艾滋病疫苗
2016-03-21
艾滋病疫苗
·编者按·
艾滋病又称获得性免疫缺陷综合症(Acquired Immunodeficiency Syndrome,AIDS),由感染人免疫缺陷病毒(Human Immunodeficiency Virus,HIV)引起。机体感染HIV后,免疫系统受到破坏,抗感染能力下降,成为许多伺机性疾病的攻击目标,促成多种临床症状,伴有机会感染、恶性肿瘤及神经障碍等多种症候群,是一种危害性极大的传染病。
自20世纪80年代初首例报道艾滋病病例以来,艾滋病在全球范围迅速感染和蔓延。2015年12月1日是第28个世界艾滋病日,全球现有大约3690万人感染艾滋病毒,其中约有200万人是2014年新增感染者。迄今为止,估计有3400万人左右因艾滋病毒或艾滋病死亡,其中120万人死于2014年。因而,尽管国际社会在防治艾滋病方面取得了重要进展,艾滋病和艾滋病病毒依然是人类健康最严峻的挑战之一。
目前尚未发现对艾滋病有效的治愈方法,研制艾滋病疫苗是控制艾滋病流行甚至根除艾滋病的理想途径,疫苗免疫接种也是控制传染病的最经济有效的措施。自1987年第一个艾滋病疫苗进入临床试验以来,按照研究的侧重点划分,艾滋病疫苗研究进程大致分为3个阶段:体液免疫的中和抗体阶段、刺激CD8 T细胞介导的细胞免疫阶段以及体液免疫和细胞免疫两者结合阶段。尚在研究的HIV疫苗包括HIV灭活疫苗、HIV减毒活疫苗、亚单位疫苗、活载体病毒蛋白疫苗和DNA疫苗等。
虽然有效的HIV疫苗还未研发出来,但新免疫策略不断涌现,泰国进行的RV-144Ⅱ期临床试验,证明了保护性的HIV疫苗的可能性,为HIV疫苗的研发带来了希望。一些医药公司也对外宣称其研发取得的新进展,比如法国生态健康科技公司称其研发出可望彻底治愈艾滋病的疫苗,2016年将确定疫苗的最终配方,从2017年起开始全球市场营销,最终的效果令人期待。
本专题得到江文正教授(华东师范大学生命科学学院)的大力支持。
·热点数据排行·
截至2015年12月1日,中国知网(CNKI)和Web of Science(WOS)的数据报告显示,以“艾滋病疫苗”为词条可以检索到的期刊文献分别为716与829条,本专题将相关数据按照:研究机构发文数、作者发文数、期刊发文数、被引用频次进行排行,结果如下。

研究机构发文数量排名(CNKI)

研究机构发文数量排名(WOS)
(数据来源:中国知网、Web of Science,检索时间:2015-12-01)
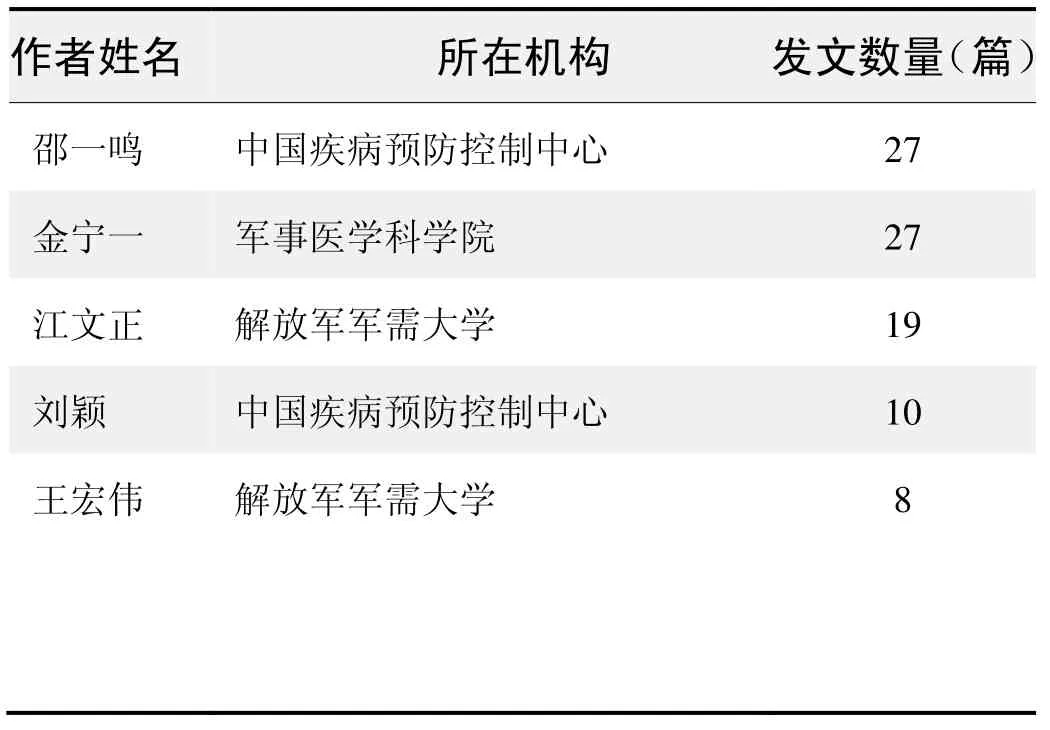
作者发文数量排名(CNKI)

作者发文数量排名(WOS)
(数据来源:中国知网、Web of Science,检索时间:2015-12-01)

期刊发文数量排名(CNKI)
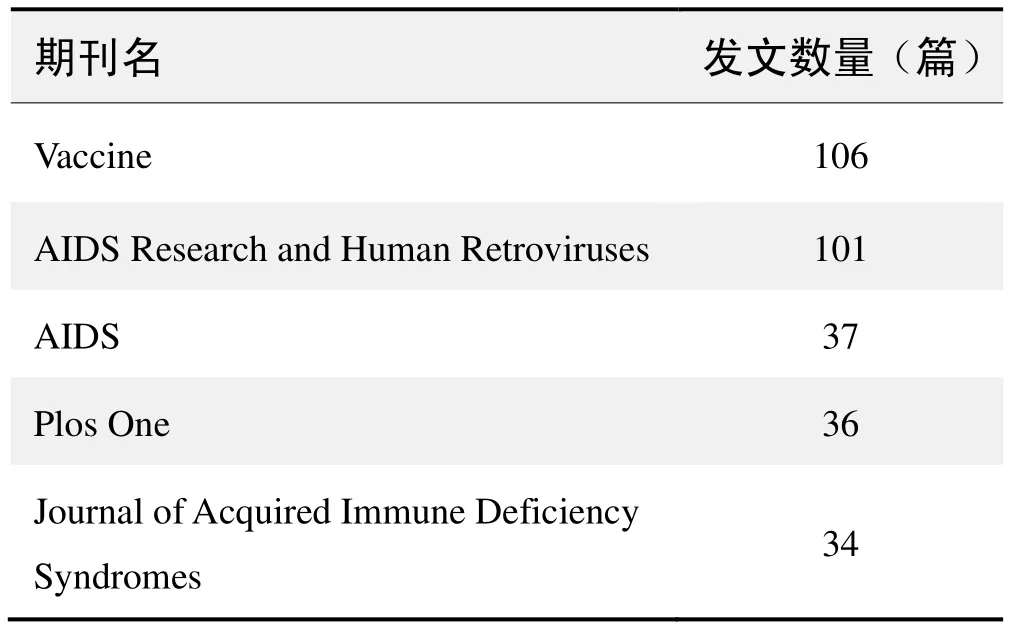
期刊发文数量排名(WOS)
(数据来源:中国知网、Web of Science,检索时间:2015-12-01)
根据中国知网(CNKI)数据报告,以“艾滋病疫苗”为词条可以检索到的高被引论文排行结果如下。

国内数据库高被引论文排行

(续表)
(数据来源:中国知网,检索时间:2015-12-01)
根据Web of Science统计数据,以“艾滋病疫苗”为词条可以检索到的高被引论文排行结果如下。

国外数据库高被引论文排行
(数据来源:Web of Science,检索时间:2015-12-01)
·经典文献推荐·
基于Web of Science检索结果,利用Histcite软件选取LCS(Local Citation Score,本地引用次数)TOP 30文献作为节点进行分析,得到本领域推荐的经典文献如下。
Development and testing of AIDS vaccines
Cleaver, JE
Abstract:Recent advances in delineating the molecular biology of human immunodeficiency virus type 1 (HIV-1) have led to innovative approaches to development of a vaccine for acquired immunodeficiency syndrome (AIDS). However, the lack of understanding of mechanisms of protective immunity against HIV-1, the magnitude of genetic variation of the virus, and the lack of effective animal models for HIV-1 infection and AIDS have impeded progress. The testing of AIDS vaccines also presents challenges. These include liability concerns over vaccine-related injuries; identification of suitable populations for phase 3 efficacy studies; balancing the ethical obligation to counsel research subjects to avoid high-risk behavior with the necessity to obtain vaccine efficacy data; and the effect of vaccine-induced seroconversion on the recruiting and welfare of trial volunteers. Several candidate AIDS vaccines are nevertheless currently under development, and some are undergoing phase 1 clinical trials. Rapid progress will depend on continued scientific advancement in conjunction with maximum use of resources, open information and reagent exchange, and a spirit of international collaboration. Issue: Several investigators are preparing to conduct efficacy trials of human immunodeficiency virus (HIV) vaccines in the developing world. Failure to adequately address the unique ethical, behavioral, and social issues that surround vaccine testing in that setting will jeopardize the success of these trials and future acquired immunodeficiency syndrome (AIDS) research in the host nation. Description of the Project: Twelve investigators from Africa, Asia, North America, and South America reviewed previous experience with HIV trials in developing countries and explored potential solutions to these issues. Conclusions: Host country scientists, government officials, and media must be actively involved in all aspects of the trials. Minimum prerequisites for conducting the trial include the following: (1) researching vaccines active against developing world HIV isolates; (2) establishing and maintaining an adequate technological infrastructure; (3) assessing the feasibility of recruitment in countries where the existence of HIV may be denied; (4) designing methods to obtain informed consent from each individual subject, rather than exclusively from family members or community elders; (5) creating locally appropriate instruments to measure risk behavior; (6) identifying a behavioral intervention for placebo and treatment groups; (7) making available laboratory methods to distinguish between natural HIV infection and vaccine-induced seropositivity; and (8) guaranteeing that an effective vaccine is available free of charge to the placebo group and at affordable prices to other host country residents. Omitted. An anonymous cross-sectional paper-and-pencil survey was used to assess incentives and disincentives to participate in a Phase I preventive human immunodeficiency virus (HIV) vaccine trial in a potential Thai target population. A total of 255 persons employed in health care service and research settings completed questionnaires after attending informational briefings regarding the proposed vaccine product and the planned trial procedures, Willingness to participate was related to self-perceived benefits from joining a preventive vaccine trial, as well as to concerns about product safety and social discrimination that might result from participation. The distinction between positive results of enzyme-linked immunosorbent assay from vaccine administration and positivity from HIV infection was unclear for many participants. Men were more willing to participate than women, and there was a trend toward greater willingness to participate in those who were less educated, Preparations for preventive vaccine trials may be more successful if they emphasize personal benefits of trial participation, clearly address safety issues, and consider ways to prevent social discrimination against participants. Objective: To determine the willingness of populations at high risk of HIV-1 infection to participate in HIV vaccine efficacy trials, determine factors influencing decisionmaking, and evaluate knowledge levels of vaccine trial concepts. Design: Cross-sectional study. Methods: HIV-1-negative homosexual men, male and female injecting drug users and non-injecting women at heterosexual risk were recruited in eight cities in the United States (n= 4892). Results: A substantial proportion of the study population (77%) would definitely (27%) or probably (50%) be willing to participate in a randomized vaccine efficacy trial. Increased willingness was associated with high-risk behaviors, lower education level, being uninsured or covered by public insurance, and not having been in a previous vaccine preparedness study. Altruism and a desire for protection from the vaccine were major motivators for participation. Major concerns included positive HIV-1 antibody test due to vaccine, safety of the vaccine, and possible problems with insurance or foreign travel. Baseline knowledge of vaccine trial concepts was low. Conclusions: It is likely that high-risk volunteers will be willing to enroll in HIV vaccine efficacy trials. A variety of participant and community educational strategies are needed to address participant concerns, and to ensure understanding of key concepts prior to giving consent for participation.
来源出版物:Science, 1988, 241(4864): 426-432
Ethical, behavioral, and social aspects of HIV vaccine trials in developing countries
Lurie, P; Bishaw, M; Chesney, MA; et al.
来源出版物:JAMA, 1994, 271(4): 295-301
Participation of homosexual/bisexual men in preventive HIV vaccine trials: Baseline attitudes and concerns and predicted behaviors during trials
Douglas, JM; Judson, FN; Parks, JP; et al.
来源出版物:AIDS Research and Human Retroviruses, 1993, 10: S257-60
Incentives and disincentives to participate in prophylactic HIV vaccine research
Jenkins, RA; Temoshok, LR; Virochsiri, K
Keywords:HIV vaccine; vaccine trials; research participation; incentives vaccine trials; homosexual men; injecting drug users; women; HIV
来源出版物:AIDS, 1998, 12(7): 785-793
·推荐综述·
来源出版物:Journal of Acquired Immune Deficiency Syndromes, 1995, 9(1): 36-42
Readiness of high-risk populations in the HIV Network for Prevention Trials to participate in HIV vaccine efficacy trials in the United States
Koblin, BA; Heagerty, P; Sheon, A; et al.
文献编号本领域经典文章题目第一作者来源出版物1 Development and testing of AIDS vaccines Cleaver, JE Science, 1988, 241(4864): 426-432 2Ethical, behavioral, and social aspects of HIV vaccine trials in developing countries Lurie, P JAMA, 1994, 271(4): 295-301 3 Participation of homosexual/bisexual men in preventive HIV vaccine trials: Baseline attitudes and concerns and predicted behaviors during trials Douglas, JM AIDS Research and Human Retroviruses, 1993, 10: S257-S260 4 Incentives and disincentives to participate in prophylactic HIV vaccine research Jenkins, RA Journal of Acquired Immune Deficiency Syndromes, 1995, 9(1): 36-42 5 Readiness of high-risk populations in the HIV Network for Prevention Trials to participate in HIV vaccine efficacy trials in the United States Koblin, BA AIDS, 1998, 12(7): 785-793
