reported the antifertility (reduced sperm count, testosterone level and fecundity)property ofH. annuusleaves[29]. The study of Ejebeet al.[29]reported the testicular damage at day 14 post treatment and this was linked to the higher doses (0.5 and 2.0 g/kg) used in their study. The reduction in testosterone level may discourage its prolonged use in male diabetic patients to avoid compounding the problem of sexual dysfunction that is common with them[30].
The findings of the study suggest thatH. annuusis tolerated in short term administration and caution must be exercised in long-term therapy. The long-term (up to 90 days) administration at high doses,may elicit hepatic, testicular and nephrotic disorder. Therefore, the medicinal values of hydromethanol extract ofH. annuuswill be fully utilized if treatment is for a short period and low doses should be used for long term treatment.
Conflict of interest statement
We declare that we have no conflict of interest.
杂志排行
Asian Pacific Journal of Tropical Medicine的其它文章
- Role of dietary phytochemicals in modulation of miRNA expression:Natural swords combating breast cancer
- Antihyperglycemic effect of Passiflora glandulosa cav. fruit rinds flour in streptozotocin-induced diabetic mice
- Ameliorative effect of Pergularia daemia (Forssk.) Chiov. leaves extract against anti-tuberculosis drugs induced liver injury in rats
- Distribution and ecological aspects of sand flies (Diptera: Psychodidae)species in Northeastern Iran
- Nano chloroquine delivery against Plasmodium berghei NK65 induced programmed cell death in spleen
- Enhanced surveillance and cohesive policies needed to tackle Rift Valley fever
