Evaluation of yield, quality and antioxidant activity of essential oil of in vitro propagated Kaempferia galanga Linn.
2014-03-22SupravaSahooReenaParidaSikhaSinghRabindraPadhySanghamitraNayak
Suprava Sahoo, Reena Parida, Sikha Singh, Rabindra N. Padhy, Sanghamitra Nayak
1Centre of Biotechnology, Siksha O Anusandhan University, Kalinga Nagar, Bhubaneswar, 751003, Odisha, India
2Central Research Laboratory, IMS and Sum Hospital, Siksha O Anusandhan University, Kalinga Nagar, Bhubaneswar, 751003, Odisha, India
Evaluation of yield, quality and antioxidant activity of essential oil of in vitro propagated Kaempferia galanga Linn.
Suprava Sahoo1, Reena Parida1, Sikha Singh1, Rabindra N. Padhy2*, Sanghamitra Nayak1
1Centre of Biotechnology, Siksha O Anusandhan University, Kalinga Nagar, Bhubaneswar, 751003, Odisha, India
2Central Research Laboratory, IMS and Sum Hospital, Siksha O Anusandhan University, Kalinga Nagar, Bhubaneswar, 751003, Odisha, India
Objective: To determine chemical constituents and antioxidant properties of essential oil from rhizome of the medicinal plant, Kaempferia galanga (K. galanga) Linn. (Zingiberaceae) in conventionally propagated (CP) and in vitro propagated (IVP) plants. Methods: In vitro (micro) propagation of K. galanga was done by inoculating explants on to Murashige and Skoog agar medium, supplemented with suitable combinations of phytohormones; the regenerants were transferred to soil for further growth. Essential oil preparations of both CP and IVP rhizomes grown in soil, obtained by the hydro-distillation method were analyzed by gas chromatographymass spectrometry. Antioxidant activities of essential oil samples were monitored. Results: Maximum numbers of regenerated shoots were found in the medium supplemented with 1 mg/L benzyl adenine and 0.5 mg/L indole-3-acetic acid. A total of 6 compounds were identified from rhizomes from CP and IVP plants that yielded 96.9% and 97.81% of the total oil contents, respectively. The major compound of rhizome oil identified from CP and IVP rhizomes was ethyl p-methoxy cinnamate in quantities, 82.01% and 71.77%, respectively, without any compositional variation. Antioxidant properties of essential oil preparations were assessed by the 2,2-diphenyl-1-picrylhydrazyl (DPPH) and hydrogen peroxide radical scavenging assays. Moreover, antioxidant activities of rhizome-oil from IVP plants were better than that of CP oil samples. Conclusions: As IVP rhizomes had better oil yield, those could be used for a large scale commercial propagation for sustainable use of essential oil. The principal chemical in the essential oil, ethyl p-methoxy cinnamate could help apothecary, for several ailments.
ARTICLE INFO
Article history:
Received 8 August 2013
Received in revised form 15 September 2013
Accepted 24 September 2013
Available online 20 June 2014
Kaempferia galangal
Essential oil
Ethyl p-methoxy cinnamate
Free radical scavenging assays
DPPH
H2O2
1. Introduction
Kaempferia galanga(K. galanga) Linn. (aromatic or sand ginger, family Zingiberaceae) is regarded as a cash plant with several medicinal properties, as many a pharmaceutical establishments require its rhizomes for the essential oil as well as for direct uses in the preparation of Ayurvedic drugs, perfumery, cosmetics, spices and a few more[1]. Further, bulky leaves of the plant are used for flavouring foodstuffs, preparing mouthwashes and a hair tonic, locally; leaves are antinociceptive and antiulcerative[2,3]. Antioxidant activity of the plant was too reported using the ferric reducing antioxidant power, the β -carotene bleaching and the oxygen radical capacity assay by a Malaysian variety of the plant[4]. Rhizomes are used for curing bronchitis, asthma, malaria, skin disease, wounds and spleenic disorders[5]. They are used for the preparation of decoction or powders, which are used for indigestion, cold, pectoral pain, abdominal pain, headache and toothache. Its alcoholic maceration has also been applied as liniment for rheumatism[6]. The essential oil from rhizome had been known having antimicrobial activities. Crude methanolic extract ofin vitropropagated (IVP) rhizomes was screened for antibacterial activity against four bacteria, of undefined antibiotic sensitivity[7]. Antibacterial activity of the rhizome against bacteria,Staphylococcus epidermidis (S. epidermidis)andBacillus subtilis (B. subtilis), isolated from skin were demonstrated that rhizome-extracts werecomparable to various foot skin deodorant formulations that are commercially available[8]. Antimalarial activity of the plant was demonstrated against the falciparum malarial parasite by rhizome-extracts[9].
The active constituents of galangal oil are ethyl p-methoxy cinnamate (EPMC), methylcinnamate and penta decane, etc., as reported[6,10]. EPMC from the rhizome extract was reported as highly cytotoxic to He La cells[6]. Crude rhizome extracts were used for the demonstration of antineoplastic activity against human colorectal adenocarcinoma, but extracts were non-toxic to normal Vero cell cultures, the study was based on cytotoxic activity that was monitored by the standard assays with 3-(4,5-dimethyl-2-thhiazolyl)-2,5-diphenyl-2H-tetrazolium bromide (MTT) and sulforhodamine B (SRB), against four cancerous cell lines[11]. EPMC was effective against the hepatic microsomal cytochrome P450s enzyme activities in mice that mostly induce liver disorders leading to cancer; this compound has antineoplastic activity[12]. This chemical was further reported as active against fibrosarcoma in mouse model induced by benzo(a)pyrene; the study demonstrated that the chemo preventive activity of the compound against fibrosarcoma was through the inhibition of COX-2, as known fromin silicoanalysis[13].
Anti-inflammatory effect of the plant by plant derived EPMC were dose dependent in inhibiting carrageenan induced edema with an minimum inhibitory concentration (MIC) value of 10 mg/kg[14]. Hepatoprotective activities of freshly powdered rhizomes were demonstrated in carbon tetrachloride induced liver damage in rats[15]. The bioactive compound, EPMC isolated from ethanol extract of rhizome had a decreasing effect on micropthalmia associated transcription factor and tyrosinase levels in MSH-strimulated B16F1O cells, helping in immunoregulation. It also could be used as a skin lightening agent to treat hyperpigmentary disorders in man[16].
Larvicidal properties of the chloroform extract of rhizome were reported against the notorious vector of the dengue virus,Aedes aegypti (Ae. aegypti)[17]. In integrative agricultural management, the use of rhizome extract against the nematode,Meloidogyne incognita (M. incognita)reduced utilizations of nematicides, carbofuran, fosthiazate and metam sodium[18].
The price of the essential oil of the plant varies from US $ 600 to 700 per kg on the international market, and rhizomes as well as leaves are highly exploited by the local people and pharmaceutical industries[19]. The high price of the natural essential oils coupled with their limited availability has encouraged a search for substitutes. Low productivity, disease susceptibility and higher cost of seed rhizome production are the major constraints faced by growers. Furthermore, the collection of plant material for the extraction of drug is the major task, as this plant species has become endangered[20]. These problems can be alleviated through the application of a tissue culture technique, an efficient-the long-recognized tool for rapid multiplication of true-to-type genotypes that could be made available, commercially[21]. Despite intensive studies on the chemical composition of rhizome ofK. galanga, only very few reports are available on the chemical constituents of the plant oil. The essential oil samples of IVP and conventionally propagated (CP) rhizomes were analyzed by gas chromatography-mass spectrometry for the quantitative estimation of chemicals. Further, there is no report on the antioxidant activity of essential oil of this endangered plant, a comparative evaluation of antioxidant activity of the essential oil samples of IVP and CP rhizomes were done. It is anticipated that apothecary would benefit from the principal chemical, EPMC, for further drug development, since the plant has holistic medicinal properties.
2. Materials and Methods
2.1. Plant material
K. galangawas grown in our garden. Young sprouting buds from rhizomes of 10 disease-free plants were used as explants for the initiation of thein vitroculture. Explants were cleaned with a liquid detergent and were treated with a seed-dresser, 0.1% mercuric chloride for 8 to 10 min, aseptically and those were cleaned with sterile distilled water to remove traces of mercuric chloride, prior to the culture initiation.
2.2. In vitro propagation and field transfer of regenerants
Explants were inoculated into autoclaved basal Murashige and Skoog (MS) agar medium[22], with varying combinations of benzyl adenine (BA) (1-5 mg/L), indole-3-acetic acid (IAA) (0.5-1.0 mg/L), naphthalene acetic acid (NAA) (0.5 mg/L) and adenine sulphate (Ads) (50-100 mg/L), for IVP (Table 1), with 30 g/L of sucrose; the pH of the medium was adjusted to 5.7, and 0.8% agar was used. Fifteen replicate explants were used for each treatment. Culture tubes containing the inoculated explants were kept under a bank of white fluorescent lights at temperature 24 ℃, in a culture room. IVP plants with well-developed shoot and roots were transferred to field for further growth and oil extraction was done from rhizomes.
2.3. Extraction and quantitative evaluation of essential oil
Essential oil was extracted by hydro-distillation of fresh rhizomes of CP and IVP plants with a Clevenger’s apparatus. The total amount of oil in rhizomes was calculated by the following method. Fresh weight of rhizome oil yield (%) (v/ w) = (volume of essential oils (mL)/ weight of raw rhizomes taken) ×100
The distilled rhizome essential oil was dried over anhydrous sodium sulphate and stored at -4 ℃ in air tight containers for further uses. Each essential oil extraction was run in triplicates for confirmation of oil yield.
2.4. Qualitative analysis of essential oil
The component identification of essential oil was achieved by the gas chromatography-mass spectrometry (GCMS) analysis, using the 6890 series instrument (Agilent Technologies, Palo Alto, California), equipped with flame ion detector (FID) and a HP-5, fused silica capillary column (30 m x 0.25 mm) with internal diameter, film thickness, as 0.25 μm; the temperature was programmed from 50-240 ℃ at 4 ℃/min; from 240 ℃ to 270 ℃ at 15℃/min; held isothermal at 50 ℃ for 1 min and at 270 ℃ for 15 min. The temperatures of both auto-injector and detector were kept at 280℃; the sample injection volume was 1 μL and the split ratio was 100:1. The carrier gas was nitrogen at the flow rate of 1.2 mL/min. GC-MS (70eV) data were measured on the same gas chromatograph, coupled with MSD 5973. The MS source temperature was at 230 ℃, but the MS quadrapole temperature was at 150 ℃; the interface temperature was at 290 ℃; the mass scan was done at 20-600 amu; the carrier gas was helium at the flow rate, 1.0 mL/min. The retention index was calculated using a homologous series ofn-alkanes C8-C18. Chemical constituents were identified by comparison of their mass spectra and retention indices with those of National Institute of Standards and Technology library data[23]. For the GC-MS evaluation, 10 plants from each group, IVP and CP were randomly selected.
2.5. DPPH free radical scavenging activity
Essential oil samples were individually assessed for their possible antioxidative activities by employing two complementary tests, DPPH free radical-scavenging and H2O2scavenging assays. Radical scavenging activity of essential oil samples of both IVP and CP rhizomes were determined by a spectrophotometry, based on the reduction of a methanolic solution of DPPH[24]. Aliquots of 1 mL of varying concentrations of oils (1, 5, 10, 20, 50 and 100 μg/mL) in methanol were added to an aliquot 2 mL of 0.1 mM methanolic solution of DPPH, for each concentration. The mixture was shaken vigorously and left to stand at the room temperature for 30 min in dark. When DPPH was reduced, the change of colour from deep violet to light yellow occurs, which was measured at 517 nm on a UV/ visible spectrophotometer (UV1, Thermo scientific). Absolute methanol was used as the base control. The DPPH solution was freshly prepared and stored in the dark. Tests were carried out in triplicates with ascorbic acid, as the standard antioxidant. Radical scavenging activity was expressed as 50% inhibition concentration (IC50) of DPPH radical and was calculated by following equation,
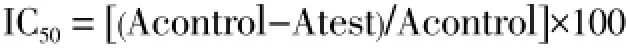
where, Acontrol was the absorbance of the control solution without the essential oil and Atest was the absorbance of oil and ascorbic acid. The antioxidant activity of eachsample was expressed in terms of IC50(i.e., micromolar concentration required to inhibit DPPH radical formation by 50%), calculated from the graph after plotting inhibition percentage against sample concentration.

Table 1 Effect of growth regulators on shoots and root in vitro multiplication of K. galanga with phytohormones.
2.6. Hydrogen peroxide scavenging activity
Hydrogen peroxide scavenging activity of both CP and IVP oil samples were estimated, as detailed[24]. A stock of 4 mM solution of H2O2was prepared in phosphate buffer saline (PBS, pH 7.4) solution. Oil samples in aliquots of 4 mL, at varying concentrations (1-100 μg/mL) were mixed with an aliquot of 0.6 mL of 4 mM H2O2solution prepared in PBS, and the mixture was incubated for 10 min, for each sample. The absorbance of the solution was noted at 230 nm against a blank solution containing the essential oil in PBS without H2O2. Ascorbic acid was too used as the positive control. The amount of H2O2radical inhibited by the oils was calculated using the following equation:
H2O2radical scavenging activity% = [(Acontrol-Atest)/ Acontrol ] x 100,
where, Acontrol was the absorbance of H2O2radical + methanol; Atest was the absorbance of H2O2radical+ sample or standard.
3. Results
3.1. In vitro propagation
Rhizome buds of CP plants were used as explants (Figure 1a) and inoculated to MS agar medium with different phytohormone combinations of BA (1-5 mg/L), IAA (0.5-1.0 mg/L), NAA (0.5 mg/L) and Ads (50-100 mg/L). The multiplication of shoot buds occurred in all hormone combinations in media, but the maximum multiplications of shoots (Figure 1b) were noted in MS supplemented with BA (1 mg/L) in combination with IAA (0.5 mg/L) (Table 1). After 90 days of culturing, fully grown IVP plantlets were transferred fromin vitrocondition (Figure 1c), as regenerants to pots containing soil, farmyard manure and sand at the ratio 1:1:1, respectively (Figure 1d). Subsequently, those were kept in a greenhouse for 30 more days for further growth. In field conditions, about 94% of plants survived successfully with normal growth (Figure 1e; Figure 1f). Rhizomes developed after 3-4 months of transferring of plantlets into pots and were subsequently harvested.

Figure 1. Different stages of in vitro propagation of K. galanga; 1a: Rhizome bud explant, 1b: Regenerated plants with shoots and roots; 1c: Multiplication of plantlets in vitro conditions; 1d: Potted plantlets in hardening stage; 1e: In vitro propagated plants growing under field condition; 1f: Flowering stage of in vitro propagated K. galanga.
3.2. Chemical composition of essential oil
The steam distillation of rhizomes yielded yellowish essential oils, which possessed the characteristic spicycampherous odour. CP rhizomes yielded 0.6% and IVP rhizomes yielded 0.9% essential oil. A detailed chemical composition of the essential oil was assessed by GC-MS analysis. From chromatograms of essential oil samples of CP and IVP rhizomes, it was discernible that 6 major identified components, accounting for 97.81% and 96.9% of the total peak area in CP (Figure 2) and IVP (Figure 3)K. galanga, respectively were recorded. The result demonstrated the presence of EPMC with the maximum peak area (82.01%)from CP rhizomes and (71.77%) from IVP rhizomes, followed by 5 chemicals, ethyl cinnamate, 3-carene, eucalyptol, borneol, pentadecane (Table 2).
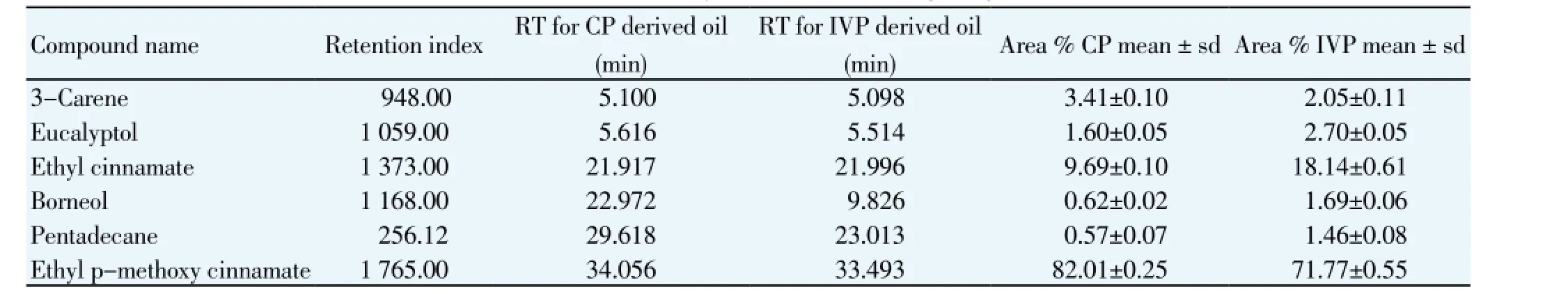
Table 2 Chemical constituents of essential oil of rhizomes from GC-MS analysis of CP and IVP K. galanga.

Figure 2. GC-MS spectrum of rhizome oil of conventionally propagated K. galanga.
3.3. Antioxidant activity
It was evident that increased antioxidative activity in DPPH free radical-scavenging assay was seen with increasing concentrations of oil samples (Figure 4), as a lower IC50value indicated higher antioxidant activity. The DPPH scavenging activity of essential oil samples indicated a concentration dependent antioxidant activity against H2O2, with IC50values, 6.6, 26, and 19.5 μg/mL for ascorbic acid, oil samples of CP and IVP rhizomes, respectively. Similarly, the H2O2scavenging activity of essential oil samples indicated a concentration dependent antioxidant activity against H2O2, with IC50values, 29, 24.5 and 21.5 μg/mL, for oil samples of CP and IVP rhizomes and ascorbic acid, respectively (Figure 5).

Figure 3. GC-MS spectrum of rhizome oil of in vitro propagated K. galanga.

Figure 4. Radical scavenging activity of essential oil of both conventionally propagated (CP) and in vitro propagated (IVP) K. galanga rhizomes using DPPH; numbers in abscissa, 1 to 6 were 1, 5, 10, 20, 50 and 100 μg/mL oil concentrations.

Figure 5. Hydrogen peroxide scavenging activity of essential oil of both conventionally propagated (CP) and in vitro propagated (IVP) K. galanga; numbers in abscissa, 1 to 6 were, 1, 5, 10, 20, 50 and 100 μg/mL oil concentrations.
4. Discussion
The maximum number of shoots was multiplied in MS agar supplemented with BA 1 mg/L in combination with IAA 0.5 mg/L. Additionally, IVP plantlets growing well in field conditions had a higher yield of essential oil, as well as peak areas in GC-MS analysis. Indeed, plant based secondary metabolites such as, essential oils are widely used in the pharmaceutical industries and are considered ‘generally recognised as safe[25]. In addition to EPMC, the plant has triterpenoids, flavonoids and resins in chloroform extract and steroids, triterpenoids, alkaloids, flavonoids, carbohydrates, resins and proteins in the methanolic extract[26]. Two novel sulfonated diarylhepatonoid epimers, kaempsulfonic acid A(1) and B(2) were isolated from this plant[27]. But, no information is available in the literature on the comparative account of the chemical composition of essential oil of CP and IVPK. galangarhizomes. In an earlier study, the compound ethyl p-methoxy cinnamate had 59.5% of thetotal compounds detected in essential oil, followed by ethyl cinnamate, 3-carene, pentadecane, borneol, bornyl acetate, delta-selinene, camphor, alpha-piene and immidazole-5-carbonylvinyl-4-nitro, using another cultivar of the plant,K. galangafrom south Odisha[6]. In this study, the values were 71.77% and 82.01% from CP and IVPK. galangarhizomes, respectively with the local cultivar of central Odisha that needs molecular typing. EPMC had also been reported, as the major constituent of rhizome oil ofK. galangaof different origins[10,28]. Our report is in close agreement with inCurcuma longa[29], another well-studied plant from Zingiberaceae, where the IVP clones were more uniform for their volatile constituents.
In this study, both IVP and CP sources of oil had remarkable antioxidant activities. In general, the essential oil obtained from IVP rhizomes showed a greater activity than that from CP rhizomes. The DPPH free radical scavenging activity of oil samples and that due to hydrogen peroxide of essential oil samples from IVP rhizomes, herein, were more than the activity level due to CP rhizomes; invariably reducing values due to ascorbic acid were the highest in each experiment. Antioxidants are compounds that neutralize chemically active products of metabolism such as, free reactive oxygen radicals, which can damage the body. Reactive oxygen species (ROS) include several metabolic by-products, hydrogen peroxide, hydroxyl radicals, nitric oxide, superoxide anions, peroxy nitrite and a few more that are potential enough to trigger degenerative processes in human body[30]. Compounds such as, phenolic acids, polyphenols and flavonoids are recognized as antioxidants, as they have the ability to scavenge free radical generating ROS. It has been documented that plant phenols neutralize free oxygen, eventually play a major role in the prevention of cancer, cardiovascular, neurogenerative diseases and a few more, with their potentiality to act as antioxidants[31]. Essential oils, despite their wide uses and fragrances, constitute an effective alternate to synthetic compounds produced by chemical industry without having any side effects[32]. Antioxidant activities of essential oils from aromatic plants are mainly attributed to inherent active compounds.
This study has shown that the yield of oil extracted from IVP plants is significantly higher than CP plants. Essential oils of both CP and IVPK. galangadisplayed strong antioxidant properties, for which this plant is used as preventive agents from various diseases in diverse cultures. However the chemical profile and relative amount of compounds were very similar in both the oil samples. The result indicated that IVPK. galanga, with enhanced oil yield and uniform constituents, could be efficiently used for large scale commercial production of drug, thereby reducing the overexploitation of natural resources.
Conflicts of interests
The authors declare that they have no conflicts of interests.
Acknowledgments
The authors are grateful to Prof (Dr) S. C. Si, Dean Centre of Biotechnology and Prof (Dr) M. R. Nayak, President, Siksha `O` Anusandhan University, for providing facilities and encouragements. S. Sahoo is an INSPIRE Fellow (IF 10244), from Department of Science & Technology, Govt. of India , New Delhi, which provided extended facilities.
[1] Rahman MM, Amin MN, Ahamed T, Ali MR, Habib A. Efficient plant regeneration through somatic embryogenesis from leaf base derived callus of Kaempferia galanga L. Asian J Plant Sci 2004; 3: 675-678.
[2] Chen C, Lim YY, Wong SK, Lim KK, Tan SP, Lianto FS, et al. Effects of drying methods on the antioxidant properties of leaves and tea of ginger species. Food Chem 2009; 113: 166-172.
[3] Sulaiman MR, Zakaria ZA, Duad IA, Hidayat MT. Antinociceptive and anti-inflammatory activities of the aqueous extract of Kaempferia galanga leaves in animal models. J Nat Med 2008; 62: 221-227.
[4] Aziman N, Abdullah N, Mohd Noor Z, Zulkifli KS, Wan Kamarudin WSS. Phytochemical constituents and in vitro bioactivity of ethanolic aromatic herb extracts. Sains Malaysiana 2012: 41: 1437-1444.
[5] Kochuthressia KP, Britto SJ, Jaseentha MO, Rini R. Antimicrobial activities of Kaempferia galanga L. leaf extract- an in vitro approach. Internet J Biol Pharm Allied Sci 2012; 1: 1734-1740.
[6] Mohanty S, Parida R, Singh S, Joshi RK, Subudhi E, Nayak S. Biochemical and molecular profiling of micropropagated and conventionally grown Kaempferia galanga. Plant Cell Tissue Organ Cult 2010; 106: 39-46.
[7] Hanumantharaju N, Shashidhara S, Rajasekharan PE, Rajendra CE. Comparative evaluation of antimicrobial and antioxidantactivities of Kaempferia galanga for natural and micro propagated plant. Internet J Pharmacy Pharma Sci 2010; 2: 72-75.
[8] Lertsatitthanakom P, Satayavongthip B. Antibacterial activity of an effective spice essential oil formulated in foot deodorant gel against Bacillus subtilis. J Biol Sci 2012; 12: 315-320.
[9] Thiengsusuk A, Chaijaroenkul W, Na-Bangchang K. Antimalarial activities of medicinal plants and herbal formulations used in Thai traditional medicine. Parasitol Res 2013; 112: 1475-1481.
[10] Tewtrakul S, Yuenyongsawad S, Kummee S, Atsawajaruwan L. Chemical components and biological activities of volatile oil of Kaempferia galanga Linn. Songklanakarin J Sci Technol 2005; 27: 503-507.
[11] Jagadish PC, Raghu Chandrashekhar H, Vinod Kumar S, Latha KP. Potent selective cytotoxic activity of Kaempferia galanga L. rhizome against cancer cell cultures. Internet J Pharma Biosci 2010; 1: 105.
[12] Sisangtragul W, Sripanidkulchai B. Effects of Kaempferia galanga L. and ethyl-p methoxycinnamate (EPMC) on hepatic microsomal cytochrome P450s enzyme activities in mice. Songklanakarin J Sci Technol 2011; 33: 411-417.
[13] Guo AJ, Choi RC, Zheng KY, Chen VP, Dong TT, Wang ZT, Vollmer G, et al. Kaempferol as a flavonoid induces osteoblastic differentiation via estrogen receptor signaling. Chinese Med 2012; 7: 10.
[14] Umar MI, Asmawi MZB, Sadikun A, Altaf R, Iqbal MA. Phytochemistry and medicinal properties of Kaempferia galanga L. (Zingiberaceae) extracts. Afr J Pharm Pharmacol 2011; 5: 1638-1647.
[15] Manigaunha A, Ganesh N, Kharya MD. Hepatoprotection by kaempferia galanga against carbon tetrachloride induced liver damage in rats. Indian Drugs 2010; 47: 55-60.
[16] Ko HJ, Kim HJ, Kim SY, Yun HY, Baek KJ, Kwon NS, Wan KW, et al. Hypopigmentary effects of ethyl p-methoxycinnamateisolated from Kaempferia galanga. Phytother Res 2013; doi: 10.1002/ ptr.4995.
[17] Satoto TBT, Maniam S, Ganesen K, Ernaningsih. Larvicidal effect of ether and chloroform extract of Kaempferia galanga against the larvae of Aedes aegypti (Diptera: Culicidae). Internat J Pharmacog Phytochem Res 2013; 5: 96-100.
[18] Hong TK, Kim SI, Heo JW, Lee JK, Choi DR. Toxicity of Kaempferia galanga rhizomen constituents to Meloidogyne incognita juveniles and eggs. Nematol 2011; 13: 235-244
[19] Chithra M, Martin KP, Sunandakumari C, Madhusoodanan PV. Protocol for rapid propagation and to overcome delayed rhizome formation in field established in vitro derived plantlets of Kaempferia galanga L. Sci Hort 2005;104: 113-120.
[20] Shirin F, Kumar S, Mishra Y. In vitro plantlet production system for Kaempferia galanga, a rare Indian medicinal herb. Plant Cell Tissue Organ Cult 2000; 63: 193-197.
[21] Mohanty S, Panda MK, Subudhi E, Acharya L, Nayak S. Genetic stability of micro propagated ginger derived from axillary bud through cytophotometric and RAPD analysis. Z Naturforsch 2008; 63: 747-754.
[22] Murashige T, Skoog F. A revised medium for rapid growth and bioassay with tobacco tissue cultures. Physiol Plant 1962; 15: 473-497.
[23] Stein SE. National Institute of Standards and Technology (NIST) Mass spectral database and software, version 3.02, USA (June 1990).
[24] Sahoo S, Ghosh G, Nayak S. Evaluation of in vitro antioxidant activity of leaf extract of Alpinia malaccensis. J Med Plant Res 2012; 6: 4032-4038.
[25] Sundarammal S, Thirugnanasampandan R, Tamil Selvi M. Chemical composition analysis and antioxidant activity evaluation of essential oil from Orthosiphon thymiflorus (Roth) Sleesen. Asian Pacif J Trop Biomed 2012; 2: S112-S115.
[26] Rajendra CE, Magadum GS, Nadaf MA, Yashoda SV, Manjula M. Phytochemical screening of the rhizome of Kaempferia galanga. Int J Pharmacog Phytochem Res 2011; 3: 61-63
[27] Wang FL, Luo JG, Wang XB, Kong LY. A pair of sulfonated diarylheptanoid epimers from Kaempferia galanga. Chinese J Natur Med 2013; 11: 171-176.
[28] Sukari MA, Mohd Sharif NW, Yap ALC, Tang SW, Neoh BK, Rahmani M, et al. Chemical constituent variations of essential oils of rhizomes of four Zingiberaceous species. Malays J Anal Sci 2008; 12: 638-644.
[29] Singh S, Kuanar A, Mohanty S, Subudhi S, Nayak S. Evaluation of phytomedicinal yield potential and molecular profiling of micropropagated and conventionally grown turmeric (Curcuma longa L.). Plant Cell Tissue Organ Cult 2011; 104: 263 -269.
[30] Wang SY, Kuo YH, Chang HN, Kang PL, Tsay HS, Lin KF. Profiling and characterization antioxidant activities in Anoectochilus formosanus Hayata. J Agric Food Chem 2002; 50: 1859-1965.
[31] Okoh OO, Sadimenko AP, Afolayan AJ. Antioxidant activities of Rosmarinus officinalis L. essential oil obtained by hydrodistillation and solvent free microwave extraction. African J Biotechnol 2011; 10: 4207-4211.
[32] Faixova Z, Faix SS. Biological effects of rosemary (Rosmarinus officinalis L. essential oil (A Review). Univ Vet Med Komenskho 2008; 73: 41-81.
ment heading
10.1016/S2221-6189(14)60028-7
*Corresponding author: Prof. Dr. R. N. Padhy, CSIR Scientist, Central research Laboratory, IMS and Sum Hospital, Siksha O Anusandhan University, Kalinga Nagar, Bhubaneswar, 751003, Odisha, India
Tel: +91 674 2432034
E-mail: rnpadhy54@yahoo.com (RN Padhy)
杂志排行
Journal of Acute Disease的其它文章
- Submersion and acute respiratory failure
- A review on some poisonous plants and their medicinal values
- Laboratory diagnosis, clinical manifestations, epidemiological situation and public health importance of cutaneous leishmaniasis in Shushtar County, Southwestern Iran
- Antihyperglycemic and antihyperlipidemic properties of aqueous root extract of Icacina senegalensis in alloxan induced diabetic rats
- Neuroprotective and antioxidant role of Phoenix dactylifera in permanent bilateral common carotid occlusion in rats
- The ameliorative effects of Averroha carambola on humoral response to sheep erythrocytes in non-treated and cyclophosphamideimmunocompromised mice
