THE EFFECT OF HIGH AMMONIA CONCENTRATION ON GILL STRUCTURE ALTERNATION AND EXPRESSION OF SOD AND HSP90 GENES IN GRASS CARP, CTENOPHARYNGODON IDELLA
2013-04-19ZHOUXinDONGYunweiWANGFangandDONGShuanglin
ZHOU Xin, DONG Yun-wei, WANG Fangand DONG Shuang-lin
(1.The Key Laboratory of Mariculture, Ministry of Education, Fisheries College, Ocean University of China, Qingdao 266003, China; 2.State Key Laboratory of Marine Environmental Science, College of Oceanography and Environmental Science, Xiamen University, Xiamen 361005, China)
THE EFFECT OF HIGH AMMONIA CONCENTRATION ON GILL STRUCTURE ALTERNATION AND EXPRESSION OF SOD AND HSP90 GENES IN GRASS CARP, CTENOPHARYNGODON IDELLA
ZHOU Xin1, DONG Yun-wei2, WANG Fang1and DONG Shuang-lin1
(1.The Key Laboratory of Mariculture, Ministry of Education, Fisheries College, Ocean University of China, Qingdao 266003, China; 2.State Key Laboratory of Marine Environmental Science, College of Oceanography and Environmental Science, Xiamen University, Xiamen 361005, China)
Histological and molecular responses of grass carpCtenopharyngodon idellato high concentration ammonia exposure were studied in this study by determining the lethal concentration, gill cellular structure, and expression patterns ofsodandhsp90 genes.Grass carps were treated at five ammonia concentrations (50, 72, 104, 151 and 220 mg/L) for 24h, and then the gill tissues were collected for histological analysis.In addition, three tissues (gill, intestine and liver) were collected to measure the expression ofsodandhsp90 genes.After exposure to high concentration of ammonia, the arrangement and structures of gill cells changed dramatically.Edema and fusion of secondary lamellae were observed, and the edge of cell and nucleus could not be clearly defined.Meanwhile, the two genes (sodandhsp90) in different tissues were unregulated significantly, indicating that high concentration of ammonia could impair the cellular structures and induce the expression of stress proteins.These results also suggested thatsodandhsp90 were suitable biomarkers for ammonia exposure assessment in grass carp.
Grass carp; Ammonia exposure; 24hLC50; Histological damage;sod; hsp90
Ammonia is a highly toxic waste product produced by plants and animals[1].Decomposition mediated by microorganisms also generates this compound.Ammoniais known to be one of the common causes for death in aquatic animals[2—4]since high concentrations of ammonia frequently occur in aquatic environments[5].It was reported that high ammonia and low dissolved oxygen concentrations during summer and spring seasons were the major factors responsible for mortality in sewage-fed fishponds[6].
In fish, ammonia is produced in white muscles when adenylates are broken down to inosine monophosphate[7]and ammonium (NH4+) during anaerobic energy production associated with burst exercise[1,8].The toxicity of ammonia is affected by several factors including temperature and pH[9], and ammonia toxicity increases with ammonia concentration, water pH, and temperature[10—12].Exposure to high concentrations of ammonia reduces survival, inhibits growth, and causes a variety of physiological malfunctions.On the organismal level, ammonia causes hyperventilation[13,14], hyper-excitability, coma, and convulsion.On the cellular level, ammonia can interfere with energy metabolism through impairing the tricarboxylic acid cycle in fish[15].In extreme conditions, high concentration of ammonia can finally cause largescale death.
When an organism is subjected to chemical, physical or biological stress, sudden shortage of oxygen causes abnormal oxidative reactions in the aerobic metabolic pathways, resulting in the formation of excessive amounts of singlet oxygen and other reactive oxygen species (ROS).Superoxide dismutase (SOD) catalyzes a rapid two-step dismutation of the toxic superoxide anion by producing molecular oxygen and H2O2[16,17], and this reaction provides protection against the damaging effects of oxidant stress in living organisms[18].It has been proved that SOD is associated with bothacute and chronic pathology.There are many biological stimulators that can regulate the expression of the sod gene, such as heat shock[19,20], shear stress[21,22], and heavy metals[23].Previous studies have shown that contaminants could induce the gene expression of Phase 2 metabolic enzyme[24]and reactive oxygen species (ROS) as the byproduct could lead to a common pathway toxicity[25,26].Meanwhile, sod gene expression is also tissue-specific, and the expression pattern and time course may differ indifferent tissues[27].
As molecular chaperones, Heat Shock Proteins assist cells in their recovery from stress and promote cytoprotection[28].Heat shock proteins consist of several families of highly conserved proteins that play an essential role in a number of cellular processes.As an important member of heat shock protein family, HSP90 responds to a wide variety of physiological and environmental stressors, including thermal shock, heavy metals, free radicals, or almost any sudden changes in the cellular environments that induce protein damage[29—31].Because of their important roles in protecting organisms from damages, HSPs are regarded as sensitive biomarkers for monitoring the health of aquatic environments[32]
Grass carp Ctenopharyngodon idella (Valenciennes 1844) is one of the most widely cultivated freshwater fish species in China.Both acute and chronic toxicities of ammonia have been extensively reviewed for freshwater species[33—36].However, few studies examined the effect of ammonia exposure on the mRNA expressions of stress proteins in the grass carp.In this study, expression of hsp90 and sod genes at mRNA levels were determined using semi-quantitative real time PCR (sqRT-PCR) with specific primers and histological changes also measured to determine the damages to the cellular microscopic structure after ammonia exposure.The integration of hsp and sod as an anti-stress system, along with the help of histological data, should be useful to study the effect of ammonia on the fish and the physiological adaptations of grass carp to ammonia exposure.
1 Materials and methods
1.1 Experiment animals and acclimation conditions
Grass carps with body length of (9.2±0.8) cm (Mean± SD) were obtained from Jiaonan Fish Farm, Qingdao, P.R.China.During the transportation, aeration to the fish containers was provided continuously.After delivery, the fish were maintained in rectangle indoor tanks filled with fully aerated running water and fed with compound feed once a day.Water temperature was controlled at (20± 0.2)℃, and one third of water was replaced daily.Light was controlled on a 12h∶12h light/dark cycle.
1.2 Test for the upper limit of ammonia tolerance
Seventy-five grass carps were divided into five groups (15 individuals per group).For each trial, one group of grass carps were placed into an aquarium (45 cm×24 cm×34 cm, L×W×H) and exposed to one of the designated ammonia concentrations (150, 186, 229, 282, 350 mg/L) for 24h.After the 24h exposure, behavior and mortality were recorded over time.During the whole experimental period, the rearing conditions were similar to those used during the acclimation and aeration was provided continuously.There were three replicates in each treatment.
1.3 Expressions of stress proteins under the ammonia exposure
Ammonia exposure and tissue collectionAfter a two-week acclimation, different treatment groups of fish were transferred into aquariums (45 cm×24 cm×34 cm, L×W×H).There were 15 fish in each aquarium and the water was exchanged daily as described above.Five ammonia concentrations were controlled (50, 72, 104, 151 and 220 mg/L) using ammonium chloride (Promega, USA).After a 24h exposure, six specimens were randomly chosen from each aquarium and anesthetized with ethyl alcohol (ETOH) before dissection.Liver, intestine, and gill were collected and frozen in liquid nitrogen for total RNA isolation.At different time points (0, 2, 4, and 24h) after the ammonia exposure, water ammonia concentrations were determined using a colorimetric method[37].
Quantification of genes expressionTotal RNA was isolated from approximately 60 mg of different tissues using the Trizol reagent (Invitrogen, USA).1 μg of total RNA was used as the template for synthesis of the first strand of cDNA.Partial β-actin gene (480 bp) was selected as an internal control.Four pairs of primers, SOD-F and SOD-R, Hsp90-F and Hsp90-R, and β-actin-F and β-actin-R were designed based on their respective sequences from Genbank (Tab.1).Semi-quantitative PCR conditions and components for hsp90, sod and β-actin were optimized, especially for the amplification cycles and annealing temperatures.PCR was carried out in 25 μL reaction solution that contained 2.5 μL of 10×PCR buffer, 1.6 μL of MgCl2(25 mmol/L), 2.0 μL of dNTP (2.5 mmol/L), 1 μL of primer (10 pmol/mL), 15.875 μL of PCR-grade water, 0.125 μL (5 U/μL) of Taq polymerase (Promega, USA), and 1 μL of cDNA reaction mix.The PCR procedure was performed at 95℃for 5min, followed by 32 cycles for and hsp90, 28 cycles for sod, and 26 cycles for β-actin at 95℃ for 30s, 55℃for 30s, 72℃ for 1min, and the final extension step was done at 72℃ for 10min.The PCR products were separated usinga 1.2% agarose gel stained with ethidium bromide (EB).To confirm the specificity of RT-PCR amplification, the products were purified usingthe agarosegel and sequenced.Electrophoretic images and the optical densities of amplified bands were analyzed using the software Gene Tools (Syn gene, USA).The abundance of mRNA was normalized to corresponding β-actin abundance in all samples and expressed as the optical density ratiosof hsp or sod and β-actin (Chsp/Cβ-actinor Csod/Cβ-actin).

Tab.1 Primer sets designed for semi-quantitative RT-PCR analysis of sod, hsp70, and hsp90 mRNA in grass carp, Ctenopharyngodon idella
1.4 Histological analysis
Gill tissues of five individual fish from each treatment including the control (fish without high concentration ammonia exposure) were collected to determine the ammonia exposure effect on the gill cellular microscopic structure.The tissue samples were fixed in the Bouin solution (70% saturated water solution of picric acid, 25% formalin, 5% glacialacetic acid) for 24h, and then transferred to 70% ethanol for storage.For microscopic examination, the samples were dehydrated by serial dilutions using alcohol and embedded in paraffin.Thick sections (6-8 μm) were cut on a microtome and stained with haematoxylin and eosin.The histological slides were examined using a microscope at 40× magnification.The images of each slide were recorded with an Olympus BX-51 camera and subsequently analyzed using the software Image J.
1.5 Statistics
The data were analyzed using the statistical package SPSS for Windows(Version 16.0; SPSS, Chicago, IL, USA).Briefly, the data were tested for homogeneity of variances using Mauchly’s Test of Sphericity.Lethal Concentration 50 (LC50) was calculated using Probit analysis.The inter-treatment differences of expression of the hsps and sod genes were analyzed by one-way ANOVA, and two-way ANOVA was applied to discern significant differences in expression among different tissues at different ammonia concentrations.The differences were considered significant if P< 0.05.
2 Results
2.1 Lethal ammonia concentration for the grass carp
There was no mortalityat the ammonia concentration of 100 mg/L.Mortality occurred at the concentration of 150 mg/L, and increased with the increase of ammonia concentration.The mortality rates of ammonia treated grass carp were 4.4%, 26.7%, 40%, 64.4%, and 88.9% at the ammonia concentration of 150, 186, 229, 282 and 350 mg/L, respectively.Probit analysis showed that the ammonia concentration had clear relationship to fish mortality (Probit value =7.6111 lg LC-13.158, R2= 0.9757, P<0.05) and the LC50of ammonia for 24h was 243 mg/L (Fig.1).
2.2 Histological damage of ammonia exposure
Without exposure to high concentration of ammonia, gill cells arranged compactly and the edge or nuclei were clearly defined.Thus the stained cells and nuclei were dark and clear (Fig.2A).After a 24h exposure at the ammonia concentration of 104 mg/L, however, the stained cytoplasm was pale pink while the nuclei remained dark, and blank in cell could also be observed (Fig.2B).After a 24h exposure at the ammonia concentration of 220 mg/L, all the stained cells were pale in color and the edge of the cells and nuclei could not be clearly defined.The edema and fusion of secondary lamellae could be observed (Fig.2C).Occurrence and severity of gill tissue damage were directly related to ammonia levels and these histological alternations were more obvious at concentration of 220 mg/L, which was near the 24h LC50(243 mg/L).
2.3 Gene expression at different ammonia concentrations
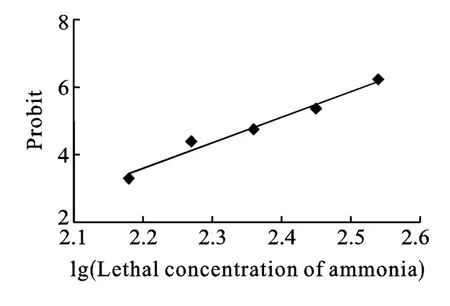
Fig.1 The relationship between the concentration of ammonia-N and the 24h mortality Ctenopharyngodon idella
Sod expressionAmmonia exposure can induce up-regulation of sod gene expression.A two-way ANOVA analysis showed that the up-regulation was affected by both tissue type (liver, intestine and gill; F2,10= 23.755; P<0.01) and ammonia concentration (F5,10= 11.159; P<0.01) (Fig.3).In the intestine and liver, the expression of sod gene increased with increasing ammonia concentration, while in liver the maximum sod gene expression occurred at the ammonia concentration of 151 mg/L.In the liver, level of sod at 151 mg/L ammonia was significantly higher than those at the 50, 72 and 220 mg/Lammonia treatments (F5,12=3.460; P<0.05).In the intestine, however, sod gene expression at 104, 151, 220 mg/L was significantly higher than the control, 50, and 72 mg/L ammonia treatments (F5,12=3.754; P<0.05).In the gill, sod level at 220 mg/L ammonia treatment was significantly higher than those of all the other treatments (F5,12=11.324; P<0.01).

Fig.2 Gills of grass carp from (A) control without exposure to high concentration of ammonia, (B) exposure to 104 mg/L ammonia, and (C) exposure to 220 mg/L ammonia

Fig.3 Effect of ammonia exposure on sod mRNA expression in grass carp Ctenopharyngodon idella
The control group was the treatment without ammonia exposure.The abundance of sod mRNA was normalized to the corresponding β-actin abundance and expressed as the optical densityratio of sod and β-actin (Csod/Cβ-actin).The values are Mean±SE(n = 3).Columns of the same tissue without common superscripts are significantly different (P< 0.05).
Hsp90 gene expressionThe responses of hsp90 gene expression to ammonia exposure were significantly different at different ammonia concentrations (F5,10= 29.085, P< 0.01).However, no significant difference in the up-regulation of hsp90 gene expression was found among the three tissue types (i.e., liver, intestine, and gill) (F2,10=2.704, P = 0.081) (Fig.4).In the liver, hsp90 gene expression reached the maximum value at the concentration of 104 mg/L ammonia and then decreased when ammonia concentration increased.At ammonia concentration of 104 mg/L, hsp90 level was significantly higher than both those under other three ammonia concentrations and the control (F5,12=21.699, P< 0.01).In the intestine, hsp90 gene expression reached the maximum value at 50 mg/L ammonia exposure and was significantly higher than all other treatments except the 72 mg/L ammonia treatment (F5,12=18.147, P<0.01).In the gill, hsp90 level increased with increasing ammonia concentration and reached the maximum value at the highest ammonia concentration (F5,12=17.241, P<0.01).

Fig.4 Effect of ammonia exposure on hsp90 mRNA expression
The abundance of hsp90 mRNA was normalized to the corresponding β-actin abundance and expressed as the optical density ratio of hsp90 and β-actin (Chsp90/ Cβ-actin).The values are Mean±SE(n=3).Columns of the same cluster without common superscripts are significantly different (P<0.05).
3 Discussion
Mortality rates across all the ammonia treatment levels went up in this study.Based on the Probit analysis, the LC50 for 24h was ~243 mg/L at water temperature 20℃ and pH 7.2, the concentration of un-ionized ammonia was ~1.25mg/L.In previous studies, the 24h LC50of un-ionized ammonia for the common carp fry was reported as 1.78 mg/L[38], and LC50 levels for beam, perch, roach, and rudd are 0.29, 0.35, 0.41, and 0.41 mg/L, respectively[39].Furthermore, tolerance limits of ammonia for different species are different, and common carp andgrass carp showed higher tolerance to ammonia than the other species (add references).However, the lethal concentration of ammonia during chronic exposure for the grass carp was lower than the acute exposure, and the allowable toxicant concentration of un-ionized ammonia for grass carp was far below the 96h LC50as described in previous studies.It has been reported that total ammonia increased directly with culture time, and can reach as high as 46 mg/L in intensive ponds[40].Therefore, ammonia is one of the major constrains for intensive culture of grass carp and the concentration of ammonia should be monitored regularly during the aquaculture period.
Gill is the most sensitive organ to external stimuli as it is directly exposed to the external environment[41].Gill damage as a result of exposure to toxicants has beenreported extensively[42—45], and gill damage has also been linked to impaired physiological functions[46].In the present study, epithelial cell hypertrophy and hyperplasia were observed (Fig.2).The gill cellular structure is very sensitive to ammonia exposure, and thus can be used as an indicator for ammonia stress[47].Based on the histological data, apparent damages from ammonia stress can be found in the treatments involving high concentration of ammonia, especially in the concentration of 220 mg/L.The damage of gill from ammonia should be a cause for the mortality at high ammonia concentration.
Superoxide dismutase, the major enzyme in phase 1, was used as an indicator to discuss the physiological defense against ammonia exposure in grass carp in this study.The expression of SOD mRNA asa protection mechanism against oxidative stress caused by ammonia exposure in gill and digestive gland was determined using semi-quantitative real-time RT-PCR analysis.The expression of sod gene is tissue-specific, consistent with are port in sliver carp[48].In the liver, the highest expression level was observed at 151 mg/L ammonia, and it was significantly higher than other treatments; in both the intestine and the gill, the expression up-regulated at high ammonia concentrations.In previous studies, various types of toxicants significantly induce the expression of SOD[49,50].In addition, the decreases in SOD mRNA expression after a specific period of exposure may be caused by reduced metabolic capacity of the organism, consistent with the results of several previous studies, indicating that antioxidant enzyme mRNA expression is both time- and dose-dependent and is related to metabolic capacity[51,52].The up-regulation of sod gene expression further indicated that sod was a useful biomarker to examine ammonia stress in fish.
Hsp90 is widely studied recently as an environmental stress indicator and is related to cellular immune functions[53—57].In grass carp, two isoforms of hsp90 have been identified, i.e., hsp90α and hsp90β.The isoform of hsp90α could be induced greatly by environmental stressors[58].In this study, the expression of hsp90 was tissue-specific.In gill, hsp90 was up-regulated and in liver the expression reached the maximum at ammonia concentration of 104 mg/L and then decreased.The tissue-specific difference of hsp90 gene expression in grass carp indicated that different tissues had different sensitivities to ammonia exposure.The decrease in hsp90 expression in liver could be due to chemical-induced injury[59], and gill is the most sensitive organ to external stimuli as it is directly exposed to the external environment[41], therefore in this study we observed that hsp90 expression in gill increased with increasing ammonia concentration.As described in previous studies, hsp90 is involved in protein folding, cytoprotection, proteomic degradation, and a number of cellular pathways.Toxicants exposure could surely induce the expression of hsp90 gene[60].It could be inferred from this study that hsp90 could be significantly induced by ammonia exposure to cope with the protein damage caused by ammonia exposure, and the protection against ammonia exposure was tissue-specific[61].
In conclusion, ammonia caused large-scale mortality in grass carp and the 24h LC50was about 243 mg/L.The ammonia exposure had dramatic impact on sub- cellular levels, including cellular structure impairments and up-regulation of stressor proteins.The up-regulations of sod and hsp90 mRNA expressions after ammonia exposure were closely related to ammonia concentration, indicating that these two biomarkers were good indicators to evaluate the effect of ammonia exposure, and ammonia exposure had an apparent impairments to grass carp even in sub-lethal concentration of ammonia.
[1] Van W A.Aerobic and anaerobic ammonia production by fish [J].Comparative Biochemistry and Physiology Part B: Comparative Biochemistry, 1983, 74(4): 675—684
[2] Baird R, Bottomley J, Taitz H.Ammonia toxicity and pH control in fish toxicity bioassays of treated wastewaters [J].Water Research, 1979, 13(2): 181—184
[3] Zhao J.Acute toxicity of ammonia to the early stage-larvae and juveniles of Eriocheir sinensis H.Milne-Edwards, 1853 (Decapoda: Grapsidae) reared in the laboratory [J].Aquaculture Research, 1997, 28(7): 517—525
[4] Harris J O, Maguire G B, Edwards S, et al.Effect of ammonia on the growth rate and oxygen consumption of juvenile greenlip abalone, Haliotis laevigata Donovan [J].Aquaculture, 1998, 160(3-4): 259—272
[5] Durborow R M, Crosby D M, Brunson M W.Ammonia in fish ponds [J].Journal of the Fisheries Research Board of Canada, 1997, 32: 2379—2383
[6] Wrigley T J, Toerien D F, Gaigher I G.Fish production insmall oxidation ponds [J].Water Research, 1988, 22(10): 1279—1285
[7] Kearney M, Simpson S J, Raubenheimer D, et al.Modelling the ecological niche from functional traits [J].Philosophical Transactions of the Royal Society B: Biological Sciences, 2010, 365(1557): 3469—3483
[8] Mommsen T P, Hochachka P W.The purine nucleotide cycle as two temporally separated metabolic units: A study on trout muscle [J].Metabolism, 1988, 37(6): 552—556
[9] Whitfield M.The hydrolysis of ammonium ions in sea watera theoretical study [J].Journal of the Marine Biological Association of the United Kingdom, 1974, 54(3): 565—580
[10] Bergerhouse D L.Lethal effects of elevated pH and ammonia on early life stages of walleye [J].North American Journal of Fisheries Management, 1992, 12(2): 356—366
[11] Ip Y K, Chew S F, Randall D J.Ammonia toxicity, tolerance, and excretion [J].Fish Physiology, 2001, 20: 109—148
[12] Augspurger T, Keller A E, Black M C, et al.Water quality guidance for protection of freshwater mussels (Unionidae) from ammonia exposure [J].Environmental Toxicology and Chemistry, 2003, 22(11): 2569—2575
[13] Hillaby B A, Randall D J.Acute Ammonia Toxicity and Ammonia Excretion in Rainbow Trout (Salmo Gairdneri) [J].Journal of the Fisheries Research Board of Canada, 1979, 36(6): 123—138
[14] McKenzie D J, Randall D J, Lin H, et al.Effects of changes in plasma pH, CO2and ammonia on ventilation in trout [J].Fish Physiology and Biochemistry, 1993, 10(6): 507—515
[15] Arillo A, Margiocco C, Melodia F, et al.Ammonia toxicity mechanism in fish: Studies on rainbow trout (Salmo gairdneri Rich.) [J].Ecotoxicology and Environmental Safety, 1981, 5(3): 316—328
[16] Falconi M, O'Neill P, Stroppolo M E, et al.Superoxide dismutase kinetics [J].Methods in Enzymology, 2002, 349(5): 38—49
[17] Liu Y, Wei L L, Li L, et al.Effect of formalin inactivated Flavobacterium columnare on expression of immune related genes in grass carp, Ctenopharyngodon idellus [J].Acta Hydrobiologica Sinica, 2008, 32(6): 794—801[刘毅, 隗黎丽,李莉, 等.福尔马林灭活柱状黄杆菌对草鱼免疫相关基因表达的影响.水生生物学报, 2008, 32(6): 794—801]
[18] Fridovich I.Superoxide radical and superoxide dismutases [J].Annual Review of Biochemistry, 1995, 64(1): 97—112
[19] Hass M A, Massaro D.Regulation of the synthesis of superoxide dismutases in rat lungs during oxidant and hyperthermic stresses [J].Journal of Biological Chemistry, 1988, 263(2): 776
[20] Yoo H Y, Chang M S, Rho H M.The activation of the rat copper/zinc superoxide dismutase gene by hydrogen peroxide through the hydrogen peroxide-responsive element and by paraquat and heat shock through the same heat shock element [J].Journal of Biological Chemistry, 1999, 274(34): 23887
[21] Dimmeler S, Hermann C, Galle J, et al.Upregulation of superoxide dismutase and nitric oxide synthase mediates the apoptosis-suppressive effects of shear stress on endothelial cells [J].Arteriosclerosis, Thrombosis, and Vascular Biology, 1999, 19(3): 656
[22] Inoue N, Ramasamy S, Fukai T, et al.Shear stress modulates expression of Cu/Zn superoxide dismutase in human aortic endothelial cells [J].Circulation Research, 1996, 79(1): 32
[23] Yoo H Y, Chang M S, Rho H M.Heavy metal-mediated activation of the rat Cu/Zn superoxide dismutase gene via a metal-responsive element [J].Molecular and General Genetics MGG, 1999, 262(2): 310—313
[24] Park H, Ahn I Y, Kim H, et al.Glutathione S-transferase as a biomarker in the Antarctic bivalve Laternula elliptica after exposure to the polychlorinated biphenyl mixture Aroclor 1254 [J].Comparative Biochemistry and Physiology Part C: Toxicology & Pharmacology, 2009, 150(4): 528—536
[25] Goeptar A R, Scheerens H, Vermeulen N P E.Oxygen and xenobiotic reductase activities of cytochrome P450 [J].CRC Critical Reviews in Toxicology, 1995, 25(1): 25—65
[26] Livingstone D R.Contaminant-stimulated reactive oxygen species production and oxidative damage in aquatic organisms [J].Marine Pollution Bulletin, 2001, 42(8): 656—666
[27] Ni D, Song L, Gao Q, et al.The cDNA cloning and mRNA expression of cytoplasmic Cu, Zn superoxide dismutase (SOD) gene in scallop Chlamys farreri [J].Fish & Shellfish Immunology, 2007, 23(5): 1032—1042
[28] Heredia-Middleton P, Brunelli J, Drew R E, et al.Heat shock protein (HSP70) RNA expression differs among rainbow trout (Oncorhynchus mykiss) clonal lines [J].Comparative Biochemistry and Physiology Part B: Biochemistry and Molecular Biology, 2008, 149(4): 552—556
[29] Li F H, Luan W, Zhang C C, et al.Cloning of cytoplasmic heat shock protein 90 (FcHSP90) from Fenneropenaeus chinensis and its expression response to heat shock and hypoxia [J].Cell Stress and Chaperones, 2008, 14(2): 161—172
[30] Cara J, Aluru N, Moyano F, et al.Food-deprivation induces HSP70 and HSP90 protein expression in larval gilthead sea bream and rainbow trout [J].Comparative Biochemistry and Physiology Part B: Biochemistry and Molecular Biology, 2005, 142(4): 426—431
[31] Palmisano A N, Winton J R, Dickhoff W W.Tissue-specific induction of Hsp90 mRNA and plasma cortisol response in chinook salmon following heat shock, seawater challenge, and handling challenge [J].Marine Biotechnology, 2000, 2(4): 329—338
[32] Mukhopadhyay I, Nazir A, Saxena D K, et al.Heat shock response: hsp70 in environmental monitoring [J].Journal of Biochemical and Molecular Toxicology, 2003, 17(5): 249—254
[33] Ruffier P J, Boyle W C, Kleinschmidt J.Short-term acutebioassays to evaluate ammonia toxicity and effluent standards [J].Journal (Water Pollution Control Federation), 1981, 53(3): 367—377
[34] Haywood G P, Canada.Dept of Fisheries Oceans, Pacific Biological Station, et al.Ammonia Toxicity in Teleost Fishes: a Review[M].Nanaimo, B.C.: Fisheries and Oceans.1983, 124—129
[35] Russo R C, Thurston R V.Toxicity of ammonia, nitrite, and nitrate to fishes [J].Aquaculture and Water Quality.World Aquaculture Society, Baton Rouge, Louisiana, 1991: 58—89
[36] Handy R D, Poxton M G.Nitrogen pollution in mariculture: toxicity and excretion of nitrogenous compounds by marine fish [J].Reviews in Fish Biology and Fisheries, 1993, 3(3): 205—241
[37] Lei Y Z, Aquaculture Water Environment Chenmical Experiment[M].Beijing: Agriculture Publisher of China, 2006: 135
[38] Hasan M R, Macintosh D J.Acute toxicity of ammonia to common carp fry [J].Aquaculture, 1986, 54(1-2): 97—107
[39] Ball I R.The relative susceptibilities of some species of fresh-water fish to poisons—I.Ammonia [J].Water Research, 1967, 1(11-12): 767—775
[40] Liu C.Effect of ammonia on the immune response of white shrimpLitopenaeus vannamei and its susceptibility to Vibrio alginolyticus [J].Fish & Shellfish Immunology, 2004, 16(3): 321—334
[41] Park H, Ahn I Y, Lee H E.Expression of heat shock protein 70 in the thermally stressed Antarctic clam Laternula elliptica [J].Cell Stress & Chaperones, 2007, 12(3): 275
[42] Rankin J C, Stagg R M, Bolis L.Effects of pollutants on gills [J].Gills.Cambridge University Press, London, 1982: 207
[43] Post G W, Textbook of Fish Health[M].Neptune City, NJ : T.F.H.Publications, 1987.
[44] Lang T, Peters G, Hoffmann R, et al.Experimental investigations on the toxicity of ammonia: effects on ventilation frequency, growth, epidermal mucous cells, and gill structure of rainbow trout Salmo gairdneri [J].Diseases of Aquatic Organisms, 1987, 3(3): 85—109
[45] Smart G.The effect of ammonia exposure on gill structure of the rainbow trout (Salmo gairdneri) [J].Journal of Fish Biology, 1976, 8(6): 471—475
[46] Woodward D F, Riley R G, Smith C E.Accumulation, sublethal effects, and safe concentration of a refined oil as evaluated with cutthroat trout [J].Archives of Environmental Contamination and Toxicology, 1983, 12(4): 455—464
[47] Lease H.Structural changes in gills of Lost River suckers exposed to elevated pH and ammonia concentrations [J].Comparative Biochemistry and Physiology Part C: Toxicology & Pharmacology, 2003, 134(4): 491—500
[48] Zhang Z W, Li Z, Liang H W, et al.Molecular cloning and differential expression patterns of copper/zinc superoxide dismutase and manganese superoxide dismutase in Hypophthalmichthys molitrix [J].Fish & Shellfish Immunology, 2011, 30(2): 473—479
[49] Park H, Ahn I Y, Lee J K, et al.Molecular cloning, characterization, and the response of manganese superoxide dismutase from the Antarctic bivalve Laternula elliptica to PCB exposure [J].Fish & Shellfish Immunology, 2009, 27(3): 522—528
[50] Zelko I N, Mariani T J, Folz R J.Superoxide dismutase multigene family: a comparison of the CuZn-SOD (SOD1), Mn-SOD (SOD2), and EC-SOD (SOD3) gene structures, evolution, and expression [J].Free Radical Biology and Medicine, 2002, 33(3): 337—349
[51] Zhang J F, Wang X R, Guo H Y, et al.Effects of watersoluble fractions of diesel oil on the antioxidant defenses of the goldfish, Carassius auratus [J].Ecotoxicology and Environmental Safety, 2004, 58(1): 110—116
[52] Jo P G, Choi Y K, Choi C Y.Cloning and mRNA expression of antioxidant enzymes in the Pacific oyster, Crassostrea gigas in response to cadmium exposure [J].Comparative Biochemistry and Physiology Part C: Toxicology & Pharmacology, 2008, 147(4): 460—469
[53] Nover L.Heat Shock Response [M].Ameraica: CRC Press, 1991: 78
[54] Parsell D A, Lindquist S.The function of heat-shock proteins in stress tolerance: degradation and reactivation of damaged proteins [J].Annual Review of Genetics, 1993, 27(1): 437—496
[55] Jakob U, Buchner J.Assisting spontaneity: the role of Hsp90 and small Hsps as molecular chaperones [J].Trends in Biochemical Sciences, 1994, 19(5): 205—211
[56] Johnson J L, Craig E A.Protein folding in vivo: minireview unraveling complex pathways [J].Cell, 1997, 90: 201—204
[57] Csermely P, Schnaider T, Soti C, et al.The 90-kDa molecular chaperone family: structure, function, and clinical applications.A comprehensive review [J].Pharmacology & Therapeutics, 1998, 79(2): 129—168
[58] Hermesz E, Ábrahám M, Nemcsók J.Identification of two hsp90 genes in carp [J].Comparative Biochemistry and Physiology Part C: Toxicology & Pharmacology, 2001, 129(4): 397—407
[59] McCuskey R S, Sipes I G.Introduction to the liver and its response to toxicants [J].Comprehensive Toxicology, 1997, 9: 1—10
[60] Shu Y, Du Y, Wang J.Molecular characterization and expression patterns of Spodoptera litura heat shock protein 70/90, and their response to zinc stress [J].Comparative Biochemistry and Physiology-Part A: Molecular & Integrative Physiology, 2010, 34(4): 487—498
[61] Ali A, Krone P H, Pearson D S, et al.Evaluation of stressinducible hsp90 gene expression as a potential molecular biomarker in Xenopus laevis [J].Cell Stress & Chaperones, 1996, 1(1): 62
急性氨氮胁迫对于草鱼sod和hsp90基因表达及鳃部结构的影响
周 鑫1董云伟2王 芳1董双林1
(1.中国海洋大学水产学院海水养殖教育部重点实验室, 青岛 266003; 2.厦门大学海洋环境学院, 海洋环境国家重点实验室, 厦门 361005)
实验通过测定草鱼的24h半致死浓度, 鳃的细胞结构以及sod和hsp90的表达模式研究了草鱼在组织学和分子生物学水平上对高浓度氨氮暴露的响应。经过半致死实验确定的氨氮24hLC50为243 mg/L试验中草鱼被置于5个浓度的处理组中(50、72、104、151、220 mg/L), 之后取鳃组织进行组织切片分析, 取肝脏、肠和鳃来测定sod和hsp90的表达情况。经过高浓度的氨氮暴露处理, 鳃组织的细胞排列和结构产生了明显的变化, 并且sod和hsp90的表达受到了显著的上调。这些结果表明, 高浓度的氨氮能够损害鳃部的细胞结构并且诱导应激蛋白的表达。这个结果同样显示出,sod和hsp90可以作为评估草鱼氨氮暴露水平的良好指标。
草鱼; 氨氮暴露; 24h半致死浓度; 组织损伤;sod;hsp90
X171.5
A
1000-3207(2013)02-0321-08
10.7541/2013.21
Date: 2012-01-09; Accepted date: 2012-11-17
Founds of the National Program on Key Basic Research Project (2009CB118706)
Brief introduction of the author: Zhou Xin, male, born in Weihai; E-mail: zhouxin19861221@gmail.com
DongYunwei, E-mail: dongyw@xmu.edu.cn
